Abstract
Background
Occupational pesticide (OcP) exposure and pathogenic GBA1 variants are established risk factors for Parkinson's disease (PD). However, whether they interact to influence disease onset or severity remains uncertain.
Objective
To determine the prevalence of OcP exposure and other lifestyle risks in relation to GBA1 status and their interactions on age of onset (AOO) and clinical scores in a well-characterized PD cohort.
Methods
We analyzed 505 people with PD (PwP) enrolled in the Ankara Parkinson's Disease Registry (ANPAR). GBA1 variants were identified using next-generation sequencing; benign or uncertain variants were excluded. Structured, face-to-face interviews collected data on history of OcP exposure, history of head trauma, smoking, coffee and tea consumption habits. PwP with and without GBA1 variants were compared using unadjusted and adjusted tests. General linear models assessed gene—environment interactions on AOO, non-motor, and motor scores.
Results
Fifty-two PwP (10.3%) carried pathogenic GBA1 variants. OcP exposure was more common in carriers than non-carriers (36% vs. 22%; adjusted OR 1.98, 95%CI: 1.07–3.67, p = 0.031). No other risk factor differed between groups. Smoking independently delayed AOO, but there were no significant GBA1 × risk/lifestyle factor interactions for AOO or for motor/non-motor scores. Head trauma, coffee, tea, and OcP exposure showed neither main nor interaction effects on severity indices.
Conclusion
OcP exposure is reported more often by individuals carrying pathogenic GBA1 variants, supporting a gene—environment “dual-hit” model. However, OcPs did not modify AOO or disease severity once PD was manifest.
Plain language summary title
Pesticide Exposure is Associated with GBA1 Mutations in Parkinson's Disease
Plain Language Summary
Parkinson's disease is a neurological disorder that mainly affects movement but also other functions. Genetic, environmental and lifestyle factors may contribute to this disease. One of the strongest genetic risk factors is carrying a harmful change (mutation) in the GBA1 gene. For environmental risks, exposure to pesticides also increases the likelihood of Parkinson's disease. However, not everybody having a GBA1 mutation develop disease. This suggests that additional risks may be necessary for the development of disease. In this study, we investigated the effects of several risk factors including pesticide exposure for their association with GBA1 mutations. We also investigated the impact of these factors on age of disease onset and disease severity. To do this, we examined 505 people with Parkinson's disease from the ANkara Parkinson's Disease Registry (ANPAR). All participants completed interviews about their lifestyle and exposures such as pesticide contact, head trauma, smoking, and coffee or tea consumption habits. We then compared people with and without GBA1 variants to see if there were any differences in these factors. We found that one every 10 patients carried a GBA1 mutation. Pesticide exposure was reported more often in patients carrying a GBA1 mutation. However, pesticide exposure did not have a significant effect on the age at which the disease started. Among the other lifestyle factors, only smoking delayed the onset of the disease. Other factors showed no effect on age of disease onset and severity. Our findings support the idea of a dual-hit model, meaning that having both a harmful GBA1 variant and pesticide exposure may increase the risk of Parkinson's disease. This highlights the complex interaction between genetic and environmental mechanisms for the development of Parkinson's disease.
Introduction
The global rise of prevalence in Parkinson's disease (PD) 1 has been linked to environmental influences collectively termed as exposome. 2 Based on this recognition, PD has been defined as an environmental disease. 3 This may be plausible because the pathology starts in the olfactory bulb and dorsal motor nucleus of vagus—structures that connect the brain to the external environment. Within this environment, occupational pesticide (OcP) exposure is a well-documented risk factor for PD, supported by evidence from cell cultures to animal studies, to prospective risk or geographical studies.4–9 Depending on the type or intensity of exposure, OcPs increase the risk as much as two-fold.10–12 However, despite the strong link, OcP exposure is a risk factor rather than an absolute determinant of a future synucleinopathy. Probably, OcPs cause an overt PD pathology only when they disrupt cells that are already predisposed or vulnerable.
Another notable risk factor for PD is pathogenic variations in the glucosylceramidase beta-1 (GBA1, formerly GBA) gene, which can be found around 5–15% of people with PD (PwP).13,14 The GBA1 encodes the lysosomal enzyme glucocerebrosidase. It has been proposed that, pathogenic variants impair enzyme activity, rendering the autophagic-lysosomal pathway vulnerable and lead to defective clearance and accumulation of alpha-synuclein aggregates.15,16 The risk of developing PD in GBA1 mutation carriers is increased by up to 20-fold, 14 depending on the variant severity. In addition, PwP carrying a GBA1 mutation (PwP + GBA1) have an earlier onset, faster disease progression, and enriched non-motor manifestations. 17
While the GBA1 is a recognized genetic risk, many individuals with a GBA1 mutation will never experience PD—even the ones carrying severe variants. This suggests the role of additional genetic, lifestyle or environmental factors that modulate the penetrance of GBA1-associated susceptibility into manifest PD, via a dual-hit mechanism. One potential candidate is the OcP exposure. OcPs may aggravate vulnerable lysosomal systems and disrupt misfolded alpha-synuclein clearance. 8 Having said that, to this day, the existing data on associations between the GBA1 variant status and OcP exposure as well as other risk/lifestyle factors are limited to few studies.18–20 Moreover, whether GBA1-related impact on age of onset (AOO) and disease severity is confounded by other factors is yet to be defined. Therefore, in this study, we investigated the i) the prevalence of OcP exposure and other risk factors with respect to GBA1 carrier status and ii) their combined effect on AOO and clinical features.
Materials and methods
The ANPAR cohort and the assessments
Since 2020, Turkish PwP have been enrolled and followed by the Department of Neurology, Ankara University School, as part of the ANkara PArkinson's Disease Registry (ANPAR). Assessments cover detailed sociodemographic information, lifestyle and environmental information, disease-related information, motor and non-motor scales and quality of life. Detailed information on the assessment protocol of the ANPAR can be found in previous reports. 21 Written informed consent was obtained from all patients for inclusion in the study. The study was approved by the Ethics Committee of Ankara University School of Medicine, and all procedures followed the Declaration of Helsinki.
Genetic investigations
GBA1 variants were identified in blood samples using next-generation sequencing. The “American College of Medical Genetics and Genomics Criteria” were used to assess the pathogenicity of the identified variations. In PwP with a detected GBA1 variant, a whole-exome sequencing was performed to rule out monogenic causes of PD. Details of the genetic assessments and the GBA1 prevalence in the ANPAR cohort were reported elsewhere. 21
Assessment of OcP exposure and other risk/lifestyle factors
As part of the ANPAR assessments, structured face-to-face questionnaires were given to capture risk factors. Among those, the “history of head trauma” was defined as head traumas causing loss of consciousness, involvement in traffic accidents or long-term contact sports. OcP exposure was defined as any contact with agricultural pesticides in farms, vineyards or gardening activities. “Smoking intensity” was calculated as lifetime pack-years. “Coffee and tea consumption” were recorded as lifetime habits of cups per day. Metabolic risk factors (diabetes mellitus type-II, body mass index and waist circumference) were excluded because they were collected after diagnosis and no temporal relationship could be constructed. Since the latency period between the last date of OcP exposure and diagnosis was available for only 65 PwP, this variable was also excluded from the analysis.
Statistical analysis
Participants were divided into two groups as PwP carrying a GBA1 variant (PwP + GBA1) and PwP who are wildtype for GBA1 (PwP-WT GBA1 ). Benign or unknown variants were excluded from the PwP + GBA1 group. First, groups were compared regarding risk/lifestyle factors using unadjusted tests. Factors reaching p < 0.05 were re-examined in regression models controlling for confounding and interactions. To test GBA1-environment interactions on AOO, we built five univariate general linear models (GLM) with sex and GBA1 status included as fixed variables plus GBA1×factor interactions (GBA1 × history of head trauma, OcP exposure, coffee consumption, tea consumption, smoking). The impact of these factors on motor (MDS-UPDRS-III) and non-motor (MDS-UPDRS-I) scores were evaluated using same models with disease duration added as an additional covariate. Because this part was an exploratory analysis, p-values were adjusted according to Bonferroni (p = 0.05/5 factors = 0.01). Main effects of the risk factors were given unadjusted. All analyses were performed using the IBM SPSS Statistics version 30.0.
Results
Frequency of factors in PwP + GBA1
Data of 505 PwP were evaluated of whom 68 (13.5%) carried a GBA1 variant. Sixteen PwP carrying benign or unknown variants were excluded, leaving 52 PwP + GBA1 (10.3%). More detail on the genetic profile of this cohort has been given in the Supplement and in previous reports. 21 PwP + GBA1 had earlier AOO and longer disease duration despite younger mean age (Table-1). Regarding the factors investigated, 21% of all PwP reported previous head trauma and 22% reported contact with OcPs. The history of OcP exposure was associated with PwP + GBA1 group with an odds ratio of 1.96 (p = 0.027). No other risk/lifestyle factor differed significantly between groups (Table-1).
Comparison of the groups with and without GBA1 variant carriers.
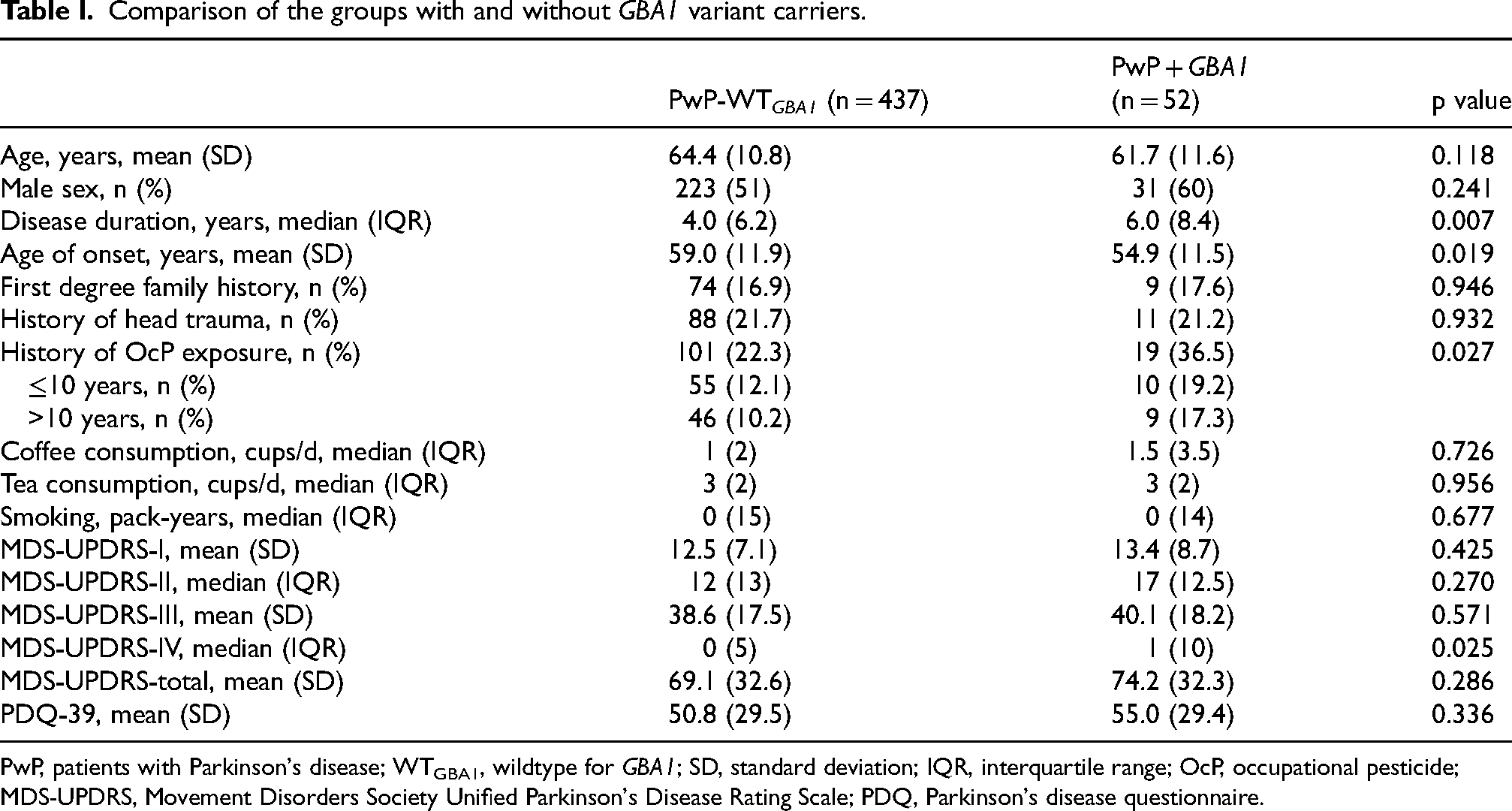
PwP, patients with Parkinson's disease; WTGBA1, wildtype for GBA1; SD, standard deviation; IQR, interquartile range; OcP, occupational pesticide; MDS-UPDRS, Movement Disorders Society Unified Parkinson's Disease Rating Scale; PDQ, Parkinson's disease questionnaire.
To confirm the unadjusted association of OcP exposure with GBA1 status, a multivariable logistic regression was performed. The model included age, sex, and OcP exposure as predictors and GBA1 status as the dependent variable. The overall model provided modest explanatory power (likelihood-ratio χ2 = 8.223 on 3 df, p = 0.042; Nagelkerke R2= 3.4%). After adjustment, individuals who reported OcP exposure had nearly twice the odds of carrying a GBA1 mutation compared with unexposed individuals (adjusted OR = 1.98, 95% CI: 1.07–3.67, p = 0.031) similar to the contingency table. Neither age, nor male sex showed a significant association. Adding coffee or tea consumption, smoking, or head trauma one at a time did not reach statistical significance to be included in the model (all p > 0.20) or change the pesticide OR estimate (all Δβ< 7%), indicating that OcP exposure is independently associated with GBA1 carrier status.
To examine the robustness of the association between GBA1 status and OcP exposure, we performed sensitivity analyses using alternative variant classifications: (i) including VUSs to the PwP + GBA1 group and (ii) restricting the PwP + GBA1 group to only severe variants. In both analyses, the proportion of pesticide exposure remained higher among PwP + GBA1 (VUS-included: 30.9% vs. 22.7%, p = 0.14; only-severe PwP-GBA1: 37.5% vs. 22.7%, p = 0.17), indicating a consistent trend across definitions. However, statistical significance was attenuated, likely reflecting smaller group sizes (only-severe PwP + GBA1 n = 16) or variant heterogeneity. These results suggest that while the association direction is robust, its magnitude is sensitive to variant classification in our cohort.
Effect of risk/lifestyle factors on age of onset and clinical scores
The main effects and the interactions of each risk factor with GBA1 carrier status were investigated with GLMs separately with sex and GBA1 as fixed covariates. First, the main effect of the GBA1 status (with covariate sex) was determined. GBA1 carriers had a younger mean age at onset (54.9 ± 11.5 years) compared to non-carriers (58.9 ± 11.9 years), corresponding to an estimated B = 3.90 ± 1.73 years (95%CI: 0.49–7.31; p = 0.025). Age at onset did not differ between men (57.8 ± 12.3 years) and women (59.2 ± 11.5 years; p = 0.19). The sex × GBA1 interaction was non-significant indicating that the impact of GBA1 variants on AOO was comparable for both sexes. Inclusion of other factors to the model showed that only smoking showed a main effect. Each pack-year of cigarette exposure is associated with a 0.082 ± 0.024-year delay in onset (95%CI: 0.02–0.14), yielding a cumulative delay of 4 years in a 50 pack-year smoker. Apart from that, history of head trauma, OcP exposure, habitual coffee or tea consumption had no significant main effect. Furthermore, no interaction effect was detected between the GBA1 status with other factors. For the assessment of motor and non-motor scores, disease duration was added to sex and GBA1 status. The results showed that the included factors showed neither a main effect nor an interaction with the GBA1 carrier status (p > 0.01). Details of the constructed models are given in the Supplement.
Discussion
The mechanisms leading to manifest disease in individuals carrying PD-associated risks are unclear. In this study, we investigated the interactions between the GBA1 carrier status and environmental or lifestyle factors for their effect on overall frequency, age of onset and clinical severity of PD. We have found that PwP carrying a GBA1 variant were almost twice as likely to report OcP exposure suggesting a potential gene-environment interaction in PD pathology via a dual-hit mechanism.
In our cohort, OcP exposure was reported in 22% of all PwP. To put this value in perspective, 14.8% of Turkey's employed population in 2024 worked in the agricultural sector—a group with higher exposure potential. 22 Population-based studies from other countries reported lower exposure rates among non-PD individuals, such as 4.1% in the Dutch Lifelines cohort 23 and 5.3–11.6% among controls in U.S. case—control studies.24,25 These values suggest that OcP exposure in our PD cohort may be higher than non-PD population, linking occupational exposure to increased PD risk. Among the PwP + GBA1, 36% reported such a history. This value exceeds that of the PPMI cohort (17%), 18 but approximates to Greek (29%) 26 and Chinese data (37.4%) 19 implying that Turkish, Greek and Chinese PwP may have more rural lifestyle than participants in PPMI. Regarding the association between the risk or lifestyle factors and GBA1 status, few reports are available in the literature. One Ashkenazi-Jewish study reported more frequent head injury in GBA1 p.N370S carriers 27 a finding could not be replicated by our data and the 23andme population. 20 Different definitions of trauma and narrow variant selection may explain the inconsistency. For coffee/tea consumption or smoking, our results are in line with previous work showing no difference in PwP with or without GBA1 mutations.19,20,26,27 A summary of these findings were given in the Table-2.
Studies investigated the frequency/association of risk/lifestyle factors in PwP + GBA1.

NE, not evaluated; OR, odds ratio.
†OR calculated post hoc based on raw counts reported in the paper ± Rural living was assessed; pesticide exposure and farming were not evaluated separately. ‡ caffeine consumption was evaluated collectively; coffee and tea consumption were not assessed separately
In terms of pesticides, most prior investigations found higher OcP exposure in PwP + GBA1, similar to ours.18,19,26 A 2018 Greek study noted a similar pattern in PwP +GBA1 compared to genetically undefined PwP, with a higher OR (6.3) than ours. 26 In 2024, the analysis of the PPMI data also showed a 5.4-fold enrichment in reports of OcP exposure in PwP + GBA1 than non-manifesting carriers (NMCs) (Table-3). 18 Of note, LRRK2 variant carriers showed no such association. Another recent Chinese study compared PwP with NMCs and reported that history of OcP (and also solvent) exposure was related to PwP in GBA1 carriers with an odds of 8.4. 19 Notably, in this study, variants of uncertain significance were also included in the GBA1 group. Reporting such findings from non-European ancestry populations is important suggesting that such an impact may be independent of ethnic or geographical influences. By contrast, the large 23andMe dataset detected no association between GBA1 carrier status and pesticide exposure, as well as between other factors. 20 However, the analysis included only three common risk allels (p.E326 K, p.T369 M and p.N370S), leaving out other risk or severe variants and relied on a single yes/no exposure question. The Parkinson's Environment and Genes (PEG) study, on the other hand, supported an association between exposure to cotton-cluster pesticides and lysosomal mechanisms. Using a geographic information system based model and an extensive data on OcP use, the authors tested a genetic panel of 85 genes related with mitophagy, autophagy and lysosomal function, and found that 36 variants of 26 genes (including GBA1) were enriched in PwP with a history of exposure. 28 This study provided important insights into gene—environment interactions in lysosomal stress using a homogeneous group of occupational pesticides (cotton cluster), and expanded the dual-hit model to individuals with genetic susceptibilities in lysosome-related pathways beyond GBA1. Independent studies should follow to test the generalizability of the findings with populations with different agricultural practices.
Studies investigated pesticide exposure in PWP + GBA1.
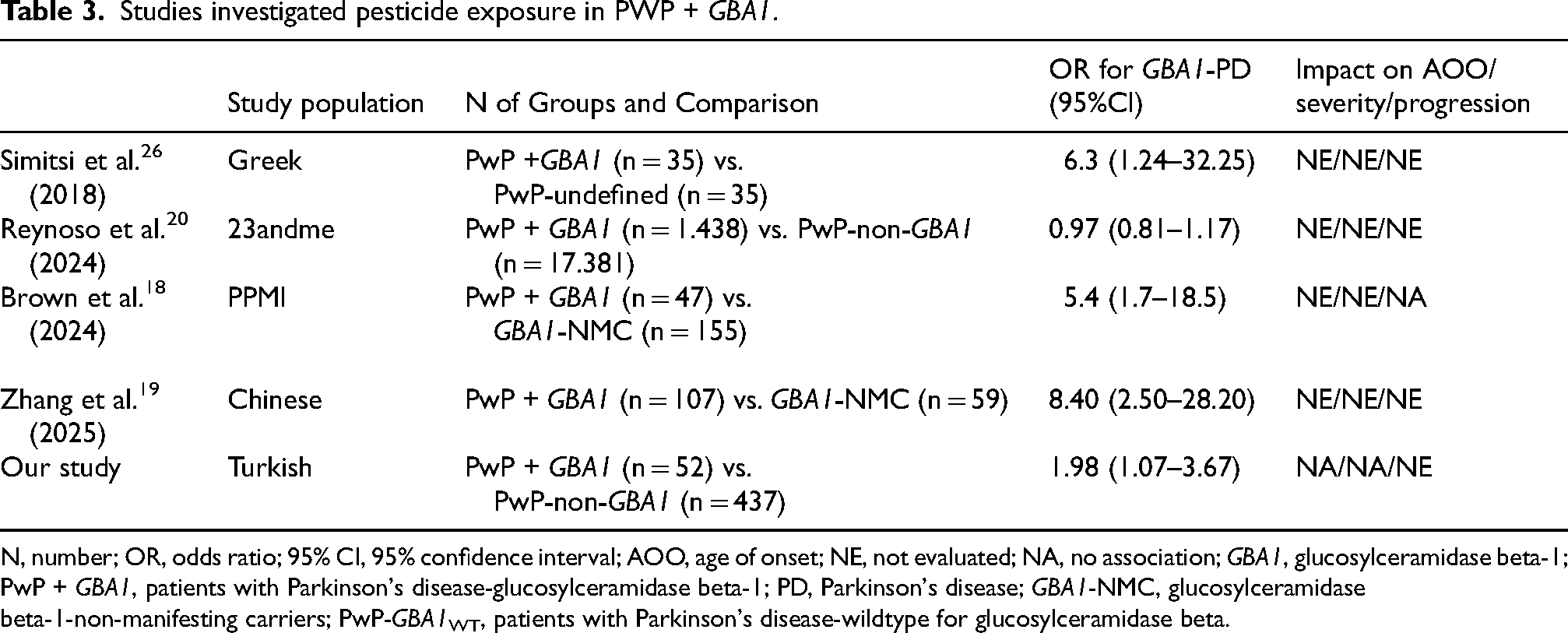
N, number; OR, odds ratio; 95% CI, 95% confidence interval; AOO, age of onset; NE, not evaluated; NA, no association; GBA1, glucosylceramidase beta-1; PwP + GBA1, patients with Parkinson's disease-glucosylceramidase beta-1; PD, Parkinson's disease; GBA1-NMC, glucosylceramidase beta-1-non-manifesting carriers; PwP-GBA1WT, patients with Parkinson's disease-wildtype for glucosylceramidase beta.
Previous studies have explored a range of gene-environment interactions on risk or AOO in PD.29–33 However, studies focused on GBA1-environment interactions are scarce. In our cohort, we tested whether the collected five risk and lifestyle factors modify the impact of GBA1 carrier status on AOO and disease severity in PwP. As expected, GBA1 variant carriers in out cohort had younger AOO, which we reported earlier. 21 In addition, smoking demonstrated a delaying effect. Other risk factors showed neither an effect on AOO nor an interaction with the GBA1 carrier status. The lack of GBA1 x smoking interaction also suggests that these two factors influence through distinct biological mechanisms. These results also differ from a single Ashkenazi-Jewish study which found a GBA1-head trauma interaction indicating a younger onset in PwP with both risks, 27 but align with a recent study (which did not investigate OcP exposure) which also found no interaction between the GBA1 status and coffee consumption or tobacco use on AOO. 20 With regard to disease severity, we found neither a main effect nor an interaction between the GBA1 carrier status and other investigated factors, similar to the analysis of the PPMI data which found no significant additional impact with OcP exposure on disease severity or progression. 18 However, while the above mentioned PEG study did not investigate the effect of lysosomal variants on AOO, it found that OcP exposure is linked to a faster progression in people carrying particular lysosomal variants, 28 underscoring the heterogeneity within the lysosomal genetic landscape. Taken together, current evidence is inconclusive on whether OcP exposure accelerates disease progression. Available studies are few, genetically diverse and used variable exposure metrics producing wide confidence intervals. To determine if OcP exposure meaningfully aggravates the disease course in lysosomal variant carriers, large-scale multicentre studies with detailed genetic stratification and uniform methods of risk quantification are required.
Limitations of the current study also need to be mentioned. First, our data on OcP captured only duration of exposure, not frequency or intensity, which prevented fully quantification of cumulative dose. Future studies should include more standardized and validated tools such as the “Agricultural Health Study”. 34 Likewise, specific classes of OcPs could not be determined, a common challenge in environmental epidemiology. The lack of a control group is another limitation, as it prevents direct comparison of OcP exposure between PwP and non-PD individuals. A potential recall bias is also unavoidable in self-reported surveys. Furthermore, although GBA1 variants were comprehensively screened, the effects of individual variant subtypes could not be analyzed separately, due to the limited statistical power. Also, other genes reported to be associated with OcP exposure33,35–38 were not evaluated. Moreover, the absence of NMCs carrying GBA1 variants precludes direct interpretation of penetrance estimation for PD. In addition, as mentioned, metabolic factors and dietary habits which could interact with OcP exposure were missing. 39 Additional confounding factors such as rural residence and socioeconomic status which influence exposure were also not assessed in this study. Despite these constraints, the comprehensive assessment of risk and lifestyle factors, a structured face to face questionnaire reducing the risk of misclassification in data are the major strengths of the study.
In conclusion, our findings suggest that OcP exposure may serve as a secondary environmental insult, exacerbating lysosomal dysfunction in genetically susceptible individuals, but does not modify AOO or disease severity. Future work should incorporate larger, variant-stratified cohorts, standardized assessment tools and biomarker-based exposure verification to create more genetically and environmentally focused samples.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251412233 - Supplemental material for Dual-Risk axis: GBA1 mutations and occupational pesticide exposure in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251412233 for Dual-Risk axis: GBA1 mutations and occupational pesticide exposure in Parkinson's disease by Zeynep Hilal Üstündağ, Hazal Ozcelik, Ipek Koker, Pınar Özkan, İrem Kar, Buse Elitas Ozmutlu, Elif İpek Gençer Mutlu, Gunal Damla Yildiz, Merve Koc Yekeduz, Fatma Tuba Eminoğlu, M Cenk Akbostancı and Rezzak Yilmaz in Journal of Parkinson's Disease
Footnotes
Ethical considerations
The study was approved by the Ethics Committee of Ankara University School of Medicine (approval number: I4–209–20), and all procedures followed the Declaration of Helsinki.
Consent to participate
A written informed consent was obtained from all patients for inclusion in the study
Funding
Funding for genetic tests achieved with the unconditional scientific support of Pfizer Inc. (Grant number: 62051201).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
