Abstract
Background
The aggregation and spread of α-synuclein within brain are associated with the loss of dopaminergic neurons and the formation of Lewy bodies as seen in Parkinson's disease. Blocking the initiation of α-synuclein aggregation, or the spread of such aggregates, may offer disease-modifying approaches to slow disease progression. Previous studies have demonstrated that modification of aggregation prone proteins, including α-synuclein, with O-linked β-N-acetylglucosamine (O-GlcNAc) reduces their aggregation. Small molecule inhibitors of the enzyme O-GlcNAcase (OGA), which removes O-GlcNAc from proteins, confers neuroprotective benefits in various preclinical disease models of Alzheimer's and Parkinson's diseases.
Objective
This study investigates the effects of long-term pharmacological enhancement of O-GlcNAcylation in a transgenic mouse model of Parkinson's disease overexpressing human α-synuclein.
Methods
Thiamet-G was orally administered to mThy1-hSNCA and wild-type (WT) mice for ten months. Behavioral assessments were conducted to examine changes in locomotion and cognition. Histological analyses were performed to analyze α-synuclein aggregates and dopaminergic neurons in brain sections. Immunoblot and ELISA analyses were performed to analyze O-GlcNAc and soluble α-synuclein using brain lysates, respectively.
Results
Thiamet-G increased the level of O-GlcNAc in the brain of both mThy1-hSNCA and WT mice. The levels of total α-synuclein in the brain were unaltered. However, Thiamet-G strongly attenuated the deposition of pS129-immunoreactive α-synuclein aggregates within the substantia nigra, prior to observable neurodegeneration. Thiamet-G also protected against locomotor decline.
Conclusions
These results support OGA inhibition as a therapeutic approach to block the pathological formation of toxic α-synuclein as a disease-modifying treatment against Parkinson's disease.
Plain language summary
Currently there are no medicines that can slow or halt the progression of Parkinson's disease. Research suggests that clumping of the neuronal protein α-synuclein within the brain is toxic and drives the advance of the disease. Slowing the clumping together of α-synuclein therefore offers a possible approach to develop a treatment to slow the disease. To test this idea, we treated mice for ten months with a compound that increases modification of proteins with a sugar known as O-GlcNAc. This molecule has been shown to be safe and well-tolerated with protective benefits in several disease mouse models. Using mice that express human α-synuclein and develop Parkinson's disease, we tested the effects of the treatment on motor control and cognition by getting these mice to perform various tasks. After treatment, we studied brain tissues for changes in the clumping of α-synuclein and other markers in the brain. We found the molecule reliably increased protein O-GlcNAc in the brain. We also found that the treatment significantly reduced the formation of toxic α-synuclein in the brain. Moreover, we observed the treatment helped preserve locomotion. These results support the idea that increasing protein O-GlcNAc in brain can slow the formation of toxic α-synuclein and may be an effective approach to slow the progression of Parkinson's disease.
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative disease after Alzheimer's disease (AD). In 2022, the number of new patients diagnosed with PD was estimated to increase by 50% in the United State alone and the total prevalence is projected to reach one million in 2030.1,2 Accordingly, there is interest in advancing new therapeutic strategies to address this disease. Postmortem analysis of patient brain tissues reveals the accumulation of aggregates known as Lewy bodies which are composed of abnormally phosphorylated α-synuclein. These proteinase-resistant aggregates are one of the primary pathological hallmarks of PD and they correlate with the extent of the degeneration of dopaminergic neurons in the brain.3–6 Currently, Levodopa (L-DOPA) is the most prescribed FDA-approved treatment for improving the quality of life for patients with PD, by transiently replenishing dopamine in the brain to restore motor control. However, the lack of direct intervention against the progressive α-synuclein pathologies results in the irreversible loss of dopaminergic neurons. Therefore, identifying a disease-modifying strategy that can effectively block the formation of toxic phosphorylated α-synuclein aggregates remains a critical need for patients.
The O-linked β-N-acetylglucosaminylation (O-GlcNAcylation) modification of nuclear and cytoplasmic proteins has gained considerable interest as a strategy to combat various neurodegenerative diseases due to the protective effects arising from increased O-GlcNAc that relieves cellular stresses and neurodegeneration.7–13 β-N-acetylglucosamine (GlcNAc) is a monosaccharide synthesized from glucose and glutamine via the hexosamine biosynthetic pathway (HBP) to generate, as an end product, the high-energy nucleotide sugar uridine diphosphate GlcNAc (UDP-GlcNAc). The enzyme O-GlcNAc transferase (OGT) uses UDP-GlcNAc as a substrate to modify proteins by attaching a GlcNAc unit onto the hydroxyl group of certain serine and threonine residues. The O-linked GlcNAc can be subsequently removed by the enzyme O-GlcNAcase (OGA), which recovers the hydroxyl group. Pharmacological elevation of O-GlcNAcylation is found to preserve neurons, strengthen synaptic plasticity, relieve neuroinflammation, and prevent cognitive decline in various cell and animal models of neurodegenerative diseases.9,14–18 Notably, AD patients exhibit hyperphosphorylation of neuronal tau protein amid compromised glucose metabolisms.19,20 Clinical studies using the fluorodeoxyglucose (FDG)-positron emission tomography (PET) as outlined by the Alzheimer's Disease Neuroimaging Initiative (ADNI) suggest diminished glucose uptake precedes tau hyperphosphorylation and cognitive decline in patients of familial AD.21,22 Most importantly, the level of O-GlcNAc-modified proteins appears markedly reduced in the brains of AD patients.9,19 These findings collectively support a protective role of O-GlcNAcylation in the brain that may delay or attenuate the intensity of neurodegeneration.
Central to several of these studies, has been the use of the high-quality small-molecule OGA inhibitor Thiamet-G, which has seen common use as a research tool for cell and in vivo preclinical studies. In PC-12 cells overexpressing human tau protein, Thiamet-G reduces tau phosphorylation while elevating the level of O-GlcNAc-modified proteins. 23 Due to Thiamet-G being a blood-brain-barrier penetrant inhibitor with high potency and specificity against OGA (Ki = 2.1 nM) [Cekic, ChemSci], chronic delivery of Thiamet-G by oral dosing in drinking water is readily feasible. 24 This tool compound has therefore been used in various rodent models of tauopathy, where oral dosing or intracranial injection of Thiamet-G reduced levels of tau hyperphosphorylation and neurofibrillary tangles.12,17,23–26 Thus, OGA inhibition shows robust and reproducible protective effects in cell and animal models of tauopathies.
In PD the initial development of Lewy bodies occurs within the substantia nigra, a dopaminergic (DA) brain region where dopamine is synthesized and released. As seen for tau, monomeric α-synuclein showed increased aggregation propensity following phosphorylation. 27 Notably, hyperphosphorylated α-synuclein, particularly phospho-serine-129 (pS129), comprises most of the α-synuclein species in Lewy bodies.6,28 Recently, α-synuclein has also been shown to be O-GlcNAc modified at several sites.29–31 Moreover, in vitro studies using synthetic α-synuclein modified with O-GlcNAc in a site-specific manner show that the presence of O-GlcNAc at some of these sites reduces its aggregation propensity.32,33 In addition, increased cellular O-GlcNAcylation induced either through OGA inhibition or OGA knock-down reduces cellular uptake of α-synuclein preformed-fibrils (α-syn-PFF). 34 Motivated by these observations, we set out to test whether OGA inhibition is an intervention that could suppress the accumulation of hyperphosphorylated α-synuclein in vivo. In particular, we aimed to investigate the efficacy of chronic Thiamet-G treatment in blocking the development of PD pathology in an α-synuclein overexpressing mouse model of PD. Notably, during our studies, findings on the enhancement of O-GlcNAc within brain using both genetic and pharmacological methods were reported, showing protective effects in two different mouse models.10,35 In one of these studies, the injection of α-syn-PFF was used to induce neurodegeneration within the substantia nigra, 36 and α-syn-PFF prepared from O-GlcNAc-modified α-synuclein monomers (gS87) induced less pathology as compared to equivalent non-glycosylated α-syn-PFF. 35 In another study using a PD mouse model that manifests disease through overexpression of a-synuclein, increasing the level of O-GlcNAcylation also delivered beneficial effects. 10 Here we found that administration of Thiamet-G in drinking water significantly reduced levels of α-synuclein aggregates without major impact on the overall level of α-synuclein in the brain. Further, mice treated with Thiamet-G showed maintained locomotion. Our findings support the emerging Targets to Therapies (T2 T) initiative led by the Michael J Fox Foundation, which has identified OGA inhibition as a promising disease-modifying target for developing treatment for PD, warranting increased preclinical attention including further preclinical animal studies. 37
Methods
Animals
We used an available transgenic mouse strain (C57BL/6N-Tg(Thy1-SNCA)15Mjff/J; line 15, #017682 from Jackson Laboratory), 38 hereafter referred to as “Line 15”, that overexpresses wild-type (WT) human α-synuclein driven by the mouse thymus cell antigen 1 (Thy-1) promoter, which leads to an accumulation of full-length α-synuclein and progressive deposition of α-synuclein aggregates in the brain. 39 Thy1-hSNCA models are best known for recapitulating the gradual α-synuclein inclusion and behavioral decline that is seen in idiopathic PD. 40 All dosing and behavioral assessments were approved by the University Animal Care Committee (UACC) at Simon Fraser University following the guidelines established by the Canadian Council on Animal Care (CCAC).
Male Line 15 and negative control C57BL/6NJ mice (hereafter referred to as “WT mice”) were group-housed upon arrival at our facility at two months of age. They were maintained under a 12-h light-dark cycle with chow pellet ad libitum. After acclimatizing to the facility for one week, mice started Thiamet-G dosing at 500 mg/kg/day (mkd) (“high dosage”) or vehicle (n = 29–31), alongside an intermediate dose group (n = 31) receiving 200 mkd (“low dosage”). A short-term preliminary study was also conducted where ten Line 15 and WT mice received either vehicle or Thiamet-G at 500 mkd for two weeks to confirm the ability of Thiamet-G to engage OGA within the brain and induce a sustained pharmacological increase in protein O-GlcNAc (Supplemental Figure 1A). Thiamet-G was delivered by dissolving the compound in autoclaved drinking water and providing ad libitum for ten months. Dosages were determined according to the average consumption of water (4.0 ml/day) and an average body weight of 30 g. No toxicity from either dosage was found in previous reports using Thiamet-G in mouse models of AD.18,24 After one month of treatment, mice were trained and assessed for their motor and cognitive performances in an alternative, bimonthly schedule as shown in Figure 1. After ten months of dosing, mice were quickly euthanized in CO2 and transcardially perfused with 15 ml ice-cold PBS (pH 7.4). Harvested brains were medially bisected, where one hemibrain was fixed in 4% paraformaldehyde for two days, and the other hemibrain was snap-frozen in liquid nitrogen and then stored at −80°C until further processing. Fixed hemibrains were cryoprotected by soaking in 30% sucrose dissolved in PBS, after which they were embedded in optimal cutting temperature (OCT) embedding medium block (Sakura Finetek USA Inc). Hemibrains-OCT blocks were coronally cryosectioned at 40 µm on a Leica cryotome for histological analysis.
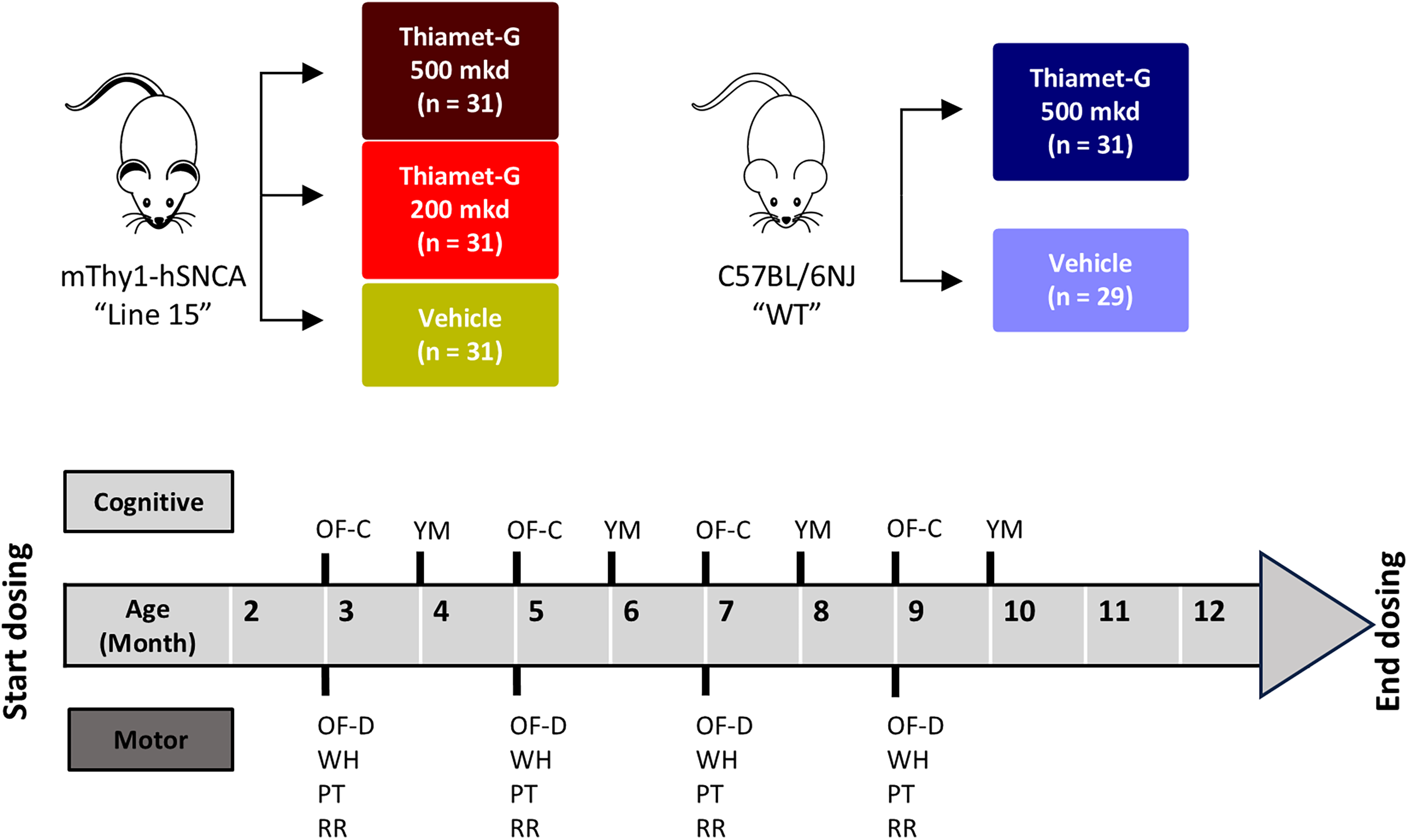
Study design. Schematic diagram summarizing the cohort breakdown and bimonthly behavioral assessments in the 10 months dosing timeline of the Line 15 and WT mice. Treatment began one week after mice arrived at the facility. After acclimatizing for one month, mice were trained in cognitive and motor assessments in an alternating, bimonthly schedule. For assessing motor performance, distance traveled in the open field (OF-D), wire hang (WH), pole test (PT) and rotarod (RR) were examined. For assessing cognitive performance, time spent in the central quadrant in open field (OF-C) and Y-maze (YM) were examined. Mice reached the termination endpoint at 12 months of age, when they were sacrificed by CO2 and brains extracted for biochemical analysis.
Behavioral assessments
Mice were assessed for effects on locomotion after 1, 3, 5, and 7 months of Thiamet-G treatment, and cognition after 2, 4, 6, and 8 months of treatment. Motor performance was assessed by open field, pole test, wire hang, and rotarod. Cognitive performance was assessed by Y-maze, and the time spent in the central quadrant during open field test, which reflects the level of anxiety. Sessions were conducted within the first 4 h of the dark cycle.
Open field
Each mouse was placed in an open field arena enclosed within a 40 cm x 40 cm x 49 cm box. The center quadrant was labeled, and a digital camera was positioned above the apparatus for tracking the movement of the mouse. Each mouse was tested for a total of 15 min per session.
Wire hang
Each mouse was placed on a sustained wire at approximately 35 cm above a pillow and allowed to have a firm grip. Then the mouse was released to test its ability to support itself on the wire for up to 120 s per session.
Pole test
Each mouse was placed onto a wooden pole at a height of 50 cm with its head facing the top of the pole. Then the investigator released the mouse and allowed it to climb to the top and reorient its body, and then descend to the base of the pole to complete the trial. The time spent reorienting its body and descending to the base of the pole was recorded separately for up to 120 s per session.
Rotarod
Each mouse was trained on the RotaRod until it was able to walk on the rod rotating at 2 rpm for 1 min. On the test day, each mouse was made to walk on the rod while the speed of rotation increased from 2 to 20 rpm within the span of 180 s. The timer stopped when the mouse fell from the rod. Each test consisted of two trials, in which the latency to fall was recorded.
Y-maze
Each mouse was placed into the long arm of a three-arm horizontal maze where each arm was angled at 120°. A digital camera was positioned above the maze to track the movement of the mouse as it explored the maze for 7 min per session. The total number of times each mouse alternates its consecutive entry into a new arm was recorded, which was then divided by the total number of entries to obtain the percentage of alternating trials.
Biochemistry
Frozen hemibrains were ground using a mortar and pestle while maintaining a low temperature by the regular addition of liquid nitrogen. One volume of powdered tissue was further homogenized using an Ultra-Turrax T25 basic homogenizer in eight volumes of homogenization buffer containing 50 mM Tris-HCl pH 8, 5 mM sodium pyrophosphate, 30 mM sodium fluoride, 30 mM glycerol-2-phosphate, 1 mM phenylmethylsulfonyl fluoride (PMSF), 274 mM sodium chloride, 5 mM potassium chloride, 2 mM EDTA, 2 mM EGTA, and one complete-mini protease inhibitor tablet (per 10 mL) (Roche). The homogenate was further sonicated at 20% output for 10 s, and then centrifuged at 11,100 g for 20 min at 4°C. The supernatant was collected, and their protein concentrations were measured by BCA assay (Pierce). The resulting tissue pellets were stored in −80°C until required for analysis.
Immunofluorescence
Brain sections at 40 μm were mounted onto Superfrost microscope slides (Fisher Scientific) and allowed to dry overnight. The mounted sections were washed three times (10 min/wash) with 1X TBS (pH 7.4), then treated in 10 µg/ml proteinase-K in TBS (10 min at room temperature) following established methods.39–41 After washing in TBS (10 min/wash), the sections were incubated in 5% normal goat serum in TBST (0.1% Triton X-100) for 1 h at room temperature for blocking and permeabilization. The sections were then incubated with primary antibodies (pS129-α-synuclein 81A: 1:2000, Abcam #184674; anti-tyrosine hydroxylase: 1:2000, Pel-Freez Biologicals #P40101) overnight at 4°C. After washing in TBST (3 × 10 min), the sections were incubated with Alexa-Fluor conjugated secondary antibodies (1:1000) for 1 h at room temperature. Nuclei were counterstained with Hoechst 33342 (1:1000; ThermoFisher Scientific, H3570) for 15 min, and cover slips placed using VECTASHIELD Vibrance antifade mounting medium (Vector Laboratories, #H1700). The sections were imaged using an Axio Scan.Z1 slide scanner (Zeiss) at 10x magnification and image optimization using the Zen software.
Immunohistochemistry and stereological assessment of TH neurons
A subset of samples (n = 49) was analyzed for unbiased stereological counting of TH-immunoreactive neurons. Every eighth coronal section (–2.5 to −3.7 mm from bregma), for four sections per mouse, were examined. Briefly, after blocking with 3% hydrogen peroxide and 5% nonfat skimmed milk diluted in 0.25% Triton X-100, hemibrain sections were incubated with anti-tyrosine hydroxylase antibody overnight. After incubating with biotinylated secondary antibody, sections were developed with ABC reagent and chromogenic development by 3,3′-diaminobenzidine (DAB). Following dehydration and coverslip mounting, sections were imaged using 20x magnification on a Axio Scan Z1 scanner. Unbiased stereological counting of TH neurons in the substantia nigra par compacta (SNc) was conducted using the DAB-stained section images and a software-based method on ImageJ as described in a previous publication. 42 Systematic random grids were overlaid onto the SNc for placing the counting frame at the size of 2500 μm2. Only TH-immunoreactive neurons within the counting frame without touching the edges were counted. The average of the estimated numbers of TH-immunoreactive neurons of four sections was reported. The experimenter was blinded to the cohort throughout the analysis.
Immunoblot
Whole brain lysate was first boiled at 95°C for 5 min, then 25 µg of protein was resolved in a 4–15% tris/glycine Mini-PROTEAN gel (Bio-Rad) by electrophoresis. Proteins were transferred onto 0.45 µm nitrocellulose membrane, which were then blocked with 5% non-fat skimmed milk diluted in PBS-T containing 0.1% Tween-20. To analyze total O-GlcNAc, the membrane was incubated overnight with primary antibody (RL-2: 1:1000, Biolegend #677902; β-actin: 1:10000 Li-COR #926-42210) diluted in 1% non-fat skimmed milk. After washing in 1% nonfat skimmed milk, the membrane was incubated with IRdye-conjugated secondary antibodies (Li-COR) at RT for 1 h. After three washes in PBS-T, the membrane was scanned and analyzed using the Li-COR Odyssey Infrared Imaging System. To analyze monomeric and oligomeric α-synuclein, a subset of brain lysate (n = 64) was resolved in Mini-PROTEAN gel with reduced SDS (0.037%) in the running buffer according to a published native SDS-PAGE method. 43 After transferring, the membrane was fixed with 4% PFA for 30 min, followed by blocking in 5% BSA and overnight incubation with anti-α-synuclein antibody (Clone 42 1:1500, BD Bioscience #610787). The membrane was developed as previously described.
Enzyme-linked immunosorbent assay (ELISA)
Total α-synuclein was quantified using a commercial α-synuclein ELISA kit (Biolegend, #844101) following the manufacturer's instructions. Briefly, whole brain lysate was diluted until reaching the sensitivity range of the kit. Antibody pre-coated plate was washed four times with the provided washing buffer, then incubated with whole brain lysate overnight in 4°C. The plate was then washed four times with the washing buffer, after which it was incubated with biotinylated anti-α-synuclein primary antibody for 1 h at RT. After four washes, samples were incubated with streptavidin-HRP conjugate before developing with chemiluminescent substrate and immediately read using the Synergy Neo2 Microplate Reader (BioTek). Each sample was loaded in triplicate, and the average of three readings was recorded.
Image analysis
Since both substantia nigra (SN) and ventral tegmental area (VTA) contain a high density of dopaminergic neurons, both regions of interest were defined according to the anatomical landmarks referenced within the Allen Mouse Brain Atlas, and the presence of tyrosine hydroxylase (TH) immunoreactivity. Every tenth coronal section ranging from −2.5 mm to −3.7 mm from the bregma was sampled, with a total of three sections per mouse brain analyzed. The immunoreactivity of pS129-α-synuclein aggregates was quantified by the number and size of puncta.
At the time of analyzing TH immunoreactivity, we noticed a signal conversion artifact arising from leaking through of pS129-α-synuclein immunoreactivity, likely caused by the incubation of proteinase-K solution prior to antibody incubation. To remedy this issue, pS129-α-synuclein signal was first converted to a filter, then applied onto the TH images to exclude the overlapping region prior to signal quantification (Supplemental Figure 3E). Dopaminergic neurons were quantified by the number of TH positive cells, average fluorescence intensity, and area of TH immunoreactivity.
Statistical analysis
All statistical analyses and generation of graphs were conducted using the GraphPad Prism 9 software. All motor and cognitive assessments were analyzed using two-way ANOVA followed by Tukey multiple comparison and reported as ‘mean ± SD’. All biochemical analyses were performed with the expectation of protective effects resulting from OGA inhibition and using one-way ANOVA followed by Tukey multiple comparison and reported as ‘mean ± SEM’.
Results
Short-term treatment of Thiamet-G robustly increases O-GlcNAcylation in mThy1-hSNCA mice
To confirm Thiamet-G penetration into the brain of the Line 15 mice, we first conducted a preliminary study by treating ten Line 15 and WT mice with Thiamet-G at two months of age for two weeks. Using the pan-specific anti-O-GlcNAc antibody RL-2 in immunoblot analysis, we measured up to a 4-fold increase in brain O-GlcNAc following two weeks of 500 mkd Thiamet-G treatment (Supplemental Figure 1B, C). Since O-GlcNAcylation influences autophagic flux,44,45 we tested whether Thiamet-G treatment may affect the total levels of α-synuclein, which is known to be degraded by autophagy. To test this hypothesis, we performed ELISA using the whole brain lysates but found no significant effect on the level of total α-synuclein following Thiamet-G treatment (p = 0.2419) (Supplemental Figure 1D). No toxicity associated with Thiamet-G treatment was observed in any of the treated mice. These experiments confirmed the engagement of OGA in the Line 15 mouse brains by Thiamet-G and suggested that total α-synuclein is unaffected by elevating O-GlcNAcylation.
Chronic Thiamet-G treatment induces sustainable O-GlcNAcylation without affecting total α-synuclein
We next investigated the long-term effect of Thiamet-G treatment on α-synuclein expression in the brain by administrating Thiamet-G for ten months. Immunoblot analysis revealed 6-fold increases in brain O-GlcNAc arising from treatment with 500 mkd Thiamet-G (Figure 2A, B). 200 mkd Thiamet-G increased O-GlcNAc by 5 folds, although a post-hoc analysis did not suggest a concentration-dependent effect (p = 0.3057). These results were in general agreement with previous studies performed in our hands using the same analysis protocols but with bigenic tau-amyloid (TAPP) mice. 18 Using ELISA analysis, no significant changes in the levels of human α-synuclein measured in Line 15 mice were associated with Thiamet-G treatment (Figure 2C). Immunoblot analysis also confirmed no major change in the levels of total α-synuclein in both Line 15 and WT mice after Thiamet-G treatment (Supplemental Figure 2A, B). These results confirmed continuous engagement with OGA by Thiamet-G in Line 15 mice and suggested that Thiamet-G treatment induced no removal of α-synuclein.
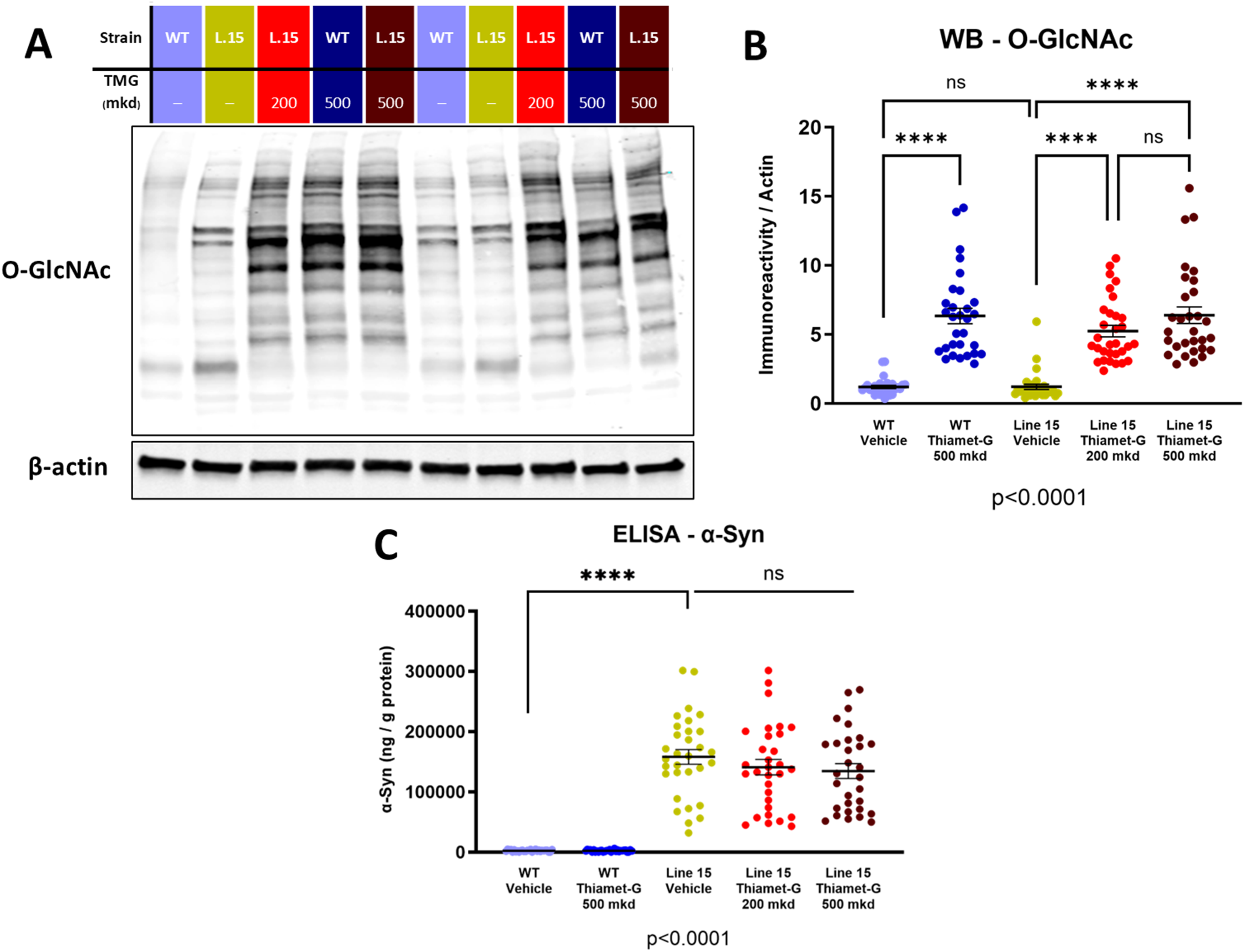
Chronic treatment of Thiamet-G increases global O-GlcNAc without decreasing overall α-synuclein in line 15 mice. (A) Representative immunoblot of whole brain lysate from Line 15 and WT mice receiving vehicle, 200 mkd or 500 mkd Thiamet-G treatment for 10 months detected by anti-O-GlcNAc antibody (RL2). β-actin was used as housekeeping control. (B) Densitometric quantification of the immunoblot. All values are normalized to the corresponding β-actin. (C) Quantification of total α-synuclein by ELISA analysis using the whole brain lysate of Line 15 and WT mice brains treated with Thiamet-G or vehicle. WT = WT vehicle (n = 29); WT 500 = WT Thiamet-G 500 mkd (n = 30); L.15 = Line 15 vehicle (n = 31); L.15 200 = Line 15 Thiamet-G 200 mkd (n = 31); L.15 500 = Line 15 Thiamet-G 500 mkd (n = 30). All results were analyzed in one-way ANOVA followed by Tukey multiple comparison. Error bar represents mean ± S.E.M. ****p ≤ 0.0001.
Thiamet-G reduces pS129-α-synuclein aggregates in the substantia nigra
Owing to the abundance of pS129-α-synuclein in Lewy bodies, inhibiting hyperphosphorylation of α-synuclein is considered one approach to slow the initiation of Lewy bodies assembly in the substantia nigra.35,46 Given the protective effects of O-GlcNAc modification of α-synuclein,32,35 we reasoned that inhibiting OGA with Thiamet-G would block α-synuclein aggregation in the brain of the Line 15 mice. By performing immunofluorescence analysis of mouse brain sections in regions spanning from the pre-frontal cortex to the cerebellum using the anti-pS129-α-synuclein antibody (clone 81A), we detected pS129-α-synuclein aggregates within the substantia nigra pars reticulata (SNr), which is consistent with previous reports showing proteinase-K resistant aggregates within the SNr of the mThy1-hSNCA mouse model starting at five months of age (Supplemental Figure 3A-D).40,47 We found that 500 mkd of Thiamet-G effectively abolished the number of pS129-α-synuclein aggregates within Line 15 mice (Figure 3A, D, F, G). The average size of the aggregates was also reduced following high dose Thiamet-G (Line 15 TMG 500 mkd vs. Line 15 Vehicle: p = 0.023) (Figure 3H). Remarkably, 200 mkd of Thiamet-G also strongly and significantly (p < 0.0001) reduced the number of pS129-α-synuclein aggregates (Figure 3D, E, G) and though a clear trend was observed, a post-hoc analysis did not suggest a significant concentration-dependent effect (Line 15 TMG 500 mkd vs. Line 15 TMG 200 mkd: p = 0.1951). Collectively, these results indicated robust inhibition of OGA leads to decreased deposition of pS129-α-synuclein aggregates within the substantia nigra.
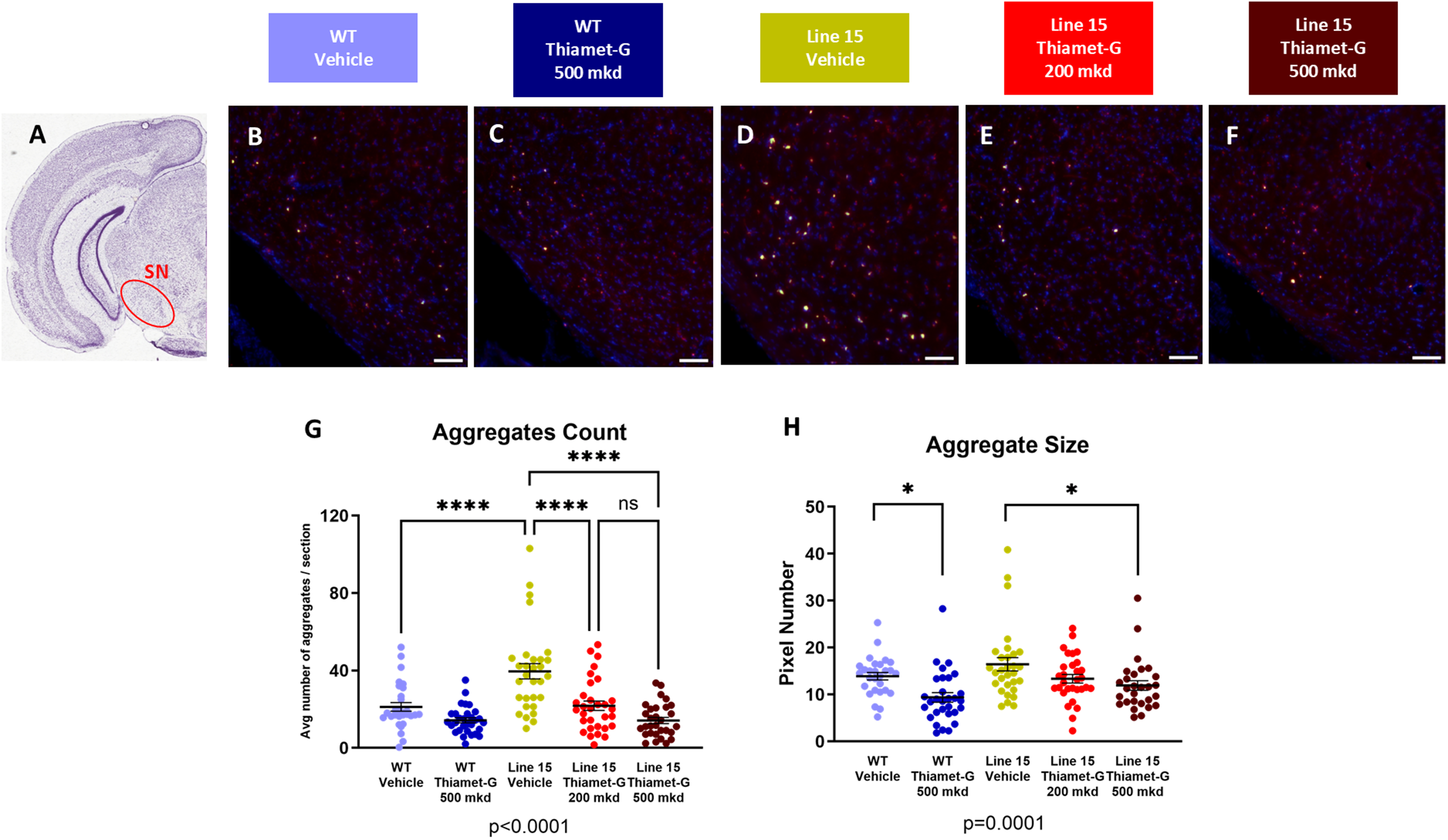
Chronic treatment of Thiamet-G drastically reduces α-synuclein aggregates in the substantia nigra. (A) Location of the substantia nigra in a coronal mouse hemibrain (atlas image from Allen Mouse Brain Atlas), using the emergence of the ventral dentate gyrus as anatomical landmark. (B-F) Representative immunofluorescence images of the substantia nigra examining pS129-α-synuclein aggregates (green puncta) in Line 15 and WT mice after chronic Thiamet-G or vehicle treatments. Nuclei are shown in DAPI (blue). Scale bar represents 100 µm. (G-H) Quantitative assessment of pS129 α-synuclein aggregates in the substantia nigra, indicating (G) the number and (H) the average size of aggregate. WT vehicle (n = 28); WT Thiamet-G 500 mkd (n = 30); Line 15 vehicle (n = 30); Line 15 Thiamet-G 200 mkd (n = 31); Line 15 Thiamet-G 500 mkd (n = 28). All results were analyzed in one-way ANOVA followed by Tukey multiple comparison. Error bar represents mean ± S.E.M. *p ≤ 0.05; ***p ≤ 0.001; ****p < 0.0001.
We also surveyed other brain regions to characterize changes in pS129-α-synuclein aggregation induced by Thiamet-G. Unlike the well-characterized Thy1-hSNCA (Line 61) mouse model of PD, which has been reported to accumulate α-synuclein aggregates in the thalamus and cerebellum in addition to the substantia nigra, 40 we did not consistently detect pS129-α-synuclein aggregates in these regions from the Line 15 mice (Supplemental Figure 4). We also found no pS129-α-synuclein aggregates within the frontal cortex, striatum, and hippocampus—regions that have been previously reported to accumulate pS129-α-synuclein protein in the Line 61 mice. 40 Native SDS-PAGE electrophoresis followed by immunoblot analysis also showed no major change in the total levels of oligomeric α-synuclein in the brain following Thiamet-G treatment (Supplemental Figure 2C, D). However, we consistently observed traces of pS129-α-synuclein aggregates within a TH-immunoreactive region between the sagittal midline and the substantia nigra (Supplemental Figure 5A). Several reports have discussed the progressive loss of dopaminergic neurons from the ventral tegmental area (VTA) in PD,48,49 which is a dopaminergic midbrain region within the mesolimbic pathway that reinforces reward-stimulated motor learning.50,51 Immunofluorescence of pS129-α-synuclein aggregates revealed a trend suggesting similar changes in the amount of pS129-α-synuclein aggregates as seen in the substantia nigra after Thiamet-G treatment (one-way ANOVA: p = 0.0596), although Tukey post-hoc analysis did not reach statistical significance (Line 15 500 mkg Thiamet-G vs. Line 15 vehicle: p = 0.3675) (Supplemental Figure 5B–G). Overall, our findings suggested Thiamet-G strongly suppresses pS129-α-synuclein aggregation in Line 15 mice within regions of the brain that are rich in dopaminergic neurons and relevant to PD.
Thiamet-G may improve locomotive performance in open field and pole tests amid early PD pathologies
Motor impairment is the most characterized clinical manifestation of PD. Progressive deterioration in motor and sensorimotor controls is observed within Line 61 mice as early as one month of age. 52 We used the open field and pole tests to determine the effect of chronic Thiamet-G treatment on voluntary locomotion in the Line 15 mice. Although two-way ANOVA analysis only indicated a weak trend on the interaction between cohorts over the time points tested (p = 0.0846), Tukey post-hoc analysis showed that Line 15 mice treated with the higher dose of Thiamet-G showed more navigation on the open field as early as after one month (p = 0.0188) and after seven month of treatment (p = 0.0002) (Figure 4A). This effect did not appear to be a result of anxiety, as both Line 15 and WT mice treated with Thiamet-G spent similar time within the central quadrant as mice treated with vehicle (Supplemental Figure 7A). We also challenged the mice on the pole test and found that in the fifth month of 500 mkd Thiamet-G treatment, Line 15 mice spent less time descending from the pole than the vehicle-treated cohort without falling (two-way ANOVA interaction: p < 0.0001; Tukey post-hoc: p = 0.0167) (Figure 4B). These results suggest that increasing O-GlcNAcylation may enhance locomotion of the Line 15 mice. We then assessed limb strength in both the wire hang and pole test (Figure 4C, D). All treatment groups performed poorly in both assessments (WH two-way ANOVA interaction: p < 0.0001; PT two-way ANOVA interaction: p = 0.0084). Tukey post-hoc analysis also suggested a dose-dependent effect on the wire hang assessment, when Line 15 mice treated with high dose Thiamet-G showed shorter latency of falling after just three months of treatment (p = 0.0003), whereas Line 15 mice treated with low dose Thiamet-G deteriorated significantly after five months of treatment (p = 0.0192). We also assessed motor coordination and balance by the rotarod test. Similarly, despite no clear interaction effect being found (two-way ANOVA: p = 0.0961), Line 15 mice treated with high dose Thiamet-G demonstrated a shorter latency of falling from the elevated rod compared to vehicle treated mice after three months of treatment (Tukey post-hoc: p = 0.0347) (Figure 4E). At the termination of the treatment and the time of tissue collection, we noticed that Thiamet-G-treated mice were larger in overall size than the vehicle-treated cohorts. We, therefore, reviewed the pre-treatment weight and monthly weight from the fifth to the ninth month of dosing, and found that both Line 15 and WT mice treated with Thiamet-G showed an up to 20% increase in body weight relative to the vehicle-treated cohorts of the same age, despite their pre-treatment weights deviating within 8% from mean (Supplemental Figure 6). We speculate that Thiamet-G treatment may increase feeding behavior in the Line 15 mice, though we could not establish this was the case as records on the amounts of food consumption were not collected. Nevertheless, our results suggested that chronic Thiamet-G treatment may improve locomotion within these transgenic mice, though its impact on preserving limb strength and motor coordination remains unclear due to this confounding effect of weight gain in the treated mice.
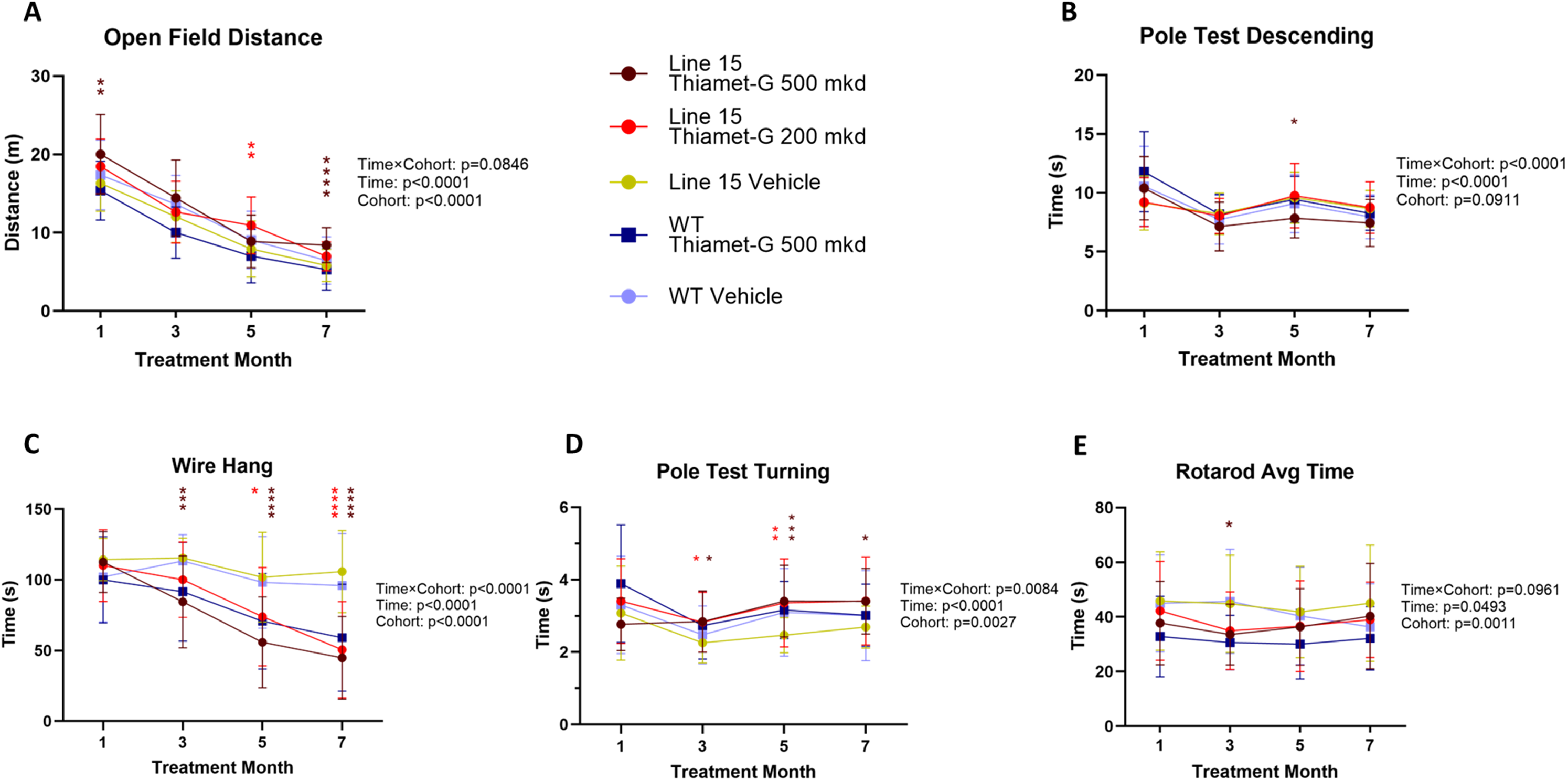
Thiamet-G may preserve locomotion within the course of treatment. (A-B) Longitudinal locomotive performances of the Line 15 and WT mice treated with vehicle, 200 mkd or 500 mkd Thiamet-G were assessed in open field and pole test. (A) The distance traveled in the open field arena was measured across four trials. Each point represents the average distance traveled by the mice in each cohort at one investigated time point. Both Line 15 and WT mice demonstrated habituation to the open field arena, as shown by a progressive decrease in exploration. (B) The time spent descending from the top of the pole was measured across four trials. Each data point represents the average time spent by mice in the same cohort at one investigated time point. (C-D) Longitudinal weight-bearing motor performances were assessed by wire hang and pole test. (C) The latency of falling from the sustaining wire was measured across four trials. Each data point represents the average latency of falling of the mice in each cohort at one investigated time point. (D) The latency of reorientation at the top of the pole was measured across four trials. Each data point represents the average latency of falling of the mice in each cohort at one investigated time point. (E) Longitudinal motor coordination was assessed by rotarod test. The latency of falling from the rotating rod was measured across four trials. Each data point represents the average latency of falling of the mice in each cohort at one investigated time point. WT vehicle (n = 28); WT Thiamet-G 500 mkd (n = 30); Line 15 vehicle (n = 30); Line 15 Thiamet-G 200 mkd (n = 31); Line 15 Thiamet-G 500 mkd (n = 28). All results were analyzed in two-way ANOVA (interaction effect of treatment and time in treatment) followed by Tukey's multiple comparison. Error bar represents mean ± S.D. For clarity purpose, only p-value(s) of comparisons between Thiamet-G and vehicle treated Line 15 mice at each time point was reported in the graphs. *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ****p < 0.0001.
Besides motor impairment, patients with PD also experience cognitive defects.53,54 To determine whether Thiamet-G improves cognition in these model mice, we monitored cognitive changes bimonthly using open field and Y-maze (Supplemental Figure 7). Throughout the treatment period, both Thiamet-G-treated Line 15 and WT mice demonstrated no significant changes in anxiety (OF two-way ANOVA interaction: p = 0.9022) and spatial memory (YM two-way ANOVA interaction: p = 0.3682). We also did not observe any cognitive abnormality in vehicle-treated Line 15 and WT mice. This may be attributed to the age of the mice used in this study and the nature of the Line 15 mice, which were not reported to manifest behavioral deficits or neurodegeneration within 12 months of age.40,55
Thiamet-G induces no adverse effect on the long-term physiological level of tyrosine hydroxylase
DA neurons generate dopamine from tyrosine by undergoing a serial conversion that involves the formation of the intermediate L-3,4-dihydroxyphenylalanime catalyzed by tyrosine hydroxylase (TH). Accumulation of Lewy bodies within the substantia nigra induces the death of DA neurons, which results in an inadequate supply of dopamine that causes impaired motor control. In vivo models and post-mortem analyses indicate that TH-deficiency is a disease driver for Parkinsonism pathophysiologies.41,56,57 Although Thiamet-G drastically reduced pS129-α-synuclein aggregates in the substantia nigra, the relatively modest changes in motor assessments suggested that the dopaminergic cascade might be largely spared by the effects of increasing O-GlcNAc in these mice at the ages examined (Figures 1 and 4). As expected, analysis of TH levels by immunofluorescence suggested no change in the number of TH-immunoreactive neurons in both Line 15 and WT mice following Thiamet-G treatment (Figure 5A–E, K). Neither TH intensity nor the total area associated with TH-positive neurons were affected by increased O-GlcNAcylation (Figure 5L, M). We also performed immunohistochemistry and stereological counting to assess TH-immunoreactive neurons in SNc, and our result confirmed no major changes on the population of DA neurons after chronic Thiamet-G treatment (one-way ANOVA p = 0.3794) (Figure 5F–J, N). No adverse effects on brain morphology were observed upon OGA inhibition, which is consistent with previous work showing that upregulation of O-GlcNAc does not alter the cell body, axonal coverage, and intensity of TH in dopaminergic neurons. 10
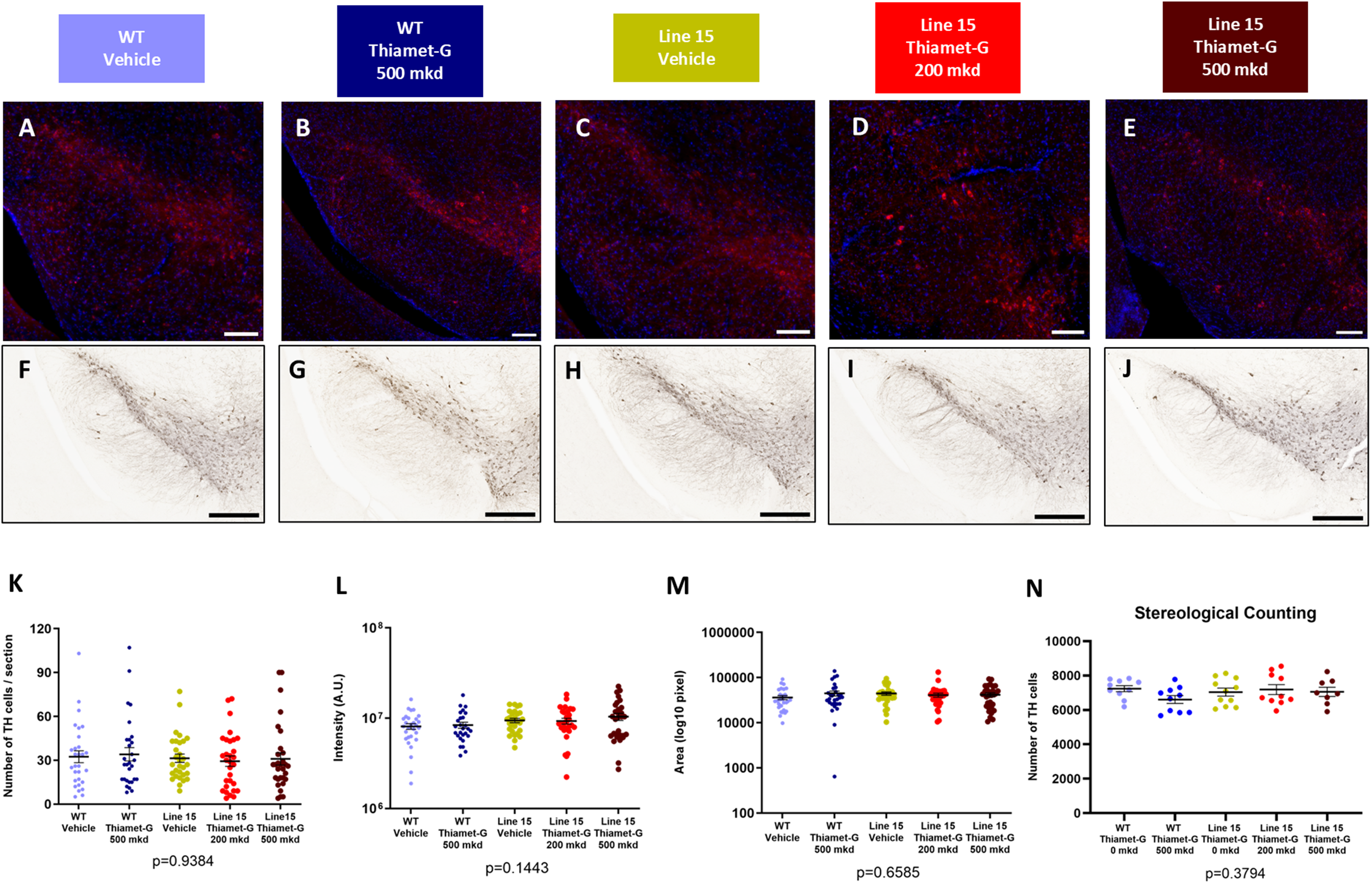
Thiamet-G does not affect the dopaminergic physiology in the substantia nigra. (A-E) Representative immunofluorescent images of TH immunoreactive neurons (red) in the substantia nigra. Nuclei were shown in DAPI channel (blue). Scale bar represents 200 µm. (F-J) Representative immunohistochemistry of TH immunoreactive neurons in the SNc. Scale bar represents 500 µm. (K-M) Quantification of TH immunoreactive neurons are reported for (F) the numbers of cell body, (G) fluorescence intensity, and (H) the size of TH-immunoreactive area representing both cell body and axon. (N) Stereological counting of TH neurons at the SNc using immunohistochemically stained images. For immunofluorescence: WT vehicle (n = 28); WT Thiamet-G 500 mkd (n = 30); Line 15 vehicle (n = 30); Line 15 Thiamet-G 200 mkd (n = 31); Line 15 Thiamet-G 500 mkd (n = 28). For stereological counting of TH neurons: WT vehicle (n = 10); WT Thiamet-G 500 mkd (n = 10); Line 15 vehicle (n = 11); Line 15 Thiamet-G 200 mkd (n = 10); Line 15 Thiamet-G 500 mkd (n = 8). All results were analyzed by one-way ANOVA followed by Tukey's multiple comparison. Error bar represents mean ± S.E.M.
Discussion
Thiamet-G combats amyloidogenic pathology
This study illustrates the neuroprotective effects of inhibiting OGA using the OGA inhibitor Thiamet-G in a mThy1-hSNCA mouse model of PD known as Line 15, which overexpresses human a-synuclein in the brain. Here we found a striking reduction in the levels of pS129-α-synuclein positive aggregates to levels seen in WT control mice. The high potency of the small molecule OGA inhibitor Thiamet-G can both acutely and chronically elevate brain O-GlcNAc through oral administration in drinking water. Accumulation of hyperphosphorylated α-synuclein, particularly the formation of phospho-serine 129, is often considered a progressive disease marker due to its prevalence within the brain of patients with advanced PD. 28 Postmortem analysis of brain tissues collected from individuals across all stages of Lewy body disorders as defined in the Unified Staging System shows a progressive increase in the levels of pS129-α-synuclein as the disease advances.58,59 Quantification of pS129-α-synuclein in the cerebral spinal fluid also shows a major increase relative to healthy individuals, with a statistically significant association with the severity of PD systems as defined by the Unified Parkinson's Disease Rating Scale (UPDRS). 60 It was also found that insoluble pS129-α-synuclein becomes detectable and gradually increased from stage IV onward according to a simplified eight-stages diagnostic scale for Lewy body dementia and PD.61,62 These clinical and postmortem studies collectively point to the accumulation of pS129-α-synuclein as PD progresses. On the other hand, the connection between the level of pS129-α-synuclein and neurodegeneration in transgenic wild-type α-synuclein overexpressing mouse models of PD remains elusive, as neuronal loss is rarely seen. 40 However, levels of pS129-α-synuclein progressively increase and condense into inclusions within the striatum and substantia nigra following intrastriatal injection of α-synuclein-PFF in wild-type rats.36,63,64 Such accumulation of pS129-α-synuclein as shown in these reports was also accompanied by a progressive loss of neurons shown in cresyl violet staining,36,63 as well as delayed motor impairment shown in grid walking and open field assessments. 64 Since neurodegeneration is mostly apparent in PFF injection models, yet not in wild-type α-synuclein overexpression rodent models, there remains some debate about the neurotoxicity of pS129-α-synuclein. 65 On balance, however, collectively the data point to pS129-α-synuclein participating in the exacerbation of synucleinopathy, and this species is therefore exploited as a disease biomarker in preclinical and clinical assessments of PD progression. Semisynthesis of a-synuclein with O-GlcNAc at Threonine 72 was shown to inhibit in vitro phosphorylation of α-synuclein at Ser129 by three relevant kinases and reduce fibrillation of α-synuclein both in vitro and within cultured cells. 32 In the present study, we confirmed that chronic increase in O-GlcNAcylation induced by Thiamet-G effectively suppresses aggregation of pS129-positive α-synuclein within the substantia nigra without affecting the overall level of α-synuclein protein nor adversely impacting TH. Our findings are consistent with earlier preclinical studies using AD model mice, where OGA inhibition reduced the formation of argyrophilic neurofibrillary tau tangles and amyloid plaques.9,18,24,26,66,67 Moreover, they are consistent with recent reports showing that both genetic enhancement of O-GlcNAc in PFF-injection model mice and OGA inhibition in Line 61 mice, using the OGA inhibitor ASN90 that differs from Thiamet-G, diminish levels of pS129-positive α-synuclein protein in the hippocampus. 67 Our work using a different mouse model (Line 15) supports and extends these findings to show that increasing O-GlcNAc by OGA inhibitors can reduce PD pathology within disease-relevant areas of the brain including the substantia nigra.
While the observed therapeutic benefits could stem from increased O-GlcNAc modification of a-synuclein, it is also possible that such modification also antagonizes the phosphorylation of serine-129. However, it is also possible that other cellular mechanisms may contribute to the observed effects. Some reports have suggested that elevating O-GlcNAc leads to increased autophagic flux, which in both cultured neurons and animal models of neurodegenerative diseases, led to decreased levels of amyloidogenic tau.12,45,68–70 Alternatively, increased O-GlcNAc also appears to reduce uptake of α-synuclein-preformed-fibrils, 34 which may be one mechanism leading to decreased pathological spread of α-synuclein aggregates that have been shown to propagate to healthy neurons in a prion-like manner throughout the brain.71,72 Finally, increased O-GlcNAc has also been shown to attenuate microgliosis and astrogliosis in the brains of preclinical mouse models of AD, resulting in reduced amyloid-β plaques and improved cognition.9,73 Though our studies are unable to distinguish which of these mechanisms drive the neuroprotective effects of OGA inhibition, the protective effects observed here are consistent with these possible mechanisms and support more focused mechanistic studies to understand which processes drive the neuroprotective effects in PD mouse models as seen here.
Effects on early α-synuclein pathogenesis by Thiamet-G
One limitation associated with using the Thy1-hSNCA mouse strain for modeling PD is the late emergence of DA neurodegeneration and consequent decline in motor and cognitive performance in this and related strains such as the Line 61 mice, which have been better characterized.40,52,74 To the best of our knowledge, only dramatic AAV-driven overexpression of wild-type α-synuclein or α-syn-PFF injection can cause clear DA neurodegeneration in mice. 75 Despite this limitation, we opted to use the murine Thy1-hSNCA (Line 15) mouse line for two main reasons: first, bulk maintenance of the Line 15 cohorts is relatively easy; second, premature mortality due to genetic manipulation, which is a problematic aspect of the A53 T mouse model of familial PD, is uncommon. Notable in this regard is that no mouse was withdrawn from this study due to disease pathology, and therefore strong statistical power could be obtained within the dataset when comparing performances between experimental trials, despite no extensive behavioral characterization of this mouse strain being available at the time of the study. Another limitation with using the Thy1-hSNCA mouse model is the differential changes in dopamine turnover and motor phenotype between various sub-strains. Notably, while preparing this manuscript, two reports by different groups have also demonstrated a lack of locomotive deficit in the Line 15 mice. For instance, Polinski and colleagues found that in the reported gripping test, the Line 15 mice showed a longer duration of gripping than their WT littermates. 55 They also found no difference in the numbers of TH-positive neurons in the substantia nigra of the Line 15 mice, in agreement with our vehicle-treated Line 15 control. However, dopamine levels in the Line 15 mice were found to be higher relative to WT control for up to 8 months of age. 55 On the other hand, Nimmo et al. conducted pole test and wire hang assessments on the Line 15 mice and WT littermate controls, and they also found no major change in the latency of descending and falling between cohorts. 76 Although these findings on the locomotive phenotype of the Line 15 mice are largely in agreement with our observation, Nimmo and colleagues found that Line 15 mice demonstrated longer traversing duration across the challenging beam test at 6 months of age, suggesting that the Line 15 mouse model develops compromised fine motor control amid an early stage of PD pathology. 76 Therefore, lacking robust assessment for examining fine motor coordination without influenced by weight as a confining factor may attribute to the absence of behavioral disease phenotypes as shown in our report. In contrast, Line 61 mice are known to manifest early motor deterioration albeit the absence of the loss of DA neurons. As early as one month of age, mice show significant increases in falling during wire hang assessment relative to their littermate controls. 52 At two months of age, mice took longer and showed increased errors to traverse in a challenging beam test, reduced cotton grasping indicative of worse fine movement, and reduced grooming behavior. 74 Lam et al. also found that at 14 months of age, Line 61 mice explored less in the open field assessment. 77 These differences in motor deficits between the Line 15 and Line 61 sub-strains of the Thy1-hSNCA mouse model may perhaps arise from the different levels of α-synuclein overexpression, as the Line 61 mice was found to harbor higher expression of human α-synuclein compared to the Line 15 mice. 78 Although the physiological function of α-synuclein remains unclear, it is largely believed that α-synuclein plays a pivotal role on regulating the trafficking of vesicles that constitutes the primary mechanism for the release and reuptake of neurotransmitters, possibly by facilitating the docking of exocytic vesicles at SNARE complexes and reformation of clathrin-mediated endocytic vesicles at presynaptic sites.79,80 An increase in the level of α-synuclein driven by the Thy1-promoter, which predominantly drives overexpression in the cortex, may spread to the midbrain that leads to a substantial increase in the abundance of extracellular α-synuclein at the substantia nigra, or even the overall basal ganglia pathway, which may impact the equilibrium of the synaptic vesicle dynamics. Hence, we speculate that even higher levels of α-synuclein expressed in young Line 61 mice perhaps accumulate at the extracellular matrix of synapses and perturb signal transmission by promoting reuptake of dopamine, which may lead to premature motor deficits that are characteristics of the Line 61 but not seen in the Line 15 mice, which overexpress lower levels of α-synuclein. Since we did not observe any locomotor deficits in our Line 15 cohort compared to WT mice, increases in locomotion induced by Thiamet-G (Figure 4A) may arise from strengthening the integrity and synaptic functionality of the basal ganglia pathway, 10 instead of inducing anxiety, which was supported by our open field assessment (Supplemental Figure 7A).
In our study, we opted to focus on the early window of α-synuclein pathology during which α-synuclein aggregates in order to ensure the study was adequately powered to observe protective benefits on biochemical markers. Accordingly, the loss of DA neurons and motor dysfunction caused by the pronounced accumulation of α-synuclein aggregates as seen in the A53 T and PFF-injection PD mice models were not expected to be well advanced in our mouse model. Indeed, mThy1-hSNCA mice overexpressing wild-type α-synuclein appear to demonstrate resilience against the toxicity of endogenously formed aggregates; however, the Line 61 mice were reported to undergo neuronal loss in the hippocampus by eight months of age, preceded by increases in both soluble and insoluble α-synuclein as early as three months of age.52,81 Another group has also reported the absence of α-synuclein inclusions or Lewy bodies in the brains of Line 15 mice at six months of age. 76 In this report, we examined multiple brain regions spanning from the frontal cortex to the cerebellum of the Line 15 mice at twelve months of age, and only consistently detected pS129-α-synuclein aggregates in dopaminergic regions without observable loss of TH-positive neurons (Figure 5, Supplemental Figures 3 and 4). These findings collectively indicate that the Line 15 mice exhibit progressive but early-stage Lewy bodies formation at the age of one year, supporting their use as a suitable mouse model for identifying disease-modifying interventions against the early pathology of PD and synucleinopathy. Nevertheless, we speculate that a similar study using the Line 61 mice strain may manifest further therapeutic benefits from Thiamet-G treatment. Indeed, as noted above, ASN90, a novel OGA inhibitor developed by Permanne and colleagues, improved motor performance of the Line 61 mice in the challenging beam assessment following 12 weeks of chronic dosing. 67 Immunohistochemistry also revealed a reduction of pS129 α-synuclein protein in the hippocampus and astrogliosis in the cortex. Together with our findings using Thiamet-G, these data indicate that OGA inhibition could broadly offer therapeutic benefits against the toxicity of monomeric and aggregated α-synuclein.
Conclusion
In conclusion, our study shows that sustained pharmacological inhibition of OGA in the Line 15 mouse model over ten months leads to a significant suppression of α-synuclein aggregation within dopaminergic regions. The robust attenuation of α-synuclein aggregation and upregulation of O-GlcNAc in a dose-dependent manner, where the low dose also exerts significant benefits, supports the exploration of lower doses of OGA inhibitor to achieve major benefits against α-synuclein aggregation. Notably, this observation is also consistent with the observation that the haploinsufficiency of OGA provides considerable protective benefits against necroptosis in amyloid model mice. 9 Such optimization of inhibition of OGA in preclinical models and even humans is now possible due to the development of positron emission tomography agents targeting OGA.82–85 In addition, the results observed here are consistent with, both supporting and extending, previous findings using OGA inhibitors in different transgenic models. Notably, the recent T2 T initiative led by the Michael J Fox Foundation also identified OGA as one of the top preclinical targets that merit increased preclinical attention. OGA was selected from among 280 proposed target candidates as a promising target that could deliver a disease-modifying therapy for PD. 37 Altogether, these findings strongly support the introduction of OGA inhibition as a disease-modifying treatment candidate for PD, which may soon be feasible given the advance of OGA inhibitors into the clinic including Phase I and II clinical trials in AD.86,87
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251410291 - Supplemental material for Pharmacological inhibition of O-GlcNAcase reduces pS129-α-synuclein positive aggregates in the substantia nigra of mThy1-hSNCA mice
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251410291 for Pharmacological inhibition of O-GlcNAcase reduces pS129-α-synuclein positive aggregates in the substantia nigra of mThy1-hSNCA mice by Jefferey Yue, Bryan Jones, Kim H Tran, Matthew Deen, Viktor Holicek, Wai Hang Cheng, Mateusz Michalik, Sarah Power, Cheryl L Wellington, Neil V Watson and David J Vocadlo in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We thank Dr Omid Tavassoly for help in initiating this study and assisting with grant preparation, performing dosing of mice, tissue collection, sectioning of brain tissues, and immunohistochemical staining. We thank Dr Ralph E. Mistlberger for resources in supporting tissue collection. We thank Dr Xiaoyang Shan for providing guidance on immunohistochemistry and data analysis. We thank the technical staff at the Simon Fraser University Animal Care Facility for the maintenance and husbandry of the animals used in this study.
ORCID iDs
Ethical considerations
Protocols for animal experiments were approved by the University Animal Care Committee of Simon Fraser University (Approval no. 1240C), in compliance with the Canadian Council on Animal Care guidelines for the care and use of laboratory animals.
Consent to participate
Not applicable
Consent for publication
Not applicable
Funding
This work is funded by the W. Garfield Weston Foundation (Weston Brain Institute) (RR162076).
Declaration of conflicting interests
D.J.V is a cofounder of and holds equity in the company Alectos Therapeutics. D.J.V serves as CSO and Chair of the Scientific Advisory Board of Alectos Therapeutics. D.J.V may receive royalties from SFU for commercialization of technology relating to OGA inhibitors.
Data availability statement
The data supporting the findings and the macros used for image quantification are available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
