Abstract
It is unclear whether Alzheimer disease pathology drives cognitive decline in Parkinson disease (PD) patients carrying GBA1 variants. We evaluated levels of serum phosphorylated tau 217 (p-tau217) in samples from 29 GBA1-PD, 32 idiopathic PD, 20 non-manifesting GBA1 variant carriers (GBA1-NMC) and 31 healthy controls. No differences were detected between PD groups. GBA1-NMCs showed higher levels than healthy controls, which correlated to worse cognition and subthreshold parkinsonism. Serum p-tau217 is not a marker of cognitive decline in GBA1-PD. Whether p-tau217 levels in GBA1-NMCs can predict conversion to PD or are a marker of cognitive decline, irrespective of PD, remains unknown.
Plain language title
Could a blood test help predict Parkinson's disease and cognitive decline in people with a GBA1 gene change?
Plain language summaries
Why was the study done? Having a change in a gene called GBA1 increases the risk of developing Parkinson's disease (PD). However, most people with a GBA1 change will not develop PD over their life, and we don’t know why only some people do. When people with a GBA1 gene change develop PD, they may have more severe cognitive problems compared to people with PD but no GBA1 change, and once again we don’t know why this happens. One idea is that changes linked to Alzheimer's disease might play a role.
What did the researchers do? Researchers looked at a blood marker called p-tau217, which is higher in Alzheimer's disease. They tested blood samples from four groups: 1) people with PD and a GBA1 gene change, 2) people with PD but no GBA1 change, 3) people with a GBA1 change but no PD symptoms, and 4) healthy people without PD or GBA1 changes.
What did the researchers find? The study found no difference in p-tau217 levels between the two PD groups. This means that p-tau217 does not seem to explain cognitive problems in people with both PD and GBA1 changes. However, people who had a GBA1 change but no PD had higher p-tau217 levels, especially those with more severe changes. These higher levels were linked to slightly worse cognitive function and very mild movement changes.
What do the findings mean? We don’t yet know what this means. It could be an early warning sign for PD, or it might just be part of having cognitive problems unrelated to PD. More research is needed to find out if this blood test could help predict who will develop PD in the future.
Background
GBA1 genetic variants are the commonest genetic risk factor for Parkinson disease (PD). Approximately 20% of GBA1 variant carriers develop PD. 1 Subtle cognitive alterations have been observed in some carriers, but their predictive value for phenotypic conversion remains unclear. 2 In the manifest phase, GBA1-PD patients, especially individuals carrying severe variants, tend to show a more ‘malignant’ subtype with faster cognitive decline compared to idiopathic PD (iPD).3,4 Developing minimally invasive biomarkers that predict conversion in pre-manifest GBA1 variant carriers and elucidate the pathophysiological pathways that mediate cognitive decline in GBA1-PD is essential for enabling earlier diagnosis and targeted therapeutic strategies.
Cerebrospinal fluid (CSF) biomarkers for Alzheimer's disease (AD) pathology have been previously investigated in manifest GBA1-PD without evidence of significant changes compared to iPD. Comparable or lower CSF levels of total and phosphorylated tau (t-tau and p-tau) were detected in 50 GBA1-PD compared to controls and iPD respectively, despite greater cognitive impairment in GBA1-PD. 5 In a recent cross-sectional study evaluating CSF α-synuclein seeding, amyloid-β1-42 and phosphorylated 181-Tau in a cohort of PD patients, carrying GBA1 variants or not (129 vs 188), none of the GBA1-PD samples showed combined α-synuclein/AD profiles, and only 2% had isolated AD profiles. 6 However, iPD participants with combined α-synuclein/AD profiles developed faster cognitive impairment compared with those with isolated α-synuclein profiles. 6
Blood-based phosphorylated tau at threonine 217 (p-tau217) has the highest discriminative power in differentiating AD from other neurodegenerative disorders. 7 Moreover, p-tau217 best identifies patients with mild cognitive impairment with abnormal Aβ pathology or those who will progress to AD, 8 and may detect AD co-pathology in Lewy Body Disease (LBD) with dementia. 9 Recently, serum p-tau217 was identified as a useful biomarker to predict the development of cognitive impairment (CI) in individuals with isolated REM sleep behaviour disorder (iRBD). 10 Thus, peripheral p-tau217 might offer a simple way to evaluate antemortem whether AD pathology drives cognitive decline in GBA1-PD and/or predicts PD development in non-parkinsonian GBA1 variant carriers.
The aim of this study was to compare serum p-tau217 levels in GBA1-PD and iPD patients, and investigate whether levels of serum p-tau217 in GBA1 variant carriers without any manifesting signs of PD (GBA1-NMC) differed from those in healthy controls (HC), and correlated with prodromal features of PD.
Methods
Participants were recruited via the RAPSODI study (University College London) in the United Kingdom. 11 Full GBA1 gene sequencing was performed on saliva samples, as previously described. 4 Four groups of participants, matched for age, sex, and education levels, were included (GBA1-PD, iPD, GBA1-NMC and HC); the PD groups were also matched for cognitive status and disease duration. GBA1 variants were classified as severe, mild, or risk, as previously described. 12
Participants completed a detailed clinical assessment, evaluating motor and non-motor functions using the following scales: Hoehn & Yahr (H&Y) scale, 13 Movement Disorder Society-Unified Parkinson's Disease Rating Scale MDS-UPDRS (part I-IV), 14 SCales for Outcomes in PArkinson's disease (SCOPA-AUT), 15 REM Sleep Behaviour Disorder Screening Questionnaire (RBDSQ), 16 University of Pennsylvania Smell Identification Test (UPSIT), 17 Beck Depression Inventory (BDI), 18 and Montreal Cognitive Assessment (MOCA). 19 CI was defined as a MOCA score <26/30.20,21 In the GBA1-NMC group, the subthreshold parkinsonism (SP) score was calculated as a MDS-UPDRS-III score, excluding postural and action tremor. 22
Venous blood was collected into BD SST II Advance tubes (REF 367958) at the time of clinical assessment. After gentle inversion (5-6 times), tubes were left upright for at least 30 min to allow clotting. Samples were centrifuged at room temperature (1800 g, 10 min, no brake), and serum was carefully aliquoted (250 µL) into 5-6 cryovials. Serum samples were stored at −80°C, and then analysed on the Simoa HDX platform using the ALZpath p-Tau 217 Advantage PLUS kit according to manufacturer's instructions at the UK Dementia Research Institute at University College London.
Statistical analysis was performed using R version 4.3.2. 23 Study data were collected and managed using REDCap electronic data capture tools. 24 The primary analysis consisted of two-group comparisons among PD groups (GBA1-PD vs iPD) and non-PD groups (GBA1-NMC vs HC). Categorical variables were compared using χ2 test or Fisher's exact test. Continuous variables are presented as means ± SDs, unless otherwise specified. Group demographics and descriptive features were compared using t-test or non-parametric Wilcoxon rank-sum test. Group comparisons for clinical features and serum p-tau217 levels were adjusted for age and sex (and disease duration for PD groups) in linear regression models using clinical scores or p-tau217 levels as outcome. For RBDSQ and MOCA scores, data were categorised using clinically validated thresholds and binary regression was used to determine associations between categorised variables and the grouping variable, with analysis adjusted for the same covariates. Correction of p-values using the Benjamini-Hochberg (BH) procedure was applied to multiple-group comparisons, when groups were stratified according to GBA1 variant severity. Correlation between p-tau217 levels and clinical variables was computed by Spearman rank tests.
Results
Demographics, clinical features and p-tau217 levels are displayed in Table 1. A total of 112 participants were included, of which 29 GBA1-PD, 32 iPD, 20 GBA1-NMC and 31 HC. GBA1-PD and iPD had similar motor and non-motor features. Within the non-PD groups, GBA1-NMC showed higher MDS-UPDRS part I (p = 0.0296, β = 2.49) and total scores (p = 0.00345, β = 5.0) and worse cognitive function (p = 0.0100, β = −1.14).
Clinical and serum p-tau217 characteristics of study cohort.

Legend: data are reported as mean ± SD, unless specified otherwise.
BDI, Beck Depression Inventory; H&Y, Hoehn & Yahr; MDS-UPDRS, Movement Disorder Society (MDS) Unified Parkinson's Disease Rating Scale; MOCA, Montreal Cognitive Assessment; NA, not applicable; ns, not significant; PD, Parkinson disease; SCOPA-AUT, SCales for Outcomes in PArkinson's disease; RBDSQ, REM Sleep Behavior Disorder Screening Questionnaire; UPSIT, University of Pennsylvania Smell Identification Test.
No differences were detected in levels of p-tau217 between GBA1-PD and iPD (Table 1). When levels of p-tau217 were compared across non-PD groups, higher levels were detected in GBA1-NMC compared with HC (p = 0.0187, β = 0.05; Figure 1A). When GBA1-NMC were stratified according to variant severity, higher levels were observed in carriers of severe variants compared with HC (p = 0.0211, β = 0.09), however this did not survive to adjustment for multiple corrections (adj.p = 0.12; Supplementary Figure 1). A trend for lower MOCA scores was observed in severe GBA1-NMC compared to HC, so it is possible that the higher p-tau217 levels might represent an indication of worse cognition, irrespective of GBA1 status or PD risk. No differences in p-tau217 levels were observed when PD patients were stratified according to GBA1 variant type. Significantly increased levels of p-tau217 were detected in individuals with CI compared to individuals who were cognitively normal (CN) in both comparisons (non-PD groups: p = 0.0026, β = 0.12; PD groups: p = 0.0028, β = 0.09; Figure 1B).
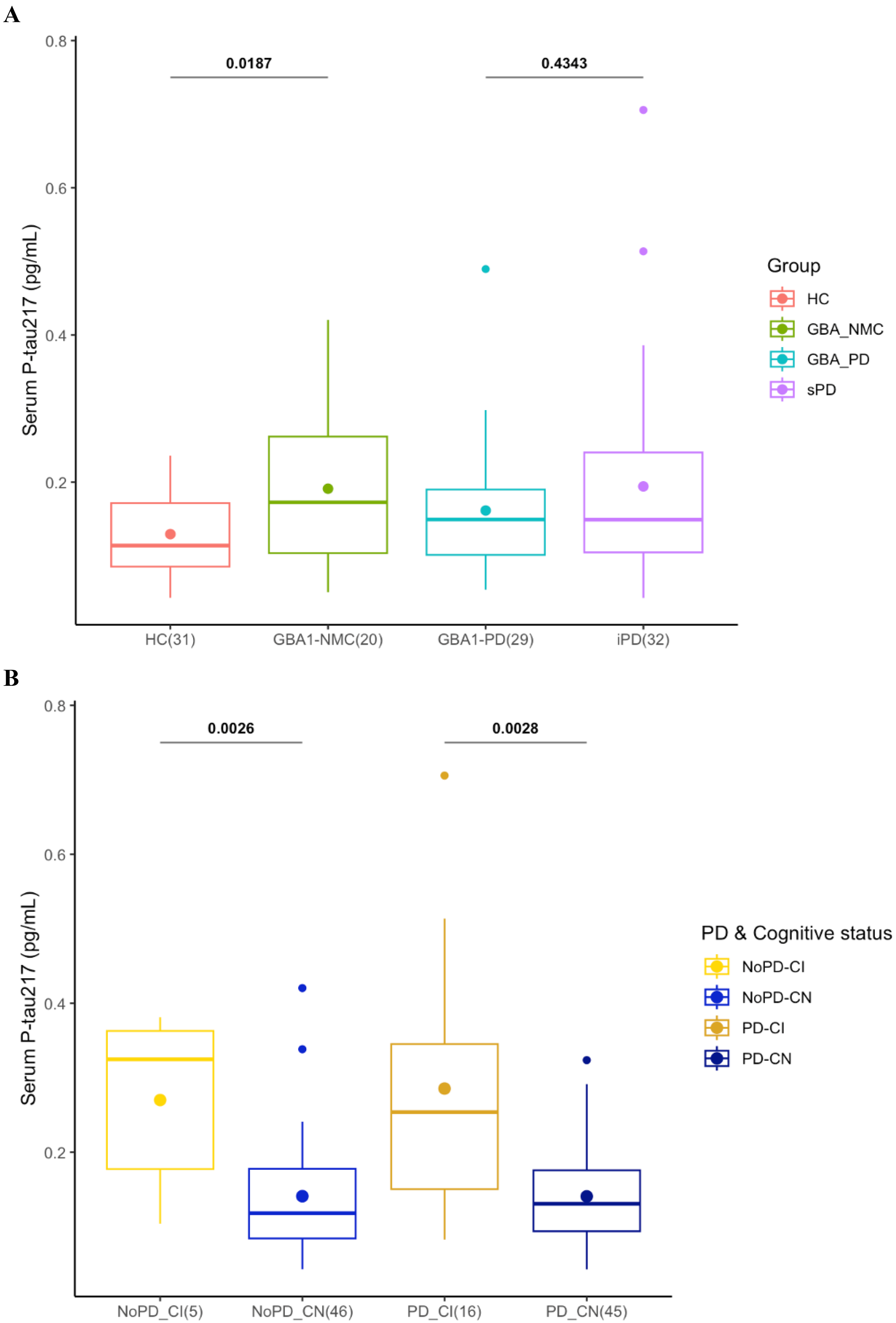
Levels of p-tau217 in the cohort. Mean levels of p-tau 217 in the four groups (HC, GBA1-NMC, GBA1-PD, and iPD) (A), and in individuals with cognitive impairment (CI; MOCA score <26/30) and individuals who were cognitively normal (CN; MOCA score ≥26/30), without PD (noPD) or with PD (B). P values from regression models are reported above the corresponding comparisons.
Within PD (Supplementary Figure 2A), levels of p-tau217 were negatively correlated with cognitive function (MOCA: p = 0.03, r = −0.28), positively correlated with age (p = 0.0006, r = 0.43) and MDS-UPDRS total scores (p = 0.03, r = 0.27). Within non-PD (Supplementary Figure 2B), levels of p-tau217 were negatively correlated with cognition (MOCA: p = 0.01, r = −0.34), positively correlated with age (p = 0.04, r = 0.29) and MDS-UPDRS part III and total scores (p = 0.03, r = 0.30, and p = 0.03, r = 0.31, respectively). Within the GBA1-NMC group specifically, levels of p-tau217 levels were negatively correlated with cognition (MOCA: p = 0.02, r = −0.53; Supplementary Figure 3A) and positively correlated with MDS-UPDRS part III and total scores (p = 0.01, r = 0.54, and p = 0.005, r = 0.60, respectively), and with SP scores (p = 0.03, r = 0.5; Supplementary Figure 3B).
Conclusions
The results from this study corroborate and expand previous findings.
We showed that AD pathology, as determined by serum p-tau217, is not significantly higher in GBA1-PD than iPD. This is consistent with data from a large postmortem study on 943 LBD cases (of which ∼43% with likely clinical Dementia with Lewy Bodies) reporting a higher prevalence of GBA1 variants in cases with greater Lewy bodies (LB) pathology and low AD pathology, particularly in those carrying the severe p.L483P variant. 25 We also showed that, irrespective of GBA1 status, higher p-tau217 levels are associated with worse cognition in PD. This aligns with results from idiopathic LBD postmortem cases, where combined LB and AD pathology predicted faster cognitive decline in patients with idiopathic LBD.26,27 Altogether, these and our own findings support alternative/additional mechanisms driving the higher burden of cognitive decline in GBA1-PD. No accumulation of glucocerebrosidase substrates was detected in GBA1-PD brains when compared to matched iPD, 28 but a more diffuse neocortical LB-type pathology has been identified in different postmortem studies conducted in GBA1-PD brains,25,29 supporting the role for α-synuclein pathology in this process. Indeed, a recent animal model showed that carrying a heterozygous p.L483P GBA1 variant alters hippocampal synaptic structure and function, leading to a pathological burden for cognitive impairment, and that the co-existence of α-synuclein overexpression with pathogenic GBA1 status can accelerate PD-associated cognitive impairment and motor symptoms. 30 Faster seeding kinetics on α-synuclein seed amplification assay was detected in GBA1-PD compared with iPD and predicted cognitive decline in two large cohorts of PD patients, 31 proposing α-synuclein seeding kinetics as a major player in faster cognitive decline in GBA1-PD.
We observed significantly higher p-tau217 levels in individuals with GBA1-PD with CI compared with those with normal cognition, as opposed to previous studies which observed comparable CSF levels in these groups. 5 It is possible that our cohort of GBA1-PD, mainly represented by risk variant carriers, resembled more the clinical and biomarker profile of iPD, whereas cohorts enriched in severe variant carriers, like the one in the CSF study, are characterised by a unique, distinctive biomarker profile. This aligns with subsequent CSF studies showing that GBA1-PD patients with severe variants were 100% CSF α-synuclein positive without AD profiles. 6
Interestingly, we identified positive correlations between serum p-tau217 levels and functional impairment in both PD and non-PD groups, and a positive correlation with subthreshold parkinsonism in GBA1-NMC. Levels of serum p-tau217 also positively correlated with subthreshold parkinsonism in a large group of individuals with iRBD. 10 Data from a large longitudinal study involving 131 newly diagnosed PD patients, showed that elevated CSF p-tau levels and p-tau/t-tau ratios accurately predicted the development of motor complications over time. 32 The exact role of AD co-pathology, especially tauopathy, as a potential predictor of motor impairment in GBA1 variant carriers warrants further investigation in larger cohorts of these individuals.
The strengths of our study include the inclusion of both PD and non-PD groups, matched for demographics and cognitive status, the extensive characterization of the clinical profile of our participants, and the use of peripheral (not CSF) biomarkers. We acknowledge some limitations to our study. First, this is a cross-sectional study, on a relatively small sample size; longitudinal measurements of serum p-tau217 levels, cognitive and motor function are needed to establish any association between blood-based biomarkers and PD risk or cognitive decline in our population. Second, we did not screen for the presence of APOE ε4 allele, or evaluate PET-based measures of tau and amyloid pathology, which would help clarify the role of AD pathology. Third, we could not determine the prevalence of positive AD profiles in our participants because of the lack of reference ranges for serum p-tau217 as opposed to plasma, 33 although it has been shown that the clinical performance in distinguishing AD from normal controls is high with both biofluids, 34 and serum t-tau was found to correlate well with both CSF t-tau and p-tau in a small PD cohort, 35 encouraging the application of serum markers in future, larger studies.
We conclude that higher serum p-tau217 levels predispose to higher risk of developing dementia in PD and healthy individuals, regardless of genetic status, and that the more aggressive and rapid progression to dementia observed in GBA1-PD, especially in severe variant carriers, is driven by distinct and specific pathological mechanisms. This information is crucial when designing clinical trials for disease-modifying therapies in this group.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251413875 - Supplemental material for Serum phosphorylated tau 217 in GBA1 variant carriers with and without Parkinson disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251413875 for Serum phosphorylated tau 217 in GBA1 variant carriers with and without Parkinson disease by Elisa Menozzi, Roxana Mezabrovschi, Aleesa Nazeer, Amelia Anderson, Nadine Loefflad, Amanda J Heslegrave, Elena Veleva, Vlada Drotsevitch, Matthew Gegg and Anthony HV Schapira in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors would like to extend their gratitude to all study participants.
Ethics approval
The study received ethical approval by the London – Queen Square REC: 15/LO/1155. All participants gave their informed consent upon enrolment.
Author contributions
E.M. and A.H.V.S. conceived and designed the study. E.M., A.N., A.A., and N.L. contributed to patient enrolment and data acquisition. E.M., R.M., and A.A. performed the metadata processing and validation. A.J.H, E.V., and V.D. processed and analysed the serum samples. E.M. wrote codes for and performed data analysis. E.M., R.M., M.G., and A.H.V.S. contributed to the interpretation of the results. E.M., R.M. and A.H.V.S. wrote the paper with contribution and editing from all authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
EM, AN, AA, NL, AJH, EV, VD and MG have no conflicts to disclose.
RM is supported by a Royal Free Charity fellowship.
AHVS has provided paid consultancy to Capsida and Axial and is the Chief Investigator of the ambroxol phase III study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author AHVS. The data are not publicly available due to privacy or ethical restrictions. The code used to generate the results of this article can be found at https://doi.org/10.5281/zenodo.16901918.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
