Abstract
This study investigated the effects of goji berry consumption on serum lipids and body fat. There were 70 healthy participants (control group: n = 35; study group: n = 35). For 8 weeks, the study group consumed goji berry (20 g/day), while the control group received no intervention. Every 15 days, 3 day records of food consumption and physical activity were obtained. At the beginning and the end of the study, measurements were obtained for total cholesterol, triglyceride, high-density lipoprotein (HDL), low-density lipoprotein (LDL), and very-low-density lipoprotein (VLDL). The intervention group showed post-intervention decreases in body weight, body fat, waist circumference, and hip circumference (p < 0.05). This group also showed decreases in total cholesterol, HDL, LDL, VLDL, triglyceride, and LDL/HDL in intra-comparisons (p < 0.001). Furthermore, the group had lower LDL and LDL/HDL and higher mean HDL than the control group (p < 0.001). Goji berry could thus improve blood-lipid profiles and decrease body weight and fat. Future studies on long-term effects and mechanisms may reveal more information about potential health benefits.
Introduction
Goji berry (Lycium barbarum) is a plant that has been used in traditional Chinese medicine for over 2000 years. Recent studies have revealed beneficial health effects of this fruit, which have been attracting attention. 1 Plant products are used as food supplements to promote health, 2 and berries are consumed globally and contain important nutrients such as fiber, vitamins, minerals, and bioactive compounds like carotenoids and phenolic compounds.3,4 In addition, berries may improve human health due to their nutrient content.5,6
Goji berry in particular is increasingly becoming an important for medicinal and pharmaceutical uses.7–9 Goji berry contains polysaccharides, flavonoids, phenolic acids, amino acids and derivatives, and carotenoids. 10 Additionally, it has shown antioxidant, anti-inflammatory, hepatoprotective, and cardioprotective effects.11–13 Substances such as polysaccharides, vitamins, pigments, and extracts derived from goji berry have been shown to have the ability to regenerate age-related retinal damage and have a protective effect on the macula. 14 Goji berry also shows positive effects on lipid metabolism and recovery from oxidative stress, which suggests that it may reduce the risk of certain disorders and diseases related to this type of stress and reduced antioxidant defense.15,16
Despite the clear advantages of goji berries, there are limited studies on their health effects in daily life. We hypothesized that regular goji berry consumption by healthy individuals would have positive effects on serum lipid levels and body fat. Therefore, the aim of this study is to evaluate the effect of goji berry consumption on serum lipids and body fat in healthy individuals.
Material and methods
Sample of study
This research was conducted for 8 weeks between May 2022 and May 2023 and included 70 healthy individuals residing in Famagusta, Cyprus. The participants were aged 19–50 years and had body mass indexes (BMIs) of 20–25 kg/m². The sample size was determined using 80% power (1-β=0.80) and α=0.05 in the software G*Power 3.1.9.2. The result indicated that a total sample size of 68 people would be needed with at least 34 people in each group. The study groups were designed to include 35 people based on this finding.
To ensure group balance, a block randomization method was employed. The blocks were structured in various sequences of XYXY, XXYY, YXYX, YYXX, XYYX, and YXXY, where X represents the control group, and Y denotes the intervention group. Due to the nature of the study, blinding was not feasible, and the study was conducted as open-label.
None of the female participants were experiencing lactation or menopause, and none of the participants had any diagnosis of chronic or acute disease or allergies to goji berries. All participants had not consumed goji berries or similar fruits regularly for the last month, and none were in the process of losing weight, doing heavy physical activities, taking any medication or anti-oxidative vitamin supplements, smokers, or consumers of alcohol.
Study design
This research involved a randomized controlled design. Participants were divided into a control group (n = 35) and study group (n = 35) with similar characteristics such as age, gender, and body weight. Both groups were asked to continue their normal eating habits. In addition, the study group was asked to consume 20 g of dried goji berry per day and not to consume any fruit with high antioxidant content other than the goji berry given during the study period. The control group was asked to eliminate goji berry and similar fruits with high antioxidant content from their diet throughout the research period.
The goji berry products given to the working group were delivered to each participant weekly in transparent bags containing 20 g. At the beginning and the end of the study, blood parameters, anthropometric measurements, and 3 day food-consumption and physical-activity records of participants were obtained. At the beginning of the study and every 15 days, food-consumption records were recorded for 3 consecutive days (2 weekdays and 1 day on the weekend), and 3 days of physical activity were recorded. Participants were informed about the study and signed a consent form certifying that they were volunteering to participate. A flow chart of the study design is shown in Figure 1.
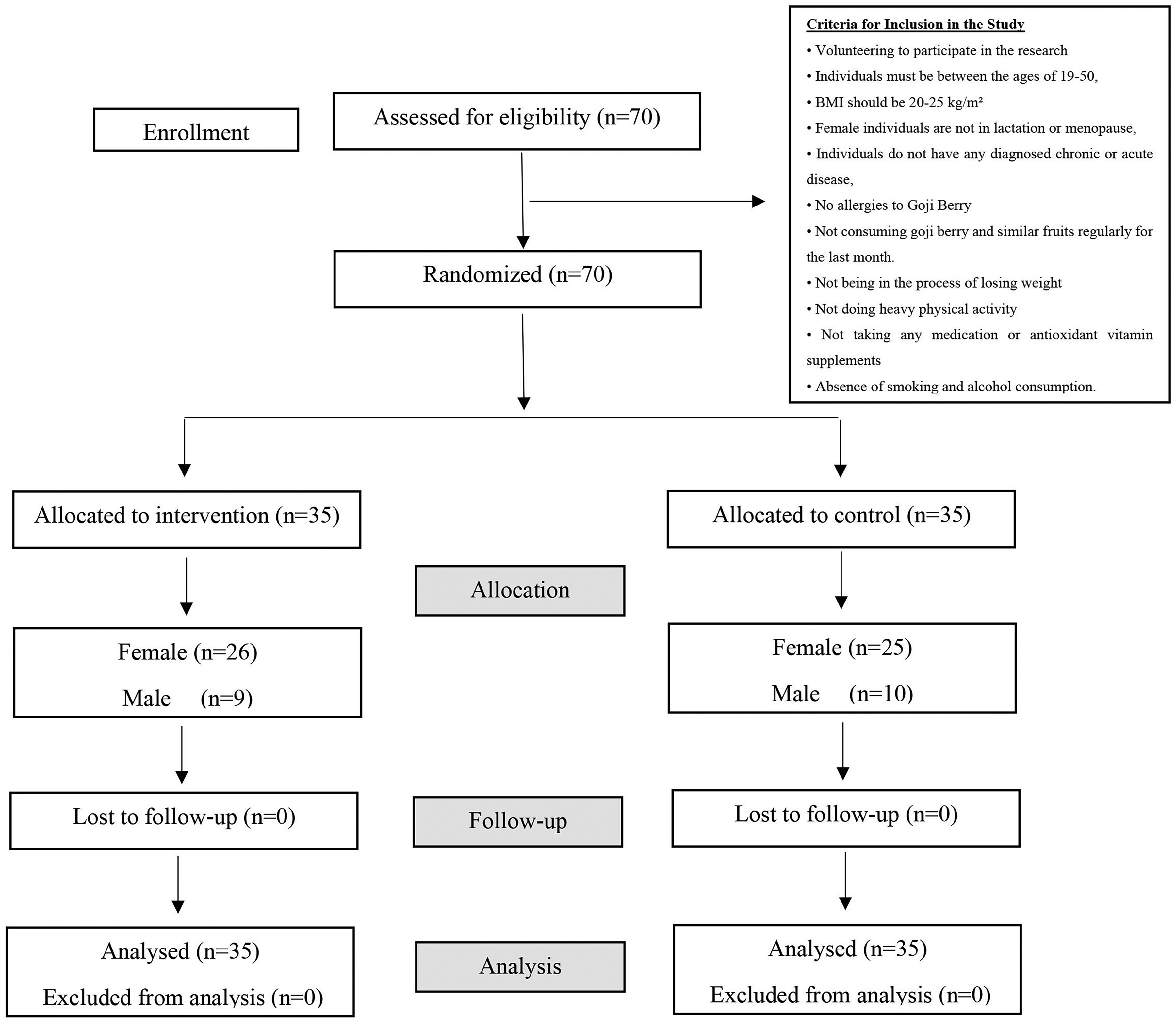
Study-design flow chart. 70 healthy individuals who met the inclusion criteria were selected for the study and divided into control (n = 35) and study (n = 35) groups with the same characteristics considering factors such as age, gender, and body weight. All 70 participants met the inclusion criteria.
This study was conducted in accordance with the guidelines of the Declaration of Helsinki, and all procedures involving human subjects were approved by the Eastern Mediterranean University Research and Publication Ethics Board (Date: 1 March 2022, Document No: 2022/22-01). In addition, the study was registered in the US National Library of Medicine Clinical Trials public registry with registration number NCT06190587.
Goji berry characteristics
The goji berry was obtained from commercial crops in Sivas. It was ensured that the goji berry fruit was from the same garden and collected in the same period. The nutritional content of dried fruit was analyzed by the company that supplied the fruit at Süleyman Demirel University Natural Products Application and Research Center.
The fixed dose of 20 g used in the study was chosen to balance both efficacy and safety. A review conducted by Amagase et al. 7 reported that the range of 10–30 g/day is widely used in clinical studies conducted with humans and that this range is appropriate in terms of both the observation of pharmacological effects and tolerability. The 20 g dose is in the middle of this range, and its scientific validity is supported.
Although some preliminary studies used doses of 30 g/day or higher, these studies are limited in number and had methodological differences. In addition, higher doses may increase the risk of mucosal sensitivity, gastrointestinal discomfort (bloating, diarrhea), and possible interactions with anticoagulants (especially warfarin). 17 Therefore, the 20 g dose was determined as a level at which both biological effects could be observed and safety limits could be maintained. The dried goji berry given to the participants for daily consumption was prepared in bags containing 20 g in a hygienic environment and given by the researcher at certain intervals. Its nutritional characteristics are shown in Table 1.
Nutritional characteristics of goji berry used in the study.

Data collection
A questionnaire form was administered, and anthropometric measurements of the participants were taken in face-to-face interviews. The questionnaire covered demographic information, 3 day food-consumption records, 3 day physical-activity records, anthropometric measurements, and blood parameters.
Food consumption record
Participants’ 3 day food-consumption records were collected and evaluated through face-to-face interviews by a researcher who is a dietitian. Food-consumption amounts were determined using the Food Photo Catalog. 18 Average daily energy and nutrient intakes and average food-consumption amounts were calculated from the 3 day records. The recorded data were entered into the program BEBIS 7.2, and daily energy and nutrient intakes were calculated.
Physical activity
Physical activity levels (PALs) of the participants in a 3 day period were determined using a form included in the questionnaire. The energy expenditure of each physical activity was multiplied by the duration of the activity, and the 24 h total physical activity ratio (PAR) value was obtained. The total PAR value was divided by 24 h for each participant to obtain the PAL values, which were classified as sedentary or light activity (1.40–1.69); active or moderate activity (1.70–1.99); or heavy activity (2.0–2.4). 19
Anthropometric measurements
A bioelectrical impedance analysis device (Tanita BC420S) was used to measure the basal metabolic rate (BMR), body weight, and body composition of the participants. Height was measured using a non-stretchable measuring tape with the feet together and the Frankfurt plane parallel to the floor. Waist circumference was measured from the midpoint between the rib cage and the crista iliaca, and hip circumference was measured from the most protruding part of the hip with a non-stretchable tape measure while the subjects were standing upright. 20 Anthropometric and body-composition measurements were obtained every 15 days during the study. The participants’ daily energy expenditure was calculated using the BMR and PAL values.
Biochemical parameters
Biochemical blood parameters were analyzed, including total cholesterol, triglyceride, high-density lipoprotein (HDL), low-density lipoprotein (LDL), the LDL/HDL ratio, and very-low-density lipoprotein (VLDL). For the analyses, venous blood samples (1 tube = 10 mL) were obtained from participants using a venipuncture method after at least 12 h of fasting and analyzed by experts in a specialized medical biochemistry laboratory. The samples were analyzed by experienced experts in a specialized medical biochemistry laboratory in order to evaluate lipid levels accurately and reliably.
Statistical analysis
Descriptive statistics were calculated for qualitative variables (frequency and percentage) and for quantitative variables (mean and standard deviation). A two-way repeated measures analysis of variance (2 Way RM-ANOVA) was used to evaluate the main effects of time, group, and their interaction. Where appropriate, Tukey's or Sidak's multiple comparison tests were applied for pairwise comparisons. Repeated Measures Analysis of Variance (RM-ANOVA) was conducted to assess changes over time (pre-test/post-test), between groups (control/experimental), and the time × group interaction. Effect sizes were determined by calculating partial eta squared (
Results
Tables 2 to 4 show the evaluation of BMR, PAL, energy expenditure, dietary energy, daily consumption amounts of food groups, and macro- and micronutrient intake pre- and post-interventions in the control and study groups. No statistically significant difference was found between BMR, dietary energy, daily consumption amounts of food groups, and macro and micronutrient intake amounts between pre- and post-intervention in both groups. The control group had significant but clinically negligible differences in PAL (pre-intervention = 1.48 ± 0.08, post-intervention = 1.50 ± 0.08) and energy expenditure (pre-intervention = 2057.67 ± 380.45 kcal, post-intervention = 2089.21 ± 363.23 kcal) between pre- and post- intervention (Table 2).
Evaluation of BMR, PAL, energy expenditure and energy intake in healthy individuals pre- and post-interventions (n = 70).
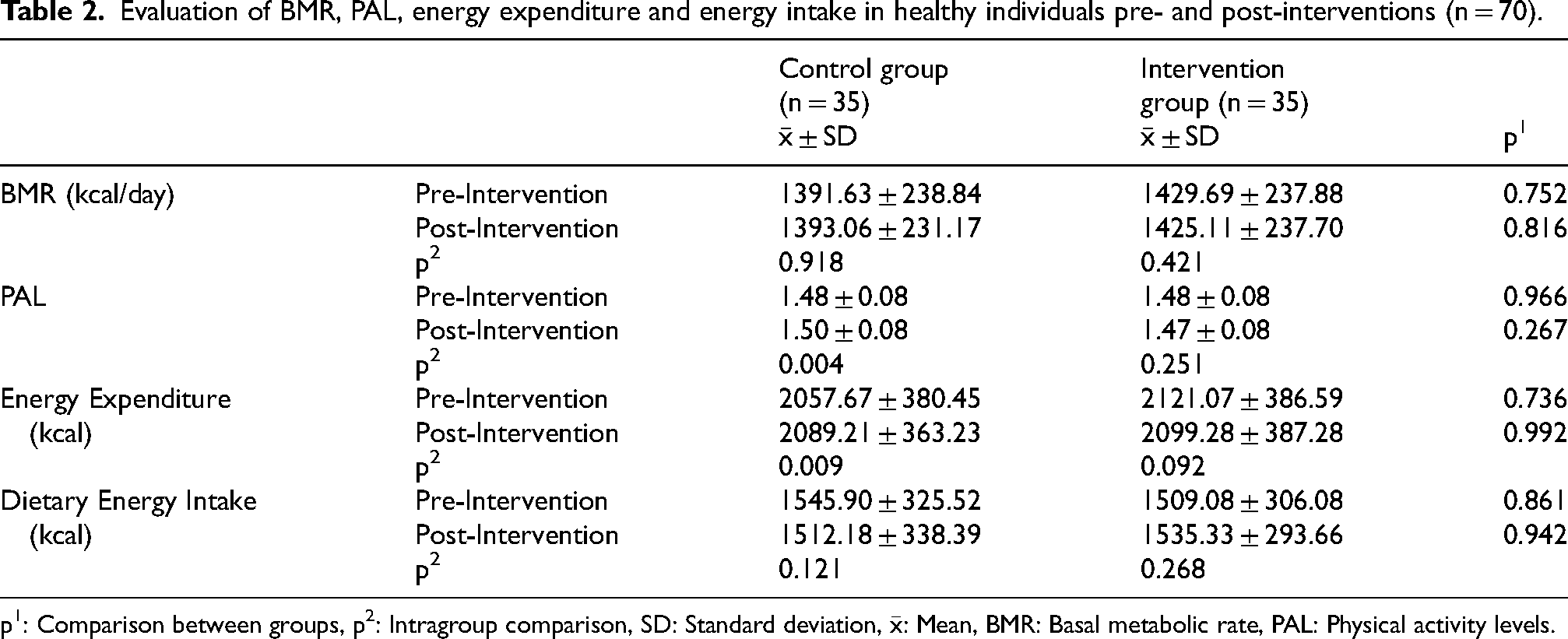
p1: Comparison between groups, p2: Intragroup comparison, SD: Standard deviation,
Evaluation of macro and micronutrient consumption at healthy individuals pre- and post-interventions (n = 70).
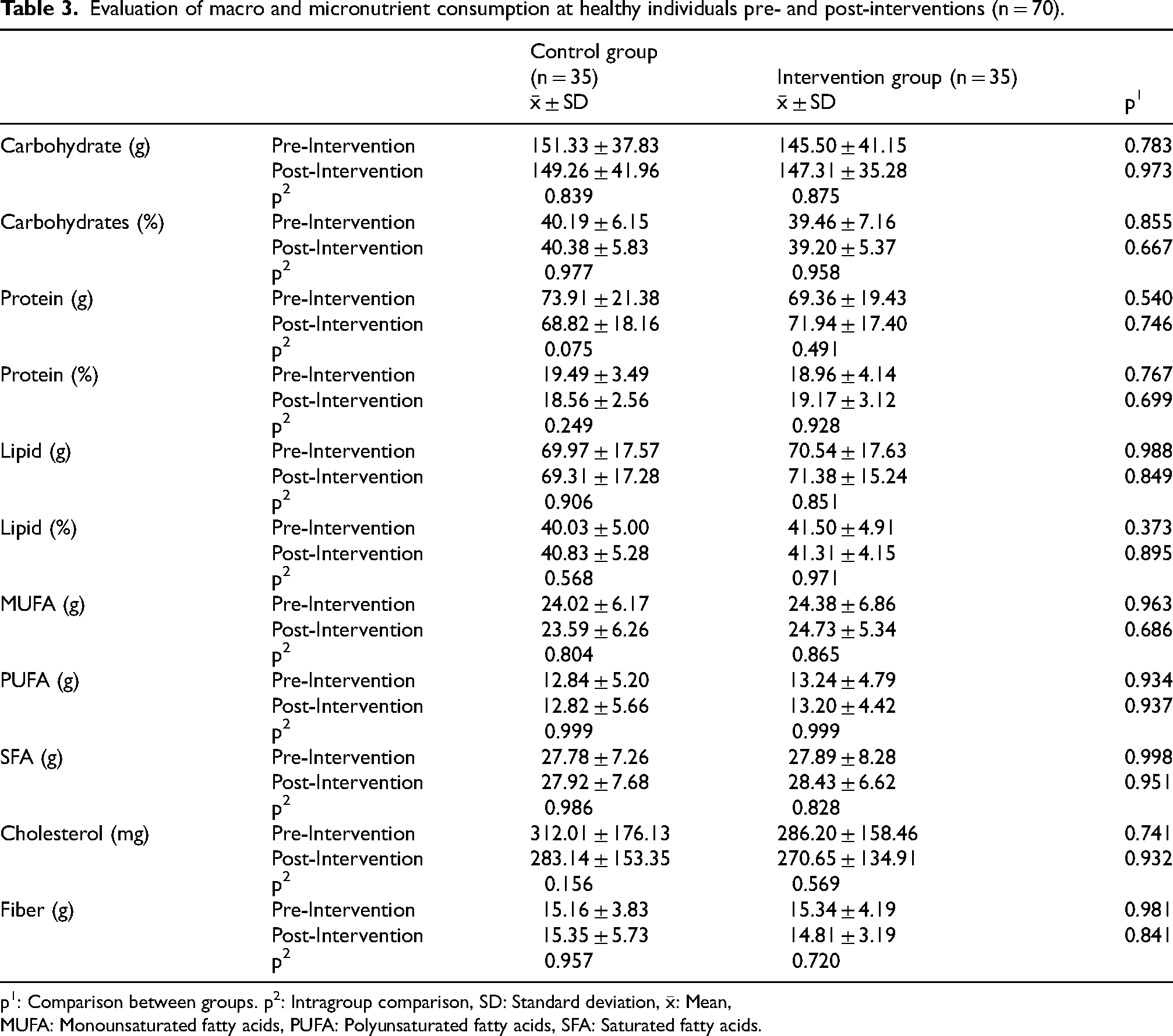
p1: Comparison between groups. p2: Intragroup comparison, SD: Standard deviation,
MUFA: Monounsaturated fatty acids, PUFA: Polyunsaturated fatty acids, SFA: Saturated fatty acids.
Evaluation of daily consumption amounts of food groups in healthy individuals pre- and post- intervention (n = 70).
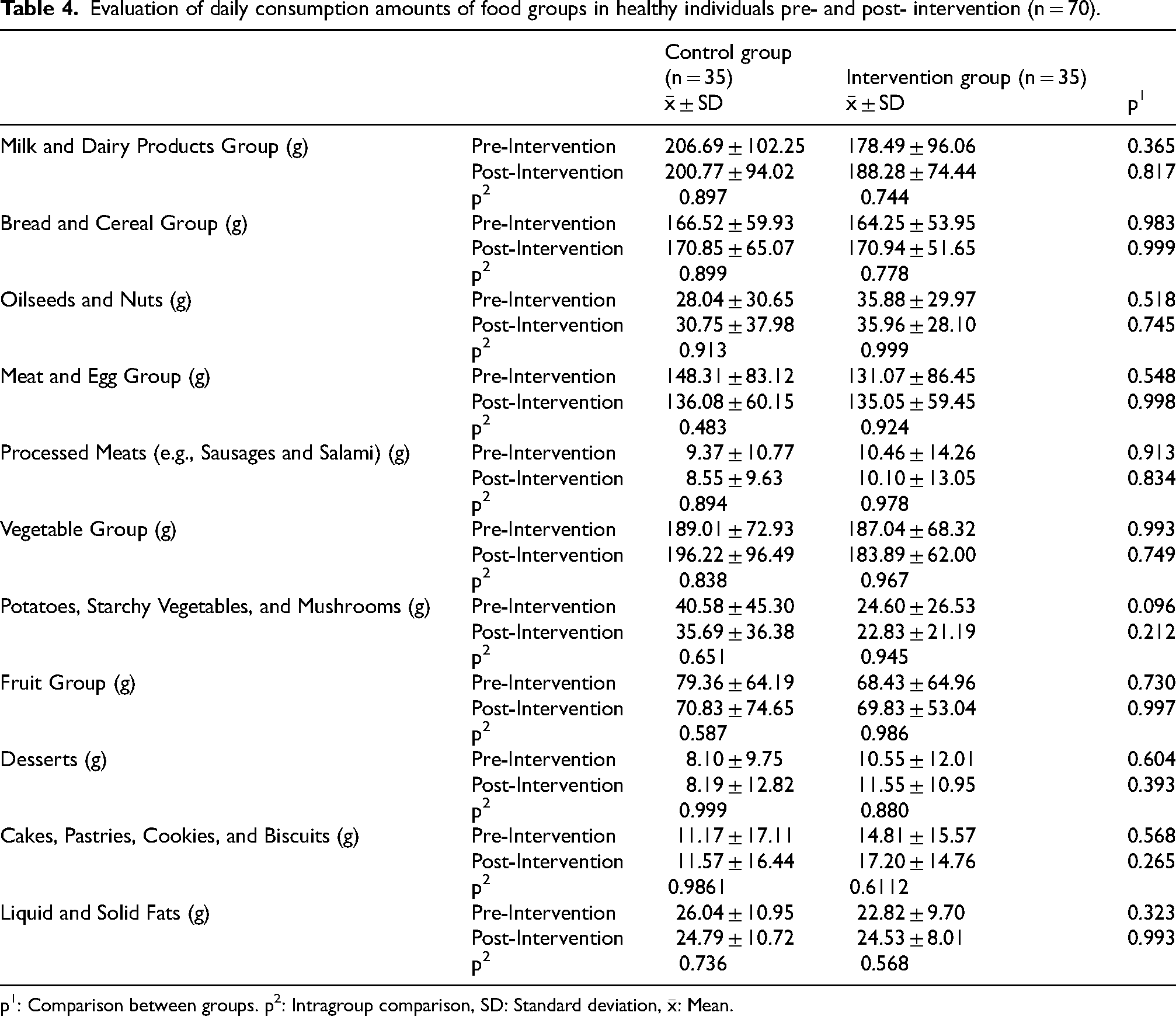
p1: Comparison between groups. p2: Intragroup comparison, SD: Standard deviation,
No statistically significant difference was found between the groups in terms of mean body weight, body fat, waist circumference, and hip circumference, but there was a decrease in mean body weight, body fat, waist circumference, and hip circumference between pre- and post-intervention in the intervention group (−1.19%, −4.19%, −1.70%, and −1.30%, respectively). Large effect sizes were found for body weight (
Evaluation of anthropometric measurements at healthy individuals pre- and post-interventions (n = 70).
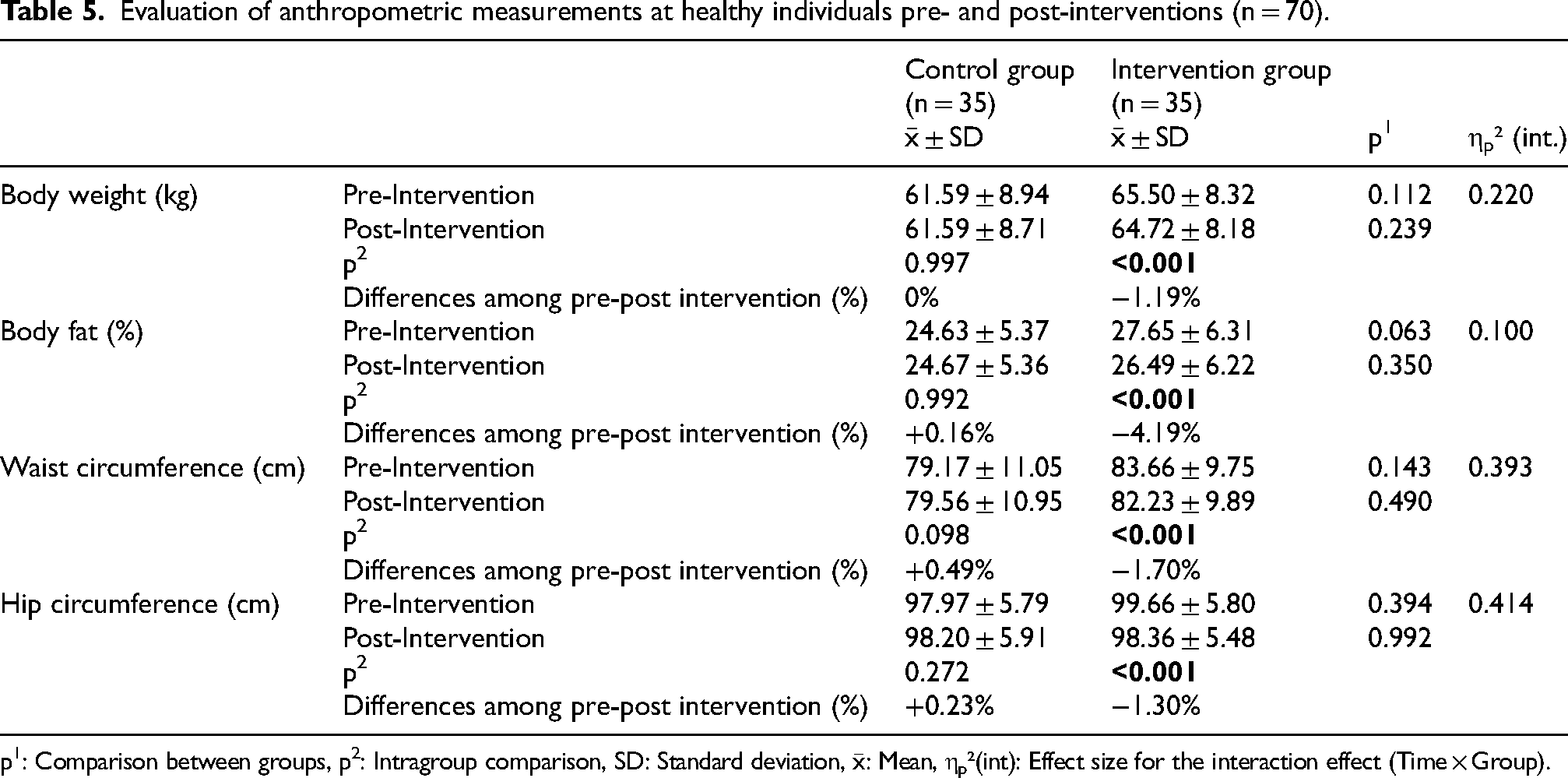
p1: Comparison between groups, p2: Intragroup comparison, SD: Standard deviation,
The mean total cholesterol and LDL values increased in the intra-comparisons of the control group, while the intervention group showed decreases in mean total cholesterol, LDL, VLDL, triglyceride, and LDL/HDL in intra-comparisons (p < 0.001). In addition, the intervention group had lower mean LDL and LDL/HDL values and higher mean HDL values than the control group (p < 0.001) (Figure 2). Large effect sizes were observed for total cholesterol (
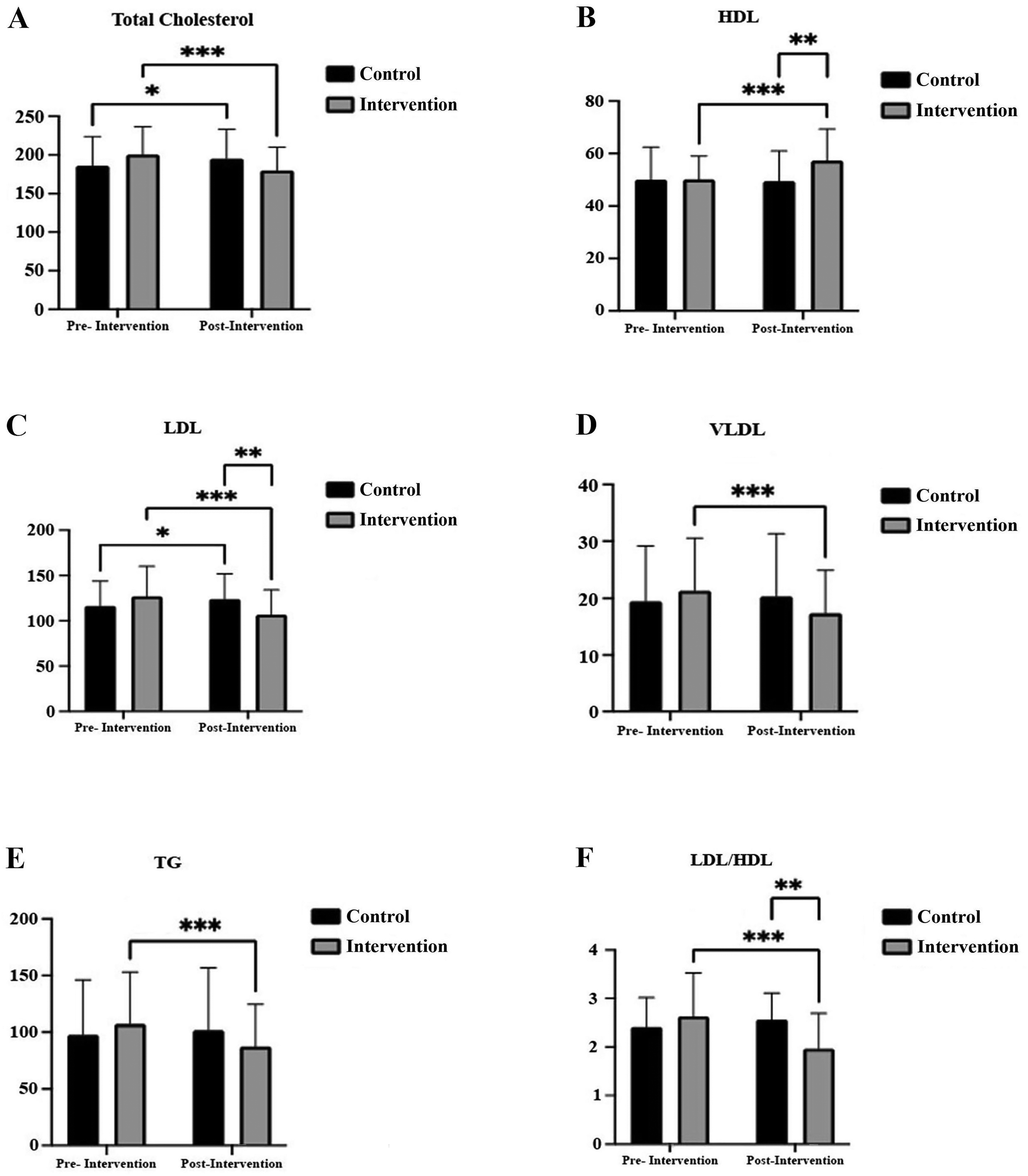
Total cholesterol, HDL, LDL, VLDL, triglyceride, and LDL/HDL measurements in healthy participants (n = 70) pre- and post-intervention. (A) shows that total cholesterol ratio decreased in the intervention group (ηp² = 0.391, pre–post difference = −10.2%), and (B) shows that HDL ratio increased (ηp² = 0.120, pre–post difference = + 14.23%). (C) shows that LDL ratio decreased in the intervention group (ηp² = 0.452, pre–post difference = −15.96%), and (D) shows that VLDL ratio decreased (ηp² = 0.249, pre–post difference = −18.50%). (E) shows that triglyceride ratio decreased in this (ηp² = 0.244, pre–post difference = −18.63%), and (F) shows that LDL/HDL ratio decreased (ηp² = 0.331, pre–post difference = −25.48%). Statistical differences. *p < 0.001 Intragroup comparison of control group, **p < 0.001 Comparison of groups at post-intervention, ***p < 0.001 Intragroup comparison of intervention group.
Discussion
This study examined the effects of goji berry consumption on total cholesterol, HDL, LDL, VLDL, triglyceride, and LDL/HDL ratio in healthy individuals. The results confirmed the hypothesis that regular consumption had a positive effect on serum lipid levels and body fat. In a previous study, individuals who consumed goji berry juice for 14 days experienced a significant decrease in abdominal circumference of 5.5 ± 0.8 cm on average. 22 Another study was conducted with overweight adults in a control group and goji berry consumption group. Both groups were directed to consume 1200 kcal/day, and the goji berry group consumed 120 ml/day of goji berry juice for 14 days. The berry group experienced a decrease of 5.54 ± 0.65 cm in abdominal circumference, while the control group had a decrease of only 0.88 ± 0.83 cm. 23
In the present study, after 8 weeks of goji berry consumption, the mean body weight, body fat, waist circumference, and hip circumference decreased in the intervention group (p < 0.05). Additionally, the intervention group showed decreases in mean body weight and body fat between pre- and post-intervention (pre–post difference = −1.19%, −4.19%, respectively) (Table 5). The decreases in body weight and fat percentage may be beneficial for metabolic health. In particular, weight loss of 5–10% may provide improvements in insulin sensitivity, inflammation, and lipid metabolism. 24 It has been indicated that goji berry can reduce body fat through its high antioxidant and fiber content. 25
Another study treated alloxan-induced diabetic and hyperlipidemic rabbits for 10 days with an oral decoction of crude and purified polysaccharide from L. barbarum. All treatments led to reduced total cholesterol, triglyceride, and LDL levels, along with increased HDL levels. 26 Zhao et al. 27 indicated that L. barbarum polysaccharide decreased serum levels of total cholesterol and triglyceride in rats with streptozotocin-induced diabetes. In the present study, total cholesterol (Figure 2A), LDL (Figure 2C), and VLDL (Figure 2D) decreased in the intervention group (p < 0.001). Our results for LDL and VLDL are in line with those of Park et al., 28 who studied Chinese individuals and found that the LDL decreased with the intake of phytosterols obtained from goji berry.
Duru and Barıt 29 showed that consumption of goji berry leaves reduced LDL. Huang et al. 30 investigated the effects of glycoconjugates and glycans obtained from L. barbarum on inhibiting LDL peroxidation and found that glycoconjugates can inhibit LDL peroxidation, but glycans had no effect on this process. Our study results suggested that consuming goji berry decreased triglyceride levels (Figure 2E) and increased HDL (p < 0.001) (Figure 2B). Previous studies have found that L. barbarum extract also decreases total cholesterol, triglyceride, and VLDL levels while increasing HDL levels in animal-model studies.31–33
Decreases in total cholesterol (10.2%), LDL (15.96%), VLDL (18.50%), triglycerides (18.63%) were observed, and a 14.23% increase in HDL levels was found. These changes were not only statistically significant but had strong effects according to Cohen's ηp² (
A systematic review showed that goji berry regulates lipid metabolism, reduces triglyceride levels, and supports cardiovascular health. In addition, polyphenols, flavonoids, and carotenoids in goji berry contribute to the reduction of LDL levels and the increase of HDL levels by reducing lipid peroxidation. Hence, goji berry consumption may exhibit anti- atherosclerotic and inflammatory effects. 36 Goji berry could be a good dietary supplement for the reduction of triglyceride levels and help to promote heart health.37,38
Earlier studies have also revealed positive benefits of goji berry supplementation by blocking the expression of factors that promote the production of fat in the liver or reducing the level of triglycerides in the bloodstream.25,39 However, De Freitas Rodrigues et al. 40 suggested that consumption goji berry juice is considered healthy but does not affect triglyceride levels. Nevertheless, goji berry has long been used in traditional Chinese medicine as a part of a healthy diet that decreases triglyceride levels. 41
Studies have mostly used animal models and goji berry extract and did not include the effects of the food matrix, dietary patterns, and human characteristics. This is a key strength of the present study, which showed the potential effects of lowering serum lipids resulting from integrating goji berry consumption into daily dietary patterns.
There are several limitations to this study. Firstly, the 20 g may not be sufficient for therapeutic effects to become more apparent. The daily dose of 20 g of goji berry used in our study showed positive effects in healthy individuals while, studies conducted in terms of therapeutic effect are generally obtained at doses above 20 g due to pharmacodynamic responses.7,42,43 Therefore, more controlled studies are needed to evaluate its effectiveness, especially in clinical patient groups. Secondly, the 8-week follow-up period may not have been sufficient to examine the long-term effects of goji berry, and the limited number of participants may have limited the generalizability of the results. Therefore, more comprehensive and long-term studies are recommended.
Conclusion
The findings of this study showed that regular consumption of goji berry can improve blood-lipid profiles and reduce body weight and body fat. These effects demonstrate the potential of goji berry in the management of cardiometabolic risk factors, especially dyslipidemia and obesity. Accordingly, goji berry could be integrated into a healthy dietary pattern and weight-management programs as a functional food to support the effects of healthy diet and reduce the risk of chronic diseases. However, to clarify the long-term sustainability of these effects, the underlying biological mechanisms, and the therapeutic effects on cardiovascular diseases and obesity, more randomized controlled studies with longer durations are needed to assess the optimal amounts of goji berry consumption.
Footnotes
Acknowledgment
Authors have no acknowledgments to declare.
Ethical
This study was approved by the Eastern Mediterranean University Research and Publication Ethics Board (Date:01/03/2022, Document No: 2022/22-01).
The study was included in the US National Library of Medicine Clinical Trials public registry with the registration number NCT06190587.
Author contributions
All authors read and approved of the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was financially supported by Research Advisory Board of Eastern Mediterranean University (Scientific research project number BAPC-0D-22-01).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data sharing
The authors do not have permission to share data.
Author declarations
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Sources of support
The research was financially supported by Research Advisory Board of Eastern Mediterranean University (Scientific research project number BAPC-0D-22-01).
