Abstract
The central cholinergic system plays a crucial role in neural communication and physiological regulation, mediated by acetylcholine (ACh) and cholinergic receptors in the central nervous system (CNS). In this review, we explore the extensive distribution and impact of the central cholinergic system and its pivotal involvement in Parkinson's disease (PD). Despite PD being traditionally perceived as primarily a dopaminergic disorder, it exhibits significant cholinergic alterations, contributing to both motor and non-motor symptoms. These PD-specific alterations manifest as neuroanatomical changes, diminished acetylcholinesterase activity, and functional disturbances across various brain regions, impacting cognition, mood, sensory perception, sleep, and motor function. Comprehension of these cholinergic dysfunctions is paramount for the development of targeted therapies aimed at alleviating these PD symptoms. To provide further insight, we explore the therapeutic potential of nicotinic acetylcholine receptors (nAChRs) in PD, highlighting their role in preventing apoptosis, modulating neuroinflammation, and mitigating CNS damage. In summary, this review underscores the critical importance of cholinergic mechanisms in PD pathology and champions cholinergic-based interventions for enhanced patient outcomes and improved quality of life.
Anatomy of the central cholinergic system
Acetylcholine and cholinergic receptors
The central cholinergic system is comprised of neurons and glia that secrete the neurotransmitter acetylcholine (ACh) in response to acetylcholinergic receptor (AChR) binding with agonists such as acetylcholine and nicotine. 1 The synthesis of ACh requires the substrates acetyl coenzyme A (acetyl-CoA) and choline to be catalyzed together by choline acetyltransferase (ChAT). 2 The amine molecule choline is derived from an ammonia molecule (NH3), whereby one or more of the three hydrogens are replaced by a functional group, maintaining the solitary nitrogen atom. The thioester molecule acetyl-CoA can be produced by several reactions, including fatty acid β-oxidation, pyruvate decarboxylation by the pyruvate dehydrogenase complex, or the catabolism of the amino acid threonine, leucine, or isoleucine. The catabolism of ACh into choline and acetate is accomplished by acetylcholinesterase (AChE) at the postsynaptic surface. 3 Subsequently, choline returns to the presynaptic terminal via high-affinity choline uptake proteins to be recycled into new ACh. Therefore, the activity rate of AChE influences acetylcholinergic synaptic transmission. The longer that synaptic ACh is available and the greater the quantity is, the greater the signal transmission to the postsynaptic neuron will be. Furthermore, Ach in the synaptic cleft signals to nearby glia to secrete their acetylcholine-binding proteins (AChBPs). 4 AChBP is a homopentamer that is about the size of the subunit N-terminus. Importantly, it contains nAChR binding sites for agonists as well as competitive antagonists, like d-tubocurarine and a-bungarotoxin. 5 Therefore, AChBP also modulates acetylcholinergic synaptic transmission through interfering with the ability of post-synaptic receptors to receive their ligand. Figure 1 presents a schema of ACh metabolism at the synaptic cleft.
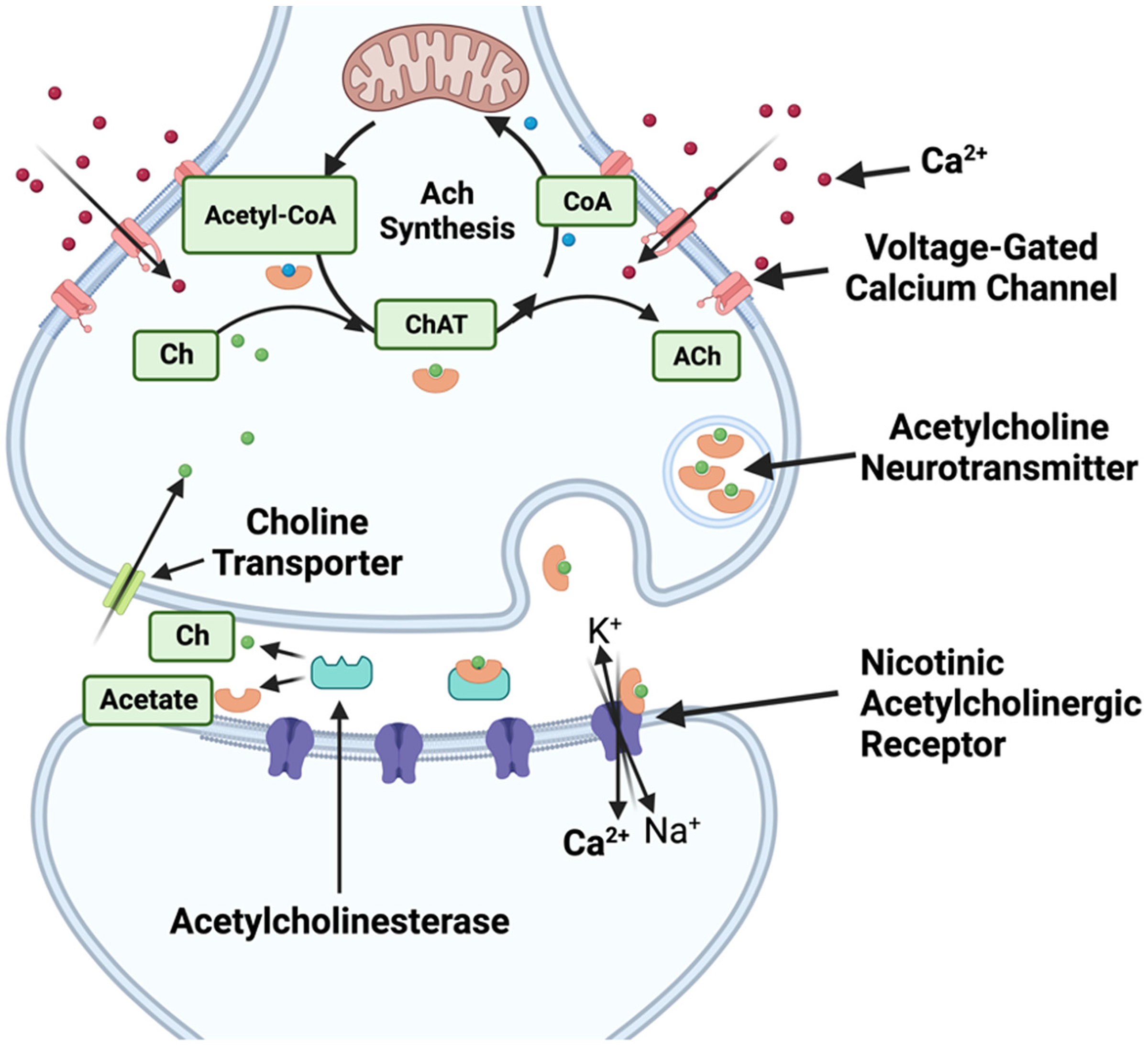
Acetylcholine synthesis and catabolism at a central neuronal synaptic cleft. Pyruvate is converted into acetyl-CoA, which exits the mitochondria. In the cytosol, acetylcholine (ACh) is synthesized by choline acetyltransferase (ChAT), using acetyl-CoA and choline (Ch) as substrates. ACh enters the synaptic cleft and is quickly broken down by acetylcholinesterase (AChE) into Ch and acetate, which exit the cleft. Ch is then recycled within the presynaptic neuron. The remaining ACh binds to either nicotine or muscarinic acetylcholine receptors.
There are two types of AChRs: muscarinic and nicotinic. Muscarinic acetylcholine receptors (mAChRs) are members of the class A G-protein coupled receptors and are responsible for initiating intracellular signal transduction cascades. There are five subtypes of mAChRs found in the peripheral and central nervous system (CNS). Predictably, they are denoted as M1 through M5 and are the protein products of genes CHRM1 through CHRM5 respectively. Parsing the specific pharmacology of the mAChR subtypes have been challenging because they share high homology among their orthosteric sites and each receptor has at least one allosteric binding sites6,7 Agonists of mAChRs include acetylcholine, alkaloids muscarine, donepezil, galantamine, nicotine, and rivastigmine. 8
Nicotinic acetylcholine receptors (nAChRs) are ligand-gated ion channels that facilitate the rapid cellular influx of cations down their electrochemical gradient (Figure 2). They belong to the Cys-loop superfamily of other ligand-gated ion channels that include receptors for serotonin, GABAA, GABAC, and glycine. The transmembrane pentameric glycoprotein structure of nAChRs is large, with a molecular mass of about 290 KDa and composed of about 2380 amino acids.
9
The five polypeptide subunits are arranged to form a symmetrical channel with a central water-filled pore. Each of the five subunits is comprised of a long extracellular N-terminus, followed by three hydrophobic transmembrane regions M1 through M3, a large intracellular loop, M4, and finally a short extracellular C-terminus.
10
The ligand-binding domain exists on the N-terminus, is about 210 amino acids in length, and contains binding sites for agonists and competitive antagonists.
5
The binding sites are found at the junction of two homomeric adjacent subunits or at the junction of two heteromeric α and β subunits.
11
In mammals, each of the five subunits can either be one of the eight α subunit types (α2–7, α9, α10) or one of the three β subunit types (β2–4).
12
In mammalian muscle, there are also
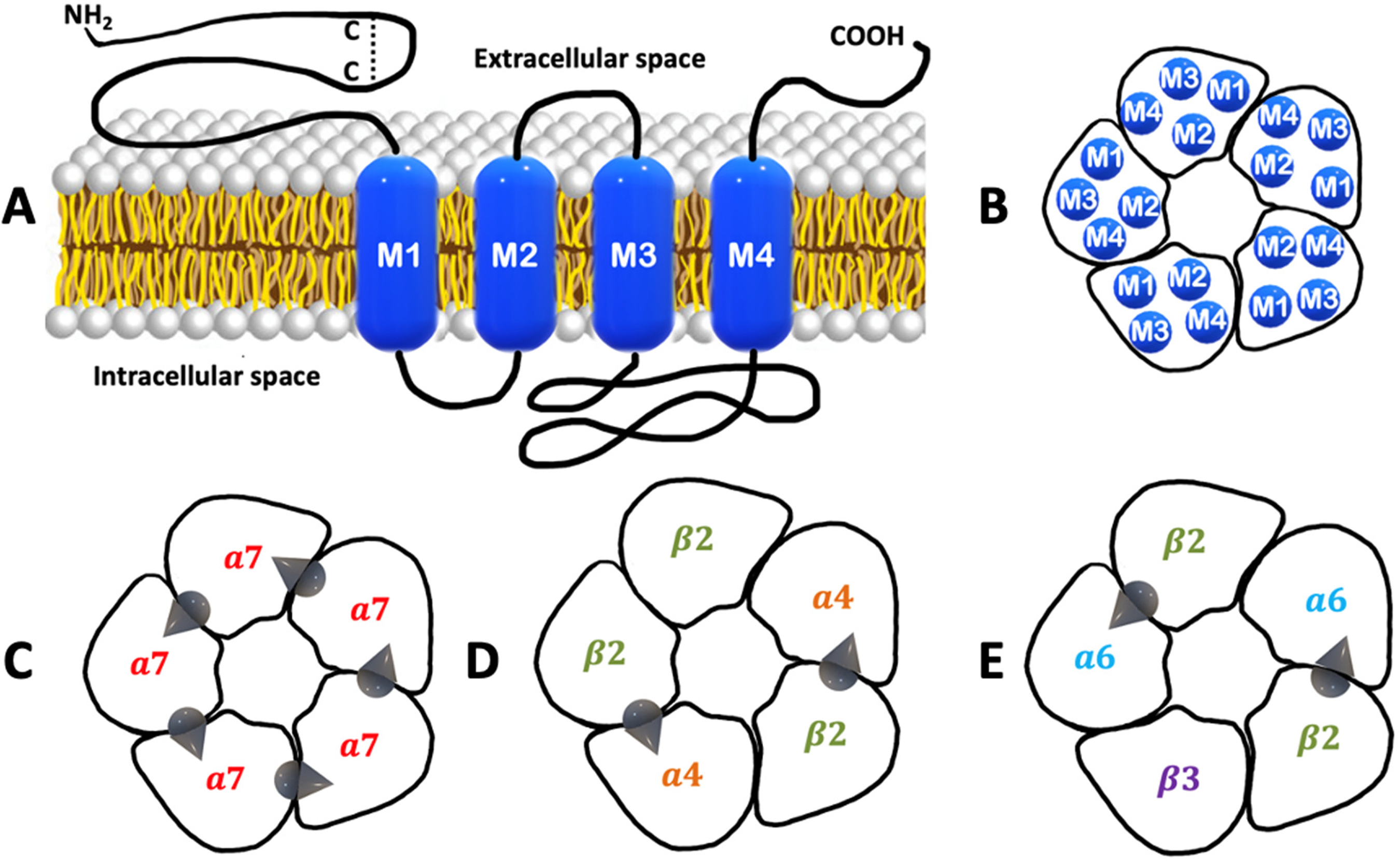
Nicotinic acetylcholine receptor (nAChR) organization. A) Topology includes a long extracellular N-terminus that contains the ligand-binding site and a Cys-loop followed by four hydrophobic transmembrane regions. Between region M3 and M4 there is a long intracellular loop. The extracellular C-terminus is relatively short. B) An overhead view of the pentameric organization reveals that hydrophobic regions are uniformly positioned and that M2 forms the walls of the inner core. C) A homopentameric receptor of α7 subunit types (α7)5.D) A heteropentameric receptor of α4 and β2 subunit types (α4)2(β2)3. E) A heteropentameric receptor of α6, β2, and β3 subunit types (α6)2(β2)3(β3)1. Ligand-binding domains only exist between two homomeric subunit types or between two heterometric α and β subunit types. The two most abundant nAChR types in the brain are example C and D.
Systemic anatomy
The cholinergic system is widely distributed throughout the human CNS with six broad primary origins: the basal forebrain, striatum, cerebral cortex, mesencephalon, cranial motor nerve nuclei, and the anterior horn of the spinal cord. 14 Cholinergic neurons in these areas have been confirmed by both in situ hybridization for ChAT mRNA and antibody immunoreactivity against ChAT. Most of the neurons that are ChAT-immunoreactive have nomenclature according to their anatomical locations, which was originally proposed for use in monkey and rodent brains. This designation was implemented because the ascending projections of cholinergic neurons to the cerebral cortex included some non-cholinergic neurons and do not involve the classic CNS nuclear groups. 15 The nomenclature includes Ch1 for the cholinergic neurons in the medial septal nucleus, Ch2 for those in the vertical limb of the diagonal band of Broca, Ch3 for those in the horizontal limb of the diagonal band of Broca, Ch4 for those in the nucleus basalis of Meynert (nbM), Ch5 for those in the pedunculopontine tegmental nucleus, Ch6 for those in the laterodorsal tegmental nucleus, Ch7 for those in the medial habenula, and Ch8 for those in the parabigeminal nucleus. 15 Simplistically, the cholinergic neurons in areas Ch1 and Ch2 project to the hippocampus, Ch3 projects to the olfactory bulb, Ch4 projects to the amygdala and the cerebral cortex, Ch5 and Ch6 projects to the basal ganglia and thalamus, Ch7 projects to the interpeduncular nucleus, and Ch8 projects to the superior colliculus. The basal forebrain includes Ch1 - Ch4, the mesopontine tegmental nuclei includes Ch5, Ch6, Ch8, and the medial habenuclar nucleus includes Ch7. 14 These structures are illustrated in Figure 3.
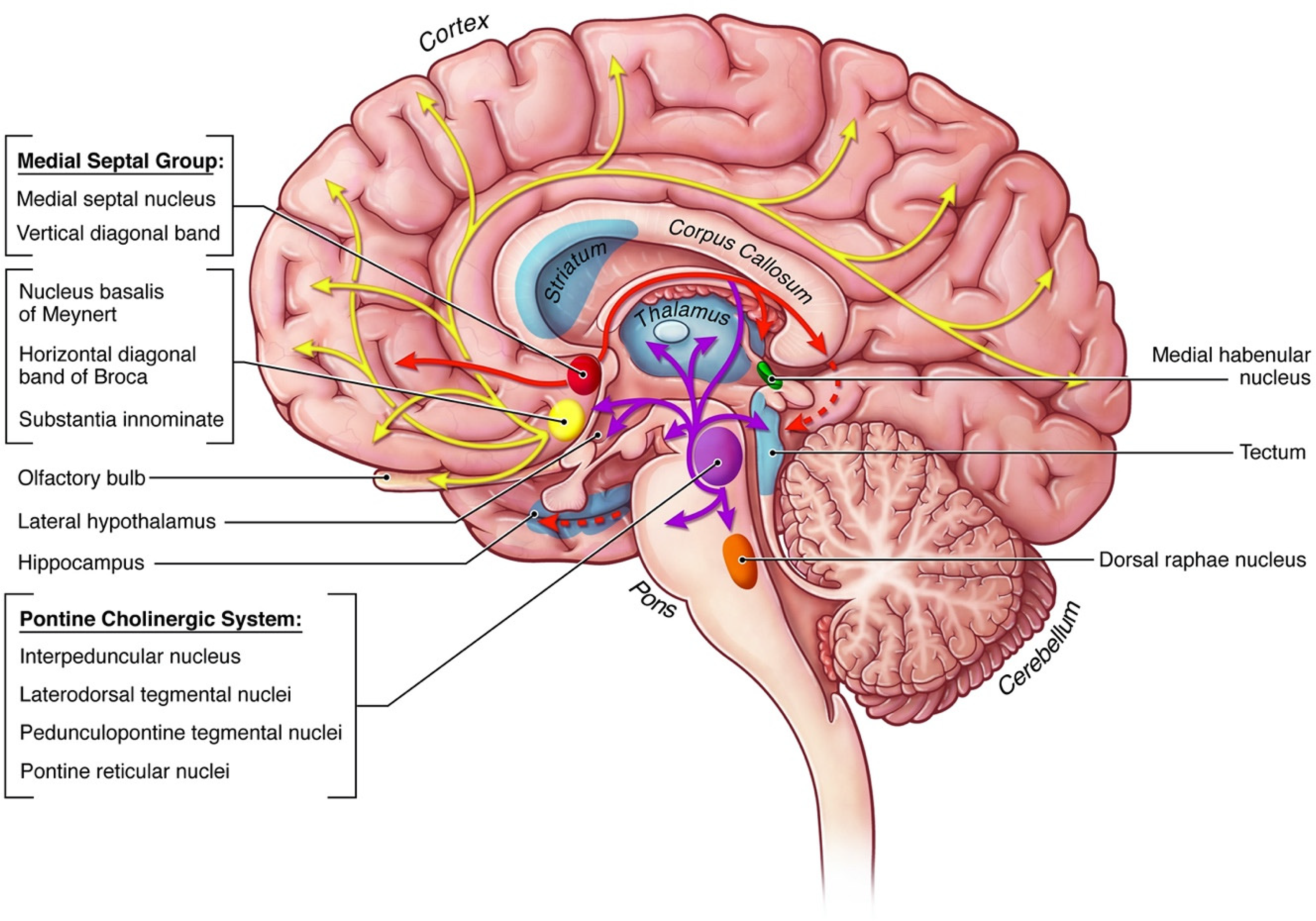
The brain's cholinergic pathways. The basal forebrain (Ch1 - Ch4) is labeled as the “Medial Septal Group”, which includes the medial septal nucleus (Ch1) and the vertical limb of the diagonal band of Broca (Ch2), both of which project to the hippocampus. It also includes the horizontal limb of the diagonal band of Broca (Ch3), which projects to the olfactory bulb, and the nucleus basalis of Meynert (Ch4), which projects to the amygdala and the cerebral cortex. The mesopontine tegmental nuclei (Ch5, Ch6, Ch8) is represented by deep midbrain tracks, labeled as the “Pontine Cholinergic System”, which includes the pedunculopontine tegmental nucleus (Ch5) and the laterodorsal tegmental nucleus (Ch6), both of which project to the basal ganglia and thalamus, and the parabigeminal nucleus (Ch8), which projects to the superior colliculus. The superior colliculus and parabigeminal nucleus are a part of the tectum, as shown in the diagram. The medial habenular nucleus (Ch7) appears green in the diagram. It sends projections to the interpeduncular nucleus. Please see the text for information on its afferent input. The diagram is used with permission from Evan Oto and the Science Photo Library, C036/5511, rights managed.
The basal forebrain (Ch1 - Ch4)
There are four structures in the basal forebrain cholinergic system: the medial septal nucleus, the vertical limb of the diagonal band of Broca (vDBB), the horizontal limb of the diagonal band of Broca (hDBB), and the nbM. These structures send cholinergic projections to the hippocampus, amygdala, and cortex. 16 The medial septum (Ch1) is one of four septal subregions, including the lateral, medial, posterior, and ventral groups of nuclei. These septal regions reside in the medial portions of the cerebral hemispheres, inferior to the corpus callosum, continuous with the septum pellucidum, and anterior to the third ventricle17,18 The medial septum region contains both the medial septal nucleus superiorly and the DBB inferiorly. The medial septal nucleus and the DBB form the septohippocampal pathway, which is composed of both cholinergic and GABAergic innervation to and from the hippocampus. 16 These projections to the hippocampus are critical modulators of hippocampal function. 19
The DBB includes the cholinergic vertical and horizontal limbs, otherwise referred to as Ch2 and Ch3 respectively. 20 The DBB is located directly superior to the optic chiasm, posterior to the anterior perforated substance, and the ventromedial portion of the lateral septum. 14 It runs diagonally from its medial origins in the paraterminal gyrus of the septal area to the lateral innervation targets of the hippocampus, the basolateral amygdala, the lateral olfactory tract, and the piriform cortex. 21 Ch2 runs within the septal gray matter of the nbM at the level of the precommissur and then continues to the hippocampus via the fornix. About 70% of the cells in Ch2 are cholinergic. 22 Ch3 runs parallel to the ventral surface of the basal forebrain and inferior to the substantia innominata towards its primary target of the hippocampus. Although Ch3 has cholinergic cells, they constitute only about 1% of the total cells for this nucleus. 23
The Ch4 complex is the largest of the cholinergic nuclei in the basil forebrain and about 90% of the Ch4 neurons are cholinergic. There is some evidence that Ch4 can be subdivided into various subregions, although there isn't a consensus as to which subdivisions are the most appropriate. 14 Unlike Ch1 - Ch3, which primarily project to the hippocampus, Ch4 provides cholinergic innervation to the neocortex and the amygdala. 24 The anatomical location of Ch4 is more difficult to describe as anatomical terms have changed throughout history and definitions of anatomical boundaries have also changed. However, most recognize Ch4 as residing in the substantia innominata, which is situated between two anterior commissure tracks, inferior to the globus pallidus and superior to the amygdala. 25
The mesopontine tegmental nuclei (Ch5, Ch6, Ch8)
The pedunculopontine tegmental nucleus (PPN) and laterodorsal tegmental nucleus (LDT), Ch5 and Ch6 respectively, have recently had their inputs and outputs reevaluated using Cre-dependent viral tracing with fluorescent micro-optical sectional tomography of whole brains in mice. Both the PPN and LDT receive cholinergic monosynaptic afferent innervations from widely distributed, bilateral, and similar brain regions26,27 Most of their afferents broadly come from the ipsilateral hemisphere and from the midbrain. Interestingly, afferents from a few locations had preferential innervation to either the PPN or LDT. Additionally, some afferents originating from the same locations and projecting equally to both the PPN and LDT exhibited signal strength preferences for either the PPN or LDT. 27
There have been several functional and anatomical divisions of the PPN described including the rostral and caudal divisions, ascending and descending projection divisions, the pars compacta, and pars dissipatus divisions, and even cholinergic and non-cholinergic projections. Cholinergic and non-cholinergic projections from the PPN preferentially innervate ascending targets associated with motor function. 28 For example, the forebrain ascending targets that receive primarily cholinergic and secondarily glutamatergic innervation from the PPN include the external globus pallidus (GPe), internal globus pallidus (GPi), substantia nigra pars reticulata (SNpr), substantia nigra pars compacta (SNpc), subthalamic nucleus, intralaminar thalamus, and dopaminergic neurons in the dorsolateral striatum29,30,31 The PPN also projects cholinergic inputs to the dorsal raphe, locus coeruleus, and the laterodorsal tegmental nuclei of the ascending reticular activating system29,28 Finally, the PPN has some descending projections to structures such as the spinal cord. Additionally, there are some reciprocal subcircuit loops between the PPN and its targets. For example, the PPN sends cholinergic and glutamatergic projections to the SNr and GPi, and in return, the SNr and GPi sends GABAergic projections back to the PPN31,32 Similarly, the PPN sends cholinergic, GABAergic, and glutamatergic projections to the STN, which reciprocates with glutamatergic innervation30,31
Some anatomists consider the rostral PPN, caudal PPN, and the LDT to be merely a rostrocaudal continuation of a single anatomical stretch of glutamatergic, GABAergic, and cholinergic neurons that project to the basal ganglia and thalamus. 33 However, the canonical view is that the LDT and PPN are separate entities, due to how much denser the neuronal populations are in the LDT compared to the PPN34,33 Although the LDT and PPN share ascending targets, the pars rostralis portion of the PPN preferentially targets the substantia nigra pars compacta and the dorsolateral striatum, whereas the pars caudalis portion of the PPN and the LDT preferentially innervates the ventral tegmental area, the dorsomedial and ventral striatum, the medial thalamus, and other targets35,36,37,38,39
The parabigeminal nucleus (Ch8) is a small cluster of cells located laterally on the edge of the midbrain. Almost the entirety of the neurons in Ch8 are cholinergic and supply the superior colliculus. 40 The anatomical and functional connection between Ch8 and the superior colliculus has led many to consider Ch8 as a satellite of the superior colliculus. 41 The circuitry between these two structures involves innervation of Ch8 from the ipsilateral superior colliculus and projections from Ch8 to both the ipsilateral and contralateral superior colliculus. Recent evidence from the rodent Octodon degus indicates that the Ch8 cholinergic neurons also produce glutamate and are functionally divided into anterior and posterior sections. 42 The posterior Ch8 of the Octodon degus exhibits the classic reciprocal network to the ipsilateral superior colliculus. However, their anterior Ch8 receives input from the ipsilateral superior colliculus and projects to the contralateral superior colliculus. The Octodon degus is a uniquely beneficial model organism for studying the CNS cholinergic systems in neurodegeneration as they have been shown to sporadically develop Alzheimer's disease (AD) in old age. 43
The medial habenula nucleus (Ch7)
Compared to other cholinergic nuclei, the medial habenula (Ch7) has not received as much attention, despite its important functions. 44 The habenula nucleus is a component of the epithalamus in the dorsal diencephalon, directly rostral and anterior to the pineal gland. It is divided into lateral and medial subregions and into the cholinergic ventral subnuclei and the dorsal subnuclei, which only secretes substance45,46 The ventral medial habenula (vCh7) receives cholinergic afferent input from the stria medullaris. The stria medullaris is a white matter tract arising from the triangular septal nuclei, the bed nuclei of the anterior commissure, and the septofimbrial nucleus in the posterior septum. It travels between the thalamus and fornix to the vCh745,47 The triangular septal nucleus is one of the primary cholinergic inputs to the vCh7, although there is some evidence that it can also release glutamate and ATP. 44 The vCh7 also receives cholinergic input from the lateral septum via the vertical limb of the diagonal band of Broca (Ch2), although Ch2 might also provide GABAergic innervation. 48 The medial septum provides strong cholinergic innervation to the vCh7, but it has also been reported to provide GABAergic input to vCh7. 49 Dopaminergic afferents from the interfascicular nucleus of the ventral tegmental area have been shown to project to the vCh7. 48 The projection target of vCh7 is the interpeduncular nucleus (IPN) via the fasciculus retroflexus. 50 The IPN receives ACh and glutamate from the vCh751,48
Introduction for cholinergic system in pd
Parkinson's Disease (PD) is a neurodegenerative disorder characterized by bradykinesia (slowed movement) with either tremors or rigidity. 52 Other physical signs and symptoms associated with PD include postural instability, gait shuffling, slurred speech, and reduced facial expressions. Cognitive and emotional dysfunction are also tied to a clinical diagnosis of PD, as they can emerge at later stages of disease and impair quality of life. 53 These non-motor symptoms include anhedonia, depression, sleep-wake cycle disturbance, and changes in sensitivity to pain. Motor deficits caused by PD are due to reduced dopamine levels in the substantia nigra, while the pathophysiology of non-motor symptoms are increasingly recognized to be linked to changes in the cholinergic nervous system.
Growing evidence points to the cholinergic system playing a key role in the progression of motor and cognitive deficits in PD. Common motor symptoms of PD such as gait disturbances and postural instability show lower responsiveness to therapies targeting dopaminergic systems compared to other symptoms like tremor and rigidity. 54 These motor symptoms frequently coincide with cognitive impairments such as dementia, which also respond less robustly to dopaminergic treatment. 55 This clinical pattern indicates the involvement of other neurotransmitter systems, like the cholinergic system, in shaping hallmark symptoms associated with PD. Cholinergic deficits in the PPN and basal forebrain have been implicated in deficits in gait, postural stability, and cognition. Dysfunction of the cholinergic pathways may be the underlying factor for continued resistance to dopaminergic treatment. Non-motor PD symptoms are also shaped by cholinergic innervation. Rapid eye movement sleep behavior disorder (RBD), characterized by muscle atonia and dream-enacting behaviors, are associated with denervation of cholinergic systems in thalamic regions and the brainstem, rather than the striatum. 56 Similarly, executive dysfunction and attention impairments have been linked to cholinergic degeneration in the basal forebrain, which is a neural hub of cholinergic input. 57 Recognizing the central role cholinergic innervation plays in both the motor and non-motor symptoms of PD will expand the therapeutic approaches that offer successful treatment outcomes.
Alterations of the central cholinergic system in Parkinson's disease
Anatomical
PD is primarily characterized and therapeutically treated as a disease of the dopaminergic system. However, the cholinergic system plays a significant role, even in the early stages of the disease. Like the chronic degeneration of the dopaminergic system during the progressive course of PD, the cholinergic system also experiences a progressive loss of neurons, disrupted communication, reduced ligand binding, and a diminished number of receptors. These changes have been observed in living PD patients using molecular imaging, such as AChE positron emission tomography (PET) scans and radio-labeled tracing (Nicolaas I. 58 AChE PET is a common means for identifying and studying the two predominant cholinergic systems in the brain: the cortical system, which has origins in the basal forebrain, and the subcortical system, which has origins in the brainstem PPN and laterodorsal tegmental nucleus. PET imaging has confirmed a reduction in AChE activity in the frontal cortex, striatum, thalamus, and especially in the parietal and occipital cortices of PD patients59,60 The reduction of AChE activity is more pronounced in PD patients with dementia compared to non-demented PD patients, and compared to mild AD patients61,62 A reduction of AChE activity is characteristic of senescent cholinergic neurons. 63 The changes of the cholinergic system in PD somewhat mirror the cadence and patterns of changes observed in the dopaminergic system.
Numerous post-mortem studies and molecular imaging studies of living PD patients have characterized their associated loss of cholinergic neurons. PD patients with dementia have a greater loss of cholinergic neurons originating from the nbM and have a greater loss of ACh receptors in the basal forebrain than PD patients without dementia, although both groups experience these changes (Nicolaas I.. 59 Furthermore, cholinergic degeneration of the nbM is more severe in PD than in AD and it has prognostic value for dementia onset in PD patients25,64 The PPN also shows loss of about 50% of the lateral cholinergic projection neurons during PD pathology65,66 One of the earliest and universal signs of PD is muted olfaction. This deficit is not due to a loss of olfactory sensory neurons, but rather, a loss of cholinergic neurons within the olfactory circuit67,68
In the striatum, interneurons, GABAergic afferents, and dopaminergic afferents from the cortex, thalamus, and midbrain have nAChRs, except for spiny projection neurons and calretinin expressing interneurons. 69 The number of striatal nAChRs are reduced in PD. 70 This has been shown in post-mortem autoradiography of PD patients, which reveals reduced binding of the nAChR-ligand 5-[125I]-A-85380. 71 Single-photon emission computerized tomography (SPECT) confirms a similar reduction of binding of the nAChR ligand [123I]5-iodo-3-[2(S)-2-azetidinylmethoxy]pyridine in non-demented PD72,73 In contrast, a PET study of muscarinic binding with the ligand [11C]N-methyl-4-piperidyl benzilate showed augmented binding values in the frontal cortex of PD patients, which is posited to counteract the loss of nAChR activity74,75
Striatal cholinergic interneurons (ChIs) have D2 dopaminergic receptors that inhibit ACh release upon activation by dopamine. 76 Conversely, ChI depolarization and subsequent ACh release causes dopamine release from dopaminergic neurons that have nAChRs77,78 Therefore, the progressive loss of nigrostriatal dopaminergic neurons in PD results in striatal imbalance between dopamine and ACh. These changes lead to a hypercholinergic state in the putamen, contributing to the bradykinesia characteristic of PD. 79
Functional
Cognition, mood, and sleep
People with PD are at an increased risk for cognitive decline. Subjective cognitive decline (SCD) is an emerging metric in PD assessment. In general, SCD is a condition where the patient reports cognitive decline in the absence of clinical evidence. However, the definition and diagnostic criteria for SCD has yet to be formally established, so the reported prevalence among PD patients varies widely. 80 Due to differences in diagnostic criteria and patient demographics, there is also a variability in the reported percentage of PD patients with mild cognitive impairment (MCI), ranging from about 10% to 63%. 81 According to the Movement Disorder Society diagnostic criteria, a recent meta-analysis concluded that 40% of PD patients have MCI, which comports with other studies. 82 Nearly half of all PD patients will develop dementia (PDD) within ten years of a PD diagnosis and more than 80% will have PDD twenty years after a diagnosis. 83 Dementia with Lewy bodies (DLB) is associated with alpha-synuclein aggregation and differs from PDD merely as a matter of timing. If dementia is diagnosed within one year or before the appearance of Parkinsonian motor symptoms, it is classified as LBD. If dementia is diagnosed after one year of the appearance of motor symptoms, it is classified as PDD84,85
Both DLB and PDD are associated with a significant loss of cholinergic neurons of the basal forebrain, especially the nbM. MRI, PET, and diffusion tensor imaging studies have shown that nbM degeneration is predictive of PDD. Recently, microstructural changes in the nbM have been shown to predict gray matter loss in Ch1 and Ch2 as well as cognitive decline in PD64,86 However, a recent MRI imaging study confirms a reduced volume of the nbM in PD compared to healthy controls, but doesn’t find a correlation between nbM volume and age, cognition, or motor impairment. 87 Perhaps this observed lack of correlation between cognition and nbM volume is due to dopaminergic circadian tone. A recent study has shown that cognition drops with daily dopaminergic OFF periods, which is worsened with the degree of severity in nbM degeneration. 88 Another explanation might be that other cholinergic structures are more important for cognitive decline. Cross sectional imaging of PD patient brains illustrates a gradual development of cholinergic degeneration from posterior to anterior cortical region as symptoms of dementia progress. 86 The cholinergic system dysfunction in the frontal and temporal lobes cause additional executive symptoms relating to attention, learning, and memory. 89
Psychiatric symptoms are also observed in PD, commonly depression, anxiety, apathy, psychosis, and disordered impulse control. 90 There is no stereotypical timing of when psychiatric symptoms will appear during the disease progression. Psychiatric signs can appear before the onset of motor symptoms or at advanced stages of the disease. Often, more than one psychiatric symptom will be present concurrently, such as depression and anxiety. There are many complex and overlapping factors involved in the etiology of mood disorders. For example, a combination of neurochemical systems such as dopaminergic, noradrenergic, serotonergic, and cholinergic systems are responsible for the emergence of these PD-related psychiatric symptoms.. 91 Furthermore, the COVID-19 pandemic was a recent source of increased psychiatric symptoms among PD patients. 92 While there are many factors involved, neuroimaging studies link the presence of these psychiatric disorders with decreased cortical AChE activity and diminished numbers of α4β2 nicotinic receptors across the brain93,94 However, the clinical use of acetylcholinesterase inhibitors to treat these symptoms in PD patients has not been very effective. 95
Sleep disturbance is among the most observed non-motor symptom of PD. However, the reported frequency ranges greatly, between 30% to 90%96,97 The most consistent reporting is about half of the PD population experience rapid eye movement (REM) sleep behavior disorder (RBD). 98 Other PD related sleep disturbances include insomnia, restless leg syndrome, excessive daytime sleepiness, circadian rhythm disorder, and disordered breathing while asleep. 96 Cholinergic degeneration of the PPN contributes to RBD and disruption of slow-wave sleep as well99,100,101 Under non-pathologic conditions, the cholinergic projections from the PPN innervate the dorsal subcoeruleus nucleus (SubCD), which is involved in producing the muscle atonia and the ponto-geniculo-occipital characteristics of REM. 102 Cholinergic innervation of the nucleus reticularis gigantocellularis also contributes to the muscle atonia of REM. 99 Furthermore, experimental studies on patients with RBD have led to the findings that stimulation of cholinergic neurons, especially PPN, can potentially promote REM sleep. 103
Visual and vestibular perception
Hallucinations occur in about 75% of PD patients during the progression of disease. 104 When combined with cognitive impairment and depression, hallucinations can cause great distress to both patients and caretakers. Visual dysfunctions are also common in PD and are a key contributor to falls. 105 These impairments are associated with disruptions in cholinergic and other neurochemical systems, as well as alterations in corticothalamic networks responsible for visual processing. 106 Neuroimaging studies in PD patients experiencing hallucinations have demonstrated decreased cholinergic neurotransmission in both cortical and subcortical visual areas. 59 One particularly intriguing aspect of visual disturbance in PD is the altered perception of visuospatial dimensions and self-motion. Patients frequently experience navigational veering while walking, which often occurs in the direction contralateral to the more affected hemisphere — a phenomenon linked not to motor asymmetry, but to disrupted optic flow perception and alterations in the perceived egocentric midline107,108,109 This rightward attentional bias may be related to subtle left hemineglect arising from dysfunctional parietal-basal ganglia interactions. Enhancing cholinergic, but not dopaminergic or noradrenergic, signaling has been shown to sharpen visuospatial perception, 110 reinforcing the importance of cholinergic tone in spatial orientation. Additionally, starburst amacrine cells, which play a central role in motion detection, lose their dopaminergic input and degenerate in PD, further impairing motion-related visual processing. 111 Abnormalities in vestibulo-ocular reflexes and vestibular signal integration have been documented in PD, suggesting central vestibular involvement that exacerbates disorientation and fall risk,112,59,113 Thus, both visual and vestibular system dysfunctions, compounded by cholinergic and dopaminergic deficits, underlie the complex visuoperceptual disturbances seen in PD.
Olfaction
Abnormality of olfactory functions such as odor detection, discrimination, and recognition functions is another prevalent non-motor symptom of PD, occurring in up to 90% of PD patients. 114 Olfaction deficit is one of the earliest symptoms of PD. It can actually predate motor symptoms by several years115,116 Importantly, olfactory bulb surroundings can help differentiate PD-related olfaction loss from non-PD olfaction loss. 114 Olfaction diminishes progressively over the course of PD, mirroring the extent of neurodegeneration. 117 Multiple studies on olfaction and cholinergic innervation of the limbic archicortex system report a significant connection between cholinergic system destruction and olfactory dysfunction118,119,120 Furthermore, impairment in acetylcholine communication in the hippocampal region is suggested for the occurrence of hyposmia in PD. 121 Performance tests of olfactory functions in PD also reveal that integrity of limbic cholinergic projections is linked with odor identification ability. 122
Vestibular dysfunction
Recent evidence highlights the contribution of vestibular dysfunction and subtle visuospatial biases to postural and perceptual disturbances. Vestibular deficits are increasingly recognized in PD and may contribute to impaired balance, gait instability, and directional veering, particularly during ambulation or visually guided tasks.112,113 These abnormalities are thought to arise from both peripheral and central vestibular processing deficits and may interact with impaired sensorimotor integration in PD. Furthermore, studies have identified a subtle left hemineglect or rightward visuospatial bias in some PD patients, independent of motor asymmetry. 112 This rightward attentional bias may reflect altered hemispheric activation or basal ganglia–cortical network dysfunction and has been proposed as a contributing factor to the rightward deviation of visual scanning or subjective visual straight-ahead, potentially compounding spatial disorientation in affected individuals. Together, these findings suggest that non-motor contributors, and more specifically vestibular and spatial-attentional dysfunction, should be considered alongside visual system pathology in the broader understanding of visuoperceptual symptoms in PD.
Motor symptoms
The primary clinical diagnostic features of PD include rigidity, bradykinesia, resting tremors, postural instability, and compromised balance 123 . People living with PD also experience disordered and freezing gait, as well as frequent falling. 124 Dopaminergic therapy isn’t guaranteed to fully address the classic motor symptoms of PD, especially in the long-term regarding gait, posture, and falling issues125,126 Indeed, a side effect of long-term use of the “gold standard” dopaminergic replacement treatment for PD, levodopa, induces dyskinesia. 127 Congruently, PET studies have associated cholinergic degeneration in the basal forebrain, prefrontal cortex, visual thalamus, and caudate nucleus with falls128,129 Cholinergic degeneration in the brainstem PPN and laterodorsal tegmental nucleus is associated with slow gait and postural reflex impairment,128,128,130; Furthermore, degeneration of cholinergic striatal interneurons and the limbic archicortex is associated with freezing in PD. 128 Acetylcholinesterase inhibitors do not increase gait speed and do not reduce fall incidence, although in a recent meta-analysis, they do improve fall rate towards approaching statistical significance. 131 However, acetylcholinesterase inhibitors have been shown to improve the variability PD patients experience in their stride and step metrics. 131
Acetylcholine receptors as therapeutic targets in Parkinson's disease
Given the widespread involvement of the central cholinergic system in cognitive, sensory, and motor dysfunction in patients with Parkinson's disease, research has focused on targeting acetylcholine receptors. Alterations in cholinergic tone, particularly degeneration of basal forebrain and brainstem cholinergic nuclei, are now recognized as key contributors to the non-dopaminergic symptomatology of PD, including cognitive decline, gait disturbances, and postural instability. These clinical manifestations, which often persist despite dopaminergic therapy, underscore the need for adjunctive strategies that address cholinergic dysfunction. As a result, both muscarinic and nicotinic acetylcholine receptors have emerged as compelling therapeutic targets. Understanding how these receptors modulate neural circuits affected in PD may offer new avenues for symptom management and neuroprotection. The following sections explore the roles of acetylcholine receptor subtypes in the pathophysiology of PD and the current state of receptor-targeted therapeutic development.
Nicotinic receptors
It has been recognized for decades that nicotine exposure reduces the risk of PD. The inverse relationship between PD risk and nicotine use has been associated with smoking tobacco, using smokeless tobacco, exposure to secondhand smoking, and with dietary nicotine132,133 The risk of PD among smokers decreases between 30% and 40%, correlating linearly to the years a person smokes and the recency of being a smoker134,135 This relationship is not due to selective mortality bias or patient sex. 136 Unsurprisingly, the well-established health detriments caused by using tobacco prevents it from being a serious contender for PD therapy. However, nicotine and other nAChRs agonists have been explored as potential disease-modifying agents. Despite many epidemiological, basic, and translational studies supporting the use of nicotine as a protective agent against PD, recent large clinical studies in early-stage PD have failed to show demonstrable benefit. 137 While this has led to some speculating on the possibility of non-nicotine components of tobacco smoke offering neuroprotection, such as carbon monoxide and monoamine oxidase-B inhibitors, nAChRs still represent critical targets for neuroprotection in PD. 136 nAChRs protect against apoptosis, mitigate neuroinflammation, and reduces the burden of chronic senescence.
The α4β2 nicotinic acetylcholine receptor (nAChR) subtype is among the most densely expressed receptor populations in the human brain, particularly in regions affected by PD, including the striatum and frontal cortex. This high receptor density suggests a critical modulatory role for nAChRs in both motor and cognitive circuits, which may become increasingly important as cholinergic deficits emerge alongside dopaminergic degeneration in PD. Varenicline, a partial agonist at α4β2 nAChRs, has been investigated in PD for its potential to enhance attention and gait stability. Clinical studies have shown that varenicline can reduce fall frequency and improve executive function, underscoring the therapeutic relevance of targeting α4β2 nAChRs in PD. 138 In addition, imaging studies have demonstrated what is likely pathologically increased binding of α4β2 nAChRs in patients with PD and decreased receptor occupancy by endogenous acetylcholine. 139 Other preclinical studies have mechanistically investigated the α4β2 receptor, demonstrating selective antagonism with dihydro-beta erythroidine (DHBE), blocks the protective effects of nicotine. 140
Recent findings have also redefined the role of α6-containing nicotinic acetylcholine receptors (α6 nAChRs) in striatal circuitry, with important implications PD. Kramer et al. 141 demonstrated that striatal ChIs can form direct, synaptic-like axo-axonal contacts with dopaminergic fibers, allowing for rapid and localized activation of presynaptic α6 nAChRs. This interaction produces EPSPs within dopaminergic axons, even in the absence of action potentials, representing a novel mechanism of fast cholinergic control over dopamine release. The highly localized and synapse-like nature of this transmission challenges the long-standing view of cholinergic signaling in the striatum as being mediated solely by diffuse volume transmission. Given the early vulnerability of dopaminergic axons in PD, this α6 nAChR-dependent axo-axonal signaling may represent a critical regulatory node in nigrostriatal communication, and potentially a therapeutic target for preserving axonal function and dopamine homeostasis in the disease. These studies highlight the need for continued investigation into nAChR-targeted therapies as a complement to dopaminergic approaches.
Muscarinic receptors
mAChRs also constitute valuable targets for PD treatment. They are widely expressed throughout the striatum and substantia nigra, where they shape the plasticity of dopaminergic neural networks critical for motor control and proper locomotion. 142 Antimuscarinic drugs, such as trihexyphenidyl and benztropine, became the first medications to treat PD symptoms. 143 These agents help restore balance between acetylcholine and dopamine by blocking excess cholinergic activity that results from dopamine depletion in PD. This mechanism of action reduces deviations in motor control. Although this mechanism of action reduces deviations in motor control, there has been a decline in usage of such drugs due to their strong association with cognitive side effects, such as acute delirium and increased risk of dementia amongst older adults,.144,145 However, mAChRs continue to remain critical targets for the development of effective PD treatments. mAChRs modulate neurodegenerative processes associated with PD disease, as different subtypes are involved in regulating neuroinflammation and oxidative stress. Some studies have pointed to the anti-inflammatory effects induced by mAChR activation. 146 Further understanding of the specific roles mAChR receptors play in relation to nAChR will expand therapeutic options.
Protection against apoptosis
Apoptosis is programmed cell death, which is critical in development, normal cell turnover, immune system function, and cellular death in response to toxins. The extrinsic pathway of apoptosis involves external signaling via death receptors such as FasR, TNFR1, DR3, DR4, and DR5. Upon binding of ligands such as specific cytokines, adaptor proteins are recruited to form the death-inducing signaling complex (DISC), culminating in the activation of caspase-8 and subsequent apoptosis. 147 The extrinsic pathway is critical in the innate and adaptive immune system, involving natural killer cells and cytotoxic T-lymphocytes respectively. 148 The intrinsic pathway is initiated by intracellular signals like DNA damage, hypoxia, or toxins, leading to mitochondrial membrane permeabilization and release of pro-apoptotic proteins like cytochrome c. 149 These proteins activate caspase-dependent and -independent pathways, leading to cell death. Regulation of this pathway involves the Bcl-2 family of proteins, including both anti-apoptotic and pro-apoptotic members. 150 The intrinsic apoptotic pathway is implicated in neurodegeneration.
Post-mitotic neurons require suppression of apoptosis to help maintain longevity. Neurons have unique apoptosis suppressing mechanisms that go awry during neurodegeneration. For example, neuronal suppression of PTBP1 allows for splicing of the Bak1 microexon 5, leading to decreased production of pro-apoptotic BAK1 proteins. 150 In PD patients, increased levels of active caspase-3, BAX, and reduced BCL-2 suggest aberrant activation of the intrinsic apoptotic pathway. 151 PRKN gene mutation is related to mitochondrial dysfunction and inherited forms of PD. Interestingly, the protein product of PRKN also potentially regulates apoptosis by targeting BAK and BAX. 152
Both in vivo and in vitro PD models have demonstrated that activation of α7 nAChRs leads to neuronal and glial protection against apoptosis in response to a variety of toxic stimuli. For example, nicotine and the selective α7 nAChR agonist PNU-282987, suppress apoptosis induced by alpha-synuclein preformed fibrils in SH-SY5Y neuronal culture by preserving Bcl-2 and Bax levels, preventing mitochondrial membrane permeability, and increasing alpha-synuclein clearance. 153 Additionally, SH-SY5Y culture treated with the electron transport chain complex I inhibitor 1-methyl-4-phenylpyridinium (MPP+), experience toxin-induced apoptosis, which is inhibited through nicotine or PNU-282987 treatment via the ERK/p53 pathway and suppressing the apoptosis-related cleaved enzymes poly(ADP-ribose) polymerase-1 (PARP-1) and caspase-3154,155 Mesencephalic neuronal primary cultures from rat fetus showed a selective vulnerability of dopaminergic neurons to rotenone, a mitochondrial complex I inhibitor commonly used to model PD neuronal impairment. This rotenone toxicity was mitigated by the simultaneous administration of nicotine via α7 and α4β2 nAChRs activating the PI3K-AKT/PKB pathways and upregulating of Bcl-2. 156
Excess extracellular glutamate is common in PD and is associated with glial activation, oxidative stress, neuroinflammation, and excitotoxicity.
157
Glutamatergic excitotoxicity in neurodegeneration is the outcome of high intracellular calcium and zinc levels as well as elevated nitric oxide (NO) synthesis in response to glutamate binding to N-methyl-D-aspartate (NMDA) receptors158,159 Activation of α7 nAChRs is effective at recruiting the Ca2+/calmodulin dependent protein kinase II (CaMKII) to phosphorylate NO synthase, and thereby, inhibit NO production160,161 Ca2+ and Zn2+ bind to allosteric locations of
The 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and 6-hydroxydopamine (6-OHDA) mouse models of PD have also confirmed the neuroprotective mechanisms of α7 nAChR agonists via mitigating the MPTP-induced Bax/Bcl-2 ratio changes, cleaved caspase-3/caspase-3 ratio, mitochondrial membrane potential, and PARP-1 changes that are characteristic of apoptosis154,153 The SNpc of chronic MPTP mouse models showed that nicotine inhibits the MPTP-induced elevation of the p-JNK/JNK ratio and increases the MPTP-induced reduced p-ERK/ERK ratio. 164 The rotenone-induced PD rat model (ROT-rat) experiences increased oxidative stress and apoptosis of dopaminergic neurons. 165 However, nicotine treatment in the ROT-rat rescues tyrosine hydroxylase and dopamine in the SNpc, reduces alpha-synuclein and glial fibrillary acidic protein levels in the SNpc, and improves motor behavior. 165 Furthermore, DMXBA (GTS-21) is an α7 nAChR agonist that protects against the loss of nigrostriatal dopaminergic neurons and microglial activation in the 6-OHDA rat model. 166 6-OHDA rat models also receive neuroprotection from nicotine combined with galantamine, a plant-derived alkaline allosteric modulator for α4β2 and α7 receptors 70 used in the treatment of cognitive symptoms. Recent work by Nozaki et al. 167 has demonstrated that galantamine reduces the accumulation of α-synuclein aggregates in vitro and in animal models of synucleinopathy. This effect appears to be mediated, at least in part, by modulation of cholinergic signaling pathways and anti-inflammatory activity, including suppression of microglial activation and oxidative stress. These findings suggest that galantamine may have therapeutic potential beyond symptomatic cognitive enhancement, possibly influencing neurodegenerative processes central to PD pathogenesis. Further investigation into the dual cholinergic and anti-α-synuclein effects of galantamine could open new avenues for its repositioning as a neuroprotective agent in PD.
Glial apoptosis is also suppressed by α7 nAChR activation. For example, the α7 nAChR agonist PNU-282987 mitigates apoptosis induced by MPP + and H2O2 in primary astrocyte cultures by upregulating Bcl-2 and downregulating Bax, cleaved-casoase-3, and JNK-p53-caspase-3 signaling. 168 Nicotine has also been shown to protect against oxidative stress-induced glial cell-derived neurotrophic factor (GDNF) reduction, inhibit cleaved caspase-9 activity, and to defend against mitochondrial membrane potential loss in primary mouse cultured astrocytes via α7 nAChRs. 25
mAChRs also exhibit protective effects against apoptosis. Caspase-3 activity, a marker of oxidative stress, increases after treatment with rotenone in PD models of neuronal impairment, a mitochondrial complex I inhibitor commonly used to model PD neuronal impairment. 169 However, activation of mAChRs protects cells from hydrogen peroxide–induced oxidative stress and prevents subsequent cell death by inhibiting caspase-3 activation. Caspase-3 is the predominant mechanism in which apoptotic signaling mechanisms propagate through to deplete ATP and NAD + energy storage. Administration of oxotremorine-M, a muscarinic agonist, leads to reduced caspase-3 activity and prevents the proteolytic mechanisms that directly induce ATP depletion associated with oxidative stress.
This protective effect mediated by mAChRs occurs through certain signaling pathways. Carbachol (Cch), a cholinergic receptor agonist, inhibits neuronal apoptosis induced by interferon-beta, a type I interferon found to exacerbate neuroinflammation. M3 receptor activation triggers the ERK 1/2 signaling pathway, which contributes to neuronal survival. 13 Thus, M3 receptors mediate this anti-apoptotic effect. Under the effect of Cch, cytochrome c is not released, caspases 3, 7, and 9 are deactivated, and DNA fragmentation is inhibited.
In addition to the ERK pathway, the Akt/Protein Kinase B (PKB) pathway also stimulates pro-survival mechanisms. These are activated through G-protein coupled receptor (GPCR) signaling coupled to muscarinic receptors. 170 Specifically, Gq and Gs – GPCRs, corresponding to M1 and M2 receptor subtypes, activate Akt kinase activity through signal transduction involving beta-gamma and alpha subunits. This leads to Akt stimulation through a PI3K-specific signaling mechanism.
Tripartite motif-containing protein 3 (TRIM3) is a tumor-suppressive protein heavily implicated in the molecular pathophysiology of PD, specifically through intracellular vesicle signaling, ubiquitin ligase activity, and transition metal ion binding. 171 Upregulation of TRIM3 expression in venous blood shows protective effects against apoptosis in PD via reduced reactive oxygen species concentration, increased expression of Bcl-2 protein, and reduced expression of Bax, caspase 3, and caspase 9. 172 These protective effects occur through the activation of the PI3 K/AKT signaling pathway, as TRIM3 upregulation leads to greater phosphorylation of PI3 K and Akt.
Modulation of neuroinflammation by nAChRs
Efforts to mitigate neuroinflammation in PD have targeted nAChRs. Inflammation plays a complex role in pathological events, as it orchestrates the removal of damaged cells, toxic agents, and pathogens. However, in neurological disorders like PD, this process becomes detrimental, exacerbating neuronal and glial dysfunction and diminishing their survival and functionality. 173 Released cytokines drive pro-inflammatory cascades and various cellular intermediates to recruit effector cells, such as activating microglia and astrocytes. 174 These effectors in turn release reactive oxygen species (ROS), reactive nitric oxide species (RNS), and proteinases, further enriching the inflammatory milieu. Furthermore, neuronal dopamine itself can be a source of oxidation-driven neuroinflammation. 175
Upon ionotropic activation, α7 nAChRs can influence synaptic ion channel activity, trafficking and cytoskeletal proteins, gene expression, and vesicle dynamics via intracellular Ca2+-sensitive kinases. 176 Glial signaling pathways such as the TLR4/NF-κB, JAK/STAT3, P13 K/Nrf2, and MAPK cascades among others, are modulated by α7 nAChRs activation to exert local anti-inflammatory effects177,178,179 The “cholinergic anti-inflammatory pathway” extends beyond the CNS via the vagus nerve to modulate inflammation in the periphery as well. 176 The gut-brain axis (GBA) can lead to central neuroinflammation stemming from dysbiosis and afferent vagus nerve signaling. Conversely, vagus efferent signaling provides cholinergic anti-inflammatory action by mitigating tumor necrosis factor alpha (TNF-α), toll-like receptors (TLRs), and macrophage-released cytokines in the gut. 180 A shared characteristic among all components of the GBA is the presence of nAChRs, which fulfill diverse functions throughout.
In microglia, activation of α7 nAChRs by either nicotine, ACh, Ca2+, or other agonists, inhibit the production of pro-inflammatory molecules such as IL-6 and NO. The α7 nAChRs mediated inhibition of these molecules can result from various signaling pathways, such phosphatidylinositol signaling, which increases the release of Ca2+ from the endoplasmic reticulum and inhibits the phosphorylation of MAP kinases involved in neuroinflammation, such as JNK, p38, and p44/42. 179 Additionally, α7 nAChR activation has been shown to reverse the pro-inflammatory phenotype of microglia induced by stimuli like lipopolysaccharide (LPS), while antagonists of α7 nAChRs exacerbate this phenotype. 181 Studies also suggest that α7 nAChR activation can inhibit the NLRP3 inflammasome, contributing to the suppression of neuroinflammation. 182 Furthermore, compounds like GTS-21, an α7 nAChR agonist, exhibit potent anti-inflammatory and neuroprotective effects in both in vitro and in vivo models of neuroinflammation and PD. 183 GTS-21 treatment reduces the expression of pro-inflammatory markers like iNOS, COX-2, and cytokines, and suppresses PI3 K/Akt and NF-κB. Concurrently, GTS-21 upregulates the anti-inflammatory factors TGF-β, Nrf2/HO-1, AMPK, Nrf2, CREB, and PPARγ in microglia, offering promising therapeutic potential for the management of neuroinflammatory conditions and PD. 183
In activated human astrocytes, nicotine agonism of α7 nAChRs reduces the release of pro-inflammatory cytokines, including IL-6, TNF-α, IL-8, IL-13, and IL-1β. 184 Similarly, GTS-21 diminishes LPS-induced secretion of inflammatory cytokines, with this effect being reversed by the α7 nAChR antagonist methyllycaconitine (MLA) or by α7 nAChR knockdown. 179 Furthermore, GTS-21 treatment of mouse astrocytes upregulates Nrf2. 185 Although Nrf2 has canonically been associated with antioxidant roles, it is also involved in anti-inflammatory signaling. 186 In the MPTP mouse model, nicotine reduces astrocyte activation, TNF-α production, and ERK1/3 activation. 187 Finally, stimulation of astroglial nAChRs leads to the inhibition of the NF-κB pathway. 180
In addition to their role in modulating striatal dopaminergic transmission, nicotinic acetylcholine receptors (nAChRs) have been implicated in the development and expression of L-DOPA-induced dyskinesias (LID) in Parkinson's disease (PD). Preclinical studies indicate that both α4β2 and α7 nAChR subtypes influence the plasticity of corticostriatal circuits and dopamine release dynamics that are dysregulated during chronic L-DOPA treatment. Nicotine and selective nAChR agonists have been shown to reduce the severity of LID in rodent and non-human primate models, likely through desensitization-mediated modulation of abnormal dopaminergic and glutamatergic signaling,.188,189 For instance, chronic nicotine administration decreases LID severity without compromising the antiparkinsonian efficacy of L-DOPA, an effect that may involve suppression of phasic dopamine release and normalization of downstream striatal signaling. 190 Moreover, α7 nAChR-selective agonists have demonstrated anti-dyskinetic effects while potentially engaging anti-inflammatory mechanisms. 191 These findings support the hypothesis that nAChR modulation represents a promising adjunctive strategy for mitigating LID while preserving dopaminergic therapeutic benefit.
While the anti-inflammatory role of nicotinic acetylcholine receptors, particularly α7 nAChRs, has been widely studied in the at of neuroimmune regulation in PD, the contributions of butyrylcholinesterase (BuChE) inhibition have received comparatively less attention. Some acetylcholinesterase inhibitors (AChE-Is), such as rivastigmine and galantamine, exhibit dual inhibitory activity against both AChE and BuChE, potentially leading to broader and more sustained enhancement of cholinergic tone in the CNS. This dual inhibition may amplify cholinergic anti-inflammatory pathways by maintaining acetylcholine availability in glial-rich regions where BuChE is more abundant, particularly in the aging and diseased brain. Moreover, BuChE inhibition has been associated with attenuation of microglial activation and reduced proinflammatory cytokine release, indicating a complementary mechanism to direct nAChR modulation. Interestingly, duloxetine, a serotonin-norepinephrine reuptake inhibitor widely used to treat depression in PD, has been reported to exert BuChE-inhibitory activity as well, 192 suggesting that its antidepressant and potential anti-inflammatory effects may partly converge on cholinergic modulation. These findings underscore the importance of considering both enzymatic and receptor-mediated pathways in the cholinergic regulation of neuroinflammation and highlight the therapeutic potential of BuChE inhibitors as adjunctive treatments in PD.
Reduction of senescence-associated secretory phenotype
The complex and heterogeneous senescence-associated secretory phenotype (SASP) of senescent cells enhances the expression and secretion of many pro-inflammatory factors. SASP components can be released due to the activation of signaling pathways, such as the mammalian target of rapamycin (mTOR), AMP-activated protein kinase (AMPK), GMP-AMP synthase-stimulator of interferon genes (cGAS-STING), p53, p38α, and DNA damage response pathways193,194,195 Additionally, transcription factors such as NF-κB, CCAT/Enhancer Binding Protein Beta (C/EBPβ), and GATA4 are involved in regulating SASP. 195 The Neuro-Immuno-Senescence Integrative Model (NISIM) suggests that the autonomic nervous system interacts with the immune system to activate the transcription factor NFκB, leading to the secretion of the cytokines IL-6 and TNF-α, subsequent ROS-induced telomere shortening, and ultimately senescence196,197 While SASP factors typically promote temporary, local inflammation essential in wound healing, tissue remodeling, and development, prolonged expression of SASP factors leads to chronic, pathologic inflammation. This chronic inflammation, along with the accumulation of senescent cells with age and pathology, is considered a driving factor of neurodegeneration. 198
The rate at which senescent cholinergic neurons and glia accumulate, and the severity of their collective senescence, increases with age. Therefore, senescent cells in the brain contribute to the chronic low-grade inflammation and oxidative stress that initiates and propagates neurodegeneration. 199 For example, the Ts65Dn mouse model experiences early stress-related senescence and subsequent degeneration of cholinergic neurons. 200 The Ts65Dn mouse model, often used to study Down Syndrome, also exhibits cognitive and motor deficits and age-related decline in cholinergic neurons, making it relevant for PD studies. This model reveals that the phosphorylation of the transcription factor FOXO1 promotes the early induction of cholinergic senescence. 200 Interestingly, LRRK2 activates Akt, which then phosphorylates FOXO1. 173 Conversely, inhibition of LRRK2 rescues early stress-related neuronal senescence. 201 Mutations in the LRRK2 gene cause a late-onset form of autosomal dominant PD, with the most common PD-associated mutation, G2019S, upregulating the p53-p21 signaling pathway and promoting senescence. 201 Therefore, PD pathology involves the convergence of cellular senescence and the central cholinergic system.
Nicotinamide adenine dinucleotide (NAD) deteriorates with age, leading to a decline in NAD + levels and associated age-related defects. The enzyme nicotinamide phosphoribosyl transferase (NAMPT), central to the NAD + salvage pathway, also diminishes in activity with age. Nicotine, a secondary metabolite in the NAD + biosynthesis pathway, has been shown to improve NAMPT activity and NAD + synthesis in aged mice. 202 In aging neurons, small doses of nicotine improved energy metabolism by reversing the dysfunction of glycolytic and mitochondrial respiration, reduced inflammation by decreasing the activation of the JAK2/STAT3 pathway and lowered oxidative stress markers. Furthermore, nicotine treatment increased telomere length and the expression of telomere-stabilizing proteins, contributing to cellular longevity. Ultimately, nicotine-treated mice exhibited lower age-related mortality, indicating its potential in mitigating senescence. Interestingly, however, this study showed that the effects of nicotine were independent of nAChRs. 202
Angiotensin II (Ang II)-induced senescence in mice vascular smooth muscle cells (VSMCs) was mitigated by the activation of α7 nAChR with the selective agonist PNU-282987. 203 Additionally, PNU-282987 reduced Ang II-induced ROS levels, lipid peroxidation, and the expression of NADPH oxidase 1, NADPH oxidase 4, and p22phox in VSMCs. It also diminished the pro-senescence signaling pathways involving p53, acetyl-p53, p21, and p16INK4a. Furthermore, PNU-282987 increased intracellular NAD + levels and enhanced SIRT1 activity without affecting SIRT1 protein expression. This effect was AMP-dependent protein kinase (AMPK)-independent. Knockdown of SIRT1 or inhibition with EX527 muted the anti-senescence effects of α7 nAChR activation, highlighting SIRT1's critical role in mediating these effects. Finally, activation of α7 nAChR reduced markers of cellular senescence induced by Ang II, such as β-galactosidase activity, and phosphorylation of H2A.X and Chk1, indicating a decrease in DNA damage and improved cellular replication. 203
Endothelial progenitor cells (EPCs) are crucial for maintaining endothelial health, participating in processes like reendothelialization and neovascularization. Nicotine delays EPC senescence in a dose-dependent manner and increases telomerase activity in EPCs. 204 The PI3 K/Akt pathway plays a significant role in nicotine-induced telomerase activation, as confirmed by the inhibitors of PI3 K, wortmannin and LY294002. Furthermore, nicotine induces a dose-dependent phosphorylation of Akt. All these effects were reversed by the nAChR antagonist mecamylamine. 204
The effect of nAChR agonists on senescence is complex and context dependent. For example, in the context of SARS-CoV-2 infection, nicotine has been shown to exert both beneficial and detrimental effects. 205 Nicotine's activation of the α7 nAChR can delay cellular senescence, promote cell viability, and upregulate pathways associated with cell growth, survival, and proliferation. However, this same mechanism can have adverse consequences during a viral infection. Nicotine-induced activation of α7 nAChR leads to the upregulation of ACE2, the primary receptor facilitating SARS-CoV-2 entry into host cells, potentially increasing susceptibility to the virus. Additionally, while delaying senescence may keep cells in a more viable state, it also means that there are more cells available for viral infection and replication. Therefore, while nAChR agonists can reduce the inflammatory response, alleviate senescence burden, and promote cell survival, they may simultaneously enhance the risk and severity of SARS-CoV-2 infection by increasing ACE2 expression and maintaining a pool of susceptible cells. 205
Furthermore, nicotine exposure induces a phenotypic switch in VSMCs, causing them to migrate and proliferate into the arterial intima, contributing to atherosclerotic plaque formation. Nicotine then induces VSMCs to become senescent and secrete SASP factors, which destabilizes the plaque and increases the risk of cardiovascular events. Nicotine's role in VSMC senescence involves activation of p38MAPK and ERK signaling pathways, upregulation of the ROS-producing enzyme Nox1, and inflammation. 206 Interestingly, this study discounted any involvement of nAChRs in generating the pro-senescence effects of nicotine. Senescence is also common in the islet β cell lesions of type 2 diabetes mellitus (T2DM). Senescent β cells lose their proliferative capacity and increase the expression of markers such as p16, p21, and p19. The effect of nicotine on nAChRs in β cells exacerbates senescence-associated β-galactosidase (SA-β-Gal) phenotype and SASP factor release. 207 Nicotine-induced β cell senescence is mediated by Ca2 + signaling and ROS production. 207
While nAChR activation can mitigate senescence and its inflammatory phenotype in various cell types, its specific effects on cholinergic neurons in PD remain underexplored. In PD, where cholinergic neuron degeneration is a hallmark, reducing senescence and SASP through nAChR modulation could help to alleviate neurodegenerative processes. However, the dual effects of nicotine and other nAChR agonists, such as increased infection susceptibility and cardiovascular risks, underscore the need for a nuanced understanding of these pathways. Modulating nAChRs presents a potential strategy for reducing senescence in cholinergic brain cells, but more targeted research is needed to fully elucidate these mechanisms and translate them into clinical therapies (Figure 4). Future studies should focus on the specific impacts of nAChR activation on cholinergic neurons in PD models to confirm these therapeutic potentials and address associated risks.
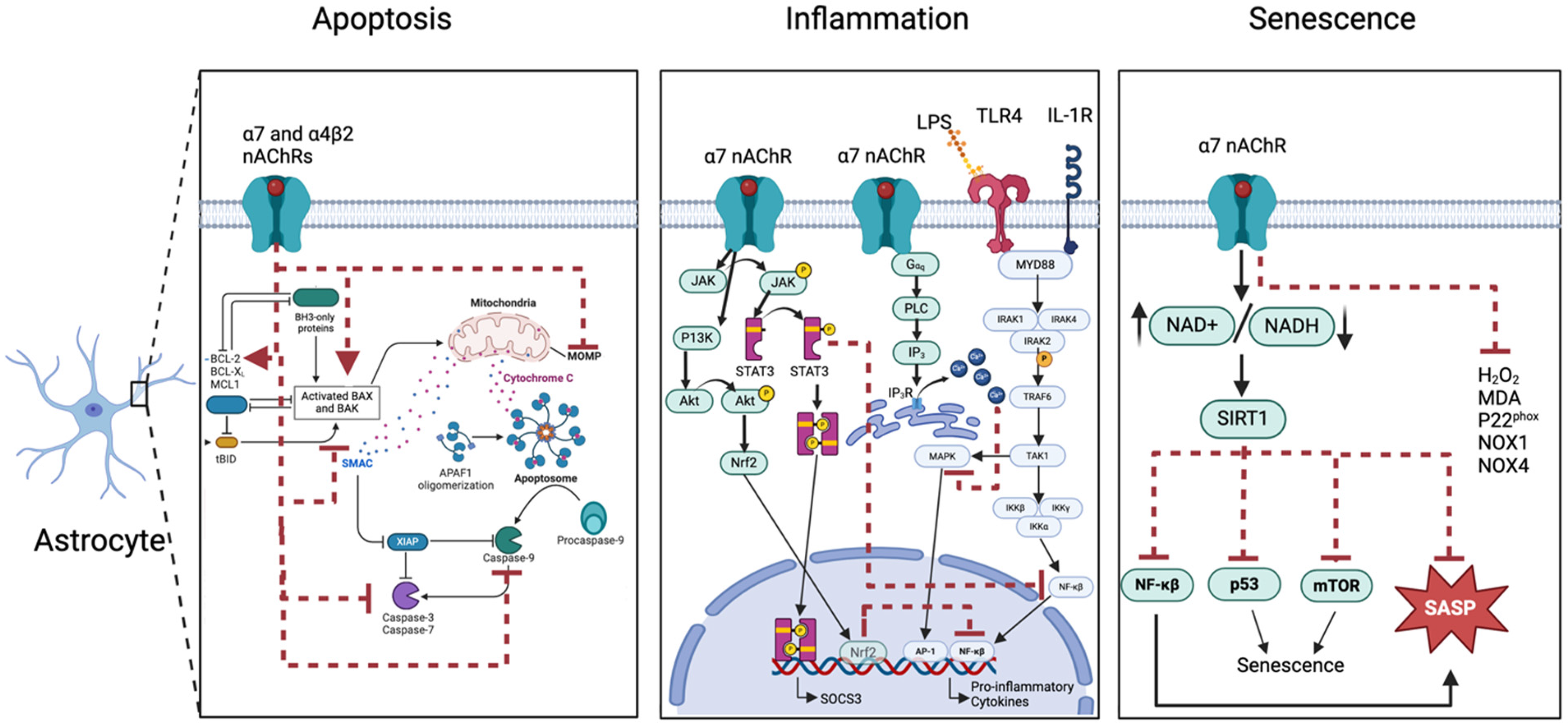
Therapeutic mechanisms of α7 nAChR activation. 1) Apoptosis involves extrinsic pathways via death receptors and intrinsic pathways via mitochondrial signals. In Parkinson's Disease (PD), dysregulation leads to increased active caspase-3 and pro-apoptotic BAX, with reduced anti-apoptotic BCL-2. Activation of α7 nicotinic acetylcholine receptors (nAChRs) protects neurons and glia by preserving mitochondrial function, modulating apoptosis markers, and reducing oxidative stress. 2) Inflammation is critical for the removal of pathogens and damaged cells, but it can also worsen neuronal and glial dysfunction in PD. Key inflammatory mediators trigger pro-inflammatory cascades, releasing reactive oxygen and nitric oxide species, as well as proteinases. Furthermore, dopamine can contribute to oxidative-driven neuroinflammation. However, activation of α7 nAChRs modulates these pathways, providing anti-inflammatory effects by inhibiting pro-inflammatory molecules and reversing pro-inflammatory phenotypes. 3) Senescence-Associated Secretory Phenotype (SASP) in aging cells increases the concentration and variety of pro-inflammatory factors, driving chronic inflammation and neurodegeneration. In PD, senescent cholinergic neurons and glial cells worsen inflammation and oxidative stress. Interestingly, LRRK2 mutations in PD activate neuronal senescence pathways. However, nicotine mitigates cellular senescence and oxidative stress, promoting cellular longevity.
Neuroprotective potential of muscarinic receptors in PD
M1 mAChRs are present in the central nervous system, including the striatum, and play pivotal roles in motor control, sleep-wake rhythms, and cognitive processes such as attention and memory. They are located on both D1 and D2 MSNs and activate the Gq/11 pathway, leading to increased production of DAG and IP3.. 208 M4 mAChRs are located preferentially on D1 MSNs. The localization of M2 and M3 receptors is less clear. M1 receptors exhibit excitatory effects on striatal MSNs. 209 Activation through the M1 agonist carbachol enhances the MSN excitability through a concentration-dependent manner. However, M1 mAChRs appear to selectively modulate cholinergic transmission in the striatum, but not other regions of the basal ganglia such as the subthalamic nucleus and substantia nigra pars reticulata. This suggests that other muscarinic receptor subtypes may mediate cholinergic innervation. In addition, selective M1 antagonists partially reversed akinesia and cataplexy induced by PD pathways but did not exhibit the full efficacy of motor symptoms reversal seen by nonselective mAChR antagonists. These findings support the involvement of M1 mAChRs in basal ganglia function implicated in PD, but there are likely other mAChR subtypes that contribute to the neural motor circuits within the basal ganglia.
M1 receptors are also implicated in the cognitive impairment associated with PD. PET imaging and gene expression analyses identified a spatial overlap between a cholinergic M1/M4 expression network and regions part of the default mode network and frontoparietal executive control network. 210 Amongst PD patients without dementia, there was a larger decline in M1 receptor expression in regions of overlap, as well as a strong correlation between M1 receptor loss and cognitive decline. Thus, cholinergic expression mediated by M1 receptor activity may shape the changes in executive control, memory, and attention commonly seen amongst PD patients.
M4 receptors are the most highly expressed subtype of the mAChRs. 211 They are preferentially expressed in MSNs of the direct pathway near glutaminergic synapses, as well as glutamatergic cortical and thalamic pre-synaptic terminals that provide neural inputs to the striatum. 212 Functionally, M4 mAChRs are coupled to Gi/o proteins, which when activated, lead to inhibition of adenylyl cyclase activity and reduced cAMP production. They are located on the presynaptic terminals of ChIs where they function as autoreceptors, exerting negative feedback control on ACh release.
M4 receptors exert an inhibitory modulatory effect over D1 dopamine receptor signaling in the direct pathway of the striatum. 213 Signaling through M4 receptor transduction regulates the excitatory influence of dopamine, regulating motor control and locomotor activity. Mice with M4 mAChR knockouts show heightened locomotor activity through disinhibition of D1 receptor transduction and enhanced activity of the direct pathway. Furthermore, administration of the selective M4 receptor antagonist tropicamide results in alleviation of PD motor deficits. 214 This is also observed after administrating selective M1 receptor antagonists. In mutant mice with lesions that abolish M4 mAChRs in neurons expressing D1 receptors, the alleviating effects of tropicamide is eliminated. Thus, M4 mAChRs mediate the motor-enhancing effects of muscarinic receptor antagonists.
On the contrary, M4 mAChRs represent a promising target for therapeutic intervention for LID. L-DOPA initially alleviates motor symptoms but eventually leads to the onset of involuntary movements categorized under LID. 215 Cholinergic transmission via M4 mAChRs in the striatum is reduced following dopamine depletion. 216 However, selectively increasing the expression of M4 mAChRs brings cholinergic transmission levels back to normal after disruptions by dopamine depletion. In the context of LID, enhancing M4-mediated cholinergic transmission, by ablation of a G-protein regulator that acts specifically through Gi/o proteins, reduces dyskinetic motor behavior while preserving the antiparkinsonian effects observed in earlier phases following L-DOPA treatment.
M1 and M4 receptors are both present on ChIs in the striatum. ACh from ChIs acts on both M1 and M4 receptors to modulate motor activity. ChIs can increase excitability of D2 MSNs through M1 receptor activation and inhibit firing of D1 MSNs via M4 receptors to terminate movement and reduce motor control. 217 This differential expression of M1 and M4 receptors on MSNs allows ChIs to fine tune motor skills and balance motor initiation and termination.
Degree of protective effect from cholinergic interneurons in PD pathophysiology
ChIs in the striatum represent a critical population that influences motor function through acetylcholine (ACh) neurotransmission. ChIs are primarily responsible for the release of ACh from the striatum, which is important for motor control and learning. 218 Alteration of ChI function shapes the motor hallmarks typically indicative of PD. Dopamine exerts an inhibitory effect on ChI activity in normal physiological conditions via D2 receptor activation. 101 As a result, dopamine degeneration associated with PD leads to disinhibition of ChI activity and enhanced acetylcholine release. The motor characteristics that arise amongst PD patients are directly influenced by the hyperactivity of cholinergic systems in the basal ganglia and striatum. Heightened ChI activity subsequently inhibits further dopamine release through muscarinic receptor activation, leading to reduced dopamine levels seen in PD.
Contributions to the imbalance between direct and indirect pathways
The striatum integrates neural inputs from thalamus and cortex to target medium spiny neurons (MSNs). MSNs are subdivided into two categories based on their receptor subtypes and neural targets. MSNs of the direct pathway harbor D1 dopamine receptors and project to the basal ganglia, while indirect pathway MSNs carry D2 receptors and project to the globus pallidus and subthalamic nucleus,.219,220 Dysregulation of the direct and indirect pathways of the basal ganglia represents a prevailing pathophysiological model to explain the hallmarks of PD. Activation of the direct pathway, which involves the disinhibition of the thalamus and motor cortex, fosters motor activity and reduces freezing. 221 In contrast, movement is inhibited with activation of the indirect pathway. This leads to motor deficits commonly associated with PD, including bradykinesia, freezing, and reduced locomotion.
ChIs contributes to this imbalance between indirect and direct pathways by enhancing the excitation of MSNs of the indirect pathway. ChIs strengthen the input by thalamostriatal regions on the parafascicular nucleus (PFn), which enhances the EPSP amplitude from indirect pathway MSNs. 222 The PFn provides glutamatergic innervation to the striatum, which is important for the balanced excitability of direct and indirect spiny projection neurons. MSNs of the indirect pathway show greater EPSP strength due to thalamic excitation in dopamine-depleted mice (PD model). This was not seen in the MSNs of the direct pathway. When ChIs were silenced through optogenetic innervation, EPSPs in indirect pathway MSNs were reduced. This points to the key role ChIs play in the exacerbation of motor function seen in Parkinson's, specifically through the motor suppressive effects of the indirect pathway. Conversely, ChIs interfere with the glutamatergic transmission between the cortex and direct pathway MSNs expressing D1 receptors. 223 This relationship is evidenced by an increase in EPSP amplitude in D1 MSNs following ontogenetically suppression of ChI activity. ChIs are also implicated in the reduced robustness of long-term potentiation (LTP) in corticostriatal synapses in PD. This can limit the effectiveness of interventions such as task-based learning and physical therapy that require continual neurological plasticity.
Cholinergic innervation on motor function
Dopamine depletion seen in PD also affects the modulatory role of ChIs in basal ganglia circuits. The firing of ChIs ChI firing becomes more irregular and less frequent under dopamine-deficient conditions. 224 Hyperpolarization-activated cation (HCN) channels control the timing of spike pulses generated by ChIs, and dopamine deficiency in PD reduces HCN current and transcription of channel-specific genes. Dopamine released from residual axons in PD activate D1 receptors on ChIs. As D1 signaling can suppress ChI function, the relative concentrations of dopamine and acetylcholine become unbalanced. This leads to motor deficits characteristic to PD. Decreased current through HCN channels alters a consistent rhythm of acetylcholine release from the striatum, which ultimately impairs the plasticity of corticostriatal circuits and medium spiny neuron activity. These results point to channel-specific signaling mechanisms involving ChIs as potential therapeutic targets for PD.
Cholinergic neurons are also implicated in episodes of gait freezing and postural instability commonly seen in late stages and severe forms of PD. Loss of cholinergic neurons in the pedunculopontine nucleus (PPN) show correlations with the presence of gait disorders. 225 The PPN and cuneiform nucleus collectively comprise the mesencephalic locomotor region (MLR), and activity of specific clusters in the MLR is greater when imagined speed of gait increases. In comparison with PD patients with normal balance and gait, loss of PPN cholinergic neurons is more severe in PD patients exhibiting neurodegenerative motor deficits. Similarly, activation of the cholinergic neurons in the pedunculopontine tegmental nucleus, the rat equivalent of the PPN in humans, ameliorated motor symptoms assessed by postural instability, gait, forelimb movement, and overall motor performance. 39 This provides more evidence to the therapeutic effect PPN cholinergic neurons may bring to reduce PD motor deficits.
Furthermore, degeneration of cholinergic neurons in the basal forebrain and PPN exacerbates as PD progresses. 129 This reduction in cholinergic neuronal abundance contributes to the decline in cognitive processing, as well as postural instability and gait dysregulation. Conversely, the striatum exists in a hypercholinergic state in PD due to the loss of dopamine neurons, which leads to dyskinesia and onset of tremors. There is a greater loss of cholinergic neurons in the nucleus basalis of Meynert and medial septal-vertical limb of the diagonal band of Broca (regions of the basal forebrain) amongst patients with diagnosis of Parkinson's with dementia compared to PD patients without dementia. 226 Cholinesterase inhibitors show modest improvement in cognitive function amongst PD patients. In a randomized control experiment with 541 PD patients, cognition, attention, and executive function significantly improved after administration of rivastigmine, an inhibitor to both acetylcholinesterase and butyrylcholinesterase. 227 However, adverse side effects, such as nausea, may arise, but these may be alleviated through dose adjustments. 228 Furthermore, the administration of cholinesterase inhibitors rivastigmine and donepezil leads to a decrease in overall fall rate compared to placebo group. 229 However, the proportion of individuals who experience at least one fall does not differ between patients administered with cholinesterase inhibitors and placebo patients. However, cholinesterase inhibitors increased adverse events unassociated with falls by 60% compared to patients receiving the placebo. These events were mild and temporary.
Deep brain stimulation (DBS) on the substantia nigra and globus pallidus has been successful in targeting tremors, slowed movements, and rigidity amongst PD patients. 230 However, evidence for the efficacy of DBS on reversing gait disturbances and postural irregularities is less consistent. Some patients in trials show improvements with postural stability, freezing, and falls, 231 but oftentimes trials are limited by small participant sizes, lack of uniform assessment metrics (stimulation parameters, etc.), or do not show motor improvements. 232 For example, low frequency DBS stimulation on the PPN gave rise to moderate restoration of gait, posture, and balance in a small cohort of patients. 233 Since the PPN is a cholinergic nucleus involved in gait and motor control, the cholinergic system is a valuable neurological target to further explore the therapeutic potential of DBS in reducing motor symptoms outside of tremors, bradykinesia, and motor rigidity. Thus, further experimentation can clarify how the cholinergic system may serve as a therapeutic target for addressing gait disturbances, postural instability, and falls associated with PD.
The cholinergic system presents numerous neurological targets that can broaden the range of potential approaches for the symptomatic treatment of PD. However, results are currently mixed and require further understanding of the pathophysiology connected to the cholinergic system.
Conclusion
Although PD is primarily a dopaminergic disorder, PD has emerged as a complex disease with substantial involvement of the brain's cholinergic circuitry. This review has explored the neuroanatomical and functional changes associated with the cholinergic system in PD, highlighting their role in both motor and non-motor symptoms (Table 1). Both cortical and subcortical cholinergic systems, originating from the basal forebrain and brainstem PPN, respectively, undergo degeneration. Notably, the severity of cholinergic neuronal senescence increases with age, exacerbating chronic inflammation and oxidative stress, further propagating neurodegeneration. Cholinergic degeneration presents a diverse array of non-motor symptoms, encompassing cognitive impairment, mood disorders, visual disturbances, sleep disruptions, and olfactory dysfunction. Moreover, the cholinergic system's implication in motor symptoms like gait disturbances and freezing of gait highlights its multifaceted involvement in PD pathophysiology.
Clinical manifestations of the cholinergic system in PD.

The importance of expanding PD treatment beyond dopaminergic pathways has been recognized for decades. Perhaps the most promising non-dopaminergic therapeutic target is the cholinergic system. Preclinical and epidemiological studies have robustly demonstrated the neuroprotective and anti-inflammatory effects of cholinergic receptor agonists, offering potential avenues for disease-modifying therapies. For example, α7 nAChRs have garnered substantial attention for their involvement in protecting the cell against apoptosis, modulating neuroinflammation, and reducing chronic senescence-associated damage. There have been two clinical trials aimed at targeting nAChR in PD. The phase II NIC-PD study (ClinicalTrials.gov Identifier: NCT01560754) investigated the potential of a transdermal nicotine treatment of 7 or 14 mg for 52 weeks. Unfortunately, this intervention failed to slow the progression of early-stage PD. 235 A pilot study (ClinicalTrials.gov Identifier: NCT03865121) of pulsatile nicotine administration, up to 10 mg/day through a nasal spray (Nicotrol NS®), showed no significant change176,236 This study only included six patients and lasted only a month. However, it pursued an idea that is worth further exploration, since pulsatile administration of nicotine could avoid possible nAChR desensitization to continuous nicotine exposure, as is provided through a patch. Also, it mimics the rhythm of nicotine dosing by smokers.
In conclusion, the cholinergic system represents a critical facet of PD pathophysiology, and it should be receiving more attention. Its involvement in both motor and non-motor symptoms offers potentially novel insights into PD pathology and enhanced therapeutic strategies. Future work on leveraging the cholinergic system in understanding PD pathology and developing improved therapeutic interventions will rely on advanced imaging techniques, preclinical models, and bioinformatic approaches. Moreover, exploring the synergistic effects of cholinergic-targeted therapies with existing treatments holds promise for enhancing clinical outcomes in PD patients. The therapeutic potential of cholinergic receptors warrants continued efforts towards fully harnessing them in the fight for improved quality of life for individuals living with PD.
Footnotes
Acknowledgements
We would like to thank our colleagues for fruitful discussions.
Funding
B.P.H. receives funding from NEI R01-EY034234, the H. Eric Cushing Foundation, the Nancy Lurie Marks Family Foundation, the C.J.L. Charitable Foundation, the Reynold and Michiko Spector Award in Neuroscience, and Sanofi Pharmaceuticals.
Declaration of conflicting interests
B.P.H. is a consultant for BioAge Labs, Biohaven Pharmaceuticals, and RxSight.
