Abstract
Background
Fatigue in Parkinson's disease (PD) is a common, debilitating symptom often overlooked in research and clinical practice. Effective interventions are needed to mitigate its impact on people with PD.
Objective
This pilot study evaluated the feasibility of the individual videoconference version of the Packer Managing Fatigue program for people with PD and explored its preliminary effectiveness versus usual care to inform the design of a definitive trial. Here we report on the second objective.
Methods
A two-arm, assessor-masked, randomized controlled pilot study recruited participants with PD who experience severe fatigue, have English proficiency, and internet access. Outcome measures included occupational performance, satisfaction with performance, occupational balance, fatigue impact, quality of life, and sleep. Mixed repeated-measures ANOVA and non-parametric tests were used for analysis.
Results
Mixed-design ANOVA (N = 25) showed an exploratory trend toward significant for the Time × Group interaction effect differences in satisfaction with performance between groups over time (p = 0.09). Paired t-tests within the intervention group indicated significant improvement in satisfaction with performance (p = 0.04). The effect size for this outcome was moderate. Small to moderate effect sizes were observed for occupational balance, occupational performance, and subscales of the Multidimensional Fatigue Inventory. Other measures showed negligible effects.
Conclusions
The results provide preliminary evidence of the program's benefits for people with PD. Larger, more rigorous studies are needed to confirm its effectiveness. Despite the small sample size and challenges posed by COVID-19, this study offers valuable insights into recruitment strategies and effect sizes to inform future trial designs.
Plain Language Summary
People living with Parkinson's disease (PD) often feel extremely fatigued. It can make daily activities difficult and reduce quality of life. Our study tested a program called “Packer Managing Fatigue” which helps people manage their energy better. This program teaches strategies like planning rest breaks, simplifying tasks, and improving sleep habits. There is proof that the program helps people with multiple sclerosis. This is the first time testing it for people with PD. We worked with 25 people from Nova Scotia and Ontario, Canada. They were divided into two groups: one group used the Packer Managing Fatigue program, and the other group continued with their usual care. We measured their level of performance and satisfaction doing meaningful daily life activities, whether they felt they led a balanced life, the impact of fatigue on their life, quality of life, sleep quality, and other factors before and after the program. We compared the two groups to see if there was a difference. The program helped improve peoples’ satisfaction with their daily activities. However, we did not find significant changes in other areas. This first, small study suggests that the program has potential to help people with PD. A new study with a larger group of participants is recommended to confirm its effectiveness. This study is an important first step in understanding how to better support people with PD in managing fatigue. Future research will help refine the program and determine the best ways to deliver it to those who need it.
Keywords
Introduction
Fatigue is the most common non-motor symptom reported by people with Parkinson's disease (PD) 1 and significantly impairs their occupational particpation and overall quality of life. 2 Fatigue is associated with early retirement, reduced work hours, financial distress, social isolation and poor quality of life.3,4 Consequently, fatigue management is a top priority in PD.5,6
While pharmacological treatments like doxepin and rasagiline may reduce PD-related fatigue, their side effects and inconclusive efficacy limit their use.7–9 This highlights the need for alternative, non-pharmacological interventions, such as cognitive-behavioral therapy, 10 physical exercise, 11 and energy conservation.12,13 However, their application to PD-related fatigue remains underexplored. Recent systematic reviews underscore the need for rigorous studies, particularly randomized controlled trials, to evaluate these approaches.7,14,15
Our study evaluated a self-management program centered on energy conservation strategies, originally developed by Packer et al. (1995). 16 This program, a pioneering intervention for managing fatigue, has been recognized for its effectiveness across a range of chronic neurological conditions.14,15,17 The six-week, in-person, group-based program empowers individuals with strategies such as strategic resting, activity simplification, task analysis, environmental modification, communicating about fatigue, planning, and prioritizing. 16 Tested in several randomized controlled trials (RCTs) it has been found effective in reducing fatigue impact, improving quality of life, and enhancing participation.13,18–21 Despite extensive evidence of its effectiveness, little is known about its effects for people living with PD.14,15,17
The one-on-one format is feasible for routine clinical practice when group sessions are not possible and beneficial for addressing specific client needs. However, it has only been evaluated in two studies using investigator-adapted versions with variations in length and content.22,23 One study found no significant differences in fatigue and participation between the experimental group and an information-only control group. 22 Conversely, Van Heest et al. 23 reported significant post-test improvements in fatigue, quality of life, and self-efficacy. The heterogeneity in program delivery and outcomes complicates interpretation. To provide a standardized and consistent approach to one-on-one delivery of the program, the original author developed a protocolized individual version of the program in 2020, named Packer Managing Fatigue: The Individual Self-Management Program. 24
Participant perspectives on the feasibility of the program have been previously reported. 25 The main objective in this study is to lay the groundwork for a full-scale RCT with three specific aims: (1) to assess the preliminary effectiveness of the Packer Managing Fatigue program; (2) to determine effect sizes to inform sample size requirements for a definitive RCT; and (3) to evaluate the effectiveness of the recruitment strategies and plan.
Methods
Study design
This was an assessor-blinded, two-arm pilot randomized trial study. Eligible participants were randomized, using sealed numbered envelopes, in a 1:1 ratio to either the intervention group, which received the study program in addition to their usual healthcare services, or the control group, which received usual care only. After study completion, control group participants were offered the program manual and one online training workshop.
Due to the behavioral nature of the intervention, participants could not be blinded to group allocation. However, interventionists (3 trained occupational therapists) were blinded to the study design and the nature of the control arm. The study was registered with ClinicalTrials.gov (ID: NCT04267107) and received ethical approval from the Nova Scotia Health Sciences Research Ethics Board (ref: 1027048). As this was an exploratory study, no primary versus secondary outcomes were pre-determined. The outcomes measured included occupational performance and satisfaction, occupational balance, fatigue impact, quality of life, and sleep quality. This approach aligns with guidelines for feasibility testing, which can focus on multiple primary outcome measures to determine if one or more are not acceptable or understood as intended. 26 The detailed protocol of this study is published elsewhere. 25 The level of statistical significance was set at p < 0.1 due to the pilot nature of the study and small sample size, allowing identification of trends warranting further investigation. Using a higher significance level in pilot studies allows researchers to detect potential effects that might be missed with more stringent criteria.26,27 This study is part of the first author's manuscript-based dissertation, which was published by Dalhousie University. 28
Participants
All participants were adults residing in Nova Scotia or Ontario, Canada, and provided informed consent prior to data collection. Inclusion criteria were having a diagnosis of PD (self-reported), experiencing severe fatigue, a score four on the Fatigue Severity Scale (FSS) 29 which is commonly used in research to indicate significant fatigue, ability to read and converse in English, access to the internet and an electronic device, and a private location for videoconferences. Exclusion criteria were prior completion of the Packer Managing Fatigue program, severe fatigue-related comorbidities such as heart failure or diabetes, or a Mini-Mental Status Exam (MMSE) score below 13. 30 Given the exploratory nature of our study and its aim to assess the feasibility of a full-scale RCT, we relied on methodological reviews that recommend a minimum total sample size of 10 to 50 for pilot studies.31–34 Multiple recruitment strategies were employed, including recruitment notification via Parkinson Canada, movement disorder clinics, patient-focused conferences, local web-based advertisements (e.g., KIJIJI websites), support group email listservs, Parkinson Canada websites, social media platforms (Twitter and Facebook), and word of mouth. Interested individuals contacted the research team via email and consented electronically before any data collection. Eligible participants were assigned an ID number and completed baseline measures. They were then randomly allocated to one of two groups using the sequentially numbered, opaque, sealed envelopes method. 35 The envelopes were opened only after each participant had completed their baseline measurements. The assessor was masked to group allocation, and participants were instructed not to disclose their group allocation during post-test assessments. Any incidents resulting in unmasking were documented and monitored. Figure 1 presents the CONSORT flow diagram of participant progress through the study.
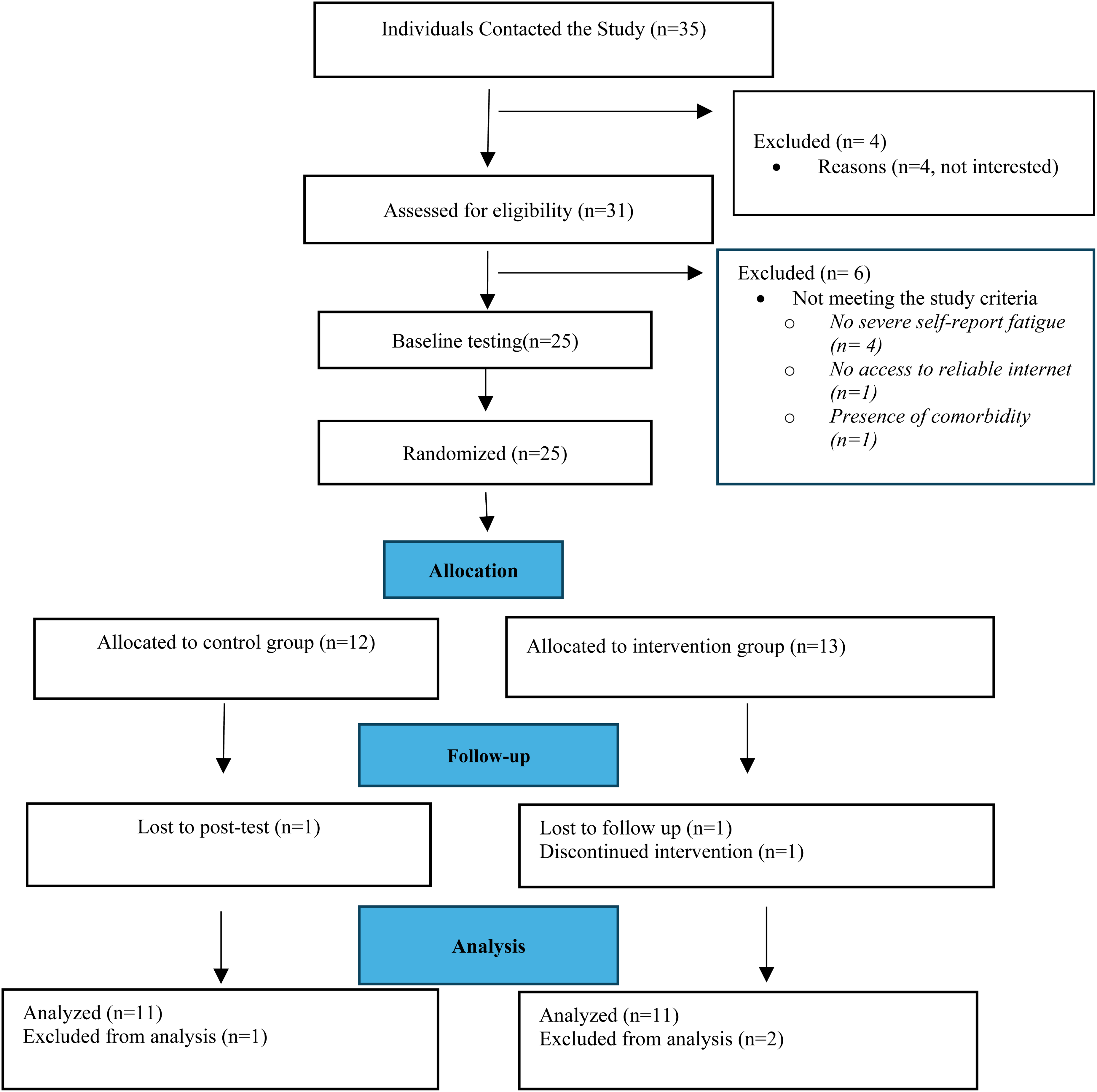
CONSORT flow diagram illustrating the progress of participants through the phases of the randomized trial.
Intervention
The program was modeled on the original program, retaining all core content as well as additional content on sleep hygiene and cognitive fatigue in accordance with more recent evidence-based recommendations for managing fatigue.36,37 The six sessions of the program focused on trialing, evaluating, and adopting energy conservation strategies: Session 1) Importance of Rest and Sleep; Session 2) Communication and Body Mechanics; Session 3) Tools, Technology and Ergonomics; Session 4) Priorities and Standards; Session 5) Putting It All Together, and Session 6) Some Closing Thoughts (including a course review and setting future goals). The program was delivered via the Zoom for Healthcare platform, 38 compliant with Canadian Data Protection regulations. 39
Each session included pre-session activities to be, completed at home to prepare participants for in-session discussions. The in-session activities focused on developing skills and strategies for energy conservation and expenditure, problem-solving and action-planning. Post-session homework assignments designed to reinforce the application of the strategies introduced during the session. Sessions were expected to last approximately 90 min but could be adjusted based on individual patient needs with the entire program completed within 6–8 weeks. A fidelity checklist, designed by researchers, was completed by OTs after each session.
Licensed occupational therapists (OTs) from Nova Scotia delivered the virtual program. All OTs (n = 3) completed an asynchronous online training course developed by the research team. This course consisted of two sections: general training to learn and deliver the program (10 modules) and PD-specific modules (2 modules). Therapists had to complete all modules and successfully complete all corresponding quizzes and case scenarios. They had the opportunity to review content and retake quizzes.
Data collection
All data were collected online using Opinio Online Survey Software 40 while participants video-conferenced with the masked assessor via Zoom for Healthcare. Upon enrollment and consent at baseline, a demographic questionnaire gathered data on age, gender, years since diagnosis, living status, employment status, and current treatments for fatigue. Outcomes were assessed at two time points: (1) at baseline, prior to randomization, and (2) post-intervention, approximately 8–10 weeks after baseline. For participants in the intervention group, this was about two weeks after completing the program. Outcome variables and associated measures included: fatigue impact (the Multidimensional Fatigue Inventory: MFI), 41 self-perceived performance in occupation in everyday livings (COPM-P) and satisfaction with their performance (COPM-S), 42 occupational balance (Occupational Balance Questionnaire:OBQ-11), 43 quality of life (Parkinson`s Disease Quality of Life: PDQ-8), 44 and sleep quality (Pittsburgh Sleep Quality Index: PSQI) 45 (Table 1). The selected outcome measures and their associated questionnaires were selected based on the program's content and intended effects, as well as evidence from previous studies that evaluated other formats of the Packer Managing Fatigue program in progressive neurological populations other than PD. They were also chosen for their relevance and suitability for persons with PD. Detailed rationale for selection and psychometric properties are reported elsewhere (188). One proposed outcome measure, the Self-efficacy for Performing Energy Conservation Strategies Assessment (SEPECSA), was not used in the analysis due to participant feedback indicating some items were not easily understood at baseline, prior to attending sessions. To assess potential confounding effects on fatigue, disease severity and depression were measured at baseline using the self-reported Hoehn and Yahr scale (HY), 46 and the short version of the Geriatric Depression Scale (GDS-15), 47 respectively.
Measurements used in the study and their properties.

To assess the effectiveness of recruitment strategies, the number of individuals who contacted the research team, met, or did not meet study criteria were documented. The tracking form also captured the date of screening and how participants learned about the study. Sociodemographic characteristics of participants enrolled were extracted from the demographic questionnaire.
Data analysis
All data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 27.0 for Windows, 48 following consultation with a statistician. Data were first examined for skewness, outliers, and systematic missing data. Out of 2596 possible values across all tests and time points, only nine values (0.003%) were missing, with no significant pattern detected. Total scores for each measure were calculated based on measure-specific guidelines. For measures without per-protocol instructions for handling missing data, mean substitution was used if less than 20% of item-level data were missing. Missing data at the outcome level were handled by listwise deletion, excluding participants who did not complete the post-test from the analysis.
No outliers, defined as greater than ±2 standard deviations (SD) from the mean, 49 were detected. All variables were normally distributed with homogeneous variances, except for the Canadian Occupational Performance Measure – Performance (COPM-P). For COPM-P at Time 2, the assumption of normality was marginally met (Shapiro–Wilk test: W = 0.91, p = 0.063; skewness=–0.92; kurtosis = 0.43), indicating a mild deviation from normality. Levene's test indicated a violation of the homogeneity of variance assumption for COPM-P at both time points (Time 1: F(1, 23) = 5.90, p = 0.02; Time 2: F(1, 20) = 3.86, p = 0.05). Given these observations and the study's small sample size, non-parametric tests (Kruskal-Wallis and Wilcoxon signed-rank tests) were applied for COPM-P analysis, while parametric statistics were used for all other measures. The level of statistical significance was set at p < 0.1 due to the pilot nature of the study and small sample size, 27 allowing for the identification of trends of effects for further investigation.
The success of randomization was assessed by comparing potential covariates (age, depression, fatigue severity, disease duration, disease stage, and gender) between groups at baseline using independent-sample t-tests (for continuous outcomes) and chi-square tests (for binary variables). Depression showed a non-significant difference between groups, t(23) = 1.79, p = 0.08, MD = 2.16, 95% CI [-0.34, 4.66] but was included as a covariate in subsequent analyses due to its clinical relevance and the exploratory nature of the study. All analyses were conducted with and without adjustment for baseline depression (Figure 2).
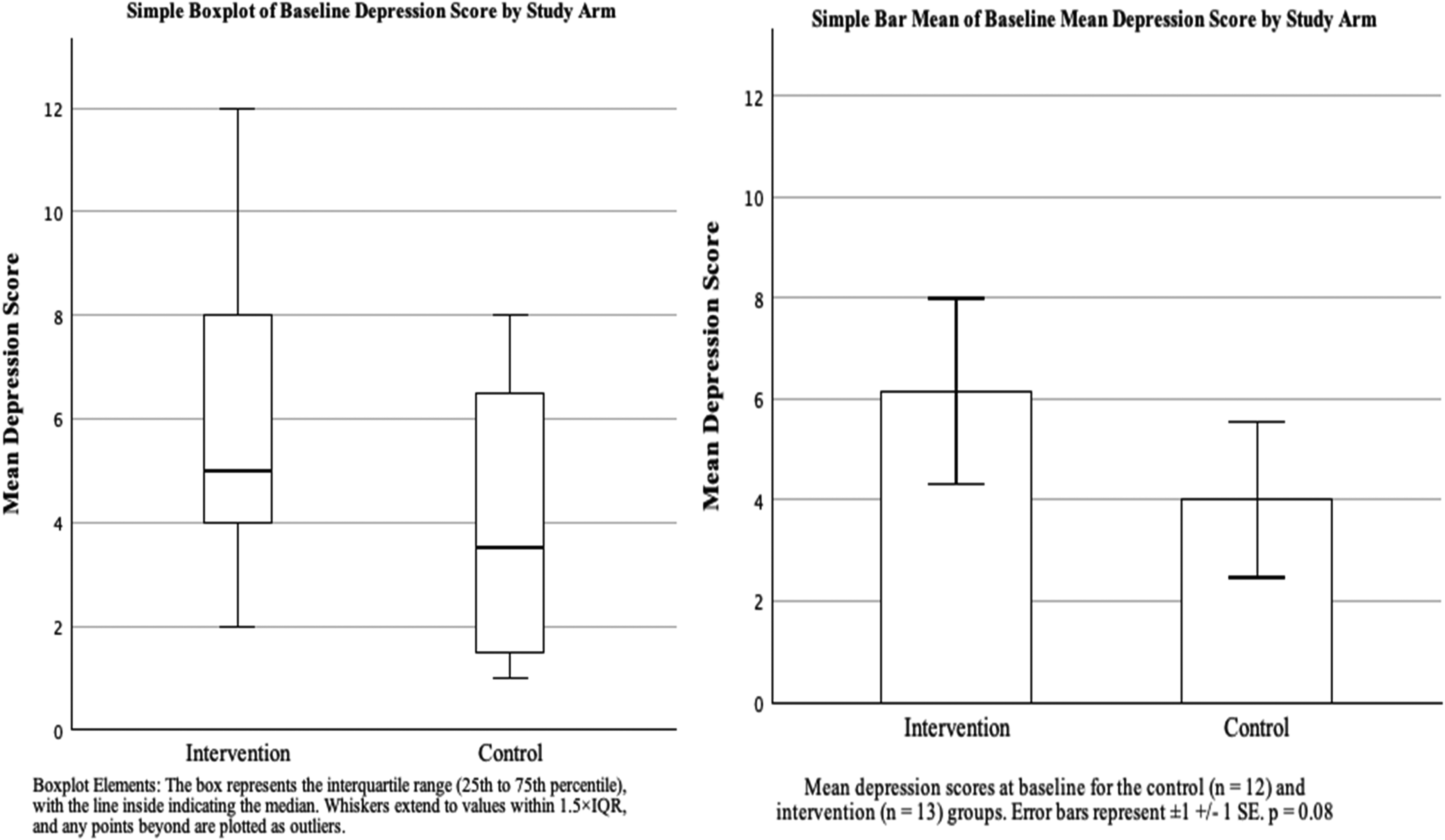
Mean differences between groups in depression scores at baseline.
The effect of the intervention on study outcomes was evaluated using a mixed repeated-measures ANOVA. Time (baseline and post-test) and group (intervention or control) were treated as independent variables, with time as a within-subject factor and group as a between-subjects factor. Time*group interaction effects were calculated. Additionally, paired t-tests were conducted separately for each group to explore within-group changes over time, appropriate for the small sample size and exploratory nature of the study. 50
Effect sizes for each outcome measure were calculated using partial eta squared (η²), interpreted as follows: 0.01 (small), 0.06 (medium), and 0.14 or higher (large). 51 To inform the sample size calculation for a future definitive RCT, η² values were converted to Cohen's f effect sizes, which are commonly used in ANOVA tests and required for calculating sample sizes using G*Power 3.0.10. 52 The conversion formula used was: f = sqrt (η²/(1−η²)). 53 The Kruskal-Wallis test value (H) was converted to partial eta squared effect size using the formula: η² [H] = (H−k + 1)/ (n−k), where k represents the number of groups and n represents the total number of observations. 54
To estimate the sample size needed for future studies, the test family and type were specified as F tests and ANOVA: repeated measures, within-between interaction. Type I error rate was set at 0.05 with a power of 0.8. To align with common RCT designs, the number of measurements was set at three time points in two groups.
Results
A total of 25 participants met eligibility criteria, consented to participate in the study, and completed baseline questionnaires (13 intervention, 12 control). There were no significant differences between the study groups in any of the baseline characteristics except for a trend toward higher mean depression scores for those in the intervention group (Table 2). Three participants (12%) withdrew from the study prior to completion, stating illness or feeling over-committed as reasons for discontinuing. One participant in the intervention group withdrew after the first session, and one participant in each group withdrew at the post-intervention measurement point. Program fidelity was measured and reported as 100% according to the program protocol. 30
Participant characteristics at baseline: mean (SD) or N (%).
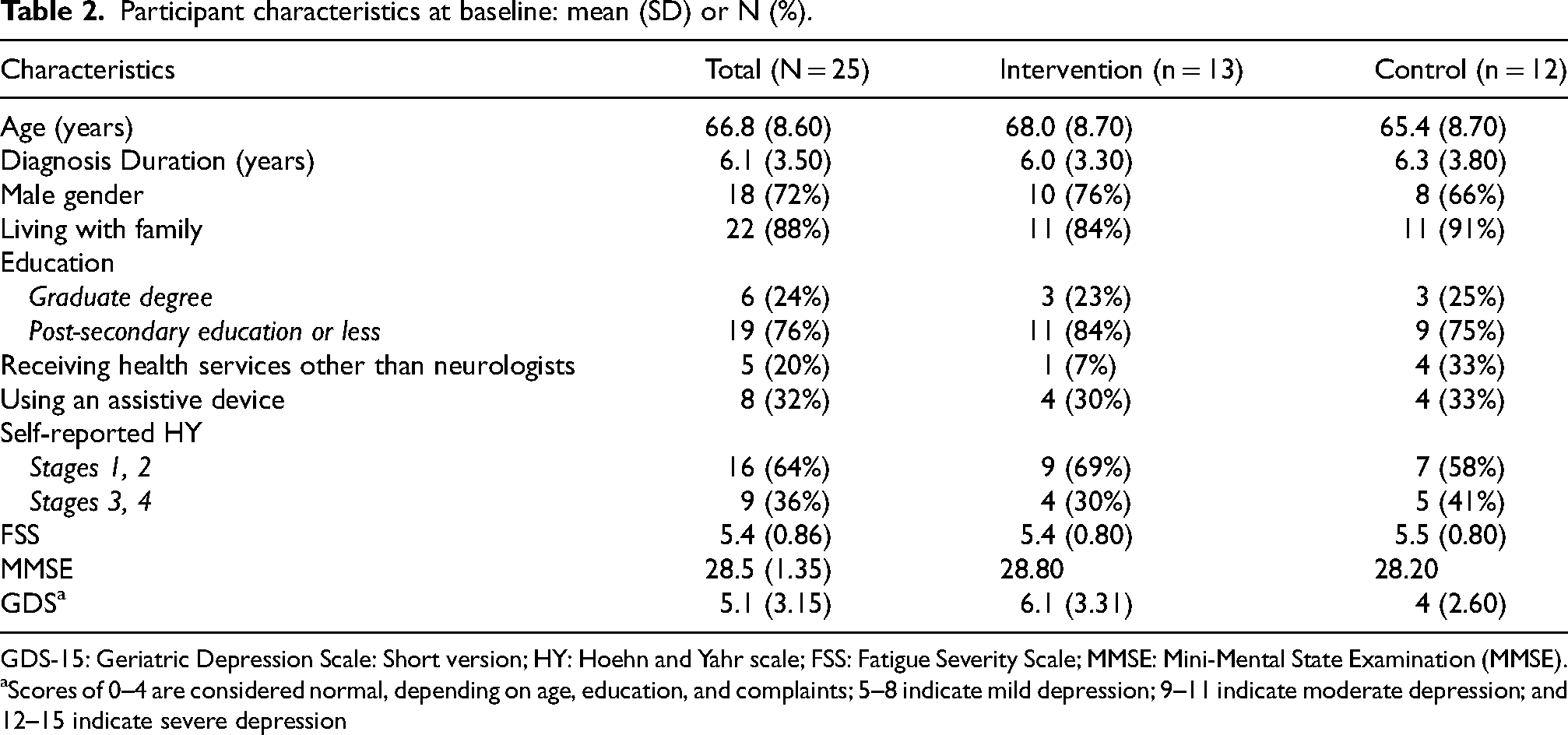
GDS-15: Geriatric Depression Scale: Short version; HY: Hoehn and Yahr scale; FSS: Fatigue Severity Scale; MMSE: Mini-Mental State Examination (MMSE).
Scores of 0–4 are considered normal, depending on age, education, and complaints; 5–8 indicate mild depression; 9–11 indicate moderate depression; and 12–15 indicate severe depression
Preliminary effectiveness of the program
Non-significant results were observed for the Time × Group interaction effect across all outcomes, except for the COPM-S. For the COPM-P, non-parametric tests were used due to unmet ANOVA assumptions. The Kruskal-Wallis test showed no significant differences in COPM-P score between groups (H = 0.24, p = 0.62). The Wilcoxon signed-rank test also revealed no significant COPM-P within-group changes for either the intervention (Z = -0.53, p = 0.59) or control group (Z = -1.55, p = 0.12). COPM-S scores changed significantly over time in the intervention group (mean change: 1.22, p = 0.04), with a tendency toward a significant time by group interaction effect [F (1,20) = 3.07, p = 0.09], suggesting that the mean differences between groups changed over time in favor of the intervention group (Table 3).
Parametric and non-parametric analysis: comparison of intervention and control group, including time effects and time × group effects.
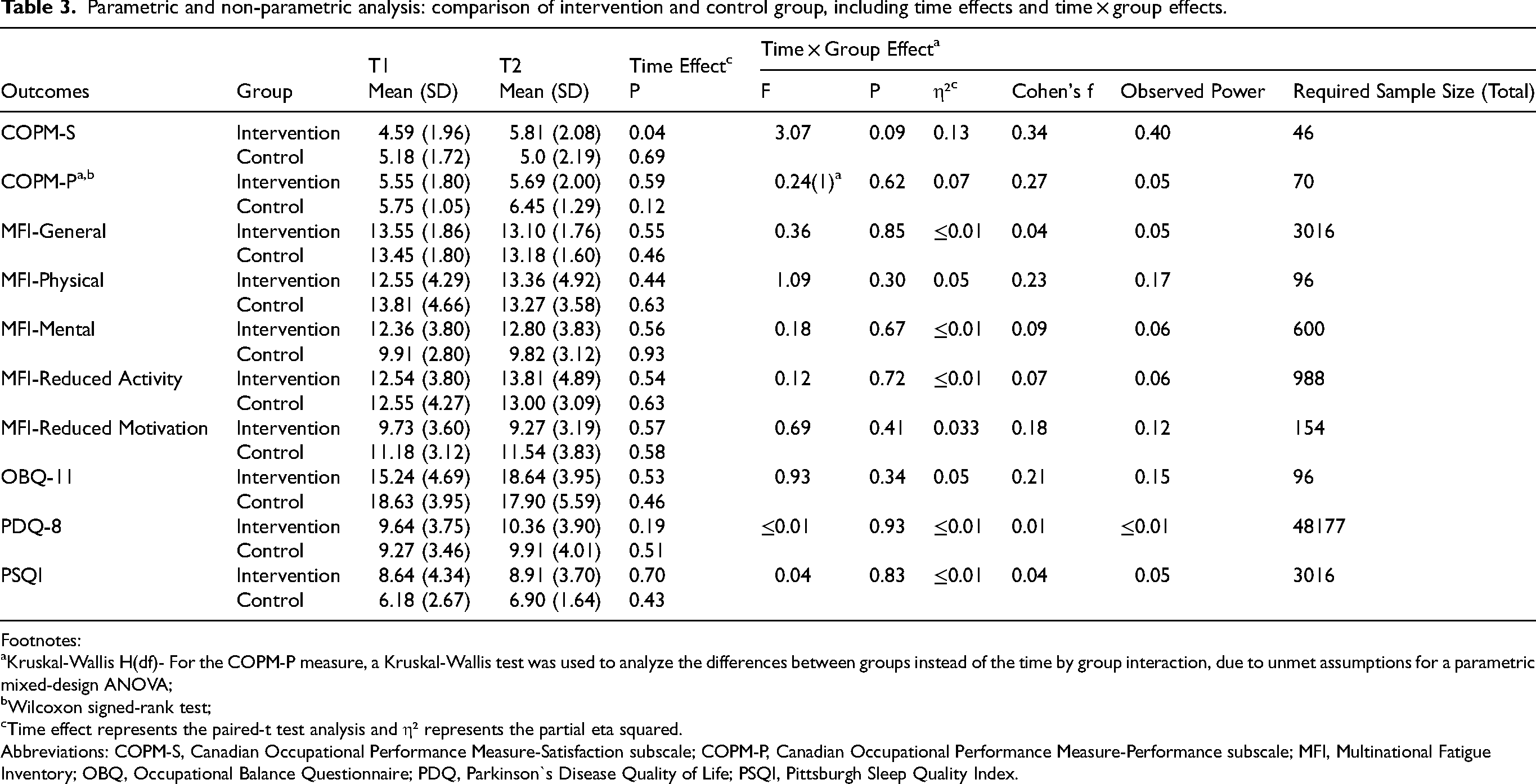
Footnotes:
Kruskal-Wallis H(df)- For the COPM-P measure, a Kruskal-Wallis test was used to analyze the differences between groups instead of the time by group interaction, due to unmet assumptions for a parametric mixed-design ANOVA;
Wilcoxon signed-rank test;
Time effect represents the paired-t test analysis and η² represents the partial eta squared.
Abbreviations: COPM-S, Canadian Occupational Performance Measure-Satisfaction subscale; COPM-P, Canadian Occupational Performance Measure-Performance subscale; MFI, Multinational Fatigue Inventory; OBQ, Occupational Balance Questionnaire; PDQ, Parkinson`s Disease Quality of Life; PSQI, Pittsburgh Sleep Quality Index.
Due to possible baseline differences in depression between the control and intervention groups, the analysis was repeated with depression as a covariate. Although the time by group interaction remained nonsignificant (Supplemental Table 1), the pattern of change on the COPM-P varied between groups. After accounting for depression, the intervention group did not demonstrate statistically significant improvements compared to the control group; however, the direction of the average observed scores changed. For the COPM-S, this trend did not change between groups even after controlling for depression. However, the differences between the two groups became more notable, changing the p-value from 0.09 to 0.05. The differences between the two groups became more notable, changing the p-value from 0.09 to 0.05. Figure 3 shows the average observed scores for the COPM-P and COPM-S across time between groups
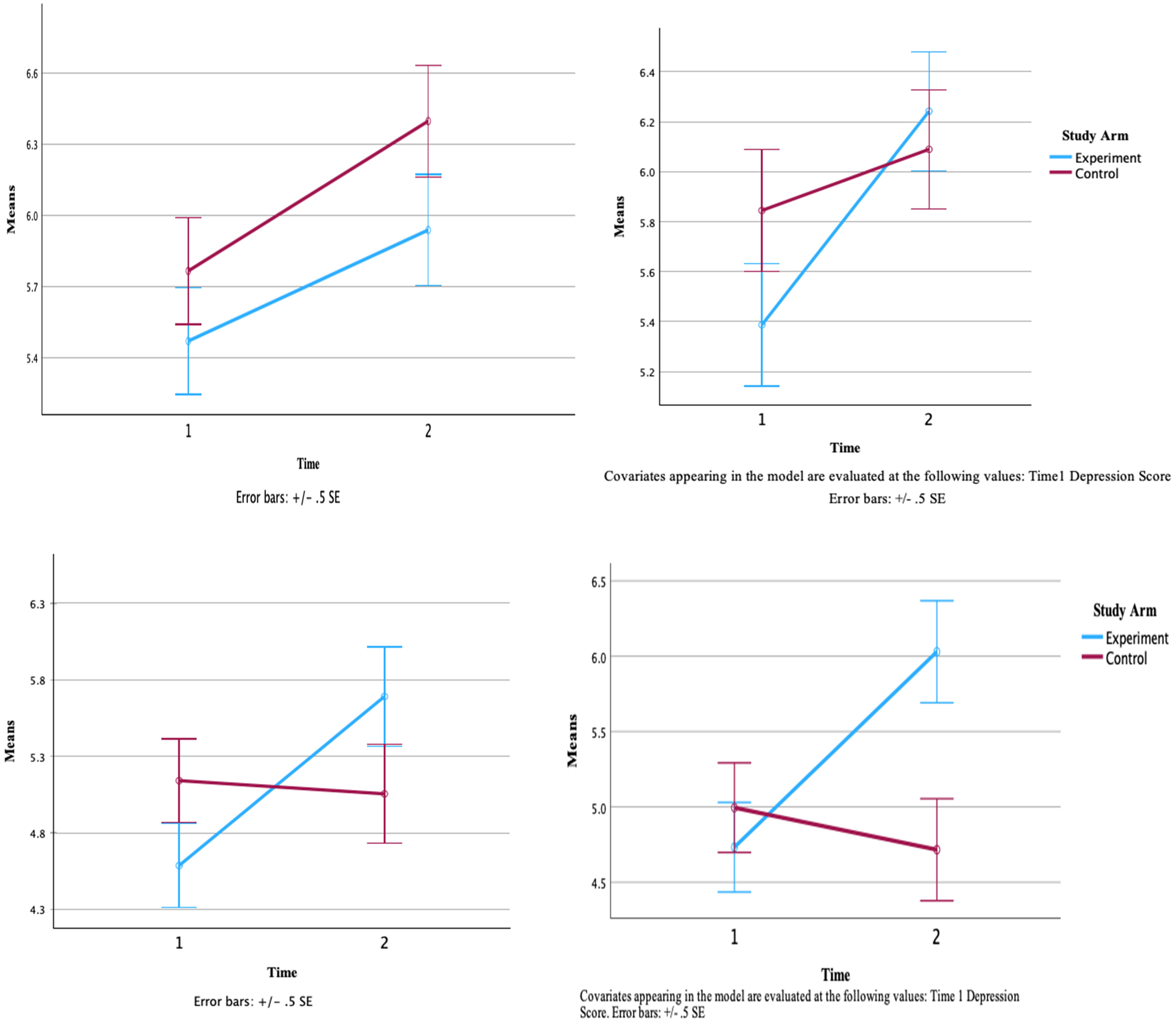
Average observed scores for Canadian occupational performance measure across time. COPM-S scores before (left) and after (right) controlling for depression. The left graph represents raw COPM-S scores without adjusting for depression, while the right graph shows COPM-S scores after statistically controlling for the effects of depression.
Analysis of effect sizes and required sample sizes
Effect sizes (η²) ranged from 0.13 (COPM-S) to 0.0003 (PDQ), as shown in Table 3. The COPM-S had a medium effect size (0.13), while the COPM-P (0.07), MFI-Physical (0.05), OBQ-11 (0.05), and MFI-Reduced Motivation (0.03) had small to medium effect sizes. The PDQ effect size (0.0003) was negligible. Based on our results, sample size calculations for future studies indicated that the COPM-S would require the smallest sample size (n = 46), while the PDQ-8 would require the largest (n = 48,177). The COPM-P, MFI-Physical, OBQ-11, and MFI-Reduced Motivation required minimum total sample sizes of 70, 96, 96, and 154 participants, respectively.
Recruitment efficacy and sociodemographic variability
Between August 2021 and February 2023, 35 individuals contacted the research team regarding study participation. Participants learned of the study through patient organizations and support groups (n = 30, 80%), social media advertisements (n = 4, 11%), and clinics (n = 1, 2%). Of those who reached out, 31completed initial screening and 26 of 31 (84%) met the eligibility criteria. A total of 25 participants completed baseline assessments (pre-intervention), and 22 (88%) completed post-intervention assessments.
Although the sample size was small, it was diverse in terms of clinical and personal characteristics. Participants ranged in age from 55 to 81 (median = 67) and disease duration ranged from 1 to12 years (median = 5) (see Table 2). However, diversity in disease stage, living status, and gender was less evident. The majority of participants (n = 18, 72%) were male and in mild- moderate disease stage (self-reported HY1–2) (n = 16; 64%).
Discussion
This pilot RCT is the first to evaluate the feasibility and preliminary effectiveness of the Packer Managing Fatigue: The Individual Self-Management Program in people with PD. Our findings showed trends toward significant improvement in satisfaction of performance in prioritized daily activities measured by COPM-S and changes within the intervention group, consistent with previous research in individuals with multiple sclerosis (MS) and neuromuscular conditions.21,55
However, unlike these previous studies, we did not observe significant differences between groups or over time for occupational performance (COPM-P). This discrepancy may be due to differences in participant diagnoses and the integration of aerobic training, 21 as well as other theories and strategies such as the Energy Conservation/Envelope Theory and techniques like goal-setting and self-monitoring 55 in the previous studies. These approaches may help individuals manage their energy levels even more effectively and set realistic goals, leading to improved performance and satisfaction with their performance. Another explanation may lie in PD participant feedback that the COVID-19 pandemic had disrupted daily occupational performance during the current study which was conducted during that time. 56 Other studies have also shown that the pandemic negatively impacted essential daily activities, and exacerbated physical symptoms, and mental health problems for people with PD.57–59 People with PD were more susceptible to COVID-19 infection and experienced a worse disease course. 60 This disruption, along with the small sample size, may explain the lack of statistically significant findings in our study. Additionally, non-parametric tests were used for the COPM-P outcome due to violations of normality and homogeneity of variance assumptions. While these tests are appropriate under such conditions, they are generally less powerful than parametric alternatives, which may reduce sensitivity to detect subtle effects. Although the use of non-parametric methods addressed the assumption violations, the lack of statistically significant findings may still be attributed to limited statistical power—potentially due to the small sample size—which could obscure true effects. 61 Given these findings, future research should further evaluate the suitability and sensitivity of the COPM-P as an outcome measure in similar intervention studies.
Baseline depression emerged as a key factor influencing outcomes. Participants in the intervention group reported higher depression scores at baseline, which affected COPM outcomes. When controlling for depression, the time by group interaction remained nonsignificant for COPM-P, but the pattern of change varied. Although the intervention group did not show statistically significant improvements compared to the control group, the direction of the average observed scores shifted positively over time. When we examined the individual data for each participant, we observed that three participants in the intervention group, who also had the highest depressive scores, reported the worst COPM-P scores. Additionally, four participants in the usual care group reported beginning other health interventions during the study, including exercise and physiotherapy, which was not observed in the intervention group and may have influenced the overall results in this low sample size study. For COPM-S, controlling for depression made the differences between groups more pronounced, changing the p-value from 0.09 to 0.05. Depression is a known predictor of fatigue in people with PD,62,63 reinforcing the need to evaluate its role in future studies to better understand its interaction with fatigue self-management programs and their effectiveness on performance in daily life activities.
Prior studies have overlooked the potential impact of the Packer Managing Fatigue program on occupational balance. This essential concept in occupational therapy refers to maintaining the right mix and variety of daily activities, including self-care, leisure, and productivity. 64 This study found a small to moderate effect size for the program's impact on occupational balance, as measured by OBQ-11, highlighting the need for future RCTs with a minimum sample size of 38 to measure this outcome. Given the program's emphasis on prioritizing and balancing activities, changes in occupational balance are anticipated. The preliminary results suggest that OBQ-11 is a valuable tool for assessing the program's impact on occupational balance. Incorporating this measure in future research can provide insights into how the Packer Managing Fatigue program enhances occupational balance and guide future studies.
Prior RCTs in MS have demonstrated the program's efficacy in reducing fatigue impact across physical, cognitive, and psychosocial domains, as measured by the FIS and MFIS.18,20,65,66 In our study, we did not find significant differences between groups for fatigue impact measured with MFI. However, small to moderate effect sizes were observed for the Reduced Motivation and Physical subscales of the MFI. Effect sizes provide valuable information about the magnitude of the differences and can offer insights that p-values alone might not reveal, especially in studies with small sample sizes.67,68 Given the strong evidence supporting the MFIS's ability to detect changes following program participation,69,70 and its emerging psychometric properties in PD, 71 future RCTs evaluating the program in people with PD should consider incorporating the MFIS to better capture the program's impact.
Recruitment for this study was impacted by several factors. COVID-19-related lockdowns and restrictions posed logistical challenges, particularly during periods of heightened public health measures. Additionally, recruitment was limited to Ontario and Nova Scotia due to occupational therapists’ licensing requirements. The most effective recruitment strategy in this study was direct communication through patient organizations and support groups, aligning with previous research that emphasizes the benefits of recruiting from patient groups to ensure a diverse sample and response rate. 72 However, the reach of recruitment via social media and other methods remains unclear.
Despite a small sample size, our study included a diverse range of participants in terms of clinical and demographic characteristics (age, living status, education, disease duration). However, we observed fewer female and late-stage participants in this study. This is expected as PD is more prevalent in males, with a relative risk 1.5 times greater than in females. 73 Given that disease and fatigue may be experienced differently between the sexes, 74 strategies to include both sexes equally could improve understanding of the program's impact. Additionally, recruitment of individuals in late stages PD is critical, as fatigue tends to progress with disease severity. 75 A longitudinal study found that severe motor disabilities predict fatigue in persons with PD, which is more expected in the later stages of the disease. 76 Difficulties in recruitment, particularly for those in later stages, may also be due to barriers that people with PD and their caregivers face in participating in research 77 and the presence of apathy. 78
Despite its contributions, this feasibility study has limitations. Conducted during the COVID-19 pandemic lockdown, the recruitment process faced significant challenges, resulting in a smaller than expected sample size. Due to the small sample size, only two time points (baseline and post-test) were analyzed and reported. Pandemic's restrictions may have influenced participants’ willingness and ability to engage in daily activities, potentially limiting the intervention effects. Using videoconferencing for recruitment may have excluded individuals without reliable internet access. Nevertheless, the findings support the feasibility of delivering the Packer Managing Fatigue program to people with PD. Focus future RCT planning should select outcome measures that align with feasible sample sizes, outcomes such as COPM-S, OBQ-11, and the MFI Physical subscale, to ensure sufficient power to evaluate the program's impact.
Conclusion
Our study provides preliminary evidence for the feasibility and potential benefits of the individual version of the Packer Managing Fatigue program in people with PD. By evaluating and reporting statistically significant levels and effect sizes for multiple expected outcomes, future trials evaluating the program in people with PD can plan more specifically for the selection of outcome measures as well as the required sample size. However, due to the small sample size and wide confidence intervals, future RCTs are necessary to draw definitive conclusions about the program's effectiveness for people with PD. Additionally, a longer follow-up with an adequate sample size may offer more insights into the program's long-term impact.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251388329 - Supplemental material for Managing fatigue in Parkinson's disease: Preparing for a randomized controlled trial
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251388329 for Managing fatigue in Parkinson's disease: Preparing for a randomized controlled trial by Neda Alizadeh, Tanya Packer, Ingrid Sturkenboom, Grace Warner and Heather Rigby in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We would like to express our gratitude to the study participants and the dedicated occupational therapists who contributed to this research. Their invaluable support and participation were instrumental in making this study a success. Additionally, we acknowledge that this study is part of the first author's manuscript-based dissertation, which was published by Dalhousie University. We also thank the Nova Scotia Health Research Ethics Board for their ethical oversight and approval of this study.
Ethical considerations
This study received ethical approval from the Nova Scotia Health Sciences Research Ethics Board (ref: 1027048) on 08,03, 2021.
Consent to participate
All participants provided written informed consent prior to enrollment in the study.
Consent for publication
Not applicable. This study reports only aggregated, non-identifiable data, and no individual details, images, or videos are included.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted as part of the PhD research of the first author. This work was supported by Abdulmajid Bader scholarship; Funding Package for PhD studies from the School of Occupational Therapy, Dalhousie University; Scotia Scholars Award (George Turnbull Student Research Award), Nova Scotia Graduate Scholarship Program (NSGS); and the LSVT Global Student Grants (LSVT-S).
Declaration of conflicting interests
The authors report the following potential conflicts of interest related to the research, authorship, and/or publication of this article: Packer is a co-founder of Think Self-Management Inc., a company that equips healthcare professionals with tools and resources to aid individuals with chronic conditions in developing self-management skills. Although Packer holds no financial interest in the company, the Packer Managing Fatigue program is distributed by Think Self-Management. The other authors have no real or potential conflicts of interest to declare concerning this research, authorship, or publication.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
