Abstract
Background
Exergaming has shown benefits in Parkinson's disease (PD) rehabilitation; however, its feasibility and potential effectiveness for people with moderate to advanced PD remain unclear.
Objective
To assess the feasibility and potential effectiveness of a universally designed exergame program in people with moderate to advanced PD.
Methods
This evaluator-blind, parallel randomized controlled trial was conducted at four specialized PD care facilities in Japan. Fifty-six participants (Hoehn & Yahr stages III–V) were randomly assigned (1:1) to the intervention or control group. The intervention group participated in an 8-week, universally designed exergame program alongside a standardized rehabilitation program. Sessions were conducted in supervised groups and lasted 15 min, thrice a week. Acceptability was assessed with a questionnaire. Adherence was based on attendance. Safety was monitored, and session-level experience (fatigue, effort, perceived progress, enjoyment) was assessed. Predefined criteria were applied for acceptability, attendance adherence, and session-level experience. The potential effectiveness was assessed by changes in health-related quality of life (HR-QoL), motor function, cognitive function, social engagement, and loneliness.
Results
The final analysis included 37 participants. The program was judged acceptable by 84% participants, and the attendance adherence was 99.7%. No intervention-related adverse events occurred. Acceptability, attendance adherence, and session-level experience met the prespecified criteria. Exploratory findings suggested potential effectiveness for HR-QoL and loneliness, whereas no significant changes were observed in motor or cognitive function scores.
Conclusion
This pilot study supports the feasibility of our exergame program for people with moderate to advanced PD and shows its potential effectiveness for HR-QoL/loneliness.
Trial Registration
UMIN Clinical Trials Registry (UMIN-CTR), https://www.umin.ac.jp/ctr/, UMIN000054292 (registered on May 1, 2024).
Plain language summary
Why was this study done?
Exercise games (exergames) can help people with Parkinson’s disease (PD) in rehabilitation. However, it has not been clear whether people with progressed PD symptoms can participate safely, continue the program, and benefit from it. We tested a universally designed exergame in people with progressed PD symptoms living in care facilities in order to confirm its feasibility (safe, practical, and acceptable) and positive effects.
What did the researchers do?
The study took place at four PD care facilities in Japan and involved 56 people with progressed PD symptoms. Participants were randomly assigned to a group that received only the usual rehabilitation or a group that received the same rehabilitation plus a 15-minute exergame session thrice a week for 8 weeks. We checked safety, acceptability, and attendance of the sessions. We also noted participants’ experiences during the sessions, such as fatigue and enjoyment, and possible effects on quality of life, movement, thinking ability, social connections, and loneliness.
What did the researchers find?
From the 56 people who joined the study, 37 were included in the final analysis. No problem-related accidents or serious problems occurred. General acceptability was rated positively by 84% of the participants, and attendance reached 99.7% of the planned sessions. All predefined targets for acceptability, participation, and session experience were met. Overall, the exergame was safe, enjoyable, and well accepted, even by people with progressed PD symptoms. It also showed possible benefits for quality of life and loneliness, with no meaningful changes in movement or thinking abilities.
What do the findings mean?
A universally designed exergame can be safe, enjoyable, and acceptable and can potentially improve quality of life and loneliness for people with progressed PD symptoms living in care facilities.
Keywords
Introduction
Parkinson's disease (PD) is a progressive neurodegenerative disorder that requires continuous efforts to maintain and enhance quality of life throughout its clinical course.1,2 While people in early stages can maintain independence through daily activities, disease progression leads to an increased dependence in activities of daily living (ADL).3,4 In advanced stages, the effectiveness of pharmacological interventions and exercise therapy becomes more limited, and management becomes increasingly complex and challenging.5–7
In recent years, technology-assisted exercise interventions, including exergames and virtual reality, have been shown to be effective in improving motor function in individuals with PD.8–12 However, most studies primarily targeted individuals with mild PD, and those with more advanced disease were not adequately investigated. In fact, people with moderate to advanced PD often face severe motor impairments, cognitive decline, and fatigue, all of which make participation in exergames, particularly those requiring complex motor tasks or standing balance, difficult. 13 Moreover, existing exergame research has predominantly focused on physical outcomes such as mobility and balance, whereas evidence addressing outcomes more directly related to quality of life (QOL), including enjoyment and social interaction, remains limited.
People with moderate to advanced PD often face physical limitations that make continuation of previous hobbies and social participation difficult. This in turn contributes to depression, loneliness, and a decline in QOL.14–16 Therefore, opportunities for safe and enjoyable activities should be provided to maintain and improve QOL. However, current exergame research has not sufficiently addressed the challenges of practical implementation and adaptation in this population. Meanwhile, an approach termed actigaming, which involves recreational and accessibility-focused use of exergames, has been described. 17 This approach is in contrast to serious exergames, which are purpose-driven and theoretically grounded, and it is positioned within the implementation framework as an activity-oriented modality. Given these characteristics, actigaming may be more accessible for people with moderate to advanced PD, who often struggle with complex motor tasks or standing balance, and could provide a promising new means of engagement and activity for this population.
However, development or introduction of exergames specifically tailored for people with moderate to advanced PD is often impractical in real-world care settings. Care facilities accommodate residents with diverse characteristics, including differences in age, disability level, and disease severity. Development of separate exergames tailored for each disease stage or symptom subgroup would require substantial resources and cannot be implemented on a large scale. However, exergames designed according to universal design principles can be used regardless of age or disability and can provide a pragmatic solution that also accommodates this population. Accordingly, we conducted this pilot randomized controlled trial to evaluate the feasibility of a highly recreational, easily accessible universally designed exergame program that enables people with moderate to advanced PD to engage in enjoyable activities.
Methods
Study design and participants
This exploratory pilot randomized controlled trial was conducted between October and November 2024 at four specialized Parkinson's disease care facilities in Osaka, Japan (PD houses in Kadoma, Joto, Yao, and Kishibe). The study protocol was approved by the Institutional Review Board in Kansai Medical University (approval number: 2024045) and was registered at the UMIN Clinical Trials Registry (UMIN000054292). To ensure transparency, the registry entry was updated with additional details. As some of this information was added after trial initiation, the registration should be considered a retrospective registration of complete information. This study abided by the CONSORT 2025 guidelines. 18 The completed CONSORT checklist is provided in Supplementary Material 1.
All participants provided written informed consent for participation and publication before enrollment. The trial was designed as a parallel-group study with a 1:1 allocation ratio that compared conventional exercise therapy with the exergame program with conventional exercise therapy alone over an 8-week intervention period. Stratified randomization was performed by facility to ensure a 1:1 allocation ratio within each of the four participating sites.
Eligible participants were residents of specialized Parkinson's disease care facilities. They were aged ≥30 years and diagnosed with PD according to the Movement Disorder Society (MDS) diagnostic criteria. 19 The lower age limit was pragmatically set to reflect the admission policy of the participating facilities, which primarily accommodate middle-aged and older adults with care needs. Inclusion criteria specified Hoehn and Yahr (H&Y) stages 3–5 20 and Mini-Mental State Examination scores ≥18/30. 21 Exclusion criteria comprised (1) medication changes within the past month, (2) neurological, orthopedic, or cardiovascular conditions contraindicated for exergames, (3) visual or auditory impairments that would limit participation, and (4) inability to provide informed consent. Initial screening was conducted through medical records and face-to-face interviews by trained medical staff.
Universal design exergame (UDe-sports)
The exergame used in this study was based on the commercial platform UDe-Sports (General Incorporated Association UDe-Sports Association, Kumamoto, Japan). 22 Developed according to universal design principles, it is operated via a four-color button interface (red, yellow, blue, and green) with output to a large display, enabling operation with either hand and facilitating use by individuals with limited mobility or severe motor impairment.
The system offers a wide range of games, from simple single-button or color-matching tasks (e.g., running race, jump rope, ball toss) to more complex tasks (e.g., rock–paper–scissors, obstacle race, spot-the-difference) that demand selective attention, timing control, and decision-making under time pressure. Difficulty is adjustable at three levels (easy/normal/hard). UDe-Sports is positioned as actigaming rather than a training-based serious exergame, 17 prioritizing universal accessibility and enjoyment so that sessions are easy to play and inviting for all participants. The system also supports multiplayer participation, providing opportunities for shared activity and social interaction. In view of these characteristics and its universal design, the program was considered applicable for people with moderate to advanced PD and implemented in this study.
Details of the game list, interface, and representative gameplay scenes are provided in Supplementary Material 2.
Interventions
Both groups received a standardized, PD-specific rehabilitation program that lasted for 20–30 min per session, three times per week for 8 weeks. The program was conducted in facility rehabilitation rooms under the continuous supervision of licensed physical therapists (PTs) or occupational therapists (OTs). The program comprised four components: (1) stretching of major muscle groups commonly affected in PD 23 ; (2) aerobic exercise at light-to-moderate intensity, guided by clinical judgment and individual tolerance 24 ; (3) resistance training using body weight or light elastic bands, primarily targeting the lower limbs and trunk 23 ; and (4) static and dynamic balance training focused on postural stability and fall prevention. 25 Exercises were individualized according to symptoms, functional status, fatigue, and medication ON/OFF fluctuations. Within this individualized approach, session frequency, setting, and duration were standardized across participants and identical between groups. Safety (e.g., fall risk) was continuously monitored during each session, with immediate assistance provided as required. All participants had been routinely receiving this standardized PD-specific rehabilitation as part of usual care prior to enrollment. All participants continued their usual medical care throughout the study period.
In addition to standard rehabilitation, the intervention group performed the exergame program three times per week for 8 weeks, with each session lasting 15 min. Sessions were delivered for small groups of 2–4 participants in facility rehabilitation rooms, and they were scheduled during the ON medication state (typically 1–2 h post-dose). The participants remained seated (wheelchair or chair) throughout, and all sessions were conducted under PT/OT supervision. The therapists received a brief device-handling orientation (functions, start/stop procedures, button layout, and basic safety).
For individualization and progression, three difficulty levels (easy/normal/hard) were available. The starting level was set by the supervising therapist according to each participant's status and adjusted on the basis of day-to-day performance (e.g., response accuracy and stability of operation). These difficulty adjustments were applied without altering the standardized session schedule (15 min, three times per week) or delivery conditions. For multiplayer sessions, participants were paired according to upper-limb/finger function and simple cognitive factors to maintain competitive balance and engagement.
Motivation was facilitated through competitive/co-operative gameplay, immediate score feedback, and therapist encouragement. No participant had prior exposure to UDe-Sports or supervised exergame programs; thus, the intervention represented their first participation in an exergame program within the rehabilitation setting.
Further details of both the standard rehabilitation and the exergame program are provided in Supplementary Material 3. Additionally, the description of the intervention followed the Consensus on Exercise Reporting Template (CERT),26,27 and the completed CERT checklist is appended in Supplementary Material 4.
Outcome measurements
This pilot, randomized controlled trial was conducted to evaluate feasibility and collect exploratory clinical outcomes to inform the design of a future full-scale trial. The primary outcome was feasibility, defined as a comprehensive construct encompassing acceptability, adherence, safety, and session-level experience. Exploratory outcomes were assessed to estimate variability and potential effect sizes, providing information on possible clinical changes. All assessments from baseline to postintervention were conducted during participants’ “ON” state, typically 1–2 h after medication, at the same time of day. For participants with advanced disease severity (H&Y stage 5), assessments were conducted with the participants seated in a wheelchair. Items requiring standing were performed under therapist supervision, with close monitoring and assistance to ensure safety. Trained evaluators, blinded to group allocation, conducted the assessments.
Following the intervention period, participants in the intervention group completed additional assessments. Program acceptability was assessed using a structured questionnaire based on the Theoretical Framework of Acceptability, which evaluated various aspects including satisfaction with the intervention, perceived benefits, challenges encountered, and willingness to continue the program. 28 If >80% participants agreed or strongly agreed with a particular statement (or disagreed or strongly disagreed with statements coded reversely), the component construct was determined to be acceptable. 29
Implementation of the exergame was evaluated by recording attendance and completion for each session over the 8-week schedule (15 min per session, three sessions per week, 24 sessions in total). Adherence was reported using two metrics: attendance adherence and completion adherence. Attendance adherence reflected compliance with the prescribed exergame schedule, excluding discontinuation for reasons unrelated to the intervention (e.g., hospitalization), and was calculated as the proportion of scheduled sessions completed during the program. An acceptable level of attendance adherence was predefined as ≥75%. 30 Completion adherence referred to trial-level follow-up, defined as the proportion of all randomized participants who completed the post-intervention assessment. Adverse events were systematically documented in both groups using standardized forms and included events that occurred during sessions as well as any incidents arising throughout the study period.
Session-level experience ratings (fatigue, effort, perceived progress, and enjoyment) were collected in the intervention group using a 6-point Likert scale. These items assessed the physical and mental effort required for the activities, fatigue, sense of progress, and level of enjoyment during participation. Target ranges were predefined as follows: (1) fatigue, expected to be lower than “4 = very much”; (2) effort, expected to fall between “1 = a little” and “4 = very much;” (3) progress, expected to fall between “1 = a little” and “5 = extreme;” and (4) enjoyment, expected to fall between “1 = a little” and “5 = extreme”. 31
To evaluate the potential effectiveness of the intervention, subjective disease-specific health-related QoL (HR-QoL) was assessed using Parkinson's Disease Questionnaire-39 (PDQ-39). 32 Motor symptoms were assessed using Part III of the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS). 33
The 12-item Short Form Health Survey (SF-12) was used to measure HR-QoL, providing separate scores for the Physical Component Summary (PCS) and Mental Component Summary (MCS). Scores are standardized to a population mean of 50 and a standard deviation of 10, with higher scores indicating better health status. 34
Cognitive function was evaluated using the following two standardized tests: the Trail Making Test Part A (TMT-A) for attention and processing speed, measured by completion time in seconds, and the Stroop Color and Word Test (SCWT) for executive function and response inhibition, providing scores for word reading (W), color naming (C), and color-word interference (CW). Each score was calculated based on the number of correct responses within 45 s.35,36
Social engagement was assessed using the Japanese version of the Lubben Social Network Scale-6 (LSNS-6), a 6-item scale measuring perceived social support from family and friends. Scores range 0–30, with higher scores indicating better social engagement. 37 The Three-Item Loneliness Scale (TIL scale) was used to measure subjective feelings of loneliness, consisting of three items rated on a 3-point scale, with total scores ranging 3–9 and higher scores indicating greater loneliness. 38
Sample size
The sample size was set with the primary objectives of ensuring adequate precision in estimating the standard deviation (SD) of the potential effectiveness outcome and assessing feasibility. To align with published ranges for external pilot trial sizes, we assumed approximately 20% attrition after allocation and recruited 60 participants.39,40
Statistical analysis
In accordance with the prespecified statistical analysis plan, all analyses involved complete case analysis. For each outcome, we analyzed randomized participants with complete baseline and follow-up measurements, and participants with incomplete data were excluded from that outcome analysis (listwise deletion). 41 Participants’ baseline characteristics were summarized as median [interquartile range, Q1–Q3] for continuous variables and as n (%) for categorical variables. Distributional assumptions were examined using the Shapiro–Wilk test and Q–Q plots to assess normality. To evaluate the potential effectiveness of the exergame intervention, between-group differences in outcome changes were estimated with 95% confidence intervals. We employed a nonparametric bootstrap method with 5000 replications to estimate the 95% confidence intervals for between-group differences in outcome changes. Change values were calculated by subtracting baseline values from follow-up values. Effect sizes for between-group differences in outcome changes were calculated using Cohen's d. No covariate adjustment was prespecified, and the analyses were therefore conducted without adjustment. All statistical analyses were performed using R version 4.1.2 (R Foundation, Vienna, Austria).
Results
Enrollment
The study design and allocation are summarized in Figure 1. In October 2024, 60 participants who met the eligibility criteria were enrolled in this study. Data from four participants were unavailable because of the lack of participation in the baseline assessment. The remaining 56 participants were randomly assigned to two groups. During the study period, five participants in the intervention group and six in the control group were lost to follow-up due to hospitalization. At the follow-up assessment, data from eight participants (four in the intervention group and four in the control group) were missing due to incomplete responses in self-reported outcome measures. As a result, the final analysis was conducted with 19 participants in the intervention group and 18 in the control group.
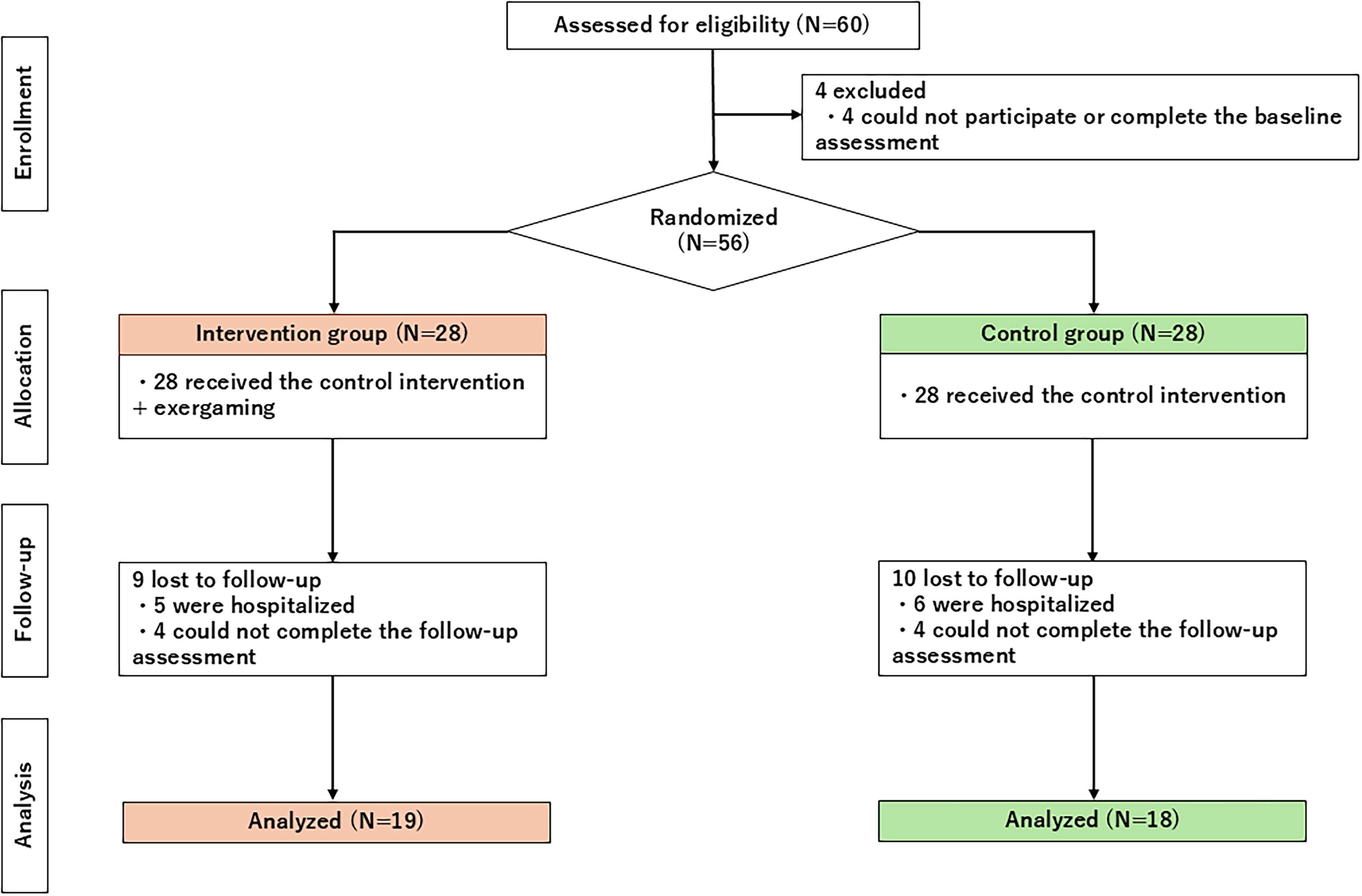
Participants allocation and enrollment.
Participant characteristics
The baseline characteristics of the participants are summarized in Table 1. No significant differences in baseline characteristics were observed between the two groups.
Baseline characteristics of study participants.

Data are shown as median [interquartile range] or n (%).
PwAPD: Patients with advanced Parkinson's disease; LEDD: Levodopa Equivalent Daily Dose; UPDRS-III: Unified Parkinson's Disease Rating Scale Part III; MMSE: Mini-Mental State Examination.
Adherence
In this study, completion adherence was 37 out of 56 participants (66.0%), and it was affected by dropouts due to hospitalization and missing data in self-reported outcome measures. In contrast, attendance adherence to the exergame intervention was relatively high. One participant in the intervention group was unable to attend for 1 week (three sessions) because of an infectious illness but resumed study participation thereafter. All other participants completed all 24 prescribed sessions as scheduled. The overall session completion rate was 99.7%, substantially exceeding the predefined adherence threshold of 75%, demonstrating a high level of compliance with the exergame intervention protocol.
Safety and session-level experience
A total of 11 participants dropped out due to hospitalization (5 in the intervention group and 6 in the control group). In total, 17 adverse events were reported among all participants, including those who dropped out. Based on assessment by an independent physician with no conflicts of interest in this study, all adverse events were determined to be unrelated to the intervention. Additionally, no intervention-related accidents or other adverse effects were reported in either group. Detailed safety outcomes are provided in Supplementary Material 5.
Averaging across participants and sessions, participants generally experienced “a little” fatigue (1.5 ± 1.4), training was reported to require “a little” to “some” effort (1.6 ± 1.0), progress was perceived as “a little” (1.1 ± 0.7), and enjoyment ranged from “some” to “much” (2.4 ± 1.3). All measures fell within the predefined criteria. Session-level experience outcomes are presented in Supplementary Material 6.
Acceptability
The results of exergame acceptability are shown in Figure 2. A large proportion of participants accepted the exergame (84%, 16/19; general acceptability) and reported that playing the exergame did not interfere with other priorities (90%, 17/19; opportunity costs), indicating that incorporating exergames into daily life under the study settings did not impose a significant burden.
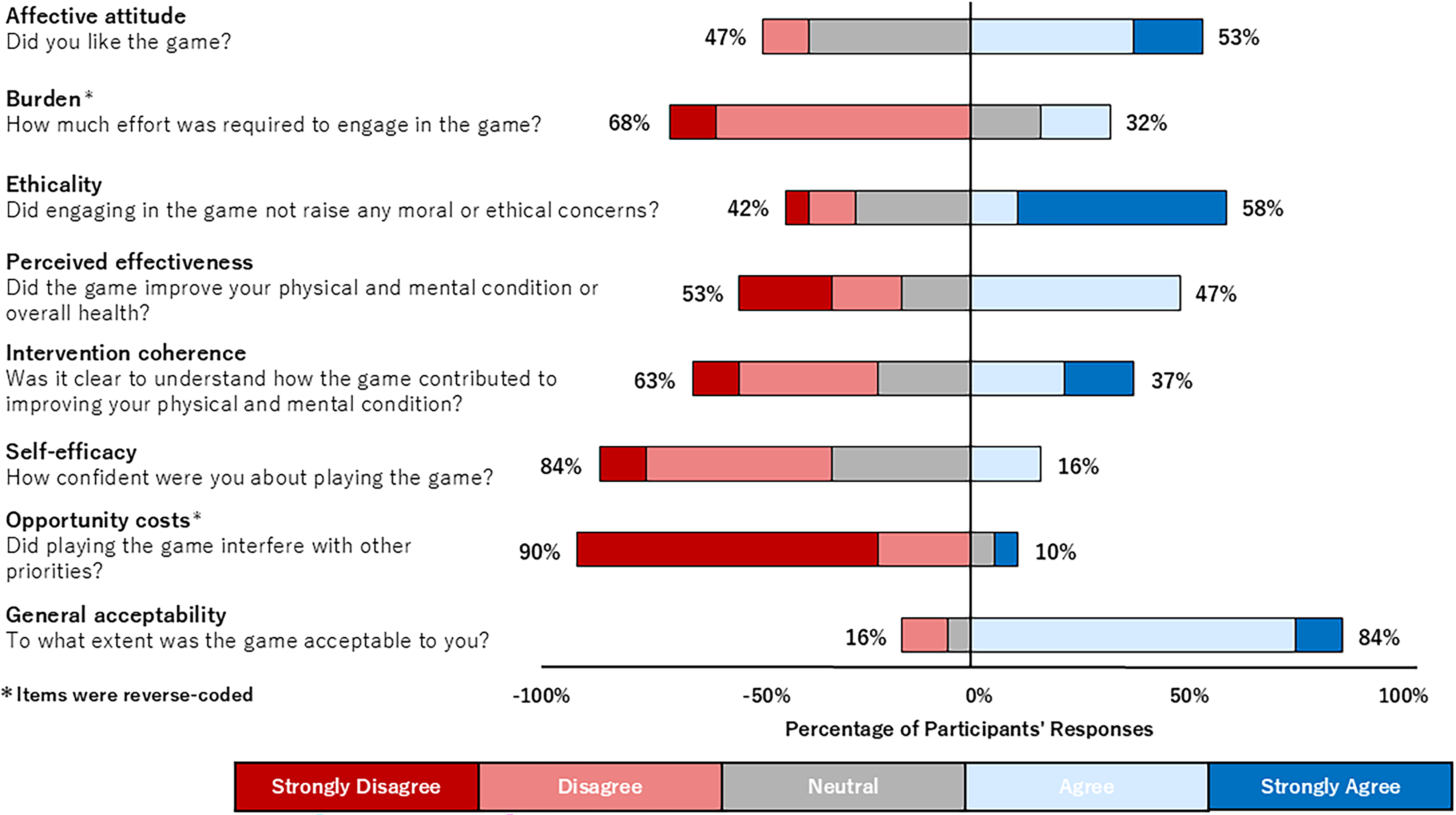
Acceptability of exergames among participants: A theoretical framework of acceptability (TFA)-based analysis.
However, some components had an acceptance rate of less than 50%. Fewer than half of the participants felt that the exergame contributed to improvements in their physical and mental health (47%, 9/19; perceived effectiveness), and an even smaller proportion found it clear how the exergame contributed to such improvements (37%, 7/19; intervention coherence). Additionally, very few participants felt confident in playing the exergame (16%, 3/19; self-efficacy).
Potential effectiveness outcome
The effectiveness of the exergame intervention was assessed for each outcome, as shown in Table 2. SF-12 PCS scores improved in the intervention group (mean change: 7.9 ± 13.8), with a moderate effect size compared to the control group (Cohen's d = 0.54). Furthermore, scores on the TIL Scale also improved in the intervention group (−0.4 ± 1.2), whereas the control group showed slight worsening (0.05 ± 1.6), indicating reduced feelings of loneliness (Cohen's d = −0.33). For transparency, median [Q1–Q3] values for all outcomes at baseline and follow-up and the corresponding changes are provided in Supplementary Material 7.
Mean differences in outcomes between the intervention and control groups.
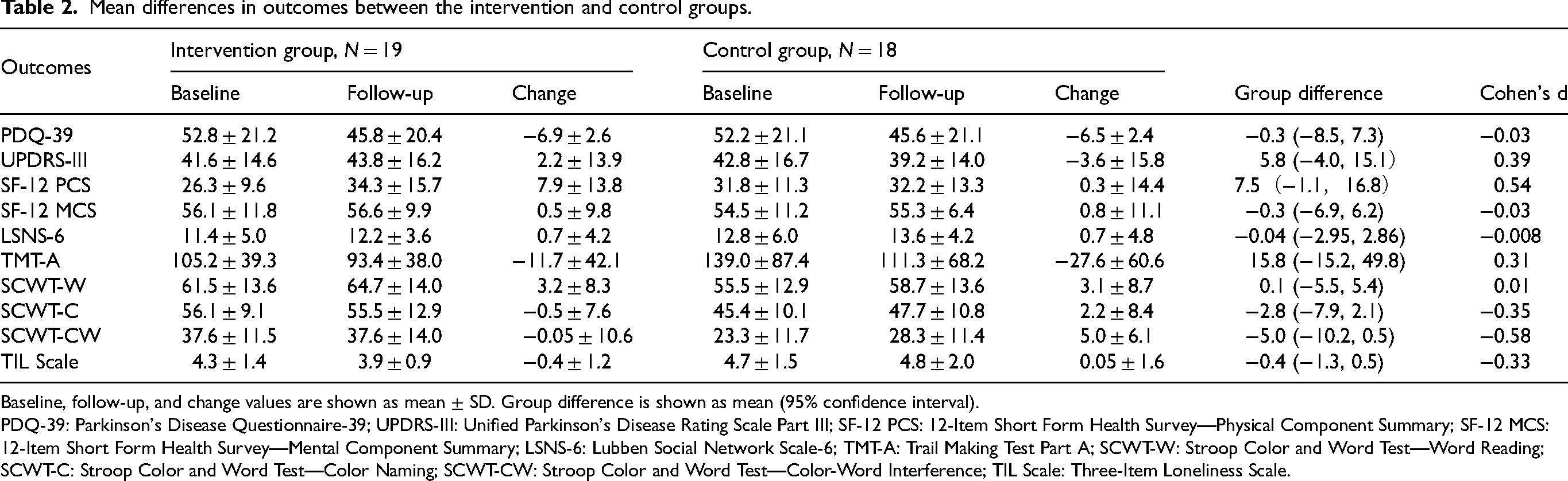
Baseline, follow-up, and change values are shown as mean ± SD. Group difference is shown as mean (95% confidence interval).
PDQ-39: Parkinson's Disease Questionnaire-39; UPDRS-III: Unified Parkinson's Disease Rating Scale Part III; SF-12 PCS: 12-Item Short Form Health Survey—Physical Component Summary; SF-12 MCS: 12-Item Short Form Health Survey—Mental Component Summary; LSNS-6: Lubben Social Network Scale-6; TMT-A: Trail Making Test Part A; SCWT-W: Stroop Color and Word Test—Word Reading; SCWT-C: Stroop Color and Word Test—Color Naming; SCWT-CW: Stroop Color and Word Test—Color-Word Interference; TIL Scale: Three-Item Loneliness Scale.
On the other hand, UPDRS-III scores improved more in the control group (−3.6 ± 15.8) than in the intervention group (2.2 ± 13.9), with a small effect size (Cohen's d = 0.39). Similarly, for cognitive function outcomes such as TMT-A and SCWT-CW, the control group showed greater improvement compared to the intervention group. Nevertheless, pre- and postintervention mean scores for these outcomes were generally higher in the intervention group than in the control group.
Discussion
This study was conducted as an exploratory pilot trial to generate feasibility evidence regarding an adaptive exergame program for people with moderate to advanced PD. The purpose of this pilot trial is not necessarily to detect significant effects but to provide sufficient evidence to determine the feasibility of proceeding to a full-scale trial. 42 In this study, attendance adherence to the exergame intervention was relatively high. Although one out of 19 participants had to withdraw for a week due to COVID-19 infection, all other participants successfully completed the scheduled intervention of three 15-min sessions per week. No adverse events related to the exergame intervention were observed; this confirmed its safety. Session-level experience ratings (fatigue, effort, progress, and enjoyment) fell within acceptable ranges, suggesting that the intervention could be sustainable without imposing an excessive burden on this population. Furthermore, high acceptability of the exergame program was observed, as 84% participants found it acceptable. With regard to potential effectiveness, improvements were observed in the SF-12 PCS score and the TIL Scale. Taken together, these findings support the feasibility of implementing this exergame program for people with moderate to advanced PD.
One of the factors contributing to the high acceptability was the convenience of the exergame program in daily life. Notably, 90% participants agreed with the “opportunity cost” item, suggesting that the exergame intervention was acceptable as part of their daily life. People with moderate to advanced PD often experience motor and non-motor difficulties in daily life, struggle with fatigue, and must incorporate leisure activities within their limited energy reserves.43–45 However, the exergame did not impose an excessive burden and allowed participants to continue it without difficulty. Session-level experience findings further supported the feasibility of the program for this population. The exergame was designed to require mild to moderate effort without excessive burden and allowed participants to perceive progression through scoring. In particular, the improvement in enjoyment scores was the highest among the evaluated items. The higher enjoyment score of the exergame can be attributed to the variety of game types, which prevented boredom, and the appropriate difficulty adjustments according to the characteristics of this population. Previous studies have identified that conventional exergames were excessively difficult for this population. 13 The exergame used in our study allowed real-time difficulty adjustments based on the player's condition, which ensured optimal balance between challenge and manageability. This design enabled participants to easily enjoy the game without feeling overwhelmed. Furthermore, appropriate feedback was provided throughout gameplay, which helped participants recognize their progress. This feedback may have contributed to sustainable motivation. 46 The opportunity for interaction and moderate competitiveness with others may also have contributed to the higher adherence rate to the exergame program.47,48 For instance, participants were naturally motivated to improve their skills and tried to get higher scores, which facilitated continuous participation.49,50 Additionally, the gaming activity was perceived as a positive and enjoyable experience rather than an obligation; therefore, participants were more likely to engage in the exergame consistently.50,51 However, some indicators of acceptability did not achieve sufficient evaluation, and the reasons for this must also be considered. Almost half (47%) the participants agreed that the exergame contributed to improvements in their physical and mental health (perceived effectiveness), and an even lower percentage (37%) fully understood the intervention's purpose and expected effects (intervention coherence). Moreover, only 16% had confidence in their ability to play the exergame (self-efficacy). These results suggest that people with moderate to advanced PD may have difficulty in immediately perceiving the benefits of the exergame program, and that the objectives and expected outcomes of the gaming intervention may not have been adequately understood. Clear explanation of the effects of the exergame program on rehabilitation is crucial for enhancing intervention coherence.52,53 Further, the lower self-efficacy scores may have been influenced by concerns regarding physical activity and participants’ unfamiliarity with gaming due to their older age (mean: 78 years), which may have led to excessive anxiety and feelings of difficulty during gameplay. 54 Moreover, this population usually exhibits excessive anxiety. 55 To overcome these issues, participants should be allowed to experience success in gaming and reduce their anxiety about the intervention. Specifically, step-by-step tutorials and intuitive operational support could help participants get accustomed with the game and ensure smooth transition into gameplay. Positive feedback to participants to generate a feeling of accomplishment is also important for enhancing self-efficacy. 56
With regard to the potential effectiveness of the exergame program for people with moderate to advanced PD, the SF-12 PCS score improved in the intervention group (mean change: 7.9 ± 13.8), whereas only a slight change was observed in the control group (0.3 ± 14.4). The improvement in HR-QoL is consistent with the results of previous research.57,58 This suggests that enjoyment, a feeling of achievement, and the promotion of social interaction between participants via exergames may have contributed to the improvement in HR-QoL scores. Unlike traditional rehabilitation exercises performed in isolation, the exergame provided opportunities for interaction through in-game competition and cooperative play, which may have reduced loneliness. This effect is reflected in the TIL Scale scores, which improved in the intervention group (−0.4 ± 1.2) and slightly worsened in the control group (0.05 ± 1.6), indicating reduced loneliness by interplay gaming. 59
UPDRS III motor scores, unexpectedly improved in the control group (−3.6 ± 15.8) but worsened in the intervention group (2.2 ± 13.9). In addition, executive function measures such as TMT-A and SCWT-CW showed lesser improvement in the intervention group (e.g., TMT-A: −27.6 ± 60.6 in the control group vs. −11.7 ± 42.1 in the intervention group). One possible explanation is the nature of the intervention itself. While the universal design (actigaming) approach enhanced feasibility in real-world care settings, there was a gap when compared with current recommendations regarding condition-specific task design and sufficient exercise dosage.8–12 These differences may have contributed to the limited and inconsistent effects in motor and cognitive domains. Going forward, while preserving the feasibility of actigaming, combining adaptive difficulty calibrated to disease severity with the inclusion of disease-specific task components and stepwise dose increases may extend benefits beyond subjective outcomes to impairment-level domains.
This study has several limitations. First, the intervention employed a universal design exergame and was not specifically tailored for people with PD. While its broad accessibility, regardless of age or level of disability, supported feasibility in real-world care settings from an actigaming perspective, the lack of task specificity and the relatively low dose and intensity may have contributed to the limited effects on motor and executive functions. Second, attrition among participants and missing self-reported assessments reduced the number of participants available for analysis; this may have lowered the power to detect between-group differences. These issues also impeded complete follow-up and affected data integrity. This pilot trial additionally provides insights into the feasibility of trial operations. Recruitment and stratified randomization across multiple facilities were successfully implemented, and blinded outcome assessments were maintained. However, the proportion of missing data in self-reported measures highlighted challenges in ensuring complete follow-up, which should be addressed in the design of a future full-scale trial. To overcome these issues, a larger sample size and strong data collection framework by improving resource allocation are necessary in future, along with re-enforcement of the exergame paradigm to increase the duration of the intervention. An additional approach is supplementation of the dataset by using the chained equations method.
Implications for research
This study was conducted as a pilot trial primarily aimed at exploring feasibility. The use of a universal design exergame allowed for broad feasibility testing in people with moderate to advanced PD and highlighted the limitations of a non-condition–specific approach, as reflected in the modest effects on motor and cognitive outcomes. These findings indicate that future research should move beyond feasibility testing and focus on developing and evaluating interventions more specifically tailored to the needs of this population. In this sense, the present study should be considered an early step in the broader line of research on exergame programs for this population, moving research in the field from feasibility testing to the development and rigorous evaluation of more tailored interventions.
Conclusion
This pilot randomized controlled trial evaluated the feasibility of a universal design exergame for people with moderate to advanced PD. No serious intervention-related adverse events were observed, and both acceptability and attendance adherence were favorable, supporting the practical feasibility of this intervention.
Potential improvements were observed in HR-QOL and loneliness, whereas no meaningful changes were found in motor or cognitive function. Among the subcomponents of acceptability, perceived effectiveness, intervention coherence, and self-efficacy were relatively low. In addition, data incompleteness and the lack of disease-specific tailoring were identified as challenges requiring further refinement.
Future studies should focus on optimizing task specificity and intensity, strengthening data management, and enhancing participant education to improve self-efficacy. These findings provide preliminary evidence to advance exergaming research for people with moderate to advanced PD from feasibility testing to the next stage.
Footnotes
Acknowledgments
We would like to express our sincere gratitude to Yusuke Yakushiji from the Department of Neurology, Kansai Medical University, for his invaluable support and dedication to this research.
Ethical considerations
The study protocol was approved by the Institutional Review Board of Kansai Medical University (approval number: 2024045)
Consent for publication
All participants provided written informed consent for participation and publication before enrolment.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was conducted with funding from the endowed chair of Sunwels Co., Ltd
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Wataru Matsushita and Koichi Nagaki are full-time employees of Sunwels Co., Ltd Makio Takahashi (Department of Neurodegenerative Disorders, Kansai Medical University) is a faculty member of an endowed chair supported by Sunwels; this support is provided to the institution and is not directed to this specific study or to any author personally. This study was conducted with funding from the endowed chair of Sunwels Co., Ltd; however, all scientific decisions regarding study design, data collection and management, statistical analysis, interpretation of results, manuscript preparation, and the decision to submit were independent of the company's business decisions. The analyses were performed by authors affiliated with an independent institution with no financial or personnel ties to Sunwels, and the de-identified analysis dataset was managed at that institution. Sunwels employees had no authority to unilaterally alter the data or analyses. Outcome assessors were blinded to group allocation. The remaining authors declare no competing interests.
Data availability
Due to privacy and ethical concerns, the data are not publicly available but can be obtained from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
