Abstract
Dysphagia is a common and often underrecognized symptom in patients with Parkinson's disease (PD) that is frequently overlooked during hospitalization. Dysphagia may be present without overt clinical signs, complicating timely identification and intervention. The absence of clear guidelines for managing dysphagia in hospitalized patients with PD often results in delays in medication administration, with subsequent deterioration in both motor and non-motor symptoms. This consensus paper, developed through collaboration among experts in otolaryngology, gastroenterology, neurology, hospital medicine, nursing, social work, speech-language pathology, pharmacy, and nutrition, presents comprehensive recommendations for the evaluation and management of dysphagia in hospitalized patients with PD. These guidelines emphasize the importance of early screening, appropriate diagnostic evaluation, and multidisciplinary involvement to support timely and safe oral intake and sustained medication schedules. Special consideration is given to the role of alternative medication formulations and compensatory strategies when swallowing impairments are identified. Recognizing the variability in access to specialty care, especially in rural and community hospital settings, these recommendations also provide practical tools for providers. They outline when to initiate swallowing evaluations, criteria for specialist referral, use of telemedicine for consultations with specialists, and strategies to bridge care from the inpatient to outpatient setting. By implementing these consensus guidelines, healthcare teams can improve the safety, outcomes, and hospital experience of patients with PD, while reducing complications and length of stay associated with dysphagia-related issues.
Plain language title
Consensus expert recommendations for management of swallowing difficulties in patients with Parkinson's disease during hospitalization
Plain language summary
Parkinson's disease is a common condition that causes motor symptoms such as slowness, stiffness, and shaking, along with non-motor problems such as difficulties with memory, sleep, and digestion. One common digestive issue is trouble swallowing (dysphagia). This can happen without obvious signs, such as choking while eating, which makes it harder to detect. When admitted to the hospital—either for emergencies or planned procedures—patients with Parkinson's disease may not get their medications at the same times they do at home. In this scenario, deterioration in swallowing function can increase the risk of aspiration (food or liquids entering the lungs), leading to longer hospital stays and complications. This paper brings together recommendations from a team of specialists—including doctors, nurses, speech-language pathologists, and dietitians—who work in neurology, gastroenterology, otolaryngology, and hospital medicine. Their goal was to develop practical recommendations to help hospital staff better recognize and treat swallowing problems in patients with Parkinson's disease. The recommendations cover when to screen for swallowing issues, when to call in specialists, how to manage medications safely, and how to plan for care after the patient leaves the hospital. These tools are especially helpful for hospitals that may not have access to Parkinson's specialists. By following these guidelines, healthcare teams can improve the care and safety of people with Parkinson's during hospital stays.
Introduction
Parkinson's disease (PD) is a neurodegenerative disorder impacting nearly 12 million people worldwide.1–4 Patients often experience a variety of motor symptoms such as bradykinesia and rigidity, as well as non-motor symptoms including gastrointestinal and sleep dysfunction. 5 Hospital admissions for patients with PD may be directly related to the disease, such as elective admission for deep brain stimulation surgery, indirectly related in the case of injurious falls, or unrelated when due to other medical comorbidities such as elective joint replacement surgeries or infections. 6 Hospitalized patients with PD often present with swallowing dysfunction.7,8 In many cases, there may be new onset of dysphagia, (e.g., admissions related to altered content or level of consciousness), while in others the presence of dysphagia may be exacerbated by alterations in medication administration leading to more OFF time or related to worsening of parkinsonism due to the primary cause of admission. 9 Monitoring swallowing function requires substantial outpatient and at home care and ideally begins before hospital admission; however, timely management during hospitalization remains critical for patients with PD. If delayed, the risk of serious complications such as aspiration pneumonia, dehydration, and prolonged hospitalization increases.10–13 Diagnosis and management are essential to prevent these potentially life-threatening complications and improve quality of life during and after hospitalization.14–17 To that end, we present consensus expert recommendations for management of dysphagia in hospitalized patients with PD, the application of which can improve care and prevent harm. 18 These recommendations aim to guide all clinicians involved in care of patients with PD during hospital admissions and suggest step by step use of screening tools, further diagnostic tests, and consideration of referrals to other teams for management of new onset or worsening of existing dysphagia in PD. The statements are not mandates, but they are a roadmap toward current best practice. Implementing these recommendations can prevent serious complications and improve hospital stay outcomes. While having access to comprehensive multidisciplinary resources is ideal, in hospitals with limited resources, judicious coordination of available expertise is recommended to optimize care for patients with PD and swallowing dysfunction. It is important for hospitals with limited resources to identify gaps in care for patients with PD and advocate for resources that can potentially prevent long hospitalizations and complications for this population.
Methods
The clinical consensus statements (CCS) presented in this paper were developed using a modified Delphi protocol. 19 This topic was prioritized for CCS development by the Parkinson's Foundation and proposed to the Parkinson Study Group, Other Non-Motor Working Group (PSG ONMWG) with a target audience defined as all medical providers involved in the care of hospitalized patients with PD, including physicians, speech-language pathologists, dietitians, and nurses. The specialists participating in this modified Delphi panel were selected for their topic expertise and publications in the area. The group was sub-divided into five working groups wherein specialists performed a narrative literature review and developed draft statements. Draft statements were initially vetted within the working groups through online meetings and offline communications. Five virtual meetings were held with the attendance of most authors, during which draft statements were shared on screen, discussed by members, revised as needed, and ultimately voted (agree/disagree). The process was repeated until unanimous (100%) agreement was achieved for the final statement. If full agreement was not achieved, the statement was excluded but discussed in the text. Since not all authors were able to attend all meetings, a final polling of all statements was conducted in one online meeting with the attendance of all authors. The final manuscript was drafted with participation and final review from all members. Below, we present statements regarding each topic, followed by discussion points from online meetings.
In-hospital dysphagia screening for patients with PD.
All hospitalized patients with PD should be administered a standardized screening instrument for oropharyngeal dysphagia unless there is a compelling reason (e.g., intubation) not to do so. Inpatients with PD who have a history of oropharyngeal dysphagia or who fail the swallowing screen should also undergo a bedside evaluation by a speech-language pathologist. A validated malnutrition screening tool should be used for all inpatients with PD to assess the need for evaluation by a registered dietitian. Inpatients with PD with signs and symptoms that suggest oropharyngeal dysfunction or evidence of malnutrition should undergo a modified barium swallow or flexible endoscopic evaluation of swallowing (FEES) with the involvement of a speech-language pathologist. Patients with PD admitted with new or worsening dysphagia should be evaluated by a neurologist. For patients with new onset dysphagia, evaluation of esophageal structure and function should be performed. Tests can include radiological evaluation (modified barium swallow/barium esophagram), upper endoscopy, and esophageal manometry. A GI consultation may help guide and interpret investigations.
Hospitalization represents a point of care to evaluate function and risk from oropharyngeal dysfunction. 20 This is more of a challenge than appreciated because symptomatic dysphagia in PD can grossly underestimate the degree of dysfunction. 10 In a meta-analysis comparing clinically overt dysphagia vs. objective demonstration of swallowing dysfunction, there was a 47% (35 vs. 82%, respectively) discordance. 21 Thus, devising a simple bedside evaluation not requiring an instrumental assessment of swallowing in PD can be difficult. Challenges also exist in devising a scale that covers all aspects of oral preparation and swallowing that may be affected in PD.22,23 Several oropharyngeal dysphagia scales have been implemented that measure either swallowing function or the risk of aspiration. 24 Scales that have been evaluated for patients with PD and can be used at the bedside are summarized in Table 1. Additional measures that are more general in assessing the severity of PD, such as presence of depression or dementia or the Hoehn and Yahr Scale, are also associated with greater degrees of oropharyngeal dysfunction.25,26 Since the ability to administer scales and/or bedside objective testing is limited, a recent consensus conference suggested the following symptoms be elicited as a prompt for further evaluation: increased eating time (meal duration), post-swallowing coughing, post-swallowing gurgling voice, drooling, choking, breathing disturbance, unintentional weight loss, difficulty in swallowing pills, sensation of retention of food, pneumonia episode(s) or if patients answer yes to either of the following questions: “Have you experienced any difficulty in swallowing food or drink?” or “Have you ever felt choked with food?” 27
Tools for screening for dysphagia in PD.
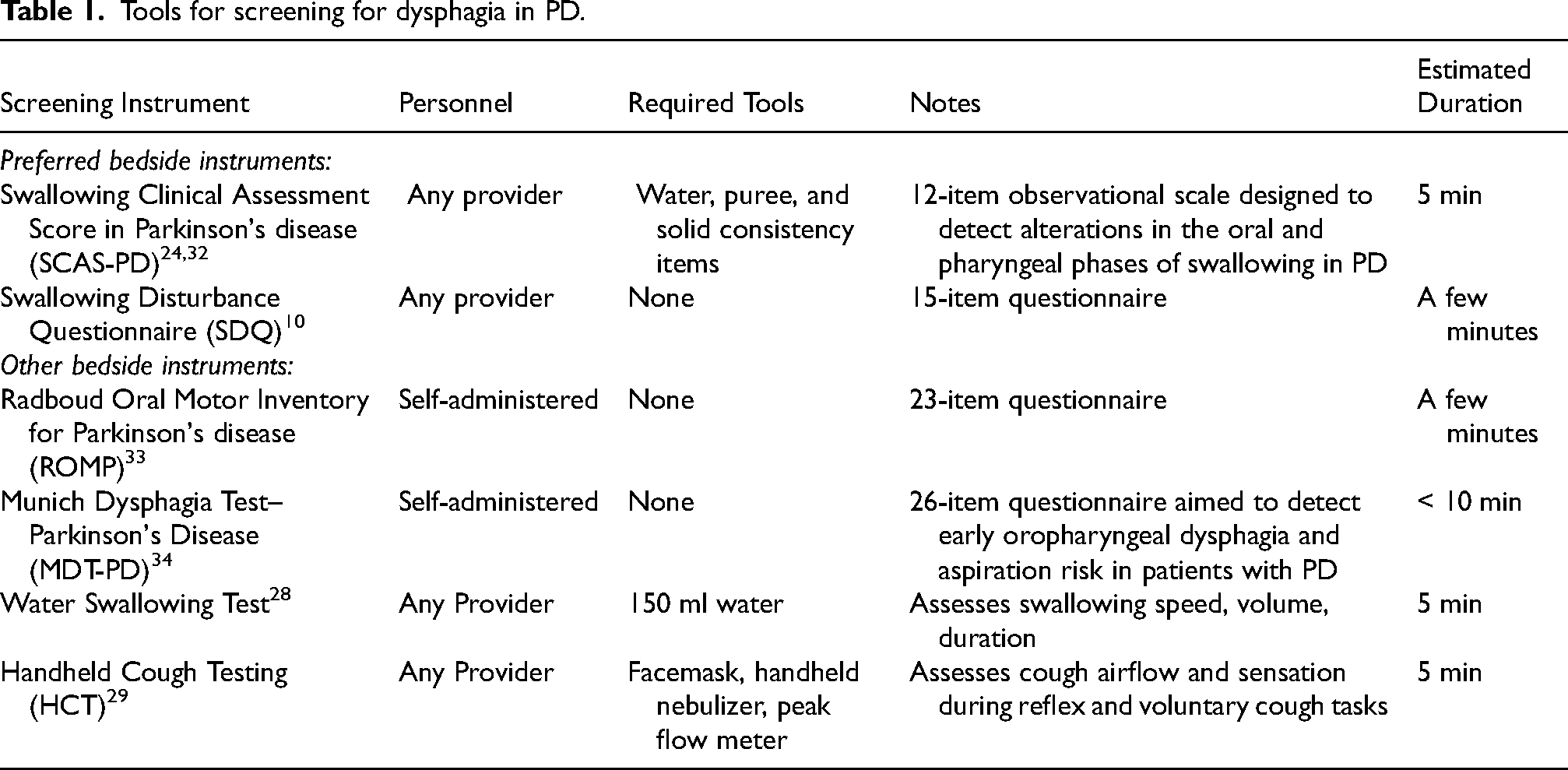
Once a patient with PD has symptoms and/or signs of oropharyngeal dysfunction identified on screening, particularly with risk for aspiration, further objective testing is indicated. 15 A simple bedside test that has been proposed is the water swallowing test. 28 This test was endorsed in a recent international consensus meeting on dysphagia in PD. 27 Another bedside test is Handheld Cough Testing (HCT). 29 There is no consensus on which of these tests should be applied on hospital admission, but one objective test prompted by symptomatic screening is recommended. 30 Objective screening measures are especially important given that other gold standard approaches for the assessment of oropharyngeal swallowing function, such as videofluoroscopy or FEES, cannot be performed at the bedside without specialized equipment or radiology facilities in addition to specific expertise of a radiologist and speech-language pathologist. 31
Malnutrition is common in patients with PD.35–37 A systematic review of malnutrition, defined by the presence of protein calorie malnutrition, 38 revealed that up to 25% of patients with PD are malnourished. 38 Notably, BMI is not used to define malnutrition, as up to 40% of patients with PD may have an elevated BMI yet still meet criteria for and experience the consequences of protein-calorie malnutrition. 39 Similar limitations apply to using weight loss alone as a screening tool. 40 The Global Leadership Initiative on Malnutrition (GLIM) developed consensus criteria for malnutrition in adults, which include five factors: weight loss, low body mass index, reduced muscle mass, reduced food intake, and disease burden/inflammation. 41 In PD, weight loss concerning for malnutrition is defined as ≥5% of usual body weight over 3 months or ≥10% over 6 months; additionally, any unexplained or unexpected weight loss is considered a red flag.35,40
Patients with dysphagia secondary to PD are at risk for malnutrition due to insufficient proteincalorie intake and inadequate knowledge of how to facilitate intake of these calories in the presence of oropharyngeal dysphagia.35,40 Also, patients may reduce their intake of protein due to concern that protein may interfere with absorption of levodopa, which can further increase their risk of malnutrition. 40 The plethora of causes unrelated to dysphagia (such as severity of the disease and depression) emphasizes the need for malnutrition assessment in any hospitalized patient with PD regardless of the presence of oropharyngeal dysphagia.42,43 However, data specific to malnutrition in hospitalized patients with PD are not available. 37
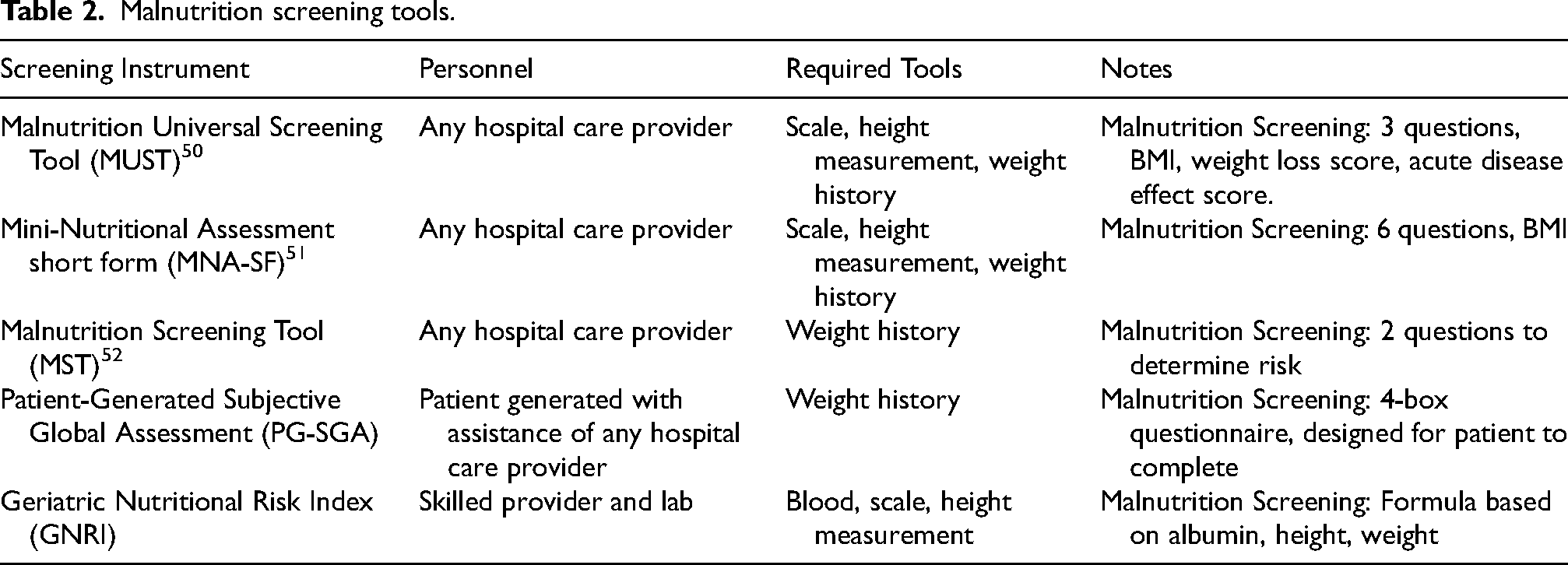
Many proposed tools are available for screening for those at risk for malnutrition in hospitalized patients with PD, some of which are detailed in Table 2 below.36,44,45 These screening tools can help identify patients at risk for malnutrition so that a referral to a registered dietitian can be made to better evaluate malnutrition status. These range from general assessment of PD and general measures of malnutrition to screening tools used specifically for PD. 45 Tests such as the Malnutrition Universal Screening Tool (MUST), the Mini-Nutritional Assessment Short Form (MNA-SF) the Malnutrition Screening Tool (MST), and the Patient-Generated Subjective Global Assessment (PG-SGA) are all methods used in the general patient population. 37 The MUST involves anthropometric measurements for weight and height, as well as identifying any unplanned weight loss and acute disease activity score. MUST is a popular and easy to use nutrition screening tool that can be used by all care workers and has similar reliability as compared to the MNA-SF and MST. The MNA-SF tool includes six screening questions regarding recent decline in food intake, weight loss, mobility, acuity, psychological problems, and BMI. There are additional questions in the full form that can help guide the assessment by a registered dietitian but are not required for purposes of screening. 46 The MST is a two-item questionnaire on recent weight loss and changes in appetite. This screening questionnaire is validated in acute, long-term, rehabilitation, and ambulatory care settings regardless of age and medical history. The PG-SGA relies on the patient to answer the screening questions, which may not always be feasible in a hospital setting. The four questions include information about recent weight changes, symptoms that have kept the patient from eating, changes in food intake, and functional status. The Geriatric Nutritional Risk Index (GNRI) is calculated from a formula using serum albumin, calculated body weight, and ideal body weight. 47 The advantage of this scale is a lack of reliance on a history, which can be difficult to obtain in some patients with PD. 47 Its limitation is relying on albumin, which is not a valid tool for assessing malnutrition and is not included in current malnutrition guidelines, and thus, the GNRI is not recommended for use. 47 The position of the Academy of Nutrition and Dietetics recommends the use of the MST to screen adults for malnutrition regardless of their age, medical history, or clinical setting. While most of these screening tools have not directly been validated in patients with PD, they are validated in hospitalized settings where patients with PD are treated. 48 The MUST, MNA-SF, and PG-SGA are also validated screening tools to use in hospital and community settings, but may be more lengthy to complete despite having similar validity as the MST. 49
Malnutrition screening tools.
Symptoms and signs of oropharyngeal dysfunction in PD often do not match the degree of dysfunction present. 10 Prior studies demonstrated that 60–80% of patients with PD are not cognizant of their oropharyngeal dysfunction, and most do not report symptoms unless specifically questioned.53,54 Dysfunction may occur at any stage of PD.21,55–57 Although a simple bedside test, as discussed previously, is helpful for screening, a detailed and comprehensive evaluation of oropharyngeal function, as provided by a videofluoroscopy or FEES, is an essential next step for assessing specific swallowing impairments, risk of aspiration and the ability of a patient to sustain adequate oral alimentation. 58 Both are considered to be gold standards.59,60 In addition to global assessment of these endpoints, more precise evaluation may be needed, since PD may variably affect oropharyngeal function, may involve both motor and sensory dysfunction, and may cause symptoms in all segments of the oropharynx.12,61 This is likely because of the multiple levels of neuromuscular control of swallowing that can be affected by PD, including cortical, brainstem, peripheral sensory, and motor nerves. 12 Furthermore, both videofluoroscopy and FEES tests yield anatomic and functional information in response to a range of textured boluses mirroring the patient's symptoms in response to specific foods and liquids.62,63 With documentation of the dysfunction, an opportunity for simultaneous and/or follow-up therapy is provided. 64
Both videofluoroscopy and FEES have similarities in their abilities to record and review images and test swallowing in response to the administration of foods and liquids of different consistencies. 15 They also provide a setting in which therapy can be initiated through taught maneuvers. 15 Videofluoroscopy provides a more direct evaluation of all swallowing phases. 65 Its disadvantages are the need for a speech-language pathologist in addition to a radiologist, radiology facilities, and radiation exposure to perform the study. 65 In contrast, FEES requires only one operator (an otorhinolaryngologist or speech-language pathologist), offers more direct visualization of pharyngeal structures, particularly the larynx, and is relatively easy to perform in inpatient settings. 66 Disadvantages include that this expertise is not as commonly available as videofluoroscopy, an increased risk of procedure-related complications due to the instrumentation involved, inability to visualize the bolus during the moment of the swallow, and visualization being limited to pharyngeal, upper esophageal sphincter (UES) and laryngeal function. 67 Another criticism of FEES has been a lack of standardization of results. 60 A recent study has validated the use of the Dynamic Imaging Grade of Swallowing Toxicity for Flexible Endoscopic Evaluation of Swallowing (DIGEST-FEES) for PD. 60 This score consists of elements from the Penetration Aspiration Scale to determine safety grades and percentage of pharyngeal residue to determine efficiency grades. 60 The choice of which of these tests to use for inpatient evaluation of PD will likely depend on available technology and expertise.68,69
For patients with new-onset dysphagia, esophageal structure and function should be evaluated. 70 Tests can include radiological evaluation (modified barium swallow/barium esophagram), upper endoscopy, and esophageal manometry. 59 In some patients GI consultation might help guide and interpret investigations. 71
Although patients with PD, particularly at more advanced stages, have altered oropharyngeal function, additional potential etiologies of dysphagia in the older adults are numerous. 22 As a result, an important question is when other causes of dysphagia in patients with PD should be investigated by a gastroenterologist. 72 To answer this question, it is important to determine if PD involves esophageal function distal to the oropharynx.22,72 There is precedent to expect esophageal dysfunction in PD. 73 For example, PD can be associated with gastroparesis and colonic inertia.74,75 Nevertheless, a clear association between PD and esophageal dysfunction is elusive. Indications for the use of high-resolution manometry for the investigation of PD-associated esophageal dysfunction have not been established, and the need for this study should be discussed with gastroenterologists.
Although PD does not convincingly predispose to clinically impactful esophageal disorders, common causes of dysphagia, such as strictures related to gastroesophageal reflux disease (GERD), Zenker's diverticulum, esophageal cancer, and achalasia may occur.76,77 Clinical prompts that suggest a need for further evaluation by GI include a history of symptoms of gastroesophageal reflux; volumetric regurgitation (i.e., an esophageal content volume that clearly exceeds that associated with UES dysfunction); a lack of oropharyngeal symptoms associated with dysphagia, such as nasal regurgitation, drooling, prandial coughing, and voice dysfunction; and a patient's description. Similarly, a negative evaluation for oropharyngeal dysfunction in a PD patient with dysphagia should alert the clinical staff.78,79 Evaluation may include an esophagram or endoscopy.80,81 It is also important to emphasize that false localization is common in esophageal dysphagia with patients with achalasia or distal stricture not infrequently pointing to the upper esophagus or area of the UES as the location where food sticks. Depending on the severity of dysphagia, the presence or absence of malnutrition and weight loss, or presence of other red flags such as pain with swallowing, rapidly progressive dysphagia evolving from solids to liquids, complete inability to swallow foods, regurgitation of undigested food or food impaction, palpable lymphadenopathy or history of cancer, these diagnostic tests may need to be performed during the admission or may be postponed until following discharge from the hospital.
Summary points:
All patients with PD should receive screening for oropharyngeal dysphagia upon hospital admission. Hospitals should use available resources and screening tools to detect new-onset or worsening symptoms of dysphagia and malnutrition in PD. Upon detection, referral to a speech-language pathologist for further testing—such as FEES, videofluoroscopy, or modified barium swallow—should be considered. Consultation with GI for further diagnostic testing should be considered in cases with unidentified etiologies of dysphagia on initial testing, or in the presence of other comorbid GI symptoms. Consultation with neurology should be considered if medication adjustments are required due to swallowing dysfunction.
Diagnostic approaches to swallowing dysfunction in inpatients with PD.
If dysphagia is identified on screening, consultation with speech-language pathology and other available specialists should be considered to prevent aspiration and guide treatment. For any patient with PD who has been admitted to the hospital, the presence of the following symptoms should trigger an assessment for dysphagia even if they pass the initial screening swallow test: malnutrition, frailty, and drooling, especially in the presence of axial/cranio-cervical dyskinesia and postural abnormalities. These patients should be considered high risk. Proper reconciliation and timely administration of PD medication in hospitalized patients with PD is critical to mitigate aspiration risk. PD medications ideally should not be changed during hospitalization, but if a change is deemed necessary, neurology input should be considered. In the evaluation of patients with oropharyngeal dysphagia, difficulties with oral medication, with or without difficulties with solids and/or liquids, should be addressed. Dysphagia evaluations, such as a videofluoroscopy by speech-language pathology, when deemed necessary, should be completed as soon as possible. Consideration should be given to the timing of carbidopa/levodopa (C/L) administration; preferably, evaluations should be performed in the ON state after taking medications. Elective procedures for patients with PD should preferably be scheduled early in the morning, allowing them to continue to take C/L with a sip of water up to the time of the procedure and administering the next dose of medication as soon as deemed safe.
When dysphagia is identified in a patient with PD during a hospital admission screening, a comprehensive management plan should be implemented to address potential swallowing impairments.15,82–84 A comprehensive dysphagia management plan, including medical treatment and behavioral rehabilitation, is necessary to reduce sequelae, especially aspiration pneumonia, a leading cause of death in people with PD that is largely attributable to dysphagia.85,86 Silent aspiration or aspiration without an appropriate cough response may occur, with or without a positive dysphagia screening.87,88 As such, a negative screening, as with a positive screening, should prompt ongoing surveillance and close monitoring of symptoms and follow-up screenings, so that risks and symptoms not captured during the initial screening are identified as early as possible.54,86,89,90 Patients with PD have a > 3 times higher risk of aspiration pneumonia than their peers, with an average 2.74% prevalence and 10% hospital mortality. 91 Higher hospital mortality rates among individuals with PD and dysphagia may be addressed with immediate engagement (as resources allow) of available specialists, prioritizing speech-language pathologists, dieticians, neurologists, gastroenterologists, and pharmacists.70,92 Other disciplines that should be considered to optimize a comprehensive approach may include movement disorder specialists, neurologists who have experience in PD, and occupational therapists, who can begin to develop and implement medical and physical rehabilitation interventions that may protect against dysphagic symptoms.93,94
Although multidisciplinary approaches to treatment are ideal for managing dysphagia in patients with PD, 95 this recommendation assumes access to a full complement of specialists, which may not be available in smaller or rural hospitals. In these situations, practical strategies may include leveraging telehealth for remote consultation with speech-language pathologists or neurologists and use of validated swallow screening tests. Implementation of basic compensatory strategies such as upright positioning, modified diet textures, minimizing distractions during feeding, adhering to proper medication timing, and careful monitoring during meals and oral medication administration should be considered when access to specialists is limited.
Individuals with PD showing symptoms of malnutrition, frailty, and drooling, especially in the presence of axial/cranio-cervical dyskinesia and postural abnormalities, during hospital admission should be considered high risk for both aspiration and aspiration pneumonia. 96 Since dysphagia is frequently neither identified nor addressed until it is more severe, conducting further investigation in the presence of these risk factors may help with earlier identification and timely treatment response.57,97 It is also important to consider the effects of motor impairments such as tremor, postural instability, and bradykinesia, which can impair self-care and the ability to feed oneself in patients with PD.96,98–100
Additional diagnoses that may predict the development of dysphagia during hospital admission may include GERD and/or pulmonary disease.101,102 Patients with PD are 4.05 times more likely to develop GERD, indicating a greater risk for developing GERD-related dysphagia. 102 Although the precise nature of the defects in esophageal motor function linked to PD remains to be defined, impaired peristalsis and lower esophageal sphincter dysfunction likely promote and exacerbate GERD and related dysphagia. Co-occurring motor and autonomic dysfunction may heighten the need for proactive management strategies during acute care episodes, which include screening to identify signs and symptoms of dysphagia as soon as possible. The presence of complications of GERD, such as esophagitis, stricture, and Barrett's esophagus, is best assessed by esophago-gastro-duodenoscopy (EGD).
Addressing dysphagia that is identified during hospital admission screening may be optimized with proper and timely medication reconciliation. 103 Results from a recent medication reconciliation program during hospitalization prevented the erroneous administration of 2716 doses of PD medications, representing 60% of the total number of doses prescribed. 103 Delays with medication reconciliation can have adverse health effects for hospitalized patients with PD. 103 Delayed doses, with consequent lengthening of the dosing interval, may result in problems related to wearing off, such as worsening of motor signs (bradykinesia, tremor, dystonia) and nonmotor symptoms (anxiety, shortness of breath), 104 all of which can result in worsening swallowing function. As such, timely and efficient care coordination among members of the clinical team, prioritizing medication reconciliation, should be performed during the first 24 h of admission or as soon as resources allow. 105 The clinical team assisting with medication reconciliation should include pharmacy and nursing, and, as resources allow, neurologists and/or movement disorder specialists who may be able to provide more specialized care. 103 Speech-language pathologists and dietitians should also be included (as staffing allows) to inform consistency of modifications as appropriate for safe swallowing and consumption.103,106,107 In hospitals with limited resources, the primary team should use the most recent outpatient clinical notes from patients’ neurologists and review them with their care partner for timing and accuracy. In cases of discrepancy, communication with outpatient neurologists or primary care or use of a telemedicine consult with neurology is recommended.
In patients with PD with motor fluctuations, swallowing assessments during the “ON” phase of medication (while patients are experiencing improved motor function in response to medications) may allow for a more accurate determination of baseline swallowing capacity and differentiation between swallowing impairments that are typically mitigated by medication versus those that persist even when medications are most effective. In addition, assessment during the ON phase may also clarify and distinguish dysphagia origin, including whether dysphagia derives from other factors such as aging and other comorbidities; this differentiation may be necessary for determining and optimizing treatment responses. In the presence of axial/cervical-cranial dyskinesias, the clinical examination should not be performed during exacerbation periods of dyskinesias interfering with the ability to feed. 27
Hospital admission for patients with PD may be prompted by elective procedures (related or unrelated to PD), during which dysphagia may develop or be identified, prompting the need for dysphagia treatment intervention. 108 Assessment for dysphagia among these patients should include an evaluation of feeding performance and arrangement of corresponding follow-up care as needed and as resources allow. 108 If dysphagic symptoms and/or a positive screening for dysphagia are found, a more thorough instrumental assessment should be completed, and treatment should be started to address swallowing safety and/or improve swallowing efficiency. 109
Patients with PD who are hospitalized for elective surgical procedures or who require emergency operations, may need to follow an NPO (nil per os) status before anesthesia. In most cases, taking oral medications with a sip of water is permitted until shortly before surgery and in the recovery room after the surgery (as permitted at the discretion of the anesthesiology team) 110 ; however, gaps in provider knowledge about managing NPO status can lead to unnecessary or longer-than-necessary withholding of medications. 110 To prevent complications such as dopamine withdrawal syndrome in patients with PD, which is clinically similar to the neuroleptic malignant syndrome (NMS), it is important to administer PD medications as closely as possible to their regular schedule. 110 Scheduling operations and other procedures early in the day can reduce missed doses and minimize the risk of complications. 110
When possible, involvement of a neurologist with expertise in movement disorders (either through hospital consult or tele-neurology) should be obtained for any necessary adjustments to the medication regimen, including alternative therapeutic options if needed. 110 Managing outpatient neurologists can partner with inpatient surgical teams in advance of elective surgeries to clarify individualized medication regimens; this approach can also be used with primary care offices in areas where outpatient neurology is not available. 110 Repeated assessment of a patient's ability to take solids and liquids orally may be necessary to determine the earliest time to reinitiate oral intake safely. 111 In cases where there is limited time to conduct repeated assessments due to staffing challenges and limited available providers (which may be the case in rural environments or smaller hospitals), clinical staff should provide detailed documentation of initial findings and clinical reasoning to help guide care and compensate for possible delayed reassessment opportunities.
Summary points: (Figure 1)
Patients with PD who fail bedside dysphagia screening tests, or those who are identified to be high risk (due to presence of malnutrition, frailty, and drooling, especially in the presence of axial/cranio-cervical dyskinesia and postural abnormalities), should have further dysphagia evaluations. Hospitals should use available specialists, including speech-language pathologists, hospitalists, gastroenterologists, and ENT specialists, for further diagnostic tests for dysphagia. Use of telemedicine is recommended in limited access settings. It is ideal to perform dysphagia evaluations in the ON state without significant dyskinesia in patients with PD. Nurses may consider coordinating with specialists to plan for the timing of testing in the ON state. Patients with PD should take their PD medications as scheduled, even during the NPO state in preparation for elective procedures. Resuming PD medications should start as soon as possible after procedures, to maintain the usual timing of the PD medications.
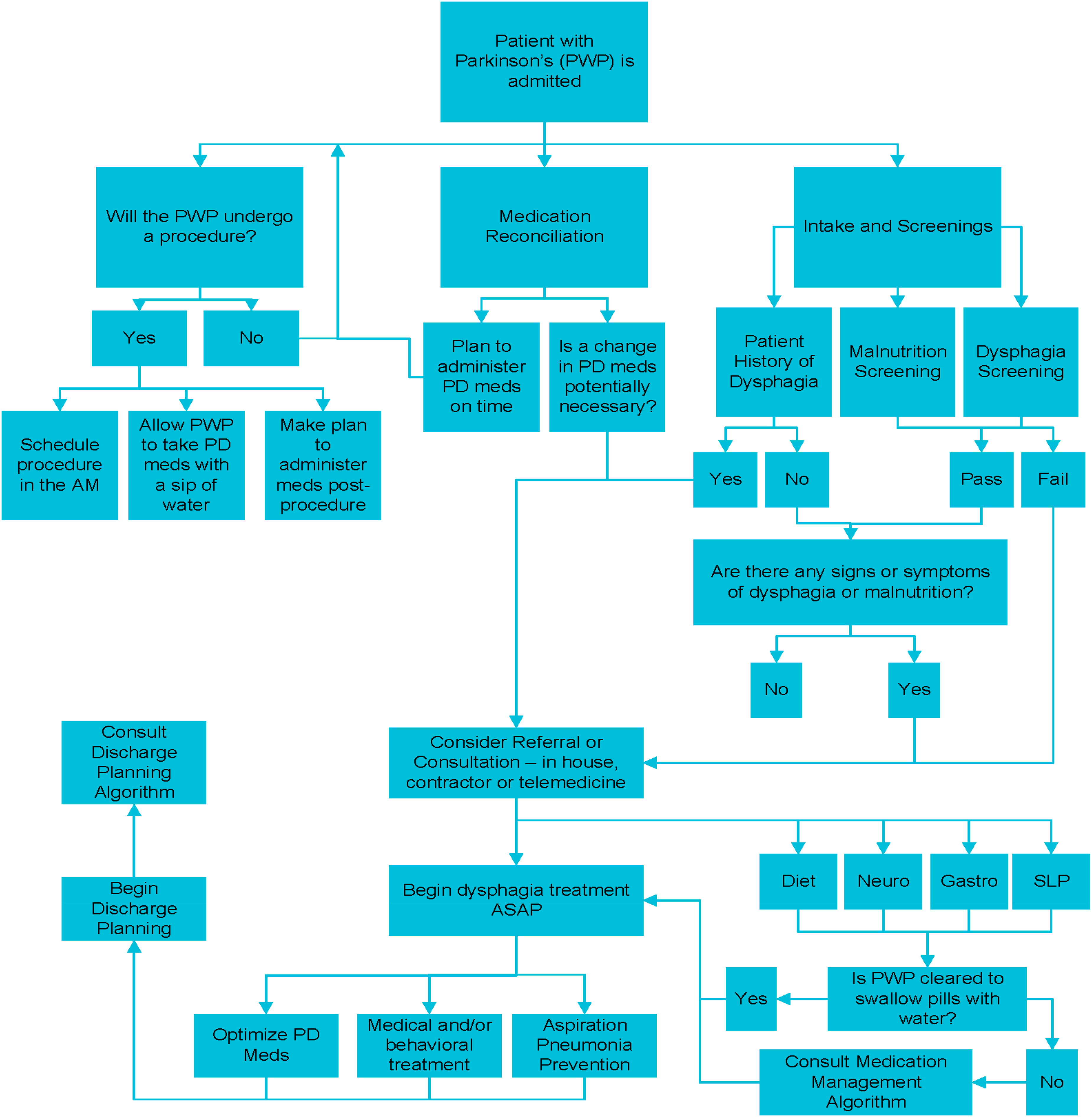
Inpatient management Algorithm for Patients with PD.
Medication delivery for inpatients with PD.
Patients with PD who are not cleared to swallow pills should have an alternative and equivalent method approved as soon as possible. To minimize risk of NMS-like withdrawal syndrome, oral C/L, dopamine agonists, and amantadine should be resumed, ideally, within 24 h. For patients who cannot safely swallow pills but are cleared to swallow crushed tablets (or capsule contents sprinkled onto food of an appropriate consistency), current medication or an equivalent dose should be resumed as soon as possible. When patients with PD are not cleared to take oral medications, but are cleared to receive medications enterally, an equivalent dose of the patient's outpatient medications should be resumed via a nasogastric or nasojejunal feeding tube. When patients with PD are unable to receive medication by oral or enteral routes, a rotigotine patch can be considered in place of outpatient oral C/L. Enteral C/L suspension, subcutaneous foscarbidopa/foslevodopa or subcutaneous apomorphine infusions should be continued from patients’ home medications if already prescribed.
Patients with PD and motor fluctuations can experience profound motor and nonmotor (anxiety, pain) OFF time dysfunction if their medications are delayed.112,113 Within 24 h after amantadine, dopamine agonists, or levodopa are withheld, patients with PD may develop an NMS-like withdrawal state characterized by severe rigidity, disorientation, and elevated serum creatine kinase114,115;therefore, an alternative method of medication administration should be implemented as soon as possible if oral medications need to be withheld due to dysphagia. For patients with PD whose symptoms are not well-controlled in the hospital, it is very important to involve a consulting neurologist. It is not uncommon for patients with PD to hallucinate and/or be agitated during hospitalizations. This may be due to the primary indication for admission or to changes in the schedule or dose of PD medications. 116 Consultation with a neurologist may play a significant role in preventing mortalities related to psychosis during admission. 116 Medications that are often used in hospitals for management of psychosis and agitation, such as haloperidol, may worsen parkinsonian symptoms and should be avoided if possible. Other dopamine blockers, such as quetiapine, are preferred in the hospital setting and may be used at the discretion of the consulting neurologist. Table 3 shows acceptable alternative delivery methods and dose conversions for commonly prescribed PD medications. Immediate-release (IR) tablets can be crushed, or extended-release (ER) capsules can be opened, and contents given in an approved consistency of food or administered via feeding tube. When substituting C/L IR tablets in place of C/L ER capsules (Rytary or Crexont), the interval between doses should be shortened by one hour for Rytary, while for Crexont the frequency of dosing should be increased by at least once per day. Amantadine may be available as a liquid formulation, and oral dopamine agonists (e.g., ropinirole and pramipexole) may be switched to an equivalent dose of the dopamine agonist administered via patch (rotigotine) if oral intake is not safe. When patients with PD are not cleared to take oral medications, they should be encouraged through shared decision making to agree upon an alternative enteral delivery method. Due to the potential to impair levodopa absorption, high-protein intermittent bolus feeds should ideally be administered at least 30 min after crushed C/L tablet administration (starting/stopping of continuous feeds is not necessary). If patients refuse all suggested methods, or are not cleared for any enteral route, transdermal rotigotine or injectable apomorphine may be used on a short-term basis to prevent an NMS-like withdrawal state, ideally in consultation with a neurologist, given the complexity of dose conversion, and higher risk of psychosis with dopamine agonists. Patients who are prescribed enteral C/L or subcutaneous foscarbidopa/foslevodopa or apomorphine pump systems should resume these as soon as possible according to their usual regimen. In the United States, limitations in timely insurance approval often hinder the inpatient use of infusion pumps, making them less feasible as bridging strategies during interruptions in oral medication administration; however, when resources and approval are available, these pumps can be considered as potential treatment options for patients unable to take their oral PD medications. 117 Where appropriate, PRN inhaled levodopa or injectable apomorphine can be continued from home medications at the discretion of a consulting neurologist.
Summary points: (Figure 2)
Delays in administration of PD medications for patients with PD who are not cleared for PO intake due to dysphagia during hospitalization can result in worsening of motor and non-motor symptoms and in severe cases, NMS-like symptoms can occur. Medications such as carbidopa/levodopa IR and ER, dopamine agonist tablets, and amantadine can be crushed and administered via a feeding tube. (Note: crushing ER C/L tablets eliminates the extended-release properties.) If enteral access is not possible, a rotigotine patch may be considered. If long-acting formulations of C/L (e.g., Rytary or Crexont) are not available on formulary, the dose should be converted to immediate release and started as soon as possible. In patients with PD with increased agitation or hallucinations requiring medication therapy during hospitalization, quetiapine is the preferred dopamine blocker. Other typical or atypical dopamine blockers (e.g., haloperidol, olanzapine) should be avoided whenever possible.
Alternative delivery methods and dose conversions for commonly prescribed PD medications.
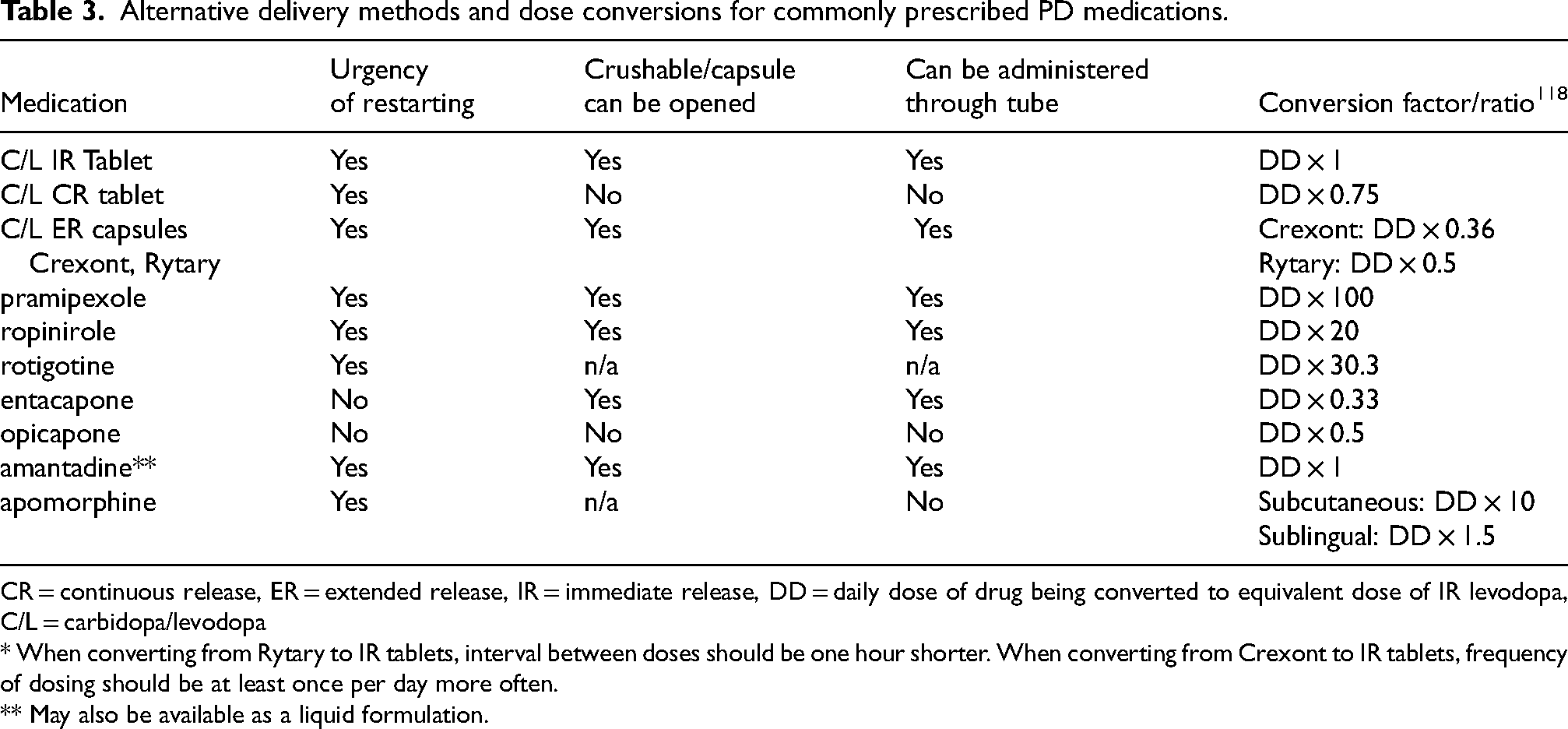
CR = continuous release, ER = extended release, IR = immediate release, DD = daily dose of drug being converted to equivalent dose of IR levodopa, C/L = carbidopa/levodopa
* When converting from Rytary to IR tablets, interval between doses should be one hour shorter. When converting from Crexont to IR tablets, frequency of dosing should be at least once per day more often.
** May also be available as a liquid formulation.
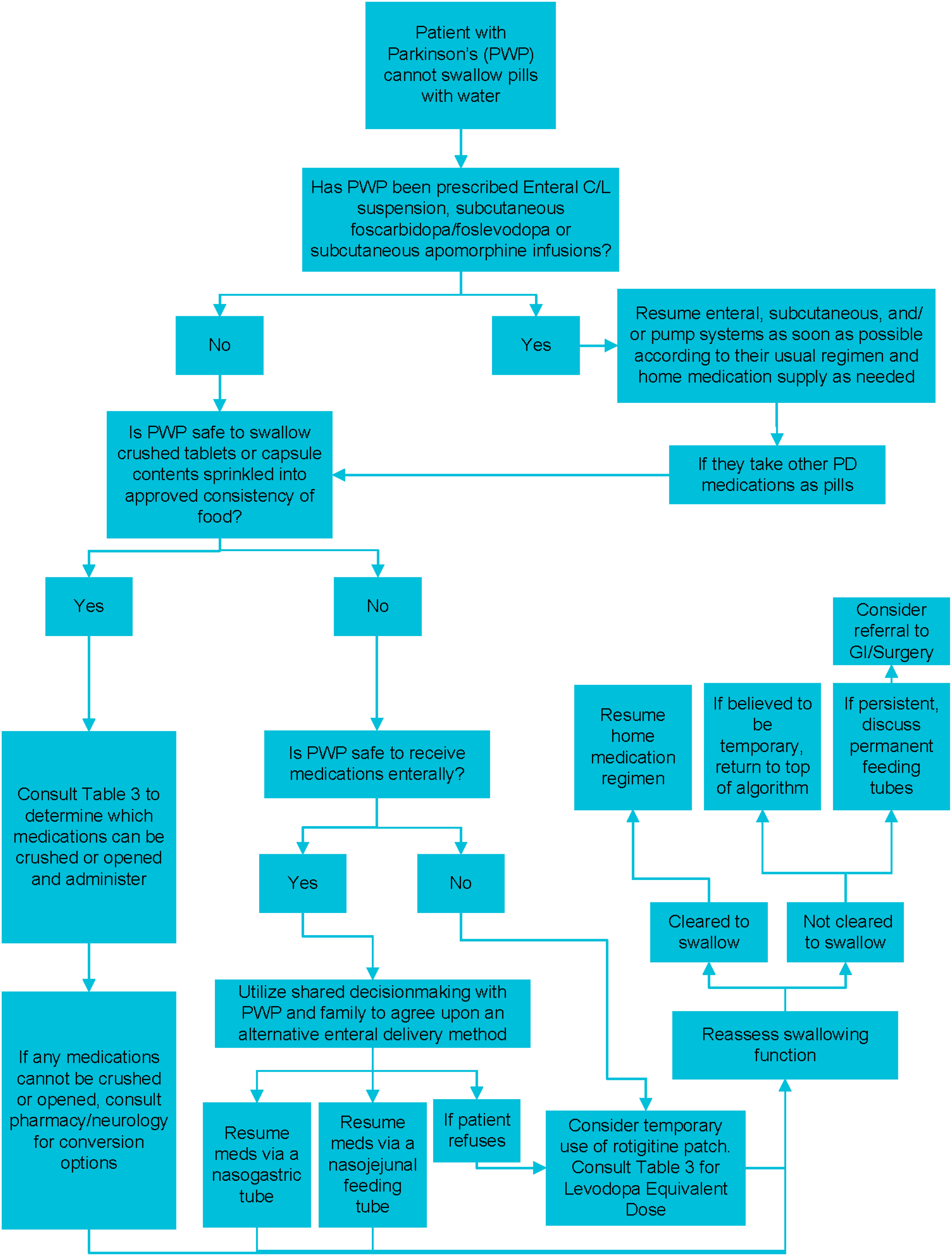
Clinical pathway for managing PD medications when swallowing is impaired.
Strategies for treatment of dysphagia during hospital admissions for Patients with PD.
Treatment of dysphagia should be started as soon as possible; treatment can begin during hospitalization and continue after discharge. Treatments may include optimizing PD motor treatments, dietary modification, thickening fluids, adopting certain postures or swallowing maneuvers, behavioral or device-driven exercises, and medical treatments. A temporary feeding tube should be considered for medication delivery and/or nutrition. If swallowing problems persist, the insertion of percutaneous endoscopic gastrostomy (PEG) tube, PEG-J, or J tube should be considered. Patients with documented or suspected gastroparesis should be considered for a jejunal extension (PEG-J) or a direct jejunostomy for post-pyloric feeding. If UES dysfunction is identified as the main cause for dysphagia, EMG-guided botulinum toxin injections, dilation, or endoscopic myotomy may be considered after consultation with ENT and/or GI. If the clinical context suggests that safe swallowing function is unlikely to return, placement of a percutaneous endoscopic gastrostomy (PEG) tube should be considered, as it can improve quality of life. PEG placement does not eliminate the risk of aspiration. Pre-procedural counseling should include this advice and address patient and care partner expectations, nutritional and hydration status, respiratory function, and the patient's overall prognosis, goals, and preferences.
A prior expert panel developed consensus statements to guide the multi-disciplinary treatment of dysphagia and related nutritional management in PD. 109 To avoid overlap, we refer the reader to that publication and will herein present a brief overview and updates. Dysphagia treatments supported by the 2021 consensus guidelines included optimizing PD motor treatments and feeding during best ON times, standardized and skill-based swallowing therapy, dietary modification, thickening fluids, adopting certain postures or swallowing maneuvers, behavioral or device-driven exercises, including expiratory muscle strength training (EMST), and medical treatments. Strength and skill (or task)-based swallowing treatments should address specific impairments identified by imaging and/or endoscopic testing of swallowing and be guided by patient-specific factors and goals. 109 These treatments are often directed by speech-language pathologists and dietitians with input from neurology and gastroenterology.119,120 Occupational therapy may also be useful in some cases. For example, when postural adjustments require muscle activation and anatomical coordination beyond the orofacial and cervical regions, occupational therapy can support activity tolerance and coordination to achieve ideal posture for feeding; in the absence of occupational therapy, existing evidence characterizes postural adjustments as low-burden, cost-effective interventions that all members of the care team can implement. 121
Since the 2021 consensus guidelines, there has been an additional randomized controlled trial that confirmed the efficacy of EMST – but also the efficacy of cough skill training (smTAP) for dysphagia in PD. 122 There are currently insufficient data to support the use of neuromodulatory techniques such as transcranial direct current stimulation, repetitive transcranial magnetic stimulation, or transcutaneous electrical stimulation for the treatment of dysphagia in PD.
If swallowing difficulties persist and impair nutrition and/or delivery of medications, a nasogastric or naso-jejunal feeding tube may be needed as a temporary measure, and if swallowing problems persist, a PEG tube, PEG-J, or even J tube may need to be considered. Enteral feeding is an option for patients with PD treated with levodopa/carbidopa intestinal gel and the feeding tube can be used both for PD treatment and feeding. 123 It is critical to ensure that patients and their families understand the difference between temporary and permanent feeding tubes as part of the informed consent discussions. Permanent measures are not appropriate for patients with advanced directives clearly articulating refusal of permanent feeding tubes and those with advanced dementia.
Summary points:
Treatment of dysphagia is guided by diagnostic evaluations and may include optimizing PD medications, adopting specific postures or swallowing maneuvers, dietary modifications (e.g., thickened fluids), behavioral or device-assisted exercises, and medical therapies. A temporary feeding tube should be considered promptly if patients are not cleared for oral intake.
If dysphagia persists, counseling patients and care partners regarding permanent feeding tube placement (e.g., PEG, PEG-J, J tube), including potential benefits and effects on quality of life, should be initiated.
Referral to GI or surgery should be considered for the above procedures.
Transition to outpatient care.
When dysphagia is identified via hospital screening and assessment, providers should develop discharge plans that include referrals to appropriate follow-up medical appointments, including home health and/or outpatient rehabilitation for swallowing therapy and self-care training, neurology (established or new referral), and outpatient, specialized nutrition care based on results of malnutrition screening and dysphagia assessment.
During inpatient discharge planning, patients and care partners should be provided patient-specific education on safe swallowing techniques, dysphagia risk, dietary modifications, enteral feedings (as appropriate), oral/dental care, and ongoing management to prevent post-discharge complications.
Care partner education and training should be completed by members of the team to facilitate adherence to dysphagia management recommendations; this includes education/training on safe feeding techniques, recognizing signs and symptoms of aspiration, compensatory strategies for safely managing at home, and safe medication administration.
Alternate medications or formulations should be considered to compensate for swallowing difficulties, with the goal of medication adherence and safe consumption.
If the presence of dysphagia requires medication changes during hospitalization, these should be discussed with the patient/care partner and communicated to managing physicians.
Preparations for hospital discharge and creation of individualized discharge planning should begin as soon as resources allow to promote comprehensive, proactive care, enhance patient safety and reduce transitory vulnerability post hospital discharge.124,125 This may include promoting early discussions about discharge needs126,127 and referrals to social work and initiation of home health services during inpatient discharge planning, which can optimize access to critical outpatient care.126,128
Follow-up communication about dysphagia care during the hospital (including screening/assessments/treatment) should be provided to the managing physician and/or home health/outpatient rehabilitation agencies.129,130 This provides updates for the outpatient medical providers on the status of the patient. 131 Dysphagia treatment recommendations that are not clearly communicated and documented are unlikely to be continued and carried over beyond the hospital care setting, and benefits may be lost post-discharge. 132 There is evidence that, in some cases, 80–100% of dysphagia recommendations made by a speech-language pathologist were omitted from hospital discharge communications, including approximately 50% of dietary recommendations. 132 Care providers managing patients with dysphagia should review documentation by all members of the treating team to extract and include dysphagia recommendations in hospital discharge communications. 132
Dysphagia, alongside polypharmacy and medication errors, has been identified as a barrier to home discharge in the general population. 83 This negative association is exacerbated among individuals with PD who are more likely to be discharged to a long-term care inpatient setting rather than returning home.133,134 In many cases, multimorbidity, frailty, the need for complex medical management, and heightened susceptibility to hospital-related complications decrease the capacity for individuals with PD and their care partners to manage their care at home 135 ; as such, long-term care is sometimes the safest option for transitioning out of the acute hospital.136,137
In patients with documented dysphagia, discharge plans should prioritize routine dysphagia monitoring to track new, resolving, or changing symptoms and ensure ongoing management post-discharge.15,138 The optimal discharge planning for dysphagia management among individuals with PD may involve (when available) advice from a team of specialists, including neurologists, physical and occupational therapists, speech-language pathologists, psychologists, social workers, pharmacists, dieticians, and other experienced medical professionals. 139
However, not all patients have equal access to quality outpatient facilities and specialized care, and there is evidence that providers’ limited experience with PD and dysphagia further limits outpatient access for patients. 140 Since variations in local resources upon hospital discharge have been linked to hospital readmission rates, 141 a comprehensive assessment of patients’ access to community resources and connections to free or low-cost post-acute services when appropriate may be helpful. 140
Care partners of individuals living with PD are at a high risk for care partner burnout, especially if they are also managing dysphagia.135,142–144 To minimize the care partner burden as much as possible, healthcare providers should engage care partners through training that may include education, tangible materials like handouts, interactive methods like visual demonstration, and role-play and teach-back techniques to reinforce trained concepts and maximize carryover among care partners.145,146 This education may be provided by all available members of the treating team depending on availability and topic knowledge.145,146
Since the acute hospitalization stay may result in medication changes due to dysphagia, medication reconciliation before hospital discharge is a critical step for safe outpatient transitions and may help reduce medication discrepancies after discharge. 147 This should include reviewing and adjusting the patient's medication regimen to ensure consistency and safety. 147 Involving pharmacy staff in completing medication reconciliation has been shown to have positive effects and may reduce adverse drug events that can hinder transitions to outpatient care. 148 When access to pharmacists is limited, medication reconciliation by the nurse in the presence of the patient and/or care partner(s) is essential.
It is also essential that any modifications made to medication regimens during hospitalization are communicated clearly to both the patient and their care partners, as well as to outpatient healthcare providers, to maintain continuity of care and prevent errors.149,150 For example, if a patient with PD is admitted to the hospital and unable to safely take their PD medication during the admission, and they are switched to rotigotine patch, this should be communicated to the managing outpatient neurologist who can subsequently advise on appropriate adjustments once the patient returns to their normal swallow function. In addition, community care providers, such as home health agencies that provide nursing, occupational therapy, and speech therapy services, often rely on a reconciled medication list to provide comprehensive home healthcare services. 150
Summary points: (Figure 3)
Patient-specific education on safe swallowing should begin during hospitalization; both the patients and their care partners should be included. Safe discharge planning for patients with PD and dysphagia includes documentation of test results, a summary of recommendations and suggested modifications, as well as medication changes during hospitalizations. Such a discharge summary should be communicated to the patients’ primary care provider, and outpatient neurologist.
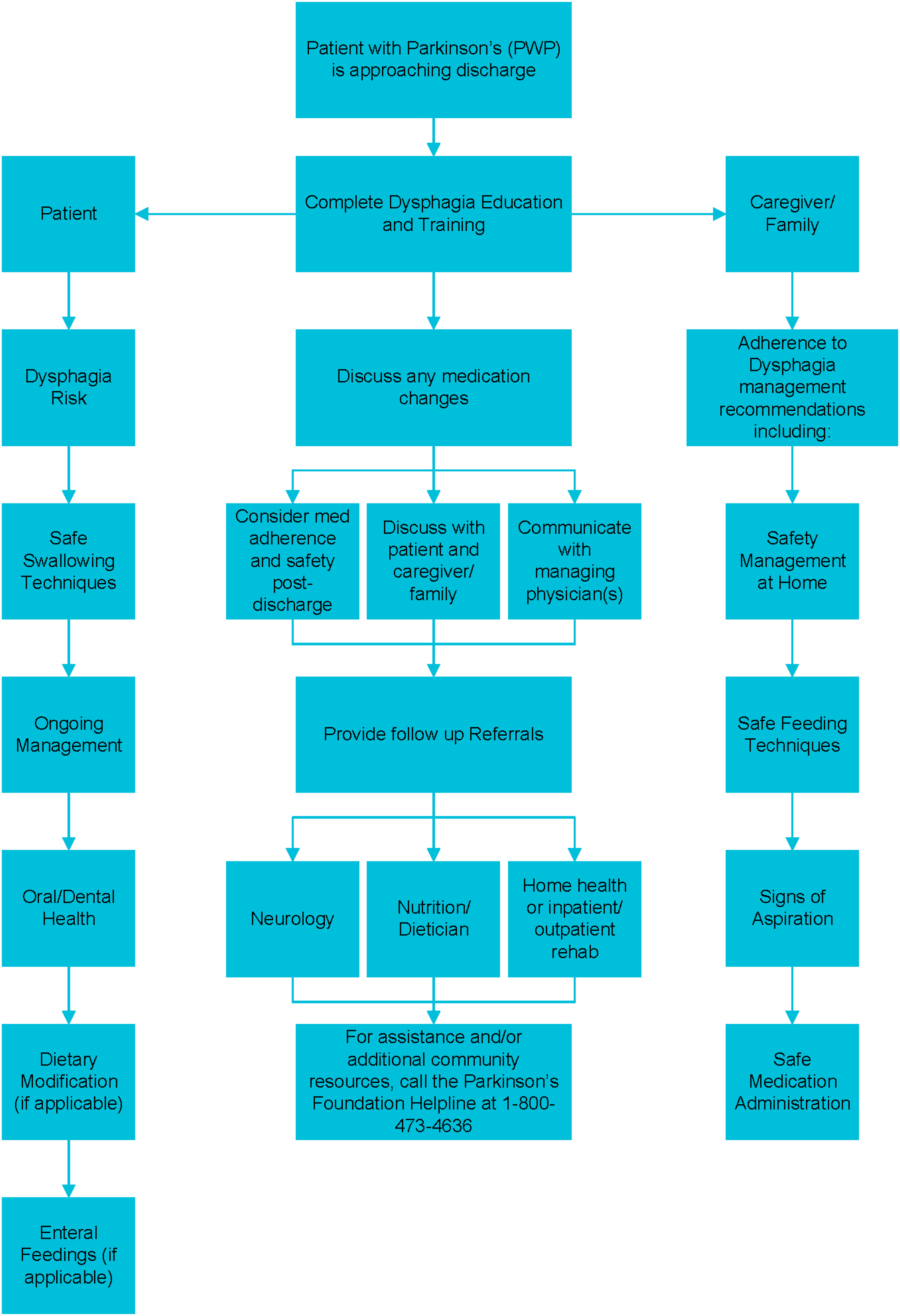
Transition of care pathway: Dysphagia management for PD.
Conclusion
Dysphagia is a common symptom in PD that can worsen chronically with progression of the disease and acutely in the hospital setting. Admissions to the hospital often result in changes in the timing of administration of PD medications; this may lead to increased OFF time, which itself can result in worsening of swallowing function. Delays in swallowing evaluations during hospital admissions can result in unnecessary delays in medication administration as well as worsening motor function in patients with PD. Such delays often result in prolonged hospitalizations due to silent aspiration and aspiration pneumonia. There has been a lack of clear guidelines for managing dysphagia among patients with PD during hospital admissions. This interdisciplinary Delphi panel collaboration aimed to close this gap by suggesting clear recommendations for screening, diagnostics, and treatment that can be used by healthcare providers involved in the care of patients with PD during hospital admissions. We acknowledge that such an interdisciplinary team may not be available to all hospitals, and, therefore, the recommendations from this Delphi panel team can be used by primary hospital teams to bridge gaps in access. Also, in the era of telemedicine, access to specialized consults, including neurology and speech-language pathology, can be facilitated remotely, expanding availability of expertise for patients with PD.
Adherence to interdisciplinary discharge plans in addition to proactive monitoring and management of evolving acute symptoms, supports early engagement of care partners and timely identification of post-discharge needs.121,151
Footnotes
Acknowledgements
This topic was prioritized for Clinical Consensus Statement development by the Parkinson's Foundation and supported by the Parkinson Study Group (PSG) Other Non-Motor Working Group. The Expert Panel included several clinicians with personal connections to Parkinson's disease including one retired clinician who is living with PD.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during this study.
