Abstract
As the use of artificial intelligence (AI) in healthcare becomes more pervasive, its application in the clinical care for those with Parkinson's disease (PD) presents exciting opportunities to improve the timeliness and accuracy of diagnosis and to optimize treatment. Perhaps more than many other neurological conditions, PD lends itself to the creation of rich data from which AI can surface actionable insights that hold the potential to revolutionize PD care in a personalized manner. For direct clinical care, AI has the potential to contribute to initial diagnosis, disease subtyping, symptom identification and severity assessment, progression monitoring, and treatment management. With patients and clinicians informing AI and contributing to its methods of deployment, an enormous opportunity exists to open up access to expert-level care to many more people.
Introduction
The field of medicine is beginning to benefit from the application of artificial intelligence (AI), “computer systems able to perform tasks normally requiring human intelligence, such as visual perception, speech recognition, and decision making.” 1 Machine learning (a subtype of AI that uses algorithms that can learn from and make predictions or decisions based on data instead of operating from predefined rules) and natural language processing (another subtype of AI that enables computers to understand, interpret, and manipulate human language) are increasingly being used in healthcare to assist with knowledge management, clinical diagnosis, and treatment decision-making. 2 Over recent years, increasing computing power, availability of large and ever-increasing amounts of data, and improved algorithms have converged to enable exciting opportunities to apply AI to characterize the transition from health to disease, measure disease burden, and track disease progression or regression–all useful capabilities to improve clinical care. Particularly when coupling data from various sensors on the body or in the environment with clinically validated algorithms, there is an opportunity to enable near-continuous, data-driven monitoring to empower patients and inform optimal clinical management. For disorders of the brain like Parkinson's disease (PD), these new capabilities are urgently needed, since the diagnosis and monitoring of these conditions still largely relies on relatively crude and subjective methods, such as the clinical history and examination, subjective patient reports, and symptom scales.
Though this article will highlight the use of AI for clinical care for those with PD, it is worth mentioning that other applications of AI will no doubt benefit those affected by PD, including AI applied to biomarker discovery and therapy development. For direct clinical care, AI has the potential to contribute to initial diagnosis, disease subtyping, symptom identification and severity assessment, progression monitoring, and treatment management. With patients and clinicians informing AI and contributing to its methods of deployment, an enormous opportunity exists to open up access to expert-level care to many more people.
Initial PD diagnosis
A number of other conditions can mimic Parkinson's disease, and arriving at an accurate diagnosis as soon as possible allows implementation of the most appropriate treatment plan. Clinicopathological studies have shown that clinicians diagnose the disease incorrectly in about 25% of patients. In these studies, the most common reasons for misdiagnosis were presence of essential tremor, vascular parkinsonism, and atypical parkinsonian syndromes. 3 The application of AI to various data types–whether brain scan images, sensor data, voice recordings, or body photographs–has shown the utility in improving the accuracy of the diagnosis of PD. For example, applying machine learning models to the analysis of relatively standard brain MRI scans allowed excellent discrimination of Parkinson's disease from atypical syndromes. 4 This model differentiated two atypical parkinsonian syndromes (multiple system atrophy, MSA, and progressive supranuclear palsy, PSP) from PD with excellent performance.
Beyond static brain scan images for diagnosis, time-series data from inertial sensors, such as accelerometers and gyroscopes placed on the upper or lower extremities or contained within a smartphone, generate a rich, continuous data stream that provides information on movement throughout daily life–highly relevant for diagnosing movement disorders like PD. Algorithms that extract parameters of gait, identify the presence and characteristics of tremor, or quantify kinematic features in the performance of various motor tasks, such as hand movements or walking tests, have been shown in numerous studies to be able to identify those with a clinical PD diagnosis from healthy controls or from essential tremor.5–7 Nevertheless, many of these investigations employed sensors placed in multiple locations of the body, which is not practicable for real-world use outside the lab and at scale. Thus, studies demonstrating the capacity for accurate and early diagnosis of PD by using accelerometer data merely from a single, wrist-worn device are particularly exciting. For example, machine learning models trained using accelerometry data from such a device achieved excellent performance in distinguishing both clinically diagnosed Parkinson's disease and prodromal Parkinson's disease up to 7 years pre-diagnosis from the general population. 8
Owing to the common occurrence of speech abnormalities in PD, such as hypophonia and dysarthria, it is not surprising that AI applied to voice recordings might help to diagnose PD. Indeed, several studies have demonstrated that machine learning models analyzing even brief voice recordings can distinguish individuals with Parkinson's disease from healthy controls with high accuracy.9–11
Since diminished facial expression is common in PD, analysis of facial images might be useful in diagnosing PD and, in fact, a number of studies have demonstrated that AI models analyzing facial features can distinguish people with PD from healthy controls with moderate to high accuracy. 12
Rather than relying on a single data type, applying AI to multimodal data, it stands to reason, may allow the diagnosis of PD with even greater accuracy and at earlier stages. The power of this approach was recently demonstrated in a framework that integrated audio speech patterns, motor measures, neuroimaging data, and cardiovascular signals to achieve exceptional diagnostic precision. 13
Beyond providing a binary diagnosis, methods employing AI to interrogate multimodal data sets are also emerging as a means of identifying patient subgroups that each have a similar symptom profile, disease course, and/or expected treatment response. 14 This will likely help in counseling individual patients about their expected clinical course and in efficiently personalizing treatment, overcoming some of the challenges that the heterogeneity of PD presents.
PD symptom monitoring and treatment management
Traditionally, information obtained from the neurological history and examination during a clinic visit offers only a narrow window of insight into the type and severity of PD symptoms being experienced by each individual, and it may not adequately reflect a patient's functioning at home. Additionally, ascertaining the extent of disease progression through these methods is difficult, other than when gross clinical changes have occurred.
AI-enabled analysis of cross-sectional and longitudinal data, especially of time-series data collected by sensors, provides insights about PD that are objective, quantitative, more frequently measured, assessed in the context of ordinary daily life, and of greater relevance to the patient and their family.15,16 Much can be concluded from analysis of digital measures collected passively, including quantification of tremor, gait, and dyskinesia.17,18 Passive tremor classification data is readily measured from a smartwatch, with calculation of the proportion of time with tremor being an example of a measure that can be derived for various time epochs. A variety of gait-related metrics can also be extracted from wearables and smartphones carried on the body, with reduced arm swing, increased stance time, slower gait cadence, increased total double support time, and increased turn duration being some of the most informative measures. Algorithms applied to passively collected data can also detect and quantify choreoathetotic dyskinesia, adventitial movement typically associated with peaks in dopaminergic medication that can interfere with purposeful movement.
This ability to monitor periods of increased parkinsonian symptom severity (“off time”) and periods in which abnormal, involuntary movements occur due to excessive dopaminergic medication levels (“dyskinetic time”) allows construction of digital motor diaries. Especially when these daily motor fluctuation pictures are assessed for consistent patterns over weeks or months, highly actionable insights can be derived to adjust medication dosage and timing. 19 This pattern recognition and the translation of these patterns into explanations that are understandable by patients and actionable by patients or clinicians alike is a capability well-suited for AI.
Another approach is to apply AI to data collected during specific, structured tasks that are performed periodically. Indeed, these “virtual motor exams” provide for a systematic evaluation of motor function using a series of tasks designed to measure signs and features of PD, such as rest and postural tremor, upper extremity bradykinesia, lower-extremity bradykinesia, gait, and postural sway. 20 Measures derived from these structured tasks may augment the value of measures that are collected purely on a passive basis.21,22
Environmental sensor technology is another approach to measuring a variety of aspects of health that are relevant to PD. Obviating the need to wear a sensor on the body, such systems use low-power wireless signals and measure the reflections of these signals from the environment and from people within a defined space. Machine learning algorithms are applied to analyze the reflected radio signals to compute measures of activity, such as gait speed. In one study, these measures of people with PD over the course of up to a year made at home strongly correlated with clinical scale PD assessments, provided a sensitive marker for tracking disease progression over time, and captured symptom fluctuations in response to medications and their impact on daily functioning. 23
Gaining insight into the symptoms, severity, and course of PD is helpful, but the newest and probably most practical application of AI is in the guidance of the ongoing treatment management of PD. Clinical use cases include adjustment of medications used to provide symptomatic treatment, identification of motor complications that may indicate a need to consider advanced therapies (such as drug pumps, focused ultrasound neuroablation, or deep brain stimulation), and helping to guide the programming of devices that administer advanced therapies. Combining AI-analyzed data and automated reports aimed at patients and caregivers or designed specifically for clinicians can efficiently identify opportunities for intervention (such as adding or adjusting a dose of medication) and provide clear indicators of whether or not a therapeutic or adverse response has occurred. 24
Future perspectives
As AI-enabled clinical decision support becomes more robust in its clinical validation, more sophisticated in its capabilities, and more widely used, there is an immense opportunity to enhance access to expert-informed care guided by real-time data.25–28
Not surprisingly, though, people with PD have mixed views on the value of AI for their personal care management. In one report, interviews with a small cohort of those with PD highlighted that AI tools may have the power to increase patient agency, by surfacing and analyzing data relevant to daily lives directly to patients. 29 There was skepticism, however, about the ability of AI models to take into consideration the heterogeneity of patients and variability of clinical issues in providing personalized care information. Another report highlights that patients, when considering the opportunity to receive timely insights from algorithms that analyze their data, looked upon this benefit as helpful in guiding symptom management and facilitating their care–while also expressing concerns about privacy, data ownership, and the implications for their relationships with care providers. 30
Therefore, as AI becomes increasingly incorporated into PD diagnosis and care, it will be important to ensure that patient trust is not eroded, since the presence of AI can reduce direct interaction with a patient's care team. This will require close partnership between those who develop care management systems and their users–patients and clinicians.
Two evolving principles in the development of AI applications for healthcare to help build trust in their use that will certainly be relevant to those with PD are first ensuring that AI processes and outputs are explainable and that they are developed by including those with relevant clinical expertise.
Explainable artificial intelligence refers to AI systems explicitly designed to make their analytical processes and derived outputs transparent and understandable to patients and clinicians. Rather than making predictions that seem to derive from a mysterious “black box,” explainable AI aims to make clear the rationale underpinning AI outputs, thereby enhancing trust, accountability, and safety in clinical practice. 31
As the pace of integration of AI into clinical care accelerates, it seems important to involve clinicians throughout the development of these systems to support care delivery. This concept of a “clinician-in-the-loop” model is increasingly recognized as essential for AI applications in healthcare, emphasizing the importance of embedding expert clinician oversight and judgement into AI workflows to ensure that AI-driven insights are clinically useful and usable. Recent studies support the premise that clinician involvement in AI-based decision-making processes can improve the reliability of predictions, prevent algorithmic errors, and foster patient trust.32,33
The availability of AI for use in the clinical care of those with PD has ushered in a new era that has the promise to bring patients a more timely and accurate diagnosis, better and more seamless integration and analysis of PD-relevant data, and expert-informed recommendations to guide clinical decisions and management. Already these new AI-enabled capabilities are empowering patients and caregivers to better understand their condition in real time and over time, which enables them to be more optimally informed partners in their care (Table 1 and Figure 1).
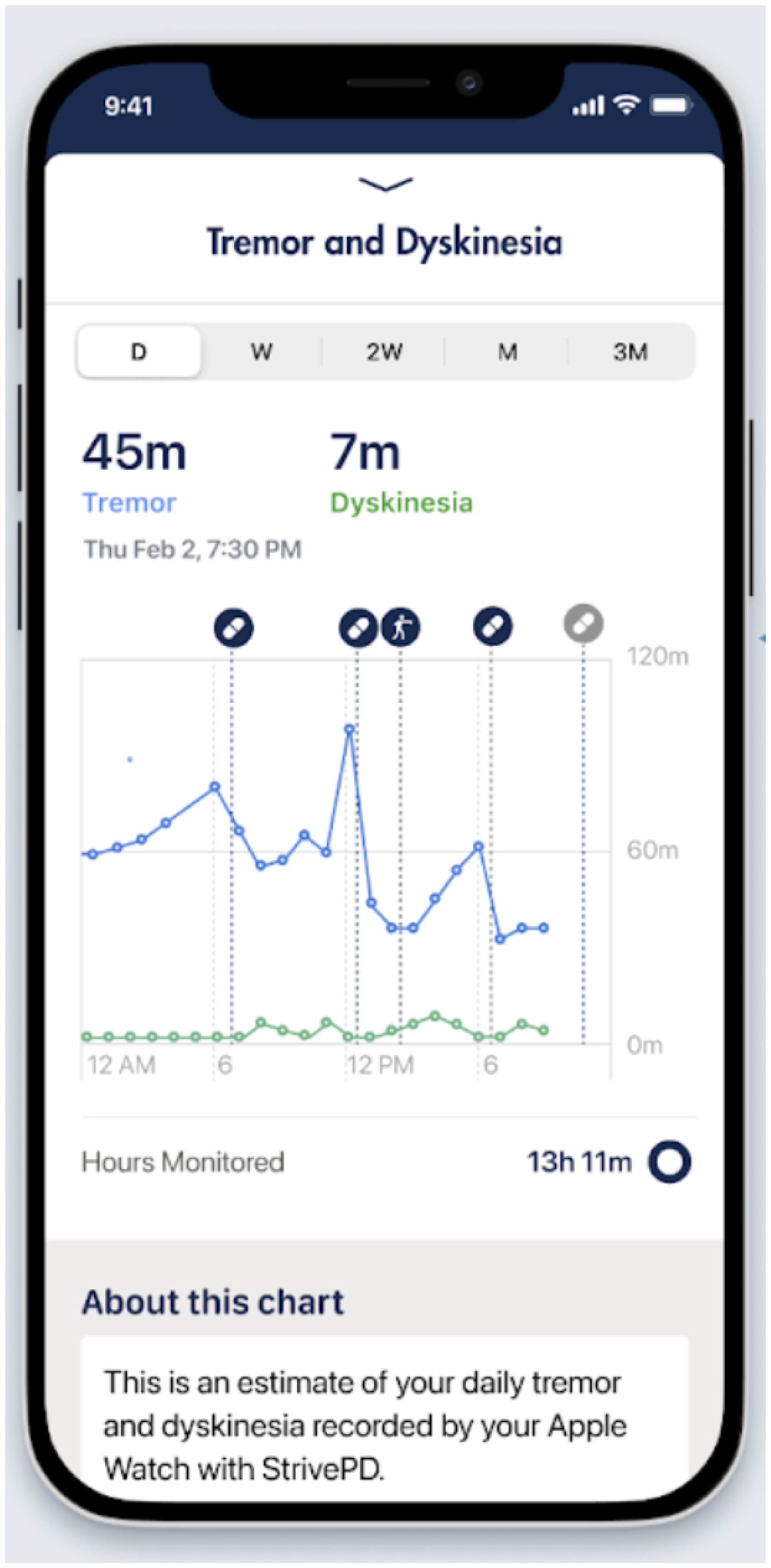
Example of PD symptom data presentation based on AI-analysis of data from wearable and smartphone sensors. 34
How artificial intelligence (AI) can improve PD care: key points.

