Abstract
Objective
Parkinson's disease (PD) frequently involves dysphagia, yet real-world longitudinal data capturing its progression and response to clinical care remain limited. Most prior studies exclude treated patients or rely solely on subjective or cross-sectional data. This study investigates long-term changes in swallowing physiology and diet in a pragmatic, treated PD population.
Methods
We retrospectively reviewed patients with PD who underwent at least two videofluoroscopic swallow studies (VFSS) ≥ 1 year apart at a tertiary laryngology center. Individuals with comorbid neurologic or structural disease were excluded. Standardized metrics included the Dynamic Imaging Grade of Swallowing Toxicity (DIGEST), Penetration Aspiration Scale (PAS), Functional Oral Intake Scale (FOIS), and International Dysphagia Diet Standardization Initiative (IDDSI), with kinematic analysis of Pharyngeal Constriction Ratio (PCR) and Maximum Pharyngoesophageal Segment Opening (PESmax). Ordinal outcomes were compared using Wilcoxon signed-rank tests; kinematic changes were analyzed with paired
Results
Nineteen patients (mean age 79.4 years, 73% male) completed 38 VFSS over a mean 2.6-year interval. Despite an average of 9.7 swallow therapy sessions per patient, significant declines were observed in PESmax (
Conclusion
Swallowing function deteriorates over time in PD, even with treatment. This study highlights the value of instrumental reassessment and supports proactive, longitudinal management in real-world care settings.
Introduction
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterized by the loss of dopaminergic neurons in the substantia nigra, resulting in hallmark motor symptoms such as bradykinesia, resting tremor, rigidity, and postural instability. In addition to these motor deficits, PD is associated with a wide range of non-motor symptoms, including cognitive decline, autonomic dysfunction, mood disturbances, and impairments in speech (dysphonia, dysarthria) and swallowing (dysphagia).1,2 Although speech and swallowing impairments arise from motor dysfunction of the oropharyngeal and respiratory musculature, they are often discussed separately from the cardinal body motor symptoms that define Parkinson's disease.
Dysphagia is a highly prevalent and clinically significant symptom of PD, affecting over 80% of individuals during the course of the disease.3,4 Its mechanism is multifactorial, involving both central and peripheral neurodegenerative processes. Degeneration of dopaminergic neurons in the basal ganglia impairs the coordination and timing of voluntary and involuntary swallowing movements, affecting oral, pharyngeal, and esophageal phases. 5 Dysfunction in the dorsal motor nucleus of the vagus disrupts motor control of swallowing muscles and esophageal motility. Peripherally, alpha-synucleiin and Lewy body aggregates have been found in the enteric nervous system and peripheral motor nerves innervating pharyngeal muscles, leading to incomplete upper esophageal sphincter (UES) relaxation, reduced UES opening, elevated intrabolus pressure and decreased contractility of the striated muscle of the esophagus.6,7 Abnormal pharyngeal kinematics such as delayed laryngeal vestibule closure and reduced hyoid bone displacement further compromise the airway. 8 Reduced activation and compensatory recruitment of motor areas may initially mitigate symptoms, but become insufficient as neurodegeneration advances. 9 Importantly, swallowing impairments often begin early, sometimes even prior to diagnosis, and may progress silently, without overt clinical symptoms.10–13 Common impairments in the oral and pharyngeal phases include delayed swallow initiation, incomplete bolus clearance, and diminished opening of the upper esophageal sphincter.10,11 These deficits increase the risk of aspiration, malnutrition, dehydration, and pneumonia, all contributing to reduced quality of life and increased healthcare burden.3,4 Nonetheless, patients often underreport symptoms, and routine instrumental evaluation is not standard in many clinical settings. 4
Although several studies have described dysphagia in PD, few have tracked its progression over time using objective, instrumental measures. Many existing studies are cross-sectional or rely on patient-reported outcomes, which may not reliably reflect physiological changes.4,10 Moreover, most longitudinal research to date has focused on untreated cohorts, limiting its generalizability to real-world clinical populations who may be receiving dopaminergic therapy or swallowing rehabilitation. 10 These gaps hinder our understanding of how dysphagia evolves in PD under standard clinical care.
Instrumental assessments such as videofluoroscopic swallow studies (VFSS) provide a means of capturing both functional and physiological aspects of swallowing. Validated metrics, including the Dynamic Imaging Grade of Swallowing Toxicity (DIGEST), Penetration-Aspiration Scale (PAS), Functional Oral Intake Scale (FOIS), and International Dysphagia Diet Standardization Initiative (IDDSI) framework, enable standardized assessment of swallow safety, efficiency, and diet level.14–18 In addition, kinematic measures such as the Pharyngeal Constriction Ratio (PCR) and Maximum Pharyngoesophageal Segment Opening (PESmax) provide insight into biomechanical changes in swallow function that may precede clinical deterioration.19–21
Given the growing prevalence of PD and the underrecognized burden of dysphagia, there is a critical need for longitudinal data reflecting real-world patient outcomes.22,23 This study aims to characterize changes in swallowing function over time in a tertiary-care cohort of individuals with PD, including those who received prior dopaminergic or swallow therapy. Analyses were exploratory to inform the role of proactive, longitudinal dysphagia management across the disease course.
Methods
This retrospective cohort study was conducted at a tertiary laryngology clinic with Institutional Review Board approval (IRB #19-29435). Patients were eligible if they had a confirmed diagnosis of PD and had undergone at least two VFSS, spaced a minimum of one year apart. These studies were designated as Time 1 (T1) and Time 2 (T2).
Patients were excluded if they had a history of respiratory disease (e.g., asthma, COPD, emphysema), cerebrovascular accident, head and neck cancer (with or without radiation), other neurodegenerative disorders (e.g., multiple system atrophy, Lewy body dementia, progressive supranuclear palsy), or parkinsonism without confirmed PD diagnosis. Additional exclusion criteria included prior swallow-related surgeries (e.g., posterior pharyngeal wall augmentation, esophageal dilation, cricopharyngeal myotomy, vocal fold augmentation, Zenker's diverticulotomy), or participation in active swallow therapy at the time of both their first and most recent VFSS. Prior deep brain stimulator (DBS) surgery was not an exclusionary criterion.
Information on swallowing therapy was gathered by review of Speech-Language Pathologist (SLP) treatment notes and descriptions. For each patient, information was gathered on type of therapy (e.g., swallow therapy only or concurrent voice and swallow therapy), targets of swallowing therapy (i.e., education, diet modification, compensatory strategies, swallowing maneuvers, oropharyngeal strengthening exercises, skill-based strategies, and/or respiratory muscle strength training), and adherence per SLP report.
Definitions
PESmax measures the maximum diameter of the pharyngoesophageal segment during swallowing, reflecting cricopharyngeal relaxation and bolus passage. 21 PCR quantifies pharyngeal contractility during swallowing, with lower PCR values indicating stronger pharyngeal contraction. 20 FOIS is a 7-point ordinal scale measuring the level of a patient's oral intake, ranging from 1 (nothing by mouth) to 7 (full oral diet with no restrictions). 16 Overall, FOIS assesses dietary independence. DIGEST is a composite scoring system based on VFSS where safety, efficiency and overall scores assess the risk of airway aspiration, the presence of residue or bolus clearance, and the combined safety and efficiency score, respectively. 14 PAS is an 8-point scale rating the depth and clearance of material entering the airway during swallow, ranging from 1 being no airway entry to 8 being silent aspiration. 15 PAS assesses airway protection during swallowing. IDDSI is a classification system from 0 (thin liquids) to 7 (regular food) for standardized diet modification in dysphagia management. 17 IDDSI reflects diet texture and thickness. Finally, the Eating Assessment Tool-10 (EAT-10) is a 10-item patient-reported outcome measure evaluating the self-perceived impact of dysphagia on quality of life and daily function. 24
Swallowing measures and VFSS protocol
Each patient's initial (T1) and most recent (T2) VFSS were assessed for the following outcome measures: swallow kinematics (PESmax 21 and PCR 20 ), FOIS, 16 DIGEST (Overall, Safety, Efficiency), 14 PAS, 15 IDDSI (solid and liquid), 17 and EAT-10. 24 Two swallowing and voice-specialized SLPs were recruited to independently evaluate each measure for reliability.
FOIS and IDDSI scores were determined by the evaluating SLP based on patient and caregiver reports of typical dietary intake, clinical evaluation, and instrumental assessment of VFSS results. VFSS was performed using a standardized Modified Barium Swallow Study (MBSS) protocol aligned with DIGEST and Dynamic Swallow Study (DSS) parameters. 25 Protocol included thin liquid, mildly thick liquid, pudding, solid (cracker coated with pudding), and a barium tablet. Imaging was performed in lateral and anterior-posterior views at 25–30 frames per second. Deviations from protocol and strategy trials were guided by observed impairments and clinical judgment. Patients took their PD medications in usual fashion on the day of the swallowing evaluation; no attempt was made to coordinate timing of this assessment to be in an intentionally off-medication state.
The PAS score recorded was the maximum score observed during the VFSS, excluding trials during which compensatory strategies were employed. DIGEST and IDDSI ratings were assigned per published criteria.14,17 For kinematic measures, PESmax and PCR were calculated manually using images from WebPACS software and integrated measuring tools, based on 1cc, 3cc, and 20cc mildly thick liquid boluses. They were interpreted relative to normative values and categorized as: within normal limits (<1 SD), mild-to-moderate deviation (1–1.5 SD, 1.5-2 SD), or significantly abnormal (>2 SD). PESmax was defined as the maximum distention of the pharyngoesophageal segment observed during swallowing. PCR was defined as the ratio of the pharyngeal area at its point of maximum constriction during a swallow to the pharyngeal area measured during the pre-swallow hold posture, as viewed laterally. 25 An EAT-10 score ≥3 was considered abnormal per validation standards. 24
Clinical and demographic variables
Demographic data collected from the electronic medical record included age, gender, years since PD diagnosis, Hoehn & Yahr (H&Y) stage, and PD-related treatments. The duration since PD diagnosis was determined based on the diagnosis date documented in the patient's initial neurology visit. H&Y stage was determined based on the neurologist's clinical note closest in time to the VFSS. Mean levodopa equivalent daily dose (LEDD) 26 was calculated from the patient's reported medication history on the day of the VFSS. History of swallow therapy was recorded, though adherence to prescribed home exercise programs was not verifiable. At this institution, initial swallowing therapy typically consists of four sessions over two months, with adjustments in frequency and duration based on individual progress. Follow-up intervals of 3–6 months are standard.
Statistical analysis
Interclass correlation coefficients (ICC) were calculated to determine interrater reliability for clinician reported outcomes based on 20% of the dataset.
27
ICC was interpreted as being poor (<0.50), moderate (0.50–0.75), good (0.75–0.90), and excellent (>0.90). Paired
Results
Nineteen patients (mean age 79.4 years, 73% male) were included in the analysis. In total, 38 VFSS studies were evaluated, with an average interval of 2.6 years between T1 and T2 (Table 1). On average, patients underwent their first VFSS 7.1 years after PD diagnosis. Overall, interrater reliability was excellent (PCR = 0.95; PESmax = 0.91; PAS = 0.98; DIGESTOverall = 0.90; DIGESTSafety = 0.94; DIGESTEfficency = 0.95).
Demographics of patients with Parkinson's dysphagia.
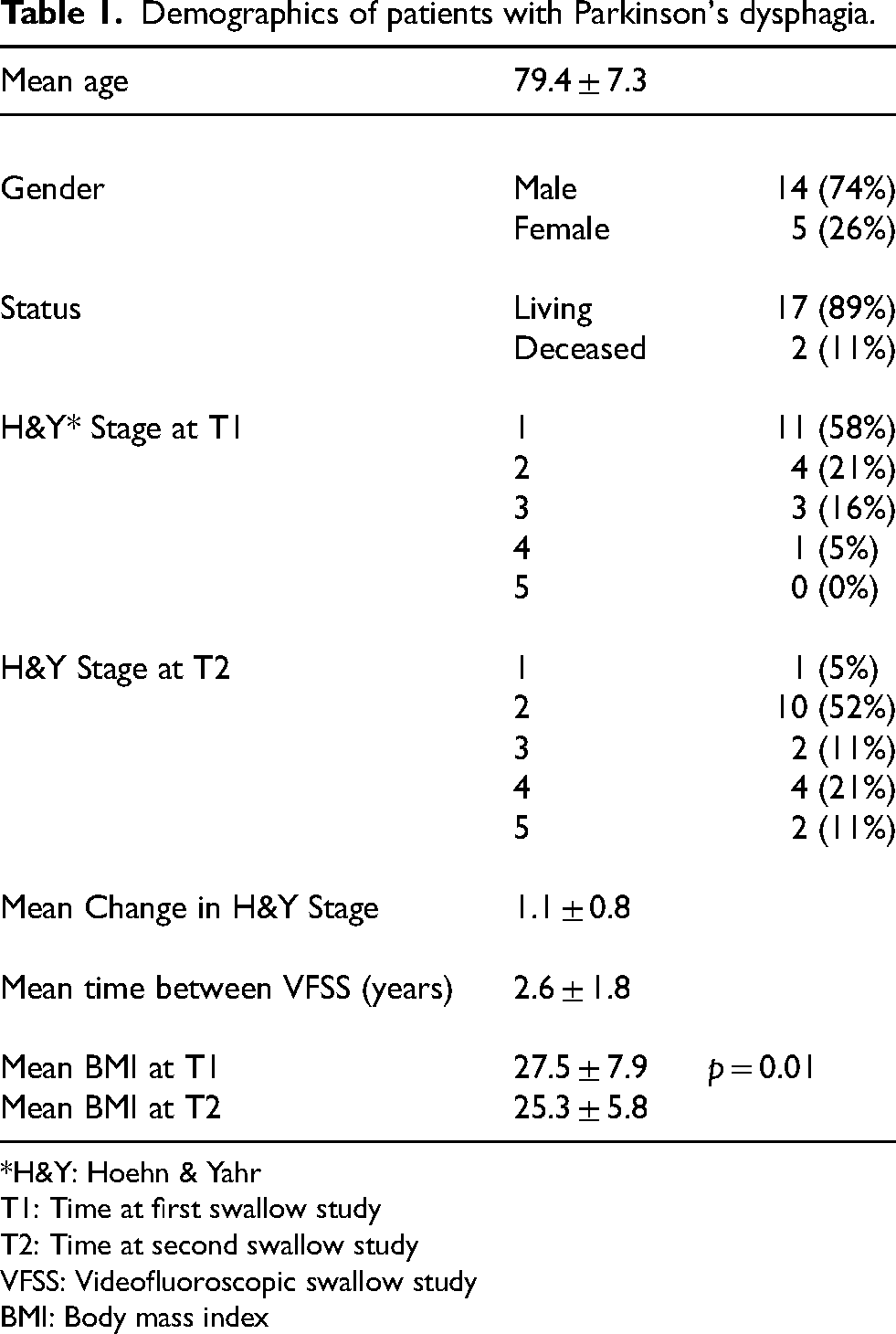
*H&Y: Hoehn & Yahr
T1: Time at first swallow study
T2: Time at second swallow study
VFSS: Videofluoroscopic swallow study
BMI: Body mass index
Swallow outcomes at T1 versus T2 in patients with Parkinson's dysphagia.
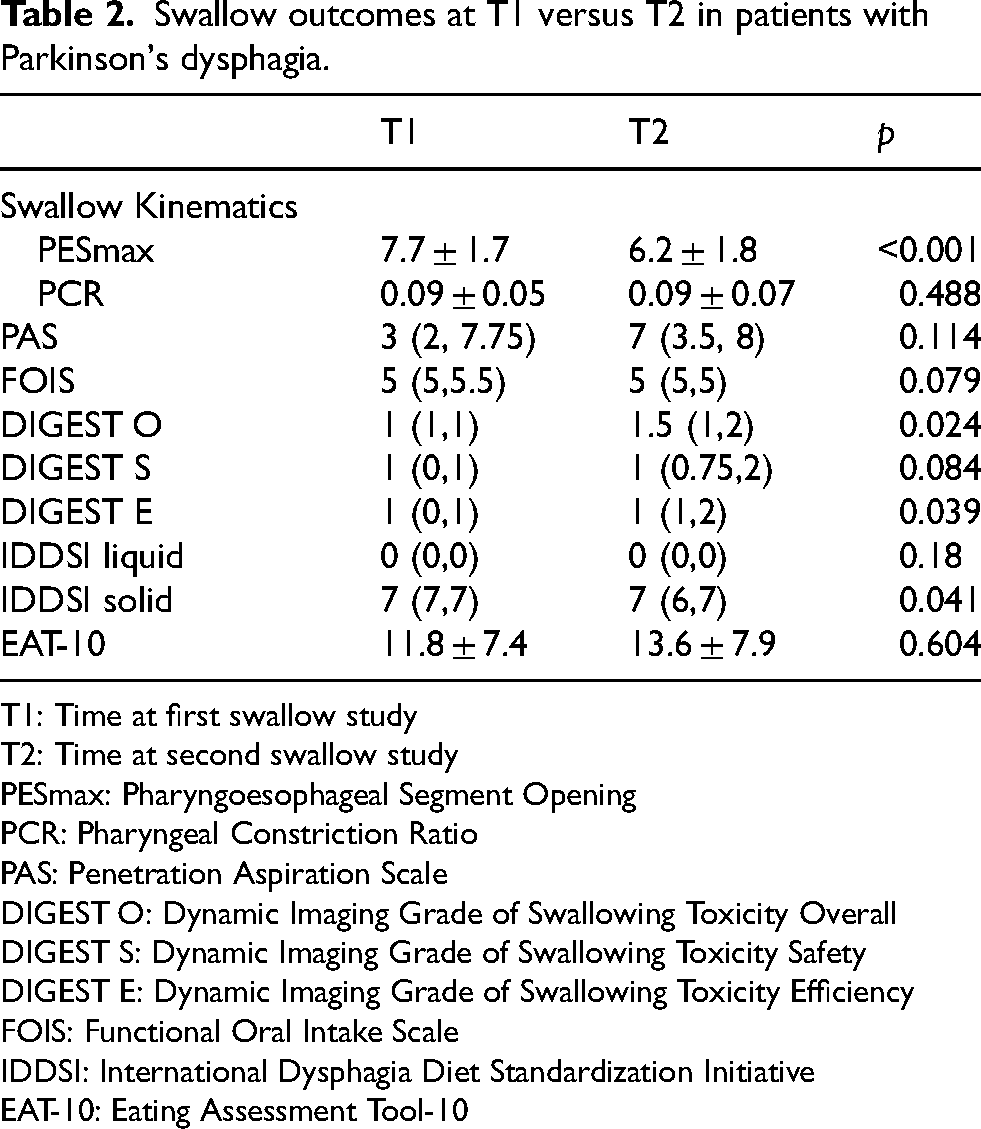
T1: Time at first swallow study
T2: Time at second swallow study
PESmax: Pharyngoesophageal Segment Opening
PCR: Pharyngeal Constriction Ratio
PAS: Penetration Aspiration Scale
DIGEST O: Dynamic Imaging Grade of Swallowing Toxicity Overall
DIGEST S: Dynamic Imaging Grade of Swallowing Toxicity Safety
DIGEST E: Dynamic Imaging Grade of Swallowing Toxicity Efficiency
FOIS: Functional Oral Intake Scale
IDDSI: International Dysphagia Diet Standardization Initiative
EAT-10: Eating Assessment Tool-10
Three out of nineteen patients (16%) were originally referred to our laryngology center for symptoms of dysphagia. All other patients were originally seen for dysphonia (13/19, 68.4%), combined dysphonia/dyspnea (1/19, 5.3%), or combined dysphonia/dysphagia (2/19, 10.5%) (Appendix). Although a minority of patients sought care originally for dysphagia, all T1 VFSS were performed for self-reported or elicited symptoms of dysphagia, such as cough (or sneeze) with foods or liquids (
All patients were on prescription medication for PD, having started treatment an average of 2.9 years before their first VFSS. All were on oral carbidopa/levodopa at T1 and T2 (additional medication details in Appendix). None received subcutaneous, inhaled, or intrajejeunal forms of levodopa and none had undergone DBS or lesional surgery for PD. Mean LEDD at T1 was 676 and at T2 was 871. The average H&Y stage increased from 1.7 at T1 to 2.8 at T2, reflecting a mean progression of 0.42 stages per year. All patients participated in swallow therapy between T1 and T2, with an average of 9.7 therapy sessions per patient. None of the patients were undergoing active swallow therapy at the time of their VFSS. There was an average of 272.7 days between their last swallow therapy session and VFSS at T2.
Regarding swallowing therapy, 15/19 (78.9%) patients participated in swallow therapy within the tertiary medical center, 2/19 (10.5%) sought treatment with SLPs outside the medical center, and 2/19 (10.5%) completed only voice therapy. Of the patients who attended swallowing therapy within the academic medical center, 9/15 (60%) completed concurrent voice and swallowing therapy, while 6/15 (40%) pursued swallowing therapy only. All of these 15 patients received education and counseling on normal swallowing anatomy and physiology, their current swallowing function, and rationale for rehabilitation. The majority of patients (11/15, 73.3%) were counseled on specific diet modifications including special preparations or compensations. Approximately half (8/15, 53.3%) of patients were trained in either compensatory strategies while swallowing (i.e., chin tuck, head turn) and/or swallowing maneuvers (i.e., supraglottic swallow, effortful swallow) to improve swallowing safety and efficiency. Almost all (12/15, 80%) swallowing therapy sessions included oropharyngeal strengthening exercises (i.e., Masako, Mendelsohn, effortful swallow), while 2/15 (13.3%) patients had limitations in ability to follow directions, in which caregiver education and training was the main focus. 28 Respiratory muscle strength training (i.e., EMST-150 or EMST-75) was provided in 9/15 (60%). Adherence was subjectively reported in the SLP notes, with 11/15 (73.3%) identified as partially adherent to the swallow therapy plan, 2/15 (13.3%) completely adherent and diligent with their exercises, and 2/15 (13.3%) lacking enough information to determine adherence.
Distribution of patients by the various swallowing outcome measures between initial and final time points are shown in Figure 1 and Table 2. At T1, despite a low average H&Y stage of 1.7 ± 0.9, swallowing impairments were already observed in our patient cohort. Eight patients exhibited elevated PAS scores (25% PAS = 6, 12.5% PAS = 7, 62.5% PAS = 8). Two patients had atypical DIGESToverall (50%
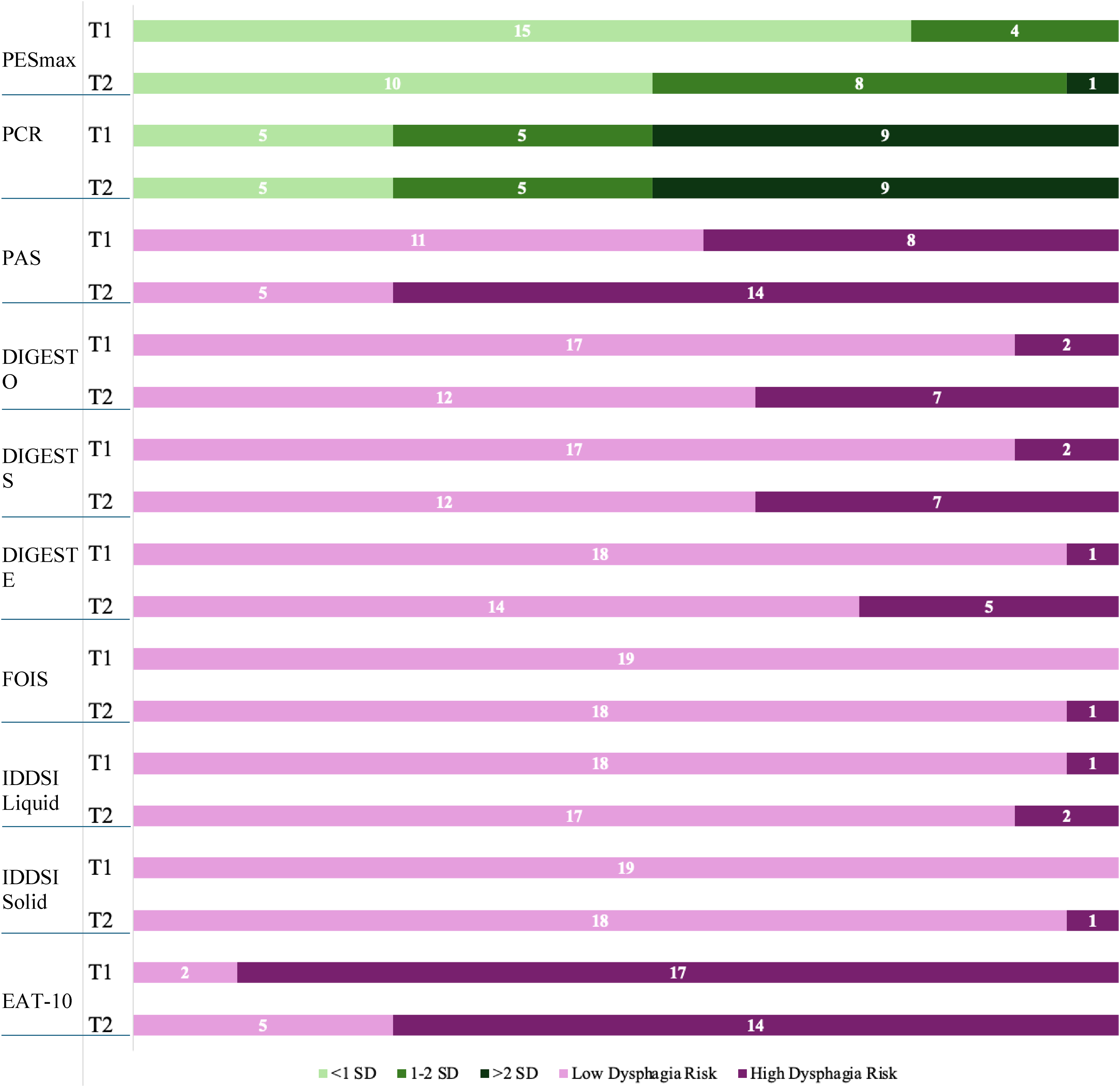
Patient distribution by outcome category from initial (T1) to final (T2) videofluoroscopic swallowing study (VFSS) in Parkinson's disease-related dysphagia. PESmax: Pharyngoesophageal Segment Opening; PCR: Pharyngeal Constriction Ratio; PAS: Penetration Aspiration Scale; DIGEST O: Dynamic Imaging Grade of Swallowing Toxicity Overall; DIGEST S: Dynamic Imaging Grade of Swallowing Toxicity Safety; DIGEST E: Dynamic Imaging Grade of Swallowing Toxicity Efficiency; FOIS: Functional Oral Intake Scale; IDDSI Liquid: International Dysphagia Diet Standardization Initiative liquid; IDDSI Solid: International Dysphagia Diet Standardization Initiative solid; EAT-10: Eating Assessment Tool-10.
At T2, swallowing impairments progressed in our patient cohort (average H&Y stage 2.8 ± 1.2). Fourteen patients demonstrated elevated PAS scores (1/14 PAS = 5, 3/14 PAS = 6, 1/14 PAS = 7, 9/14 PAS = 8). Seven patients had elevated DIGESToverall (4/7 D-O = 2, 3/7 D-O = 3), seven patients had elevated DIGESTsafety (5/7 D-S = 2, 2/7 D-S = 3), and five patients had elevated DIGESTefficency (3/5 D-E = 2, 2/5 D-E = 3). One patient exhibited a reduced FOIS score of 2. Two patients demonstrated elevated IDDSI liquid (1/2 I-L = 2, 1/2 I-L = 3). Fourteen exhibited EAT-10 (range 3–26) scores greater than 3. Nine patients demonstrated mild to moderate deviation from PESmax normative values (8 patients 1–1.5 SD, 1 patient 1.5–2 SD), and one patient demonstrated significant deviation (1 patient >2 SD). Five patients exhibited mild to moderate deviation from PCR normative values (1 patient 1–1.5 SD, 4 patients 1.5–2 SD), and nine patients continued to exhibit significant deviation (9 patients >2 SD).
A paired samples
A subgroup analysis was performed to analyze the change in outcomes in patients with mild to moderate PD (H&Y stage 1 to 3) at T2. A statistically significant decline was seen in PESmax (−2.2,
Linear regression was performed between the number of swallow therapy sessions and the overall duration of swallow therapy with the change in outcomes. EAT-10 was significantly correlated with session count (
Discussion
This study highlights the progressive decline in swallowing function among individuals with PD, underscoring the critical need for long-term, proactive dysphagia surveillance and patient-centered management. Through a retrospective review of a tertiary-care cohort, we tracked changes in swallow physiology over an average of 2.6 years using validated functional scales and kinematic measures. At the time of the most recent VFSS, no patients were actively engaged in swallow therapy, with an average of nine months since their last session. This reflects common clinical practice, which often dichotomizes swallow therapy into either (a) proactive intervention – ideally prior to significant functional decline, or (b) episodic rehabilitation, often initiated in response to emerging symptoms, rather than as a continuous treatment. 4
Most patients were initially referred to our clinic for evaluation of dysphonia. VFSS at T1 was not performed solely due to a diagnosis of Parkinson's Disease or for routine screening purposes. Instead, the decision to conduct VFSS was driven by clinical signs suggestive of dysphagia, regardless of whether patients explicitly reported swallowing difficulties. Notably, although only three patients presented with dysphagia as their chief complaint, all exhibited observable symptoms or clinical features raising concern for impaired swallow function. This underscores the subtle and often underrecognized presentation of dysphagia in individuals with PD.
In contrast, T2 VFSS was conducted for routine longitudinal monitoring in the absence of specific clinical triggers. Given the progressive nature of dysphagia in PD and the frequent disconnect between patient awareness and physiologic impairment, our clinical practice supports annual VFSS to assess oropharyngeal function. More frequent assessments are prompted by patient-reported symptoms, caregiver observations, or clinical signs suggestive of dysphagia-related sequelae (e.g., weight loss, aspiration pneumonia).
This structured approach to surveillance acknowledges the well-documented mismatch between subjective symptom reporting and objective physiologic findings in PD. Indeed, while self-reported EAT-10 scores remained stable over time, we observed significant deterioration in objective measures, including swallowing kinematics (PESmax,
Of note, interrater reliability for all outcome measures was excellent, indicating a high level of consistency between raters. These findings support the robustness and reproducibility of our rating procedures and affirm the reliability of both kinematic and categorical measures in this study. Such strong agreement enhances the credibility of longitudinal comparisons and suggests minimal measurement bias introduced by rater variability. FOIS and IDDSI were excluded from this post-hoc analysis, as these measures require comprehensive clinical context, including patient history and symptomatology, that was not accessible during blinded review.
Subgroup analysis revealed that both early and late-stage patients experienced functional deterioration, though the magnitude and nature of changes differed. Patients with advanced disease (H&Y 4–5) showed marked reductions in PESmax, while those in earlier stages exhibited worsening DIGEST scores. These findings may indicate that while the mechanisms of decline may differ by disease severity, the need for continued dysphagia surveillance applies across the PD spectrum.
In addition to functional decline, we observed a statistically significant decrease in BMI (
Our findings support the implementation of structured, long-term dysphagia management programs in PD, initiated early and maintained throughout the disease course. Strategies may include periodic instrumental assessments (e.g., VFSS) along with individualized therapy plans based on evolving physiology, caregiver training, and techniques to optimize adherence. Approaches that integrate motor learning principles, skill-based training, and respiratory-swallow coordination may be particularly valuable if introduced early and revisited regularly throughout the disease course.
It is important to note that swallowing dysfunction may occur due to the normal aging process. The progression is gradual, heterogeneous and marked by subtle physiologic changes rather than abrupt loss of function. Recognized age-related swallowing changes include increased oral and pharyngeal transit times, delayed initiation of swallow, prolonged upper esophageal sphincter opening and reduced pharyngeal pressures.29,30 Age-related sarcopenia of swallowing muscles can lead to reduced tongue control, decreased hyolaryngeal elevation and less effective pharyngeal contraction, resulting in increased pharyngeal residue. 29 Despite these changes, overt dysphagia does not occur unless compounded by disease or additional insults. This loss of functional reserve puts older adults at higher risk for clinically significant swallowing dysfunction when faced with illness or neurological disease. 30 Current swallowing assessments (including objective measures and subjective patient symptom reports or patient reported outcome measures) are unable to differentiate between various etiologies of swallowing dysfunction. The degree to which age and neurologic impairment may exacerbate each other – especially related to severity of PD – remains unknown and is an important focus of future study.
Several limitations warrant consideration. The sample size was modest, limiting generalizability. Nevertheless, post-hoc power analysis confirmed sufficient statistical power to detect moderate-to-large effects even when accounting for small sample size bias. The retrospective design limits control over the timing and content of swallow therapy. Although patients were not actively in therapy during VFSS, variability in prior therapy exposure and adherence may still influence outcomes. It is unlikely that referral patterns introduced sampling bias in this study. All but three patients were referred for speech changes and were subsequently found to have dysphagia, indicating that PD patients with dysphagia may not be referred as early as they should for that indication. Notably, three patients underwent their latest VFSS due to perceived worsening of swallow function, potentially influencing the severity of findings at T2. Finally, while all patients were receiving dopaminergic therapy, medication timing and state (on/off L-dopa) relative to VFSS was not consistently recorded. This may have influenced the degree of observed impairment given that dopaminergic therapy can improve swallowing function in some patients. Finally, the lack of a comparison control group of patients who did not participate in swallowing therapy presents a limitation. At our institution, all individuals who underwent VFSS evaluation were subsequently referred for and engaged in swallowing therapy as part of routine clinical management. While this uniformity in care reflects real-world practice, it precludes assessment of how dysphagia progression may differ in patients who do not receive intervention.
Future prospective studies are needed to clarify the long-term impact of sustained dysphagia therapy on disease progression and quality of life. Protocols should include periodic objective swallowing evaluation using tools like VFSS with special attention to parameters sensitive to disease progression (e.g., PESmax, DIGEST, IDDSI). This will allow clinicians to identify deterioration, tailor swallow interventions effectively, and improve long-term outcomes for individuals with PD-related dysphagia.
Conclusion
In this retrospective cohort of individuals with PD, longitudinal VFSS findings over an average follow-up of 2.6 years suggest a persistent progression of swallowing impairments, particularly in pharyngoesophageal segment opening and overall swallow efficiency. Notably, these changes were observed despite prior engagement in swallow therapy, underscoring the importance of routine instrumental monitoring and the potential need for ongoing, individualized dysphagia management throughout the disease course.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X261420003 - Supplemental material for Longitudinal change in swallow function in patients with Parkinson's disease (PD)
Supplemental material, sj-docx-1-pkn-10.1177_1877718X261420003 for Longitudinal change in swallow function in patients with Parkinson's disease (PD) by Camryn R Marshall, VyVy N Young, Zoe Weston, Sarah L Schneider, Tyler W Crosby, Clark A Rosen, Steve D Stockton Jr., Ian O Bledsoe and Yue Ma in Journal of Parkinson's Disease
Footnotes
Acknowledgements
none
Disclosures
Sarah L. Schneider- MedBridge (Royalties)
Clark A. Rosen, MD – AmplioVox (Co-owner), Instrumentarium (Royalties), Medtronic (Consultant), Reflux Gourmet LLC (Shareholder); Aether Devices, Inc (Shareholder); Copyright holder of the VHI-10, Dyspnea Index, SVHI-10, CSI
Ian O. Bledsoe MD – Abbvie (Consultant), Clarityco (Consultant), GLG, Inc. (Consultant)
Accepted for poster presentation at the American Broncho-Esophagological Association (ABEA), May 2025, New Orleans, LA and at the Fall Voice Conference, Sept 2025, Charlotte, NC
Ethical statement
These authors agree to uphold the highest standards of publishing ethics and affirm that there have been no misconduct related to this study or the writing of this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Data availability statement
The datasets generated during and/or analyzed during the current study are available in the attached Appendix and/or from the corresponding author on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
