Abstract
Background
Freezing of gait (FOG) in Parkinson's disease (PD) is a major cause of disability and falls and often responds incompletely to conventional therapy. Rehabilitative interventions including cognitive strategies and sensory cueing are efficacious, but difficulties in learning impair executing these strategies. Transcranial direct current stimulation (tDCS) enhances motor task learning and might enhance the efficacy of rehabilitative interventions.
Objective
We assessed whether tDCS can enhance the efficacy of physiotherapy for FOG in PD.
Methods
In a randomized, double-blind, controlled study, anodal tDCS (pre-/motor cortex) or sham-tDCS were delivered combined with a standardized rehabilitative intervention in 24 PD patients with FOG for eight sessions within four weeks. Clinical assessment included walking a Parcourse, timed gait tests, FOG questionary, and clinical scales at baseline, across the interventions with a follow-up 3-months after the last intervention.
Results
Nineteen PD patients with FOG completed the study. TDCS combined with physiotherapy reduced FOG, but not more than physiotherapy alone
Conclusion
Combining tDCS with physiotherapy did not enhance its efficacy in reducing FOG.
Plain Language Summary
Freezing of gait (FOG) in Parkinson's disease (PD) is a major cause of disability and falls and often responds incompletely to conventional therapy. Rehabilitative interventions consisting of learning cognitive and motor strategies with sensory cueing to prevent and overcome FOG are the most efficacious, but difficulties in learning and executing these strategies are the main cause of failure.
Transcranial direct current stimulation (tDCS), a non-invasive brain stimulation (NIBS) technique enhances learning in PD patients and might enhance the efficacy of rehabilitative interventions.
The study found that NIBS combined with physiotherapy reduced FOG, but not more than physiotherapy alone. We conclude that combining NIBS with physiotherapy did not enhance its efficacy in reducing FOG.
Highlights
TDCS does not increase the efficacy of physiotherapy for FOG and gait disorders in PD. Focused physiotherapy improves FOG and gait in PD A real-life parcourse may be superior in evaluating FOG than a standard evaluation.
Introduction
Freezing of gait (FOG) is a major cause of falls, immobility, and disability in Parkinson's disease (PD) and has a major impact on patients’ quality of life. The pathogenesis remains incompletely understood. The current state of knowledge suggests a disorder of a widespread network, including the frontal lobe, basal ganglia, and locomotor pattern generators in the upper brainstem, which causes FOG. 1
Therapeutic approaches remain limited. Beyond optimizing conventional medication for maintaining a persistent on-condition to improve the dopamine-responsive FOG, there is no strong evidence for a specific medication to treat dopamine-unresponsive FOG. 2 There is a rationale for using MAO-B inhibitors, L-Threo-DOPS, amantadine, antidepressant drugs, acetylcholine-esterase inhibitors, methylphenidate, 3 and pallidal (GPi), sub-thalamic (STN) and pedunculo-pontine nucleus (PPN)-deep brain stimulation (DBS) 4 –, but the scientific evidence remains weak. 5
Currently, the best treatment option is physiotherapy, combining training of cognitive strategies, cueing, and motor learning, including movement coordination and postural strategies.6–9 However, cognitive impairment with difficulties in learning and executing these strategies challenges rehabilitative interventions. The fronto-executive dysfunction –in particular– is associated with FOG 10 –and could explain the nature of conditions precipitating FOG when increased attention, capacity to integrate competing inputs, and cognitive flexibility in adapting motor programs are required.11–13 Transcranial direct current stimulation (tDCS) –a noninvasive brain stimulation method– may offer a promising approach. The tDCS modulates cortical excitability, promotes motor learning and consolidation, and may enhance long-term retention.14,15 The tDCS enhances motor learning and improves gait in PD.2,16,17 Combined with rehabilitative interventions, tDCS promotes motor recovery in chronic stroke patients 18 and could offer a novel approach to treating FOG in PD. In addition, tDCS is considered safe,19–21 well-tolerated, rarely causing local discomfort, 22 and easily applicable.
In this RCT, we investigate whether anodal tDCS over the premotor and motor cortex could enhance the beneficial effects of the state-of-the-art rehabilitative intervention for treating FOG in PD.
Methods
Study population
We enrolled men and women aged between 30 and 80 years with a Dopamine-responsive PD, Hoehn and Yahr (HY) stage of 2 to 4 during their “off” periods, who experienced FOG with an MDS-UPDRS I FOG score of ≥ 2. They had to be on an optimal conventional PD medication with a levodopa equivalent dosage equal to or more than 300 milligrams daily for more than one month before screening and considered candidates for a rehabilitative intervention to treat FOG at the discretion of the treating physician.
We excluded patients with any of the following conditions: significant concurrent medical or psychiatric diseases; a history of seizures or epilepsy; dementia or any other neurodegenerative disease besides PD; prior pallidotomy or the presence of implanted devices for DBS; pregnancy; surgically or traumatically implanted foreign bodies, such as medical pumps, hearing aids, metal plates in the skull, or metal implants in the skull or eyes, excluding dental appliances or fillings. Further exclusion criteria included conditions that would cause undue risk or stress, such as a tendency to fall, excessive fatigue, general frailty, or excessive apprehensiveness; significant postural instability leading to daily falls; inability to walk a set course, or inability to walk 10 meters; significant cognitive dysfunction as determined by a Montreal Cognitive Assessment (MoCA) score of less than 20, or if the patient was mentally impaired and could not provide their consent. We excluded patients with other co-morbid conditions contributing to gait dysfunction, clinically significant hallucinations, or had physiotherapy for FOG within the six months before screening.
We had planned testing for excluding pregnancy before starting the tDCS sessions, but no participant was considered to be of childbearing potential. Participants were recruited from the Policlinique du Service de Neurologie, CHUV, Lausanne, Switzerland. All patients provided written informed consent. The study had been approved by the local ethics committee and registered online in the Clinical Trials database (https://clinicaltrials.gov/ NCT02205216) and the Swiss National Portal (SNCTP). Participants did not receive any monetary or other forms of compensation.
The sample size calculation
We calculated the sample size based on the effect size of tDCS on motor learning, as reported in Benninger et al., 2010. 16 A minimum sample size of 16 patients was considered sufficient for each group, with a relatively conservative power at 95% (alpha 5%; two-sided test). However, the COVID-19 pandemic put the study on hold. We completed a futility analysis after assessing 19 patients, which fulfills the minimum requirement of 9 patients for each group based on a power analysis (alpha 5%; power 80%) with a priori very small effect size (Cohen's d = 0,1).
Study design (Supplementary 1)
We performed a double-blinded, parallel, randomized, sham-controlled, single-center study. Twenty-four (Supplementary 1) tDCS naive patients underwent eight sessions of active or sham tDCS (2 sessions per week). A computer-generated-block-allocation-schedule randomization procedure assigned the patients to a real or a sham tDCS treatment group (13 patients in the tDCS + physiotherapy group and 11 patients in the sham + physiotherapy group). In the active group, tDCS was applied for 20 min at 2 mA with one anode located over the premotor and motor cortex and two cathodes located bilaterally over the mastoids. In the sham group, a direct current was applied for one minute at one mA; anode and cathode were located one cm apart –over the forehead– causing a short circuit through the skin with a brief tingling sensation, but without stimulating the brain. 23 All participants received physiotherapy, simultaneously with the real or sham tDCS. The physiotherapists followed a standardized physiotherapy program 9 for 45 min containing: 1) cognitive training (mental counting and obstacle; attentional strategies, sensory cueing), 2) transfer training during everyday life activities, and 3) motor training including coordination and postural control. Each week, two sessions were applied, one focused on gait and the other on balance (Supplementary 3). The physiotherapist and the physician assessing the outcome measures were blinded, while the tDCS was applied by an unblinded rater.
Outcome measures
We assessed patients at baseline, immediately after, one month, and three months after the last intervention. For the assessment of FOG, we designed an appropriate “Parcourse” to provoke FOG 8 (Supplementary 2). The Parcourse included real-life situations triggering FOG, such as narrow, closed and underexposed spaces, double tasks, and anticipating gait obstacles. The number of FOG episodes provoked during the Parcourse and the time taken to perform the Parcourse –at one-month compared to baseline– was the primary outcome.
Secondary outcomes were the amount of FOG episodes provoked during the Parcourse, the time taken to perform the Parcourse, the 10-meter walk test (10MWT); the Timed Up and Go test (TUG); the New Freezing of Gait-Questionnaire (FOG-Q) 24 ; the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS); the 39-Item Parkinson's Disease Questionnaire (PDQ-39); and the Beck Depression Inventory (BDI), assessed on all the time points. During the TUG 25 and the 10 MWT, 16 we assessed the number of FOG episodes, the number of steps, and the time to complete the trial. During each intervention, the patients walked the Parcourse five times, but we recorded only the last two trials for the assessment.
We kept the medication state, dosage, and daytime constant; we also asked the participants to walk at a comfortable speed over the entire study.
Safety
For safety reasons, we assessed the first ten participants before and after the first intervention in the standardized walking the Parcourse; the 10MWT; the TUG; the MDS-UPDRS; and the MoCA. In addition, we asked the first ten participants to write down a diary accounting for the number of falls during the first four weeks. If a substantial impairment had been shown in gait or cognitive function and more than three patients had to be excluded, the study would have been halted.
Statistical analysis
Statistical analysis was performed using RStudio (v 1.2.1335, RStudio, Inc.).
Baseline differences in patients’ characteristics between both groups were tested with a Wilcoxon Rank-Sum test 26 and Fisher's exact test. 27
For safety reasons, we tested the effect of tDCS directly after the first intervention compared to the baseline for the first ten subjects using a Wilcoxon Rank-Sum test.
In order to analyze the primary outcome, we performed a mixed ANOVA and paired t-tests in order to compare baseline and one-month follow-up values for each group, assessing the combined effect of tDCS and physiotherapy. To analyze secondary outcomes, we performed a mixed ANOVA and paired t-tests to investigate the effect of combined physiotherapy + tDCS and tDCS alone on different secondary outcomes. We computed effect size estimates with 95% confidence intervals; when possible, we calculated standardized effect sizes as Cohen's d. 28 In addition, we computed p-values. We used residual plots and q-q plots to check the independence of observations, a non-explained trend in the residuals, linearity, normality, equality of variance, and outliers. Significance level was set at an alpha = 0.05; power values were calculated a priori to account for the type-2 error.
Results
Twenty-four patients were enrolled and nineteen completed the study. Four of the five patients who withdrew received active tDCS: four withdrew for personal reasons (n = 2), fatigue (n = 1) or orthopedic issues (n = 1), and the last patient could not terminate the study due to Covid restrictions (Figure 1). There were no significant differences in demographic and clinical characteristics between both groups (Table 1), indicating well-balanced randomization.
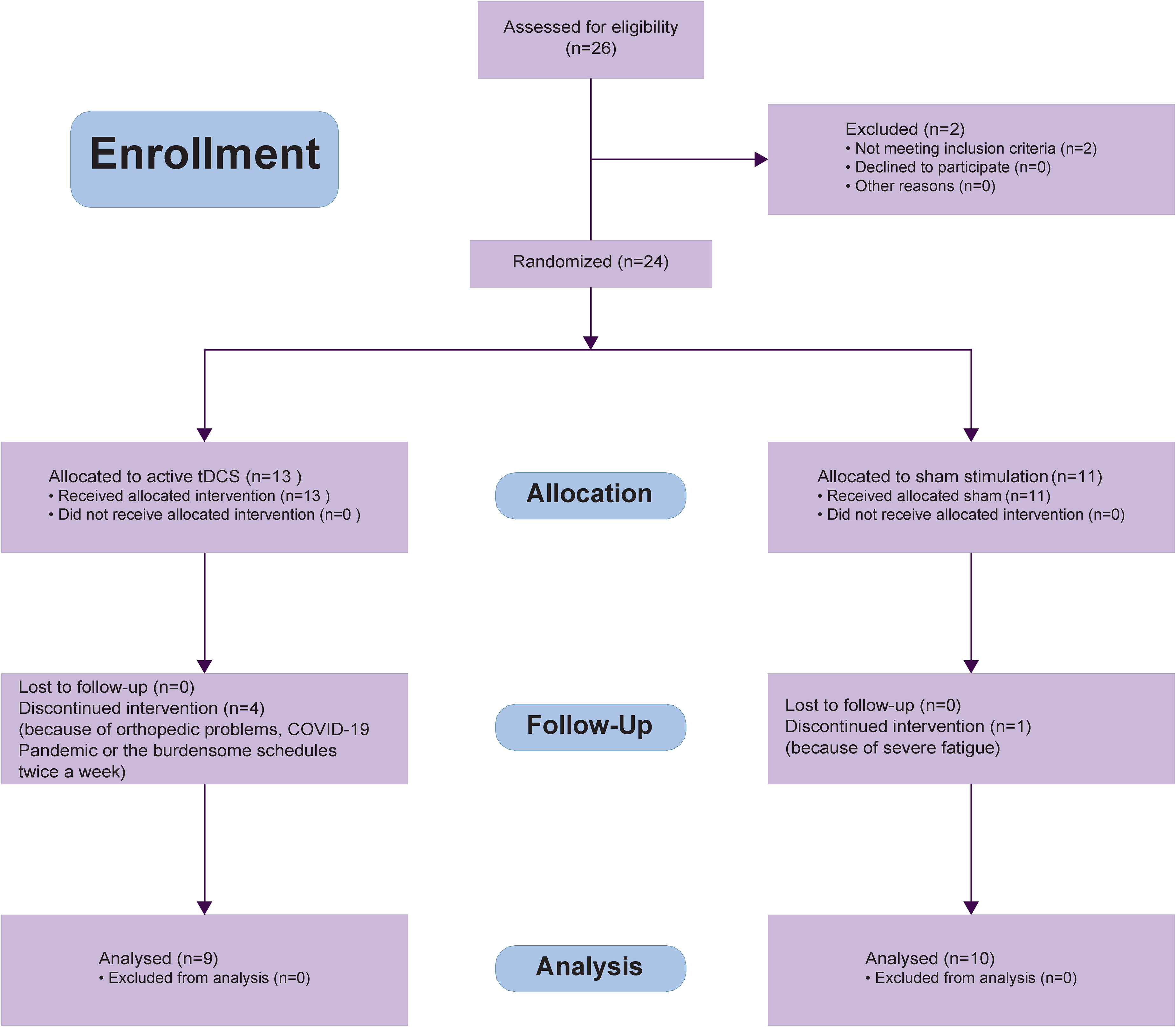
Consort flow diagram.
Demographic and clinical findings in patients with Parkinson's disease (PD) receiving tDCS (n = 9) or sham (n = 10). (Mean ± 95% CI or counts + percentages are presented; significant results are highlighted in bold).
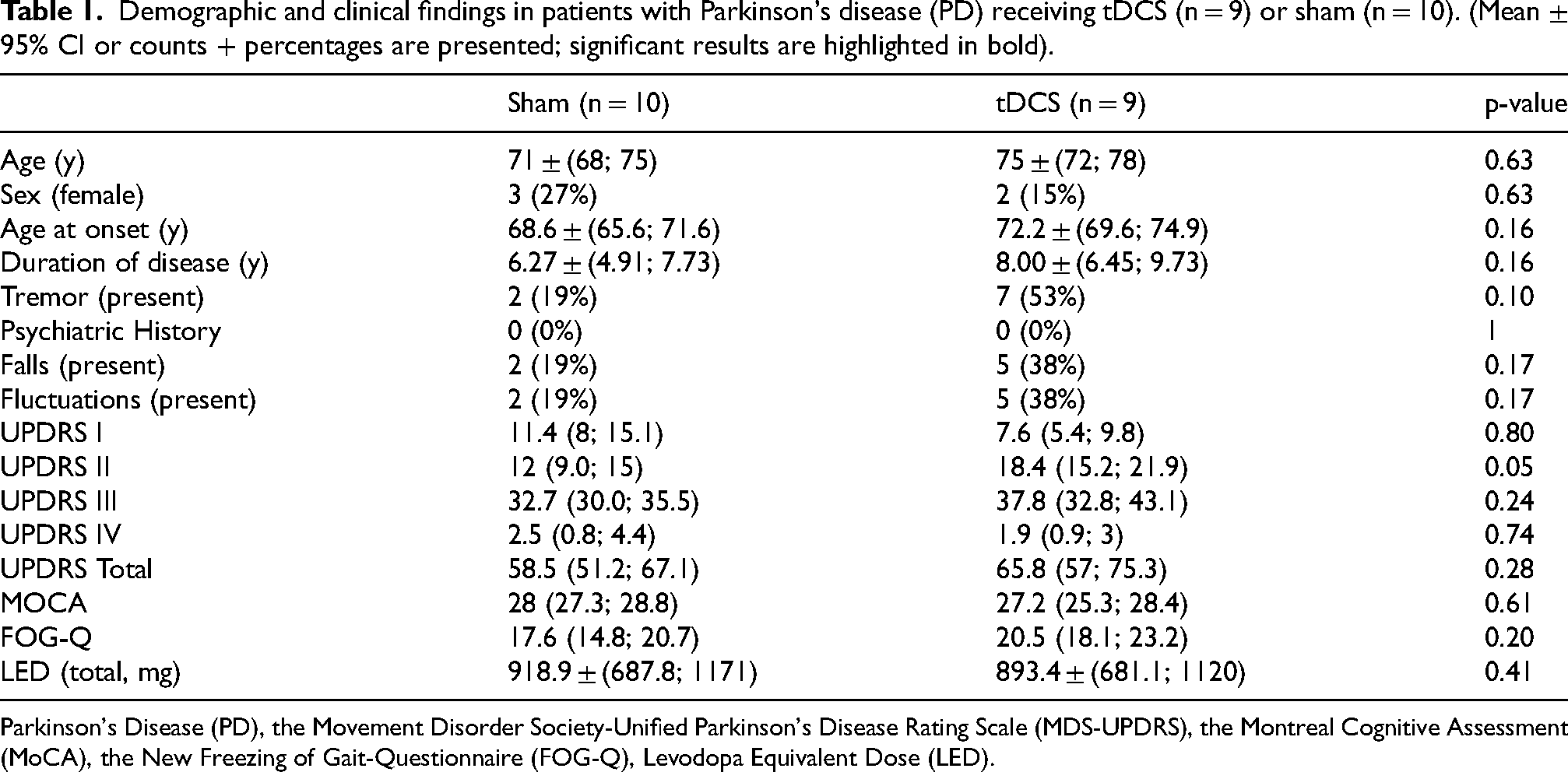
Parkinson's Disease (PD), the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS), the Montreal Cognitive Assessment (MoCA), the New Freezing of Gait-Questionnaire (FOG-Q), Levodopa Equivalent Dose (LED).
Safety testing
We observed no clinical signs of adverse events during or after the first Intervention. We found no worsening after the first intervention in parcourse duration (CI −1.85,0.88; p = 0.55), parcourse FOG (CI −1.96,0.6; p = 0.22), 10 MWT Duration (CI −170, 1.13; p = 0.75), 10 MWT Step (CI −1.46, 1.14; p = 0.61), 10 MWT FOG (CI −1.17, 1.15; p = 1), TUG duration (CI −1.71, 1.02; p = 0.40), TUG step (CI −1.57, 0.86; p = 0.42), TUG FOG (CI −0.88; 1.56; p = 0.42), UPDRS I-IV (CI −1.23,1.47, p = 0.83; CI −1.14,1.68, p = 0.75; CI −1.65,1.12, p = 0.67; CI −0.8, 1.88, p = 0.37), UPDRS total (CI −0.83, 1.88; p = 0.37) and MOCA (CI −1.07, 1.71; p = 0.67).
Primary outcomes
The improvement in the duration to perform the Parcourse from baseline to one-month follow-up (Table 2) was similar between the active tDCS + physiotherapy group and the sham stimulation + physiotherapy group (ANOVA: treatment p = 0.238, interaction p = 0.467). Both groups performed the Parcourse significantly faster at one-month follow-up compared to baseline (p < 0.0001; Cohen's d 1.37 CI 1.02, 2.12).
Primary outcome measures.
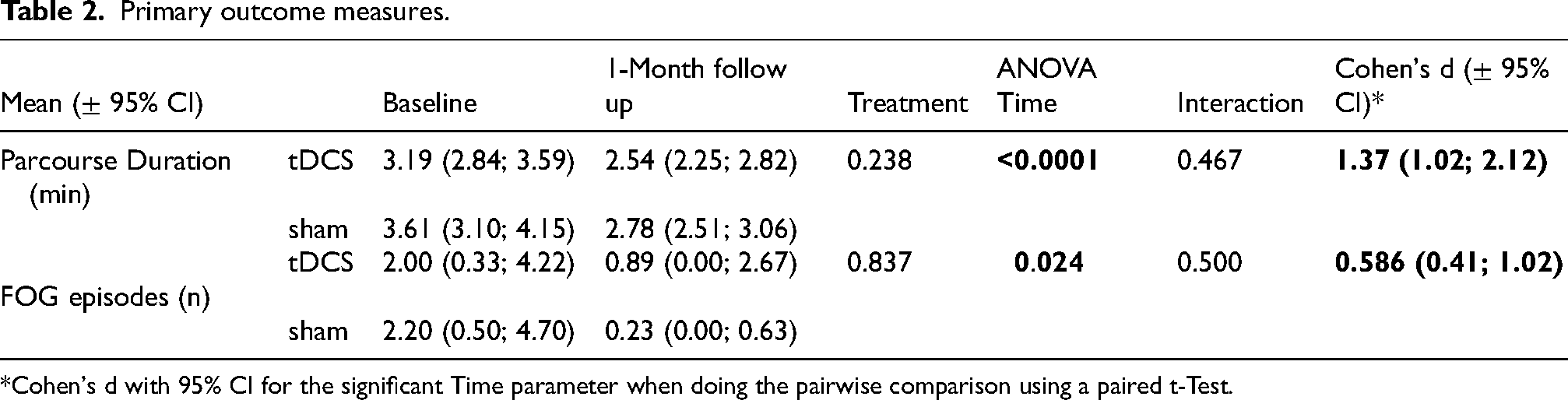
*Cohen's d with 95% CI for the significant Time parameter when doing the pairwise comparison using a paired t-Test.
The improvement in FOG episodes while performing the Parcourse from baseline to one-month follow-up was similar between the active tDCS + physiotherapy group and the sham stimulation + physiotherapy group (ANOVA: treatment p = 0.837; interaction p = 0.500). Both groups had a significantly decreased number of FOG episodes at one-month follow-up (p < 0.024; Cohen's d 0.586 CI 0.41, 1.02).
Secondary outcomes
The Parcourse duration and number of FOG episodes decreased for both groups over all sessions from baseline to follow-up (p < 0.0001 respectively p = 0.015). However, the combination of tDCS + physiotherapy had no additional benefit in improving the duration to perform the Parcourse (ANOVA: treatment p = 0.386; interaction p = 0.907 respectively treatment p = 0.693; interaction p = 0.769) (Figure 2). Patients were faster when performing the 10 MWT after Physiotherapy sessions (p = 0.004), but tDCS did not enhance the beneficial effect of Physiotherapy (ANOVA: treatment p = 0.476; interaction p = 0.259). Neither Physiotherapy nor tDCS improved the number of steps while performing the 10 MWT (ANOVA: treatment p = 0.508; time p = 0.144; interaction p = 0.145). The TUG test shortened with Physiotherapy (p < 0.0001), but there was no additional effect of tDCS (ANOVA: treatment p = 0.308; interaction p = 0.607). Physiotherapy decreased the number of steps (p < 0.0001), but tDCS did not have an additional effect on the number of steps while performing the TUG (ANOVA: treatment p = 0.761; interaction 0.784). Physiotherapy reduced FOG episodes while performing the TUG (p = 0.047), but tDCS had no additional effect (ANOVA: treatment p = 0.652; interaction p = 0.546).
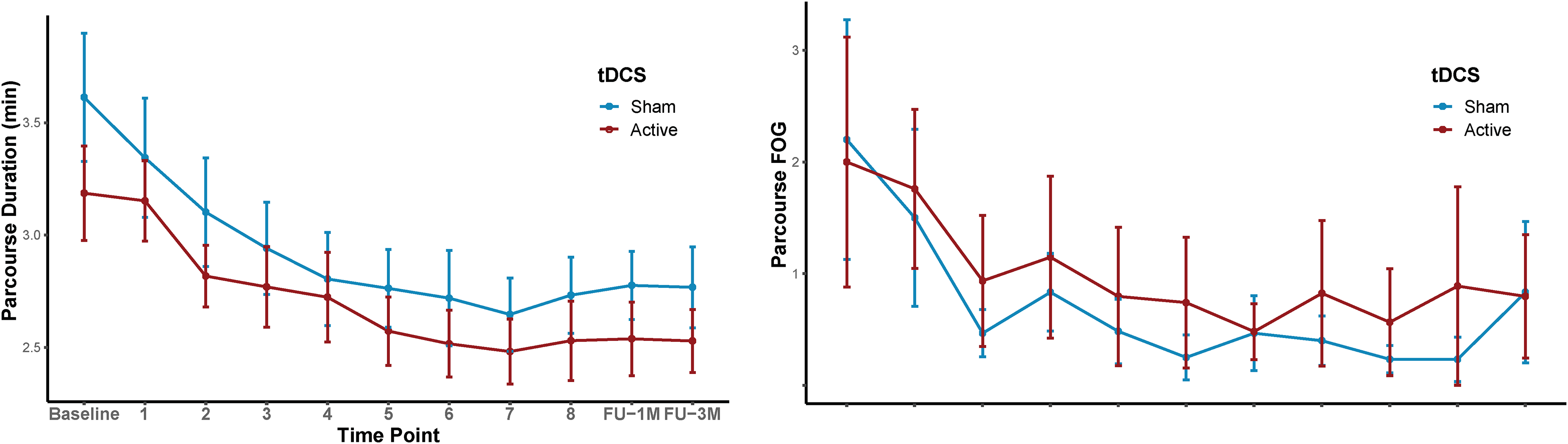
There was no effect of physiotherapy or tDCS on the UPDRS I (ANOVA: treatment p = 0.515; time = 0.211; interaction p = 0.832), UPDRS II (ANOVA: treatment p = 0.121; time = 0.162; interaction p = 0.858), UPDRS III (ANOVA: treatment p = 0.069; time = 0.289; interaction p = 0.660), UPDRS IV (ANOVA: treatment p = 0.086; time = 0.925; interaction p = 0.962), total UPDRS (ANOVA: treatment p = 0.402; time = 0.205; interaction p = 0.681), PDQ-39 (ANOVA: treatment p = 0.879; time = 0.108; interaction p = 0.537), and BDI (ANOVA: treatment p = 0.964; time = 0.389; interaction p = 0.320).
Physiotherapy improved the FOG questionnaire (p = 0.028). However, tDCS did not have an additional effect on the FOG questionnaire (ANOVA: treatment p = 0.243; interaction p = 0.200).
Our standardized parcourse was significantly superior to the 10 MWT (p < 0.0001; estimate 3.73, 95% CI 2.64, 5.03) and the TUG test (p < 0.0001; estimate 1.80, 95% CI 1.12, 2.50) in provoking FOG episodes (Table 3).
Secondary outcome measures.
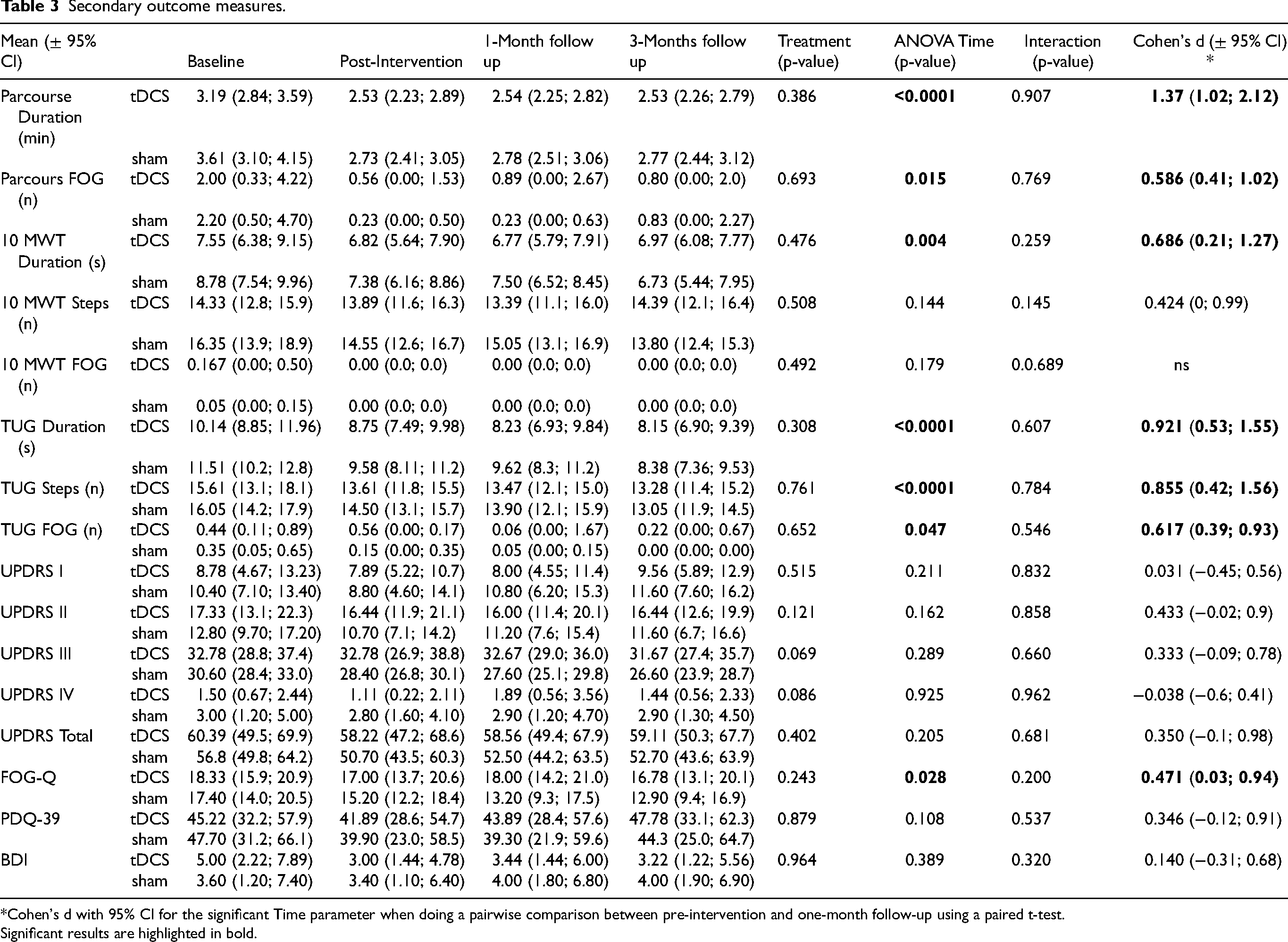
*Cohen's d with 95% CI for the significant Time parameter when doing a pairwise comparison between pre-intervention and one-month follow-up using a paired t-test.
Significant results are highlighted in bold.
Discussion
This double-blind, randomized, sham-controlled study investigated whether tDCS could enhance the efficacy of gait-focused physiotherapy for the treatment of FOG in patients with Parkinson's disease (PD). Our findings do not suggest an additional effect of tDCS, but confirm the efficacy of physiotherapy in improving FOG and gait in PD.
The literature provides robust evidence supporting the efficacy of physiotherapy for treating PD in maintaining motor and functional skills and slowing the cognitive decline.2,8,29,30 Physiotherapy with a focus on gait, including coordination training, postural control, and dynamic balance, improves walking performance and quality. Combining high-intensity gait training with learning cognitive, sensory, and motor strategies reduces FOG occurrence.2,8,30 Our findings are consistent with the current state of evidence, and the beneficial effects of physiotherapy on FOG and gait last for at least three months after intervention. Physiotherapy, therefore, remains the standard care for treating FOG in PD, while our study fails to show a beneficial effect of combining tDCS with physiotherapy.
The rationale for tDCS arises from the evidence for its potential in enhancing synaptic plasticity as shown in numerous studies. The tDCS promotes motor learning in healthy individuals. The simultaneous tDCS during a finger sequence learning task leads to a significantly better performance compared to the sham-stimulated group; this indicates enhanced plasticity. 15 This tDCS efficacy remains preserved in patients with PD. In a prior randomized controlled trial (RCT) with eight sessions of anodal tDCS over the motor and prefrontal cortices, we found a better performance of complex hand and arm movements, which persisted for three months. Since bradykinesia did not improve, this underlines the efficacy of tDCS in directly promoting motor learning, probably by enhancing plasticity. 16 This finding provided the rationale for the current study.
There are few randomized, double-blind studies investigating the efficacy of tDCS for treating FOG in PD. In a cross-over study, Valentino et al. (2014) 17 found a beneficial effect of five sessions of anodal tDCS of M1 alone without concurrent rehabilitative intervention for treating FOG. This points to a direct effect of tDCS on FOG. Though the precise mechanism remains yet unknown, the persistence of effects for a month implies the involvement of neural plasticity. This direct effect of tDCS is supported by another study by Yotnuengnit et al. (2018), 31 which compared the efficacy of a monotherapy consisting of either tDCS or physiotherapy without tDCS for treating gait in PD patients and found an improvement with either intervention. Therefore, we hypothesized a potential complementary effect of tDCS when combined with physiotherapy. However, our findings do not support such an effect. Hence, tDCS may have a direct beneficial effect on gait in PD, but no superior or complementary effect could be observed when combined with physiotherapy. The physiotherapy's beneficial effect on gait probably masked the tDCS's beneficial effect. This corresponds to the “ceiling effect”, which could explain the lack of an additional effect when combining physiotherapy and tDCS in our study. The tDCS might have improved motor learning, which we did not evaluate distinctly.
Even a single session of tDCS may be beneficial. In a study by Dagan and colleagues (2018), 32 a single session of combined anodal tDCS over both M1 and DLPFC successfully reduced FOG occurrence in PD patients, but this effect was not seen when stimulating M1 alone or in the sham protocol. Therefore, the choice of the tDCS-stimulated region may matter. This wider multi-target stimulation likely enhances more dopaminergic circuits 33 and may also attenuate the observed decoupling between the basal ganglia and the cognitive control networks during FOG episodes, in which the DLPFC may play a pivotal role. 34 The combined anodal tDCS stimulation of M1 and DLPFC may enhance executive function and influence motor control. 35 In their study, Dagan and colleagues (2018) 32 measured the effect immediately post-stimulation, but whether a single session induces a sustained effect has yet to be investigated. We found a cumulative improvement across the eight intervention sessions of combined physiotherapy with anodal or sham-tDCS. This suggests repeated sessions are more effective as plasticity may require repeated activation for a sustained beneficial effect which is relevant for clinical practice. In our previous study, we also observed increasingly better performance in the complex hand and arm movements across the sessions, even though they were only part of the testing and not trained. 16 We conclude that conducting multiple intervention sessions might be crucial for achieving lasting beneficial outcomes, which result from neuronal changes driven by mechanisms of plasticity.
The study has limitations. The study population is relatively small, as we had to interrupt the study during the COVID-19 pandemic. Yet, the futility analysis fulfilled the minimum requirement of 9 patients for each group based on a power analysis (alpha 5%; power 80%) with a priori very small effect size (Cohen's d = 0,1), pointing to the futility of this intervention. For this reason, we also opted to terminate the study for the sake of limited resources. This class II study can conclude the absence of an additional effect of tDCS combined with physiotherapy, while the efficacy of physiotherapy alone is confirmed. In addition, we could confirm the safety of tDCS. 36
This study demonstrates the feasibility of an intense focused physiotherapy consisting of two sessions a week, even in patients with more advanced stages of PD. Yet, two participants withdrew prematurely because of a schedule that finally turned out to be too “intense” for them, and others may have chosen not to participate. Furthermore, the prevalence of FOG rises and becomes a principal cause of disability along with disease progression. We had to exclude patients with more advanced disease for safety reasons and our exclusion criteria may have been too strict. However, we believe our study population is representative of a large part of our PD patients, and our findings might be extrapolated to more severely affected patients.
The blinding of participants in interventional trials may be a challenge. We have previously shown that the blinding could be maintained when applying the sham stimulation as previously described. 16
For a reliable evaluation of FOG, we faced the challenge of simulating the everyday conditions prone to trigger FOG, which may be notoriously absent in laboratory conditions. We defined an innovative and standardized walking Parcourse within the hospital setting, including doors, narrow passages, turns within a limited space, spaces of contrasting illuminations, and sliding doors. These Parcourse features correspond to real-life situations and are known to trigger freezing. Some gait maneuvers like turning are particularly suited to provoke freezing 8 and to uncover the aging process or disease states.37,38 Our Parcourse differs from a conventional obstacle course, which does not correspond to a natural everyday environment or experience. This strategy was revealed to be superior to a conventional obstacle course as all patients experienced FOG in the Parcourse. In contrast, we observed few FOG episodes in standard test conditions, including the 10-meter walk and TUG tests, which were secondary outcome measures. This observation proves the validity of the Parcourse, allowing the reliable evaluation of FOG.
Conclusions
This study provides class II evidence that pre-/motor anodal tDCS does not enhance the efficacy of focused physiotherapy for treating FOG and gait impairment in Parkinson's disease. Our study reconfirms the clinical efficacy of PIGD-focused physiotherapy, including learning mental strategies, training with sensory cueing, and transfer training during everyday life activities, and that these effects persist for at least three months after the intervention. Our innovative Parcourse proved its validity by recreating real-life conditions and triggering FOG in all participants, allowing reliable assessment of FOG in a clinical setting.
Footnotes
Acknowledgments
The Parkinson Suisse / Schweiz (Switzerland) funded this study. The study sponsors were not involved in the study design, in the collection, analysis, and interpretation of data, in the writing of the manuscript, and in the decision to submit the manuscript for publication. We thank Dr Candia for his valuable help and discussions during the preparation of the manuscript.
Funding
The study has been in part funded by Parkinson Switzerland.
Conflict of interest
None of the authors has any financial and personal relationships that could improperly influence this work.
Author roles
1. Research project: A. Conception, B. Organization, C. Execution.
2. Statistical Analysis: A. Design, B. Execution, C. Review and Critique.
3. Manuscript Preparation: A. Writing of the first draft, B. Review and Critique.
JM: 1B, 1C, 2A, 2B, 2C, 3A, 3B and should be considered first author
EB: 1B, 1C, 3B
DR: 1A, 1C, 2C, 3B
SV: 1C, 2C, 3B
KK: 1C, 2C, 3B
DHB: 1A, 1B, 1C, 2A, 2B, 2C, 3A, 3B and should be considered last author.
Supplemental material
Supplemental material for this article is available online.
