Abstract
Background
A key challenge in trials targeting disease modification in Parkinson's disease (PD) is the lack of sensitive, precise, and patient-relevant outcome measures. Digital mobility outcomes (DMOs), captured using body-worn devices, offer a novel, objective means to assess real-world gait and mobility—domains often impaired early in PD. The Mobilise-D consortium was established to develop and validate DMOs in PD and other conditions.
Objective
To describe DMOs in a large, representative international cohort of individuals with PD and compare to controls and across disease stage; and to determine compliance and feasibility.
Methods
As part of the Mobilise-D Clinical Validation and Extension Studies, real-world mobility of individuals with PD (n = 601) and matched controls (n = 232) was assessed using a single wearable device for seven days. Data were processed to yield 24 technically validated DMOs, representing different domains of real-world walking and mobility performance.
Results
DMO data were available for 531 PD and 221 controls. Significant differences between the groups were observed in 20 of 24 DMOs. Compared to controls, PD participants exhibited shorter daily walking duration and lower step counts, walking at a higher cadence and in fewer walking bouts per day. Findings also varied by disease severity, with differences observed particularly between controls vs. mild (Hoehn and Yahr stage I-II) and mild vs. moderate (Hoehn and Yahr stage III) disease. Compliance rates were high.
Conclusions
Distinct DMO patterns across PD severity and between PD and controls support their utility as sensitive, scalable outcome measures for future clinical trials and therapeutic development.
Plain language summary
Difficulty with walking is regularly highlighted by people with Parkinson's as one of their most troublesome symptoms. To identify treatments with the potential to slow or halt disease progression, researchers need to track changes to mobility in a way that is accurate and meaningful to people with Parkinson's. Currently, many measures used in clinical trials often provide only a snapshot on one day. This does not accurately reflect how fluctuations in mobility from day-to-day can affect people. One promising method instead is using wearable devices that monitor how people move during their daily lives.
A large international research group, the Mobilise-D consortium, studied over 600 people with Parkinson's and more than 200 people without the condition across five sites in Europe. Participants wore a small device either on their lower back or on a belt around their waist for a week. This tracked their walking patterns and activity levels.
The results showed clear differences in how people with Parkinson's moved compared to those without the disease. For example, people with PD walked less, took fewer steps, and had different walking rhythms throughout the day. These differences generally became more noticeable as the disease became more advanced.
Importantly, participants were happy to wear the devices and followed the study instructions well, showing this method is practical for future use. Digital measures of mobility are therefore able to measure changes in mobility and walking in Parkinson's disease. This may be a useful tool for improving development and real-world testing of new treatments.
Keywords
Introduction
Parkinson's disease (PD) is the fastest-growing neurological condition in the Western world, affecting over 8.5 million people globally. 1 Over the past 30 years, the global burden of PD has more than doubled due to an ageing population, optimised diagnosis and management, and environmental factors. 2 The consequences of PD to the individual, family and society are wide-ranging, ultimately leading to loss of independence and reduced quality of life, in addition to the significant associated healthcare costs. 3 In the absence of a cure, management is largely symptomatic. The early and profound impact on gait and mobility4,5 is recognised by people with Parkinson's, who highlight problems with walking amongst their most bothersome symptoms,6,7 leading to calls to prioritise research to mitigate mobility loss. 8 Apace with this, there is increasing interest in trials aimed at modifying the disease, with several promising studies on the horizon.9,10
One of the main challenges for disease-modifying and therapeutic trials in PD is the need for better outcome measures, that ideally can capture change over a shorter duration of time and with greater precision and sensitivity. 11 Existing clinical tools to depict PD outcomes, such as the Movement Disorder Society – Unified Parkinson's Disease Rating Scale (MDS-UPDRS 12 ), are limited by the nature of their one-off assessment, resource intensity, and lack of sensitivity.13,14 Objective measures that track changes, predict outcomes, and even help identify PD early for better patient stratification are urgently needed to complement existing clinical standards.15,16 In addition, when proposing novel measures for use in clinical trials, regulatory requirements (such as those from the Food and Drug Administration (FDA)) emphasise the need for these outcomes to be meaningful to patients. 17
As gait is impacted early and deterioration progresses over time,4,5 digital health technologies which capture measures of walking activity and gait that may complement conventional tools for diagnosis, monitoring and prognosis, offer a promising solution to these limitations. 18 More specifically, low-cost wearable devices can be used to quantify a wide range of digital mobility outcomes (DMOs), including gait speed, stride length, cadence, number of walking bouts and steps per day. 19 Digital measures of mobility captured during brief walking tests carried out in the clinic can discriminate PD, track disease progression, response to therapy and falls risk.20–25 While less well-developed, DMOs can also be collected passively in the real-world as people engage in their daily routines. 18 This offers an even greater opportunity and potential benefits, by delivering an inclusive approach that widens access to research and care and empowers remote assessments; by capturing fluctuations typical of PD in a manner that brief tests are unable to13,14; and by providing a direct and objective measure of mobility performance in daily life. DMOs can therefore encapsulate an aspect of health identified as fundamentally important to patients, as well as provide insight into mobility loss, that was previously unobtainable.18,26 Early work using remote monitoring has shown promise to detect, monitor and predict events in PD.20,24,2527–30 Collectively, this provides valuable insights into both the risk and progression of PD, as well delivering precise measures of gait. However, before these outcomes can be adopted widely in research and clinical care, further work is needed to establish their validity from a technical, clinical and patient acceptability perspective.31,32
To address these knowledge gaps, the Mobilise-D project (GA No. 820820, www.mobilise-d.eu) was funded with the aim of developing and validating real-world DMOs for use in clinical trials in PD and other long-term health conditions (multiple sclerosis, chronic obstructive pulmonary disease and proximal hip fracture). 33 This multi-centre longitudinal international study involved the use of wearable devices to objectively measure mobility and gait in real-world settings and represents the largest study of digital mobility in PD to date. The Mobilise-D Extension study (MJFF-022735) provided an age- and sex-matched control group to inform progression in normal ageing, record change in DMOs, and offered an additional follow up timepoint for PD participants.
The objectives of this paper are to provide first insights using the baseline data into real-world mobility through technically validated DMOs in a relatively large and representative international cohort of individuals with PD and healthy controls, and to raise awareness of the data, which will be made available to the wider community for further knowledge gain (see https://zenodo.org/records/13899386 for open access Technical Validation Study data as an example, and https://github.com/mobilise-d for the validated pipeline/algorithms). The main aims were to quantify real-world DMOs in PD and compare to controls and across disease stage; and to determine the feasibility and compliance of measuring mobility within this study using two devices. Taken together, these will provide an important step towards the implementation of DMOs as objective endpoints into future clinical trials and clinical practice.
Materials and methods
Study design
Individuals with PD were recruited and assessed as part of the EU Innovative Medicine Initiative (IMI) funded longitudinal observational Mobilise-D Clinical Validation Study (CVS) (ISRCTN12051706) across five European sites (Newcastle, UK; Kiel, Germany; Erlangen, Germany; Leuven, Belgium; Tel Aviv, Israel) between April 2021 and May 2022. 33 Participants were followed up every six months for a total of 24 months. An additional follow up period at 36 months also took place, with separate funding (Mobilise-D Extension Study, ClinicalTrials.gov ID NCT05874739). Age- and sex-matched controls were recruited as part of the Mobilise-D Extension Study (NCT05874739) across the same sites between July 2023 and June 2024 and followed up once at 12 months. They were identified through Public and Patient Networks and Engagement teams and banks of volunteers already known to clinical sites. Control participants could also be relatives or friends of PD participants. This paper presents the baseline data of the controls and PD cohort.
The study protocol has been published outlining the study's design and deployed assessments in detail.33,34 Briefly, inclusion criteria for the patients and controls comprised: anticipated availability for study duration; capacity to consent to the study; willingness to wear a wearable sensor; and ability to read and write in the first language of the respective country. Exclusions for both cohorts were those with a significant cardiac history or stroke within the last three months; active treatment for malignancy; and significant psychiatric disorder. Specific PD inclusions were a diagnosis of PD according to the recent Movement Disorder Society criteria 35 ; Hoehn and Yahr (H&Y) stage 1–3; and ability to walk four metres independently with or without a walking aid. Exclusions were those with an atypical parkinsonian syndrome, vascular or drug-induced parkinsonism. Additional inclusion criteria for controls were aged 50 years or over, and ability to walk four metres independently without walking aids. Exclusion for controls comprised a neurological or orthopaedic impairment that significantly impacted on gait; history of dementia/significant cognitive impairment or movement disorder; and those with a clinical diagnosis of PD, chronic obstructive pulmonary disease, proximal hip fracture or multiple sclerosis.
Ethics
The Mobilise-D CVS and Extension Study were sponsored and coordinated by The Newcastle upon Tyne National Health Service Foundation Trust, UK. Approvals from ethics committees of the five recruiting sites were obtained for both the CVS and Extension Study (CVS: Newcastle (London Bloomsbury REC. Ref: 20/PR/0792); Kiel (EC of the Medical Faculty of the Christian-Albrechts University of Kiel. Ref: D 630/20); Erlangen (EC of the Friedrich-Alexander University Erlangen-Nuremberg. Ref: 535_20 B); Leuven (EC Research UZ/KU Leuven. Ref: S64977); and Tel Aviv (Tel Aviv Sourasky Medical Centre. Ref: 0551-19-TLV). MJFF Extension Study: Newcastle (Health and Social Care REC B. Ref: 23/NI/0046); Kiel (EC of the Medical Faculty of the Christian-Albrechts University of Kiel. Ref: D 475/23); Erlangen (EC of the Friedrich-Alexander University Erlangen-Nuremberg. Ref: 23-166-B); Leuven (EC Research UZ/KU Leuven. Ref: S67777); and Tel Aviv (Tel Aviv Sourasky Medical Centre. Ref: 0551-19-TLV). Written informed consent was obtained from all participants.
Clinical assessment
Demographic and clinical variables collected for both PD and controls included age, gender, height, weight, education, employment status, use of walking aid and falls history. The Late-Life Function and Disability Instrument (LLFDI)36,37 and prospective fall frequency (using falls diaries) were the primary outcome measures in the full Mobilise-D cohort and allowed for the sample size calculation. 33 Clinical outcome assessments included 6-min walk test (6MWT), timed up and go (TUG) test, 38 Short Physical Performance Battery (SPPB), 39 mini Balance Evaluation Systems Test (mini-BEST), 40 and hand grip strength measured using a hand-held dynamometer. Patient reported outcome measures included Short Falls Efficacy Scale International (Short FES-I), 41 the Functional Assessment of Chronic Illness Therapy (FACIT) Fatigue scale, Euro-Qol (EQ-5D) 42 and the Patient Health Questionnaire (PHQ-2). Cognition was assessed using the Montreal Cognitive Assessment (MoCA). 43 The Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) parts I-IV, 12 H&Y scale 44 and New Freezing of Gait Questionnaire (NFOG-Q) 45 comprised PD specific measures. Mean levodopa equivalent daily dose (LEDD; mg/day) was also calculated for each PD participant. 46 Gait and physical performance tests were performed ‘on’ medication.
Digital mobility assessment
Mobility performance was assessed directly using a single wearable device (either McRoberts MoveMonitor (MM)+ (McRoberts B.V., The Hague, The Netherlands) or Axivity AX6 (Axivity Ltd, Newcastle Upon Tyne, UK)). Axivity AX6 was body fixed using a custom designed adhesive patch, whilst the McRoberts MM + was a device fixed to the body using a belt. These were worn continuously for 7 days, with the AX6 device instructed to be worn both day and night. Both devices were metrologically equivalent and consisted of a six Degrees of Freedom (DoF) inertial measurement unit with the following configuration: triaxial accelerometer with a range of ±8 g and a resolution of 1 mg, triaxial gyroscope with a range of ±2000 degrees per second (dps) and a resolution of 70 millidps, sampling frequency 100 Hz.
Calculation of digital mobility outcomes
To derive the DMOs, the sensor data were first standardised according to the protocol outlined in Palmerini et al. 47 Identification of walking bouts (WBs) and extraction of gait features (initial contact, cadence estimation, stride length estimation) for these bouts were carried out using the technically validated Mobilise-D processing pipeline as described in Mico-Amigo et al. 19 and Kirk et al. 48 In keeping with recommendations of the latter work, walking speed and stride length DMOs in WBs < 10 s were excluded. 48 Only the technically validated DMOs are described in this paper. A WB was defined as a continuous sequence with at least two consecutive strides of both feet. 48 This enabled the calculation of WB duration, number of strides, cadence, walking speed, stride length and stride duration for each WB. The 90th percentile (P90) values of WB duration, walking speed and cadence were calculated and are hereafter referred to as the maximum WB duration, walking speed and cadence, respectively.
Compliance and feasibility were determined by number of valid and complete datasets. A technically valid day of real-world digital mobility assessment requires >12 h wear time during waking hours (07h00-22h00). A reliable week of digital mobility assessment requires ≥ three days irrespective of weekend days. 49 Data that did not meet the predefined technically valid day and reliable week criteria were removed. Bout level DMOs were then processed according to a prespecified protocol 50 to calculate aggregated DMOs at the weekly level (see Table 1 for the weekly aggregated DMOs).
List of technically validated digital mobility outcomes (DMOs) obtained in mobilise-D.

WB = walking bout, defined as a continuous sequence with at least two consecutive strides of both feet48; P90 = 90th percentile; hereafter in the manuscript referred to as ‘maximum’.
Statistical analysis
Data were analysed in SPSS (v 29.0.1.0) using the PD (v6.0, 20 December 2024) and control database (v1.0, 31 October 2024). To describe the clinical characteristics and DMOs of the PD and control cohort, mean and SD for continuous variables with normal distribution were used; for non-normally distributed variables, the median [minimum, maximum] was reported. Group differences in categorical outcomes were evaluated using chi squared tests. Comparison of demographics, clinical outcomes and DMOs in PD vs. controls was made using independent samples t-test (parametric data) and Mann-Whitney U test (non-parametric data); comparison was then stratified by disease severity according to H&Y (H&Y I and II classified as mild disease; H&Y III as moderate) using Kruskal-Wallis test (Bonferroni corrections were used to account for multiple comparisons). This process was repeated when stratified by site. Statistical assumptions including Levene's test for equality of variances were reviewed and two tailed significance values were reported. The pre-defined level of significance was set at p < 0.05. Descriptive statistics were used to depict feasibility and compliance of devices, as determined by number with complete clean DMOs over the minimum data set.
Results
Sample overview
A total of 601 PD participants were included in the baseline analysis (Figure 1), of whom 65% were male, with a mean (SD) age of 65.7 (9.5) years (Supplementary Table 1). Mean disease duration was 6.9 (5.1) years, with a median LEDD of 543 mg/day. As per the inclusion criteria, participants had mild to moderate disease, with mean MDS UPDRS part III score of 26.6 (12.5); 67% were H&Y stage 2 and 18% freezers, the latter based on a positive response to Part I of NFOG-Q. Median SPPB was 10, median TUG was < 10 s and fear of falling scores were low (median Short FES-I 9). Despite this, over 35% reported one or more fall in the last 12 months, with over 45% reporting resultant injury. Mean MoCA score was 26, with low levels of positive screening for depression on the PHQ-2, and moderate involvement or limitations in activities as scored by the LLFDI Disability Component (median LLFDI disability-frequency score 55 and LLFDI disability-limitation score 68).
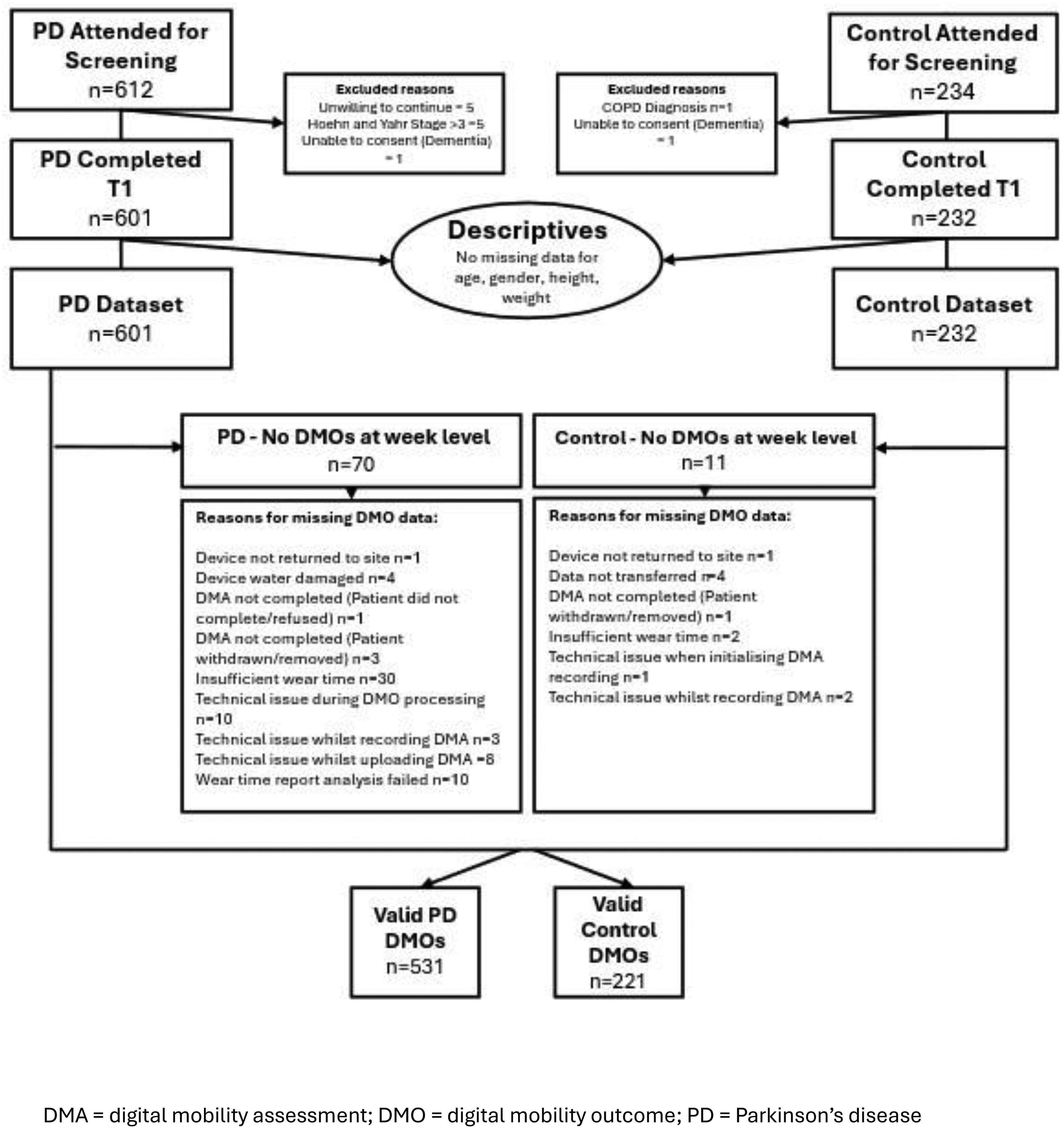
Recruitment flowchart for mobilise-D and mobilise-D extension study.
The control group (n = 232) was well-matched to the PD participants with respect to age, sex, employment status, height, weight, and BMI (Supplementary Table 1). There was minimal use of walking aids both indoors and outdoors in the controls compared to the PD participants, in addition to significantly fewer controls reporting one or more fall in the previous year (14.2% vs. 35.4%, p < 0.001). Handgrip strengths for both men and women were significantly higher for controls compared to PD. Six-minute walk distance was larger in controls, with higher scores on the Mini-BEST and a faster TUG. Control participants reported lower fear of falling, had higher cognitive scores, lower fatigue levels and less disability limitation as measured by the LLFDI.
Digital mobility outcomes in PD and control participants
Of the 601 PD and 232 control participants, valid weekly-level DMOs were available in 531 and 221, respectively (Figure 1). These were well matched for age, sex, height, weight and BMI (Table 2), and were representative of the cohorts as a whole in terms of clinical characteristics (Supplementary Table 2). Analogous to the cohorts as a whole and as expected, PD participants with DMO data available had poorer function in terms of self-reported falls, handgrip strength, measures of physical function, cognitive scores, fatigue levels, depression screening scores and functional disability compared to the control cohort (Table 2).
Baseline clinical characteristics of control and PD cohort with DMO data available (≥3 days).
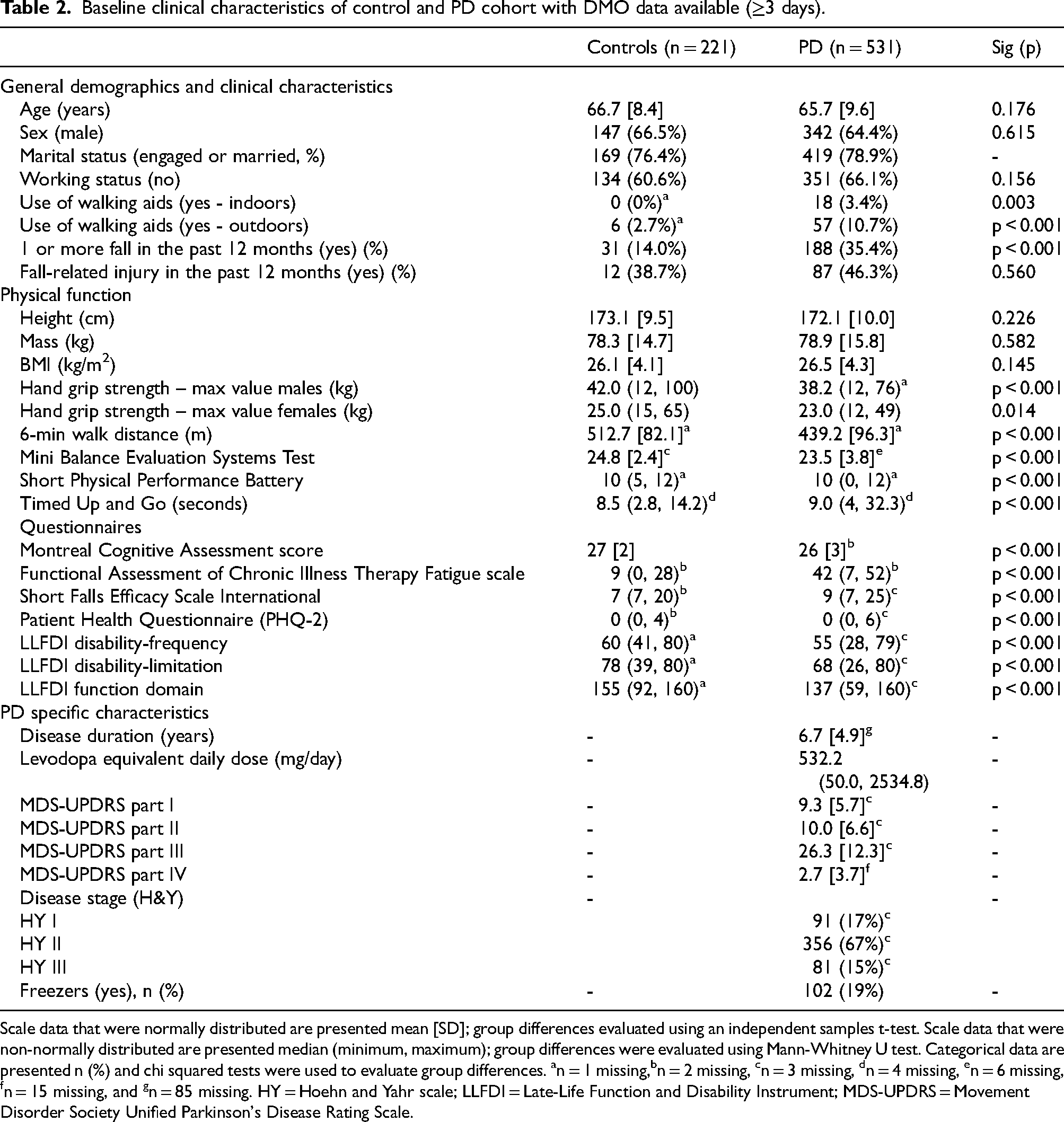
Scale data that were normally distributed are presented mean [SD]; group differences evaluated using an independent samples t-test. Scale data that were non-normally distributed are presented median (minimum, maximum); group differences were evaluated using Mann-Whitney U test. Categorical data are presented n (%) and chi squared tests were used to evaluate group differences. an = 1 missing,bn = 2 missing, cn = 3 missing, dn = 4 missing, en = 6 missing, fn = 15 missing, and gn = 85 missing. HY = Hoehn and Yahr scale; LLFDI = Late-Life Function and Disability Instrument; MDS-UPDRS = Movement Disorder Society Unified Parkinson's Disease Rating Scale.
Real-world mobility is described in detail in Table 3 and Figure 2. The results can be divided into broader measures of walking activity (amount and pattern) and discrete measures of gait (pace, rhythm and bout to bout variability). PD participants walked for 1.4 h per day at over 7733 steps, with a total number of WBs per day of 329 (Table 3). Mean gait speed in shorter WBs (10–30 s) was 0.74 m/s, maximum walking speed in all WBs was 1.0 m/s, stride length in shorter WBs 95.8 cm, and at a cadence of 92.1 steps/minute in all WBs. Importantly, when compared to controls, 20 out of 24 DMOs showed a significant difference across walking activity and gait patterns (Table 3). Key differences of broad ranging deficits were seen across multiple domains. Controls walked for longer per day, with a greater WB step count, at a lower cadence and had a larger number of WBs in bouts greater than 10, 30 and 60 s (Figure 2). Bout to bout variability differed significantly between the groups; controls had greater walking speed, stride length and cadence bout to bout variability, but lower stride duration bout to bout variability. There was no difference in many of the pace domain variables, although PD participants walked faster than controls in shorter WBs, whilst controls demonstrated a greater maximum walking speed in longer WBs.
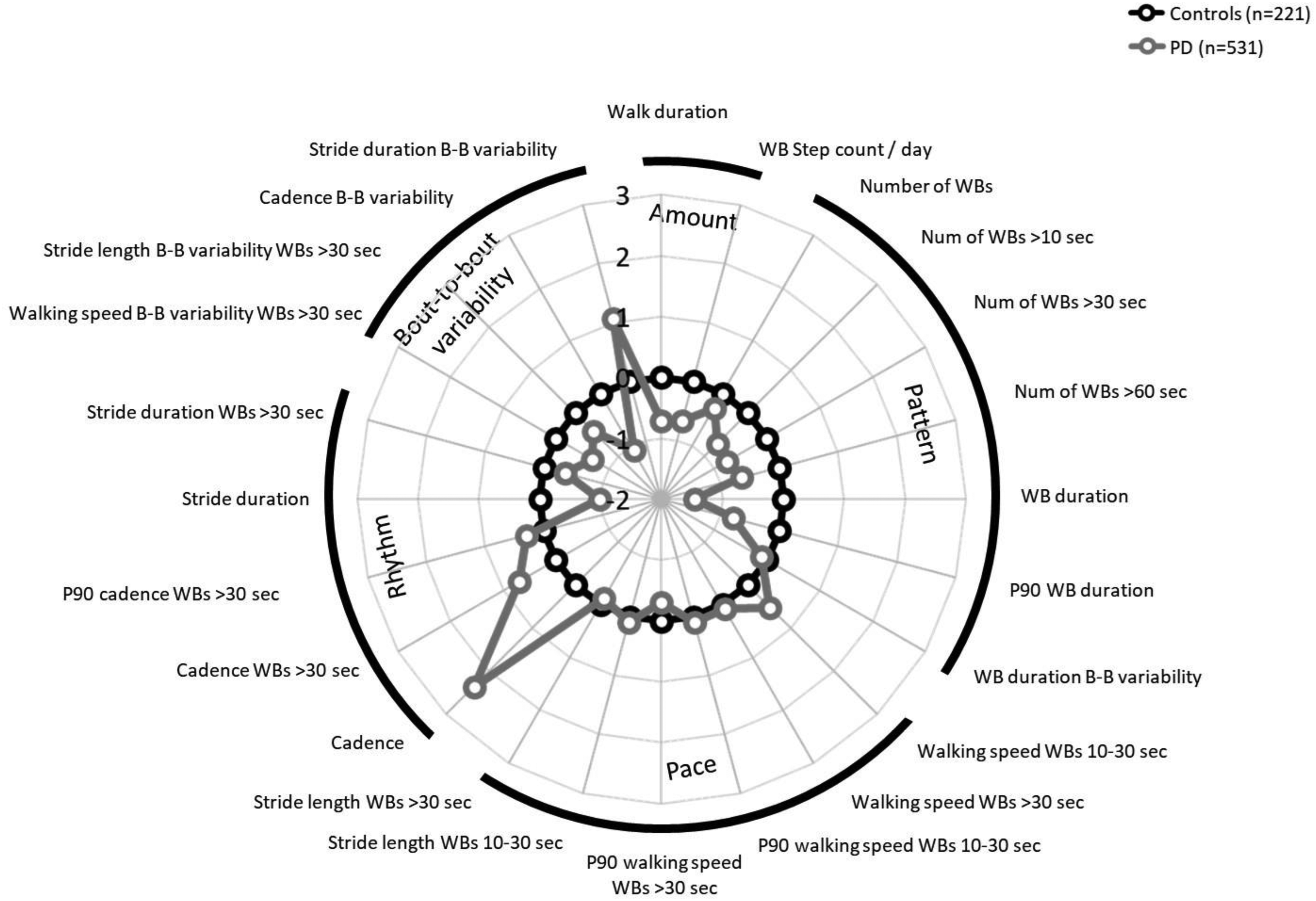
Radar plot of digital mobility outcomes in control vs. PD participants.
Comparison of DMOs in control and PD participants.
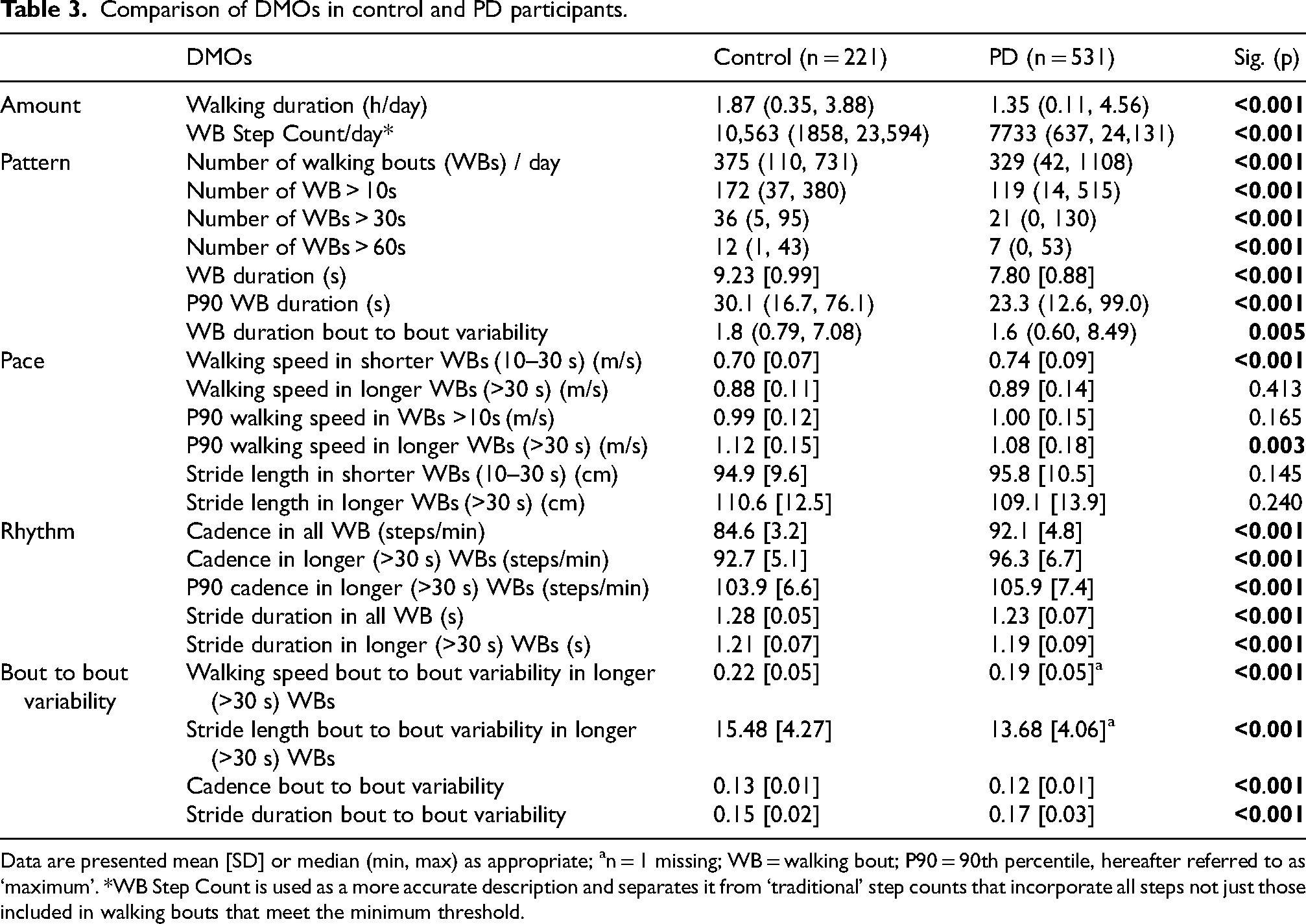
Data are presented mean [SD] or median (min, max) as appropriate; an = 1 missing; WB = walking bout; P90 = 90th percentile, hereafter referred to as ‘maximum’. *WB Step Count is used as a more accurate description and separates it from ‘traditional’ step counts that incorporate all steps not just those included in walking bouts that meet the minimum threshold.
Exploring DMOs by disease severity, Table 4 and Figure 3 outline DMOs by H&Y stage and illustrates wide ranging differences across every domain. Post hoc comparisons using Bonferroni adjustment highlighted that the differences were largely between controls vs. H&Y stages I-III and/or H&Y stage I and II vs. H&Y stage III (Table 4). Within measures of activity, those at H&Y stage III walked less, with fewer WB steps per day, and had lower numbers of total WBs and WBs >10 s. In terms of gait, walking speed, maximum walking speed and stride length were significantly lower in the more severely affected group. Maximum cadence in longer walking bouts and walking speed bout to bout variability were lower in H&Y III participants. Clinical characteristics by H&Y stage did differ somewhat (Supplementary Table 3); those at H&Y III were older, slightly shorter in height which was not clinically significant (compared to controls and H&Y II), with worse cognitive scores.
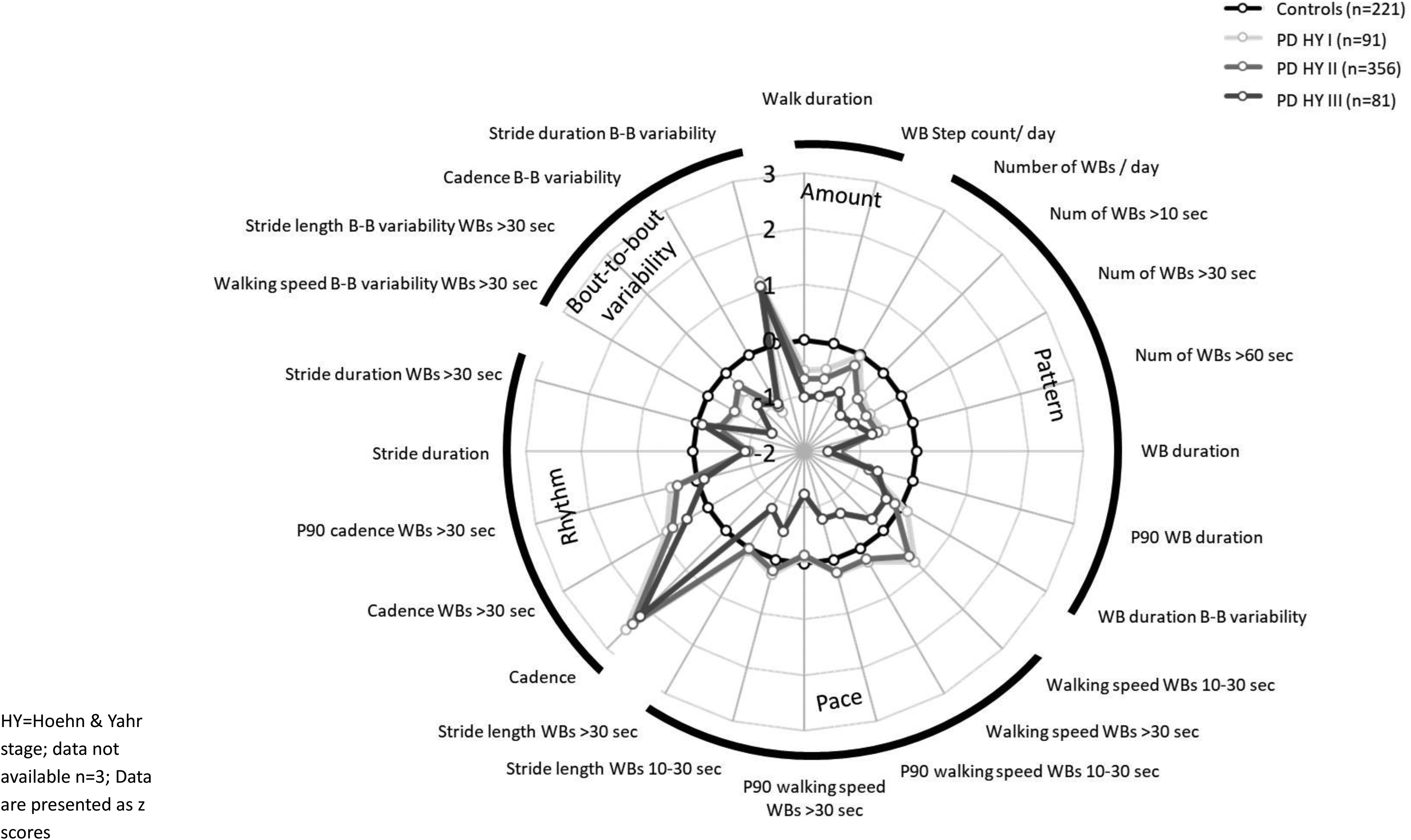
Radar plot of digital mobility outcomes in control vs. PD participants by Hoehn & Yahr stage.
Comparison of DMOs in controls and PD stratified by Hoehn and Yahr stage.
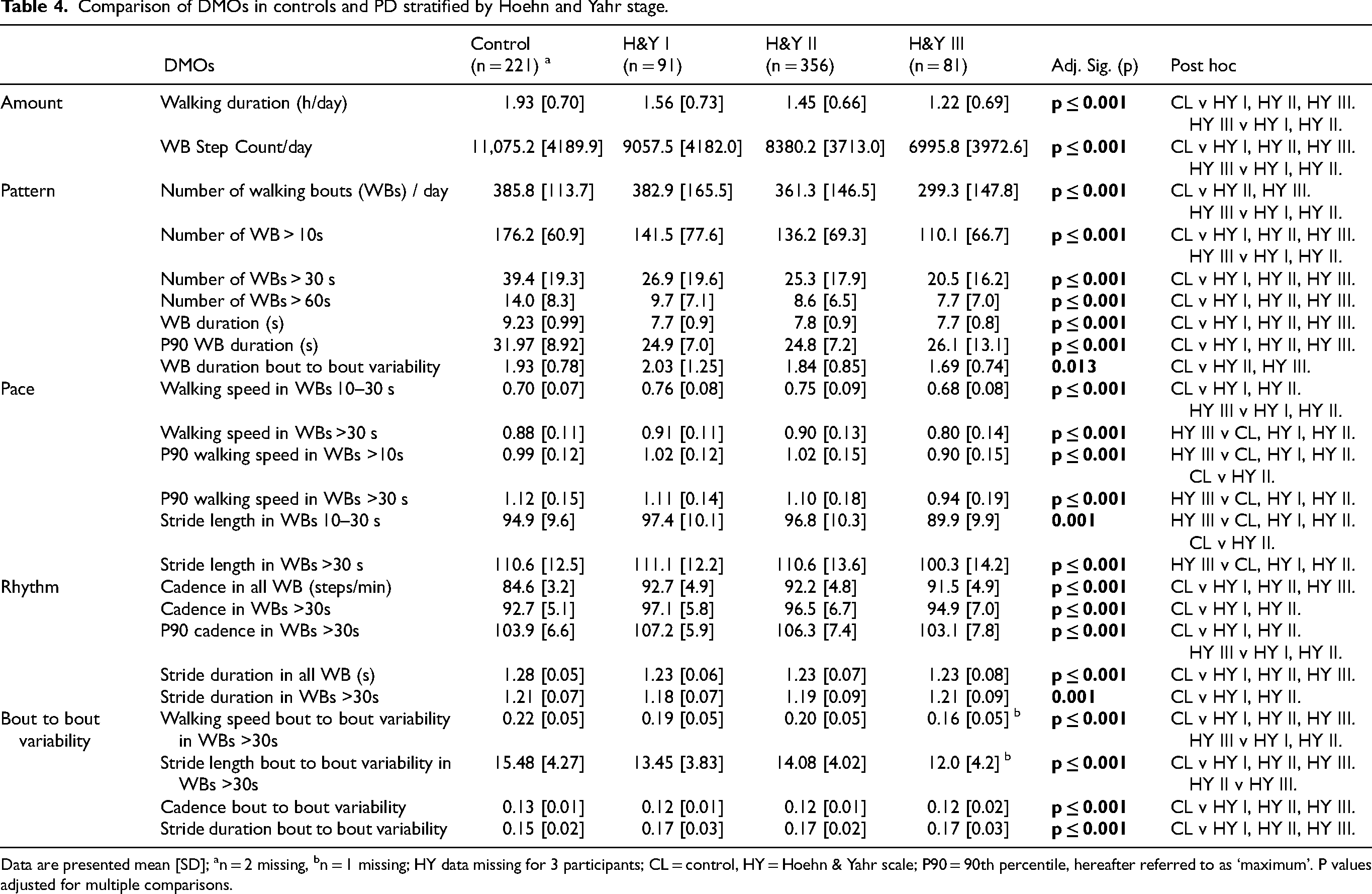
Data are presented mean [SD]; an = 2 missing, bn = 1 missing; HY data missing for 3 participants; CL = control, HY = Hoehn & Yahr scale; P90 = 90th percentile, hereafter referred to as ‘maximum’. P values adjusted for multiple comparisons.
We also explored whether certain site differences were seen in DMOs, and found differences particularly in walking activity and pace domains (Supplementary Table 4). Post hoc comparison revealed the differences largely at the Tel Aviv site, where PD participants walked for shorter durations with a fewer WB steps per day, at slower walking speeds and with shorter stride length. Some of these differences could be explained by differences in clinical characteristics, such as shorter stature, retirement status and older age, but not by disease profiles (Supplementary Table 5). In the control cohort, site differences were seen in the pace, rhythm and bout to bout variability domains (Supplementary Table 6; clinical characteristics Supplementary Table 7). Cadence and stride duration were lower in participants from Kiel compared to other sites.
Feasibility and compliance of devices
Feasibility and compliance were high in both the PD and control cohorts. Of the 601 PD participants who had clinical data collected, valid DMO data was available in 531 (88.4%); the majority of these had a full seven days of sufficient data (Supplementary Figure 1). Of the 232 controls, all but 11 had DMOs available at a weekly level (96.1%; Supplementary Figure 1). The PD participants without DMOs available (Supplementary Table 8; n = 70) had a slightly longer disease duration and minimally higher scores on the MDS-UPDRS parts I and II compared to those who underwent digital mobility assessment. DMO data loss varied by site (Newcastle 0%, Leuven 5.9%, Kiel 7.7%, Erlangen 14.6% and Tel Aviv 18.8%, p < 0.001), but remained consistent across sensor type (12.2% AX6 vs. 11.2% MM+, p = 0.717).
Within the PD cohort, 29.8% wore the AX6 device, with 99% in controls using AX6 (Supplementary Table 9). The only statistically significant differences in DMOs between AX6 and MM + in PD were a minimally longer walking bout duration in those with the MM+, shorter stride length in shorter WBs in AX6, and greater cadence in all WBs plus maximum cadence in longer WBs with the AX6 device (Supplementary Table 9). Clinical characteristics of device cohort were well-matched, although those using the MM + device were slightly taller, with minimally higher scores on the MDS-UPDRS parts I, III and IV (Supplementary Table 10).
Discussion
By presenting the largest real-world DMO dataset for individuals with PD and controls, this study enables robust, objective and generalisable mobility quantification on an international scale. Notable findings were wide-ranging differences in walking activity and discrete gait characteristics that allowed differentiation between PD and controls, where PD participants walked for briefer periods, at a slower maximum walking speed in longer WBs, with a greater cadence. Importantly, when stratified by disease stage, those with mild disease (H&Y stage I-II) significantly differed in their walking and gait patterns to the control cohort; there were also clear differences between mild (H&Y I-II) and moderate (H&Y stage III) disease. This is important in the context of both disease-modifying and symptomatic treatment trials, where deployment of robust measures such as DMOs could feasibly be utilised to determine differential change compared to placebo, and thus facilitate faster evaluation and approval of emerging therapies.15,51 Also of relevance, both body-worn devices exhibited excellent compliance, with this and the device-agnostic nature highlighting their suitability for clinical trial use.
It is notable that there was a degree of DMO data loss, particularly in the PD cohort, with some disease profile features suggesting those without DMO data were more severely affected. However, nearly half (n = 31) of these were due to technical issues recording, uploading, processing or analysing the DMOs. Lessons were learnt from this in between the Mobilise-D CVS starting in 2021 and the Extension Study for controls in 2023, with technical issues leading to DMO data loss only affecting three control participants. Although the number of participants with complete data at seven days differed from the suggested minimum data set, 49 this remained high in both cohorts and lends support to implementation as part of large clinical trials.
The DMOs obtained in Mobilise-D used accurate and technically validated algorithms with lumbar devices that met minimum specified criteria, which is likely to explain some of the differences with other studies. 52 A greater WB step count in both the control and PD cohort was demonstrated here compared to earlier work, 53 where step counts of almost 8000 steps/day in controls vs. over 5000 steps/day in an early PD group can be attributed to more accurate algorithm development in Mobilise-D, in addition to the definition of WB upon which they are developed.19,48 WB step count was used in Mobilise-D as a more accurate description and separates it from traditional step counts that incorporate all steps, not just those included in walking bouts that meet the minimum threshold. WB step count per day was comparable, albeit still below recommendations, 54 to those seen at baseline in a large exercise interventional study (7733 in the present work vs. 8874), 55 where participants walked for longer per day (almost two hours), in fewer (233) but longer (29.8 s) bouts than in Mobilise-D. Participants were older in this previous study (71.7 years), and its interventional nature may additionally account for some of these differences. 55 Other studies in PD which reported disparate step count utilising diverse body worn sensor positions, including wrist (WATCH-PD; step count 3494 steps/day in PD vs. 4930 steps/day in controls) 29 and ankle (9328 steps/day), 56 can be explained by these different sensor positions and corresponding algorithms.
Although walking speed in both the PD and control cohorts were slower than previously reported with differences in the order of 20 cm/s,30,57,58 there are several factors that preclude direct comparison between Mobilise-D and other studies. The impact of different walking bout definitions, newly validated algorithms, new pipeline, different cleaning methods of DMOs, different aggregation methods and summary metrics are all of critical importance. The pipeline procedures are described in detail as part of the Mobilise-D Technical Validation Study, 48 which includes the gold standard / reference data of the multi-sensor reference system integrating pressure insoles (INDIP); of note, the reference walking speed as measured by the INDIP was lower across all walking bout durations. 48 Moreover, a stride level approach rather than step level was used as a consensus, with walking speed evaluated using cadence and stride length. It should also be noted that weekly aggregated DMOs were calculated. This aggregation approach ensured stability of the outcomes and helps reliability and clinical interpretation of the DMOs. 50 Future work could investigate the value of between-day or within-day variability of the DMOs to track disease progression. Further technical details of the methodological aspects of the Mobilise-D pipeline are provided in Supplementary Methods.
Some results presented here merit further discussion. Somewhat unexpectedly, walking speed in shorter WBs was significantly faster in the PD compared to the control cohort (0.74 vs. 0.70 m/s, respectively). Maximum walking speed in longer WBs was faster in the control group, as would be anticipated; this suggests that some gait impairments may not be as pronounced in shorter walking periods in PD. A similar reversal of walking speeds at shorter bouts was also seen in the aforementioned study in early PD (walking speed 1.05 m/s in PD vs. 1.0 m/s controls). 30 One potential explanation is that during shorter WBs, individuals with PD produce power bursts to overcome bradykinesia and difficulties with motor initiation that cannot be sustained over longer periods 59 ; thus overall speed appears faster than controls in specific contexts. This effect here seemed to be particularly driven by moderate (H&Y III) stage disease. However, it seems less likely that this occurs during home mobility; it is also possible that in longer bouts there is a sequence effect and thus the PD-control differences emerge in the expected direction.
The majority of gait outcomes from the pace domain did not differ significantly between the PD cohort as a whole and controls, which was unexpected. By contrast, when stratified by disease stage, walking activity, rhythm and bout-to-bout variability all differed between controls and even mild PD, suggesting that these could offer options for objective, remote measurement in future disease-modifying trials in PD. With increasing neurodegeneration and motor severity, people with PD walked slower, with shorter stride length. Future research could investigate whether specific subsets of the Hoehn and Yahr staging system differ in their DMOs (e.g., H&Y 2 vs. H&Y 2.5). Taken together, these results indicate that DMOs in future work should be selected depending on the context of use; for example, a trial of disease modification that was looking to slow progression to a specific milestone, such as the development of postural instability (progression of H&Y I and II to stage III) may examine walking activity (amount) and pace characteristics.
Many of the individual clinical characteristics in this large PD cohort are of interest; note the scores of physical function such as SPPB and TUG are below the cut-off levels for traditional falls risk in older adults, but there were high self-reported fall rates in the PD cohort over the preceding year. This points to a different approach needed for fall risk detection within PD. In keeping with the literature,60,61 falls were reported even at H&Y stage I. Significant differences in physical function assessments were observed in PD compared to control participants. This might raise the question of whether DMOs are required, given that these brief, conventional and inexpensive tests can discriminate between the groups. However, the utility of DMOs lies in their ability to provide sensitive, valid, objective, and reliable measures that can be collected continuously and remotely within an individual's home environment, which avoids in-clinic visits and is more representative of actual mobility performance. Such characteristics are essential for the development and implementation of robust outcomes in clinical trials.
Strengths of this study include the collection of objective real-world gait data using robust, technically validated and open-source available algorithms 19 in a relatively large number of participants with PD and matched controls. While site-specific differences were observed, potentially influenced by sociodemographic characteristics such as age and retirement status, these variations enhanced the study's strength by capturing a broad range of walking behaviours across different environments. This diversity supports the generalisability of the findings and contributes to a better understanding of DMOs across different contexts and cultures.
Several limitations should be acknowledged. This study did not account for environmental factors, such as geographic location or weather, which may influence real-world mobility in both PD and control groups, and should be considered in future research. Additionally, early recruitment of PD participants occurred during the end of the COVID-19 pandemic, which may have influenced walking activity. The study's longitudinal follow-up and extended baseline protocol may have introduced a self-selection bias, potentially explaining some of the better function in this PD cohort compared to other studies. Initial technical difficulties led to some data loss among early PD recruits; however, this issue was mitigated through effective monitoring and data cleaning / processing strategies. It is of note that pace and rhythm characteristics can be measured in the clinic or laboratory setting; bout-to-bout variability cannot. There is some evidence to suggest that in contrast to within bout variability, higher values of bout-to-bout variability may be beneficial. 62 The present analysis does not include any measures of asymmetry or within-bout variability, measures that have been previously shown to be interesting in PD. These (and others, including signal-based characteristics) will be added in the future. Finally, the exploration of covariates, including moderating and mediating factors, and their influence on DMO patterns was beyond the scope of this study, but will be addressed in future analyses.
In conclusion, walking activity and gait in the real-world can be objectively and feasibly measured in a relatively large cohort of individuals with PD and matched controls. DMOs were found to differ between PD participants and controls, and many of these differences increased with worsened motor severity. Future work will explore the predictive value of these DMOs as the cohort is followed longitudinally. This includes refining the set of DMOs to reduce redundancy and define a core dataset suitable for future clinical studies—an essential step toward regulatory acceptance of DMOs. The algorithms and processing code used in this study has already been made available for commercial and research use under a permissive Apache 2.0 license (https://github.com/mobilise-d), and the dataset will be released during the next year (https://zenodo.org/communities/mobilise-d/).
The sensitivity of DMOs to assess disease progression and predict adverse outcomes will be evaluated in separate analyses to ensure that the most appropriate digital outcomes are selected for specific clinical questions. This work represents a foundational step in the objective measurement of mobility in PD, particularly in the context of emerging disease-modifying therapies.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251407026 - Supplemental material for The future of clinical trials? Monitoring mobility as an outcome measure in Parkinson's disease: The mobilise-D study
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251407026 for The future of clinical trials? Monitoring mobility as an outcome measure in Parkinson's disease: The mobilise-D study by Alison J Yarnall, Lisa Alcock, Christian Schlenstedt, Claudia Armengol, Clemens Becker, Philip Brown, Joren Buekers, Brian Caulfield, Andrea Cereatti, Laura Cordova-Rivera, Silvia Del Din, Laura Delgado-Ortiz, Mark Forrest-Gordon, Judith Garcia-Aymeriche, Pia Goerrissen, Clint Hansen, Hanna Hildesheim, Hugo Hiden, Heather Hunter, Carl-Philipp Jansen, Sarah Koch, Jennifer Kudelka, Michael Long, Isabel Neatrour, Basil Sharrack, David Singleton, Lou Sutcliffe, Thierry Troosters, Juergen Winkler, Pieter Ginis, Heiko Gassner, Alice Nieuwboer, Jeffrey M Hausdorff, Anat Mirelman, Lynn Rochester, and Walter Maetzler in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X251407026 - Supplemental material for The future of clinical trials? Monitoring mobility as an outcome measure in Parkinson's disease: The mobilise-D study
Supplemental material, sj-docx-2-pkn-10.1177_1877718X251407026 for The future of clinical trials? Monitoring mobility as an outcome measure in Parkinson's disease: The mobilise-D study by Alison J Yarnall, Lisa Alcock, Christian Schlenstedt, Claudia Armengol, Clemens Becker, Philip Brown, Joren Buekers, Brian Caulfield, Andrea Cereatti, Laura Cordova-Rivera, Silvia Del Din, Laura Delgado-Ortiz, Mark Forrest-Gordon, Judith Garcia-Aymeriche, Pia Goerrissen, Clint Hansen, Hanna Hildesheim, Hugo Hiden, Heather Hunter, Carl-Philipp Jansen, Sarah Koch, Jennifer Kudelka, Michael Long, Isabel Neatrour, Basil Sharrack, David Singleton, Lou Sutcliffe, Thierry Troosters, Juergen Winkler, Pieter Ginis, Heiko Gassner, Alice Nieuwboer, Jeffrey M Hausdorff, Anat Mirelman, Lynn Rochester, and Walter Maetzler in Journal of Parkinson's Disease
Footnotes
Acknowledgements
This paper presents independent research supported by the NIHR Newcastle Biomedical Research Centre (BRC). The NIHR Newcastle BRC is a partnership between Newcastle Hospitals NHS Foundation Trust, Newcastle University, and Cumbria, Northumberland and Tyne and Wear NHS Foundation Trust and is funded by the National Institute for Health and Care Research (NIHR). The authors wish to thank Michèle Bartlett for her review of the manuscript and lay summary.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Mobilise-D project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement no. 820820. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation program and the European Federation of Pharmaceutical Industries and Associations (EFPIA). This publication reflects the authors’ views and neither IMI nor the European Union, EFPIA, or any Associated Partners are responsible for any use that may be made of the information contained herein. The Mobilise-D Extension study was funded by the Michael J Fox Foundation (MJFF-022735).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AJY has received research support from the NIHR Newcastle Biomedical Research Centre, Dunhill Medical Trust, EU IMI, NIHR, Parkinson's UK, Michael J Fox Foundation, Weston Brain Institute, Lewy Body Society, Intercept Pharmaceuticals, Electrocore; and has received funding and/or honoraria from Britannia, UCB, Abbvie, GSK, Teva-Lundbeck, GE Healthcare and Genus for attending or speaking at educational events. LS reports funding from EU IMI, NIHR Newcastle Biomedical Research Centre and Michael J Fox Foundation. HG was supported by the Fraunhofer Internal Programs under grant No. Attract 044-602140, No. Attract 044-602150, and No. SME 40-09311. He further received personal fees from Zambon GmbH outside of the submitted work. LR receives research funding from MRC, NIHR, EU, EPSRC, PDUK and is a shareholder of Enoda Health Ltd. SDD is a shareholder in Enoda Health Ltd. This shareholding did not have any relationship with the work in this paper. HH is a director of Inkspot Science, a consultancy that provides study management and cloud services to support medical studies. Inkspot had no involvement in the Mobilise-D project.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
