Abstract
Background
Anxiety is a common non-motor symptom in Parkinson's disease (PD) and has previously been associated with changes in cortical thickness of various brain regions.
Objective
To identify changes in cortical thickness in PD-related anxiety.
Methods
148 patients from an ongoing cohort study were included: 30 PD patients with anxiety, 73 PD patients without anxiety and 45 healthy controls. Anxiety was measured with the Parkinson Anxiety Scale (PAS). A 7 T structural MRI scan was performed and used to compare cortical thickness between these groups. Region of interest (ROI) as well as whole-brain analyses were performed to identify differences.
Results
ROI analyses revealed a strong negative association between the cortical thickness of the left lingual gyrus and the severity of anxiety in PD patients (R = −0.71; p = 0.006). Additional significant strong negative associations with the severity of anxiety in PD patients were observed in the frontal and cingulate regions (R between −0.56 and −0.65). Whole-brain analysis revealed a significant cluster of cortical thinning in the left anterior cortex and the dorsolateral prefrontal cortex weakly associated with PAS total score across all groups (R = −0.25, p = 0.00201).
Conclusions
This study is the first to report a strong negative association between left lingual gyrus thickness and anxiety severity in PD. Additionally, anxiety in early PD is associated with cortical thinning in the fronto-cingulate region, mainly affecting left sided structures. Future studies should examine whether these cortical changes can predict the anxiety progression patterns or the treatment response in PD patients.
Plain language summary
Anxiety is a common symptom in Parkinson's disease (PD) and has been linked to changes in thickness of certain cortical areas in the brain. Our study examines whether specific cortical areas show an altered thickness in early diagnosed PD patients that suffer from anxiety compared with those not suffering from anxiety. 148 participants were included: 30 PD patients with anxiety, 73 PD patients without anxiety, and 45 people without PD. Each participant underwent an ultra-high-field 7 T MRI-scan to determine regional cortical thickness. By comparing the thickness of different regions between the groups we found that people with both PD and anxiety displayed thinning of the ‘lingual gyrus’, a specific area in the back part of the brain, compared to the other two groups. Secondly, the people in the PD group with more severe anxiety showed thinning of the ‘fronto-cingulate region’, an area located more toward the front of the brain, compared to the other two groups. Finally, in all three groups, people with a higher anxiety score showed thinning of the ‘prefrontal cortex’, an area in the front of the brain, compared to the participants with a low score for anxiety. The exact role of these areas in the brain of anxious people with PD is not clear yet. Still, the results are interesting and should be further investigated.
Introduction
Parkinson's disease (PD) is a neurodegenerative disease characterized by both motor and non-motor symptoms, including neuropsychiatric symptoms. Among these neuropsychiatric symptoms, anxiety is one of the most frequent with approximately 31% of PD patients experiencing significant anxiety symptoms. 1 The underlying mechanisms of PD-related anxiety are still poorly understood. In PD, increased anxiety levels have been linked to structural and functional changes in anxiety-related circuits, consisting of the ‘fear circuit’ and the ‘limbic anxiety circuit’. 2 The fear circuit is a neuronal circuit involved in fear processing and includes the amygdala, anterior cingulate cortex (ACC), the medial prefrontal cortex (mPFC), the insular cortex (IC), the hippocampus and the striatum. The limbic loop of the cortico-striato-thalamo-cortical circuit, also called the “limbic anxiety circuit”, is involved in emotional control. While altered functional connectivity has been observed between structures within these circuits, little is known about alterations of the cortical grey matter structures. 2
In the general population, most studies on fear and anxiety disorders reported a decrease in cortical thickness of the ventromedial (vmPFC) and dorsolateral (dlPFC) prefrontal cortex, as well as cortical thinning in several other cortical regions, such as the lateral orbitofrontal cortex (OFC), medial OFC, ACC and caudal middle frontal gyrus.3–6 In PD patients, a reduced cortical thickness in the bilateral ACC and left parietal regions is reported in patients with anxiety compared to patients without anxiety. 7 Additionally, higher levels of anxiety correlate with a reduction in grey matter volume in the ACC and precuneus. 8 These findings suggest that cingulate and parietal areas may be involved in the underlying mechanisms of PD-related anxiety through disrupted emotion regulation, reflecting dysfunction in the fear circuit. However, studies focusing specifically on cortical thickness in patients with PD-related anxiety are limited, and so far, none was based on ultra-high field 7-Tesla MRI, which can reveal subtle cortical alterations that may be undetectable at 3 T resolution. Identifying changes in cortical thickness in these patients could help to decipher the underlying mechanisms of PD-related anxiety, which is essential for development of more effective treatment options.
The aim of this study was to identify changes of brain cortical thickness in PD patients suffering from anxiety using ultra-high field 7-Tesla MRI scans. For PD patients with anxiety symptoms, we hypothesize a reduction of cortical thickness in areas involved in the anxiety circuits, such as the ACC and mPFC, as well as in the parietal regions, which were previously associated with anxiety.7,8
Methods
Study design
We analyzed data from the TRACK-PD study. 9 This is an on-going longitudinal observational study that includes recently diagnosed PD patients without dementia (≤ 3 years after diagnosis), and healthy controls (HC), with 4-year of follow-up assessments. All data were collected at Maastricht University Medical Centre. At each visit (baseline, 2- and 4-year follow-up) participants were extensively assessed with (non)motor-scores, questionnaires neuropsychological battery and underwent 7-Tesla brain MRI scanning. In this cross-sectional study, we only used the baseline dataset. The study design is described in more detail by Wolters et al. 9 Ethical approval was provided by the local medical ethical committee, and the study has been registered (Dutch Trial Register, NL7558). Written informed consent was obtained from all participants.
Study population
Patients were recruited locally and nationwide. Local recruitment targeted the PD population of the movement disorder clinic in Maastricht University Medical Centre, Maastricht, the Netherlands. Nationwide recruitment was conducted through online advertising. Patients included in this study had received a diagnosis of idiopathic PD in the past three years and were between 35 and 80 years old. The diagnosis of PD was made by a neurologist based on the MDS clinical diagnostic criteria. 10 Patients with other neurological disorders, dementia or severe cognitive impairment (Montreal Cognitive Assessment (MoCA) score <24), contra-indications for MRI, major depressive disorder (MDD) or abuse of alcohol, drugs or benzodiazepines were excluded.
At baseline, demographic and clinical variables were collected including sex, age, handedness, and disease duration.
The non-motor, motor symptoms and disease severity were assessed by the Movement Disorders Society - Unified Parkinson's Disease Rating Scale (MDS-UPDRS) part I and III and the Hoehn & Yahr scale (H&Y), respectively. The ‘Beck Depression Inventory’ (BDI) was used to assess depression. 9
Assessment of anxiety
The Parkinson Anxiety Scale (PAS) was used for the assessment of anxiety symptoms. It has subsections for persistent anxiety (PAS-A), episodic/situational anxiety (PAS-B), and avoidance behavior (PAS-C). 11 A PAS-total score was calculated by summing the three sub-scores. PD-patients were considered to have significant PD-related anxiety if they had a score above the cut-off in at least one of the three subparts of the scale: PAS-A > 9, PAS-B > 3, or PAS-C > 3. PD participants were divided into two groups: 1) PD-patients with anxiety (Anx-PD), 2) PD-patients without anxiety (noAnx-PD), alongside a third group of healthy controls (HC).
Image acquisition
MRI-data was acquired using a 7-Tesla MRI-scanner (Magnetom, Siemens, Erlangen, Germany) equipped with a Nova Medical 32-channel head coil. Dielectric pads were applied to enhance the signal in the temporal brain regions. Cardiac and respiratory physiological signals were measured synchronized with the scan start. A whole-brain MP2RAGE (Magnetization Prepared 2 Rapid Acquisition Gradient Echoes) acquisition was used with an acquisition time of 10:57 min, resulting in a T1-weighted image and a quantitative T1 map. These settings were used: TE = 2.51 ms, TR = 5000 ms, TI = 900 ms and 2750 ms, flip angle = 5° and 3°, FoV = 208 mm, resolution (x-y-z) = 0.65 × 0.65 × 0.65mm3, slices = 240, orientation = sagittal (more details in the published protocol) (9).
Image processing and cortical thickness extraction
After dicom to nifti conversion using dicomx, structural brain MRI scans were processed using FreeSurfer (version 6.0.0) (https://surfer.nmr.mgh.harvard.edu/) for extraction and segmentation of the cortical thickness. 12 Pial and white surfaces were automatically segmented. The “Desikan-Killiany” cortical atlas, a gyral based atlas including 34 cortical areas bilaterally, was used to define the cortical regions of interest (ROI) in MNI-space. 13
Quality control was performed by trained researchers (ML, GC) and each surface was visually inspected and corrected manually as needed. Quality control steps recommended for FreeSurfer were performed.
The average cortical thickness of each cortical region of the atlas was extracted and provided in mm for each participant.
Cortical thickness analysis at vertex level
The open-access multimodal processing and data co-registration pipeline micapipe (V.0.2.0) was used to process anatomical images. 14 T1w images were deobliqued, reoriented to standard neuroscience orientation (LPI), corrected for intensity nonuniformity, intensity normalized, and skull stripped. Cortical features were projected from the native space to the fsaverage5 midsurface (with approximately 10,000 surface vertices per hemisphere) and smoothed at 5 mm full width at half maximum (FWHM), using nilearn python library and Workbench tools (v. 1.4.2). Finally, all cortical surface maps were individually z-scored.
Statistical analysis
To investigate the relationship between anxiety and cortical thickness, we employed two separate analyses: a whole-brain analysis and a region of interest (ROI) analysis.
The ROI analysis was employed to assess mean cortical thickness within pre-defined brain regions. Using the mean cortical thickness of each region instead of focusing on individual voxels makes this analysis suitable for the detection of regional alterations in cortical thickness. Additionally, it facilitates comparison to existing literature on cortical thickness as it uses pre-defined cortical areas.
The whole-brain analysis served as an exploratory analysis to observe differences in cortical thickness on a vertex level without a priori assumptions about regions that could possibly be affected. Conducting this analysis allowed the detection of diffuse alterations in cortical thickness that might not be identified in a more targeted analysis.
The significance threshold was set at p-value < 0.05 and corrected for multiple comparisons –using the False Discovery Rate (FDR) when necessary. The numerical variables were described as means and standard deviations, the ordinal and the categorical variables as frequencies and percentages. The normality of distribution was assessed using a Kolmogorov-Smirnov test.
Descriptive analyses
Group differences were assessed using Chi2 tests for categorical variables and ANOVA tests for continuous variables, using SPSS, version 29 (SPSS, Chicago, 2023). A Mann-Whitney U test was used to compare the H&Y stages.
Region of interest analysis
Cortical thickness was analyzed across all 34 bilaterally defined regions from the Desikan-Killiany atlas. 13 The mean cortical thickness of each region was normalized using Z-scores.
First, group comparison analyses of those z-scores between the three groups were performed using MANCOVA tests with age, sex and BDI-score as co-variables. No other covariables were added.
Secondly, hierarchical multiple regression analyses were performed to examine the relationship between the PAS-total score and the mean cortical thickness (Z-scores) of the cortical regions. This regression-based approach was chosen over simple correlational analyses to control for potential confounders and to quantify the effect of PAS-total score on cortical thickness alterations.
In this analysis, age, sex and depressive symptoms (BDI-score) were set as nuisance regressors in the first block (model 1) of all regression models, whereas PAS-score (independent variable) was separately added to the second block of the model (model 2). The mean cortical thickness (Z-score) of each cortical region was set as a dependent variable.
We ensured that all models met the assumptions for multiple regression analyses, including normality of the residuals, multicollinearity, and homoscedasticity.
Whole-brain analysis
Surface-based linear models (SLM in python brainstat toolbox) were performed to investigate the relationship between two key variables: BDI score and PAS score. Both variables were included as fixed effects in a single model, allowing assessment of their individual contributions to cortical thickness while controlling for the other variable. Age and sex were added as nuisance covariates. No other covariates were added.
This model was then fitted to our neuroimaging data, specifically the cortical thickness measurements. We applied a cluster-wise correction for multiple comparisons using both FDR and Random Field Theory (RFT) methods, with a cluster-defining threshold of p < 0.01.
Results
Demographic and clinical characteristics
Of the 151 participants originally included in the study, one was excluded at baseline due to withdrawal of consent. Therefore, 150 participants were included in this analysis. After MRI image quality control, 2 additional participants were excluded because of too poor quality of the MRI data despite correction and reprocessing steps. The 148 participants consisted of 30 PD patients with anxiety (Anx-PD), 73 PD patients without anxiety (NoAnx-PD), and 45 healthy controls. Baseline characteristics of these 148 subjects are reported in Table 1.
Demographic, cognitive and clinical characteristics in Parkinson patients with anxiety, without anxiety and healthy controls.
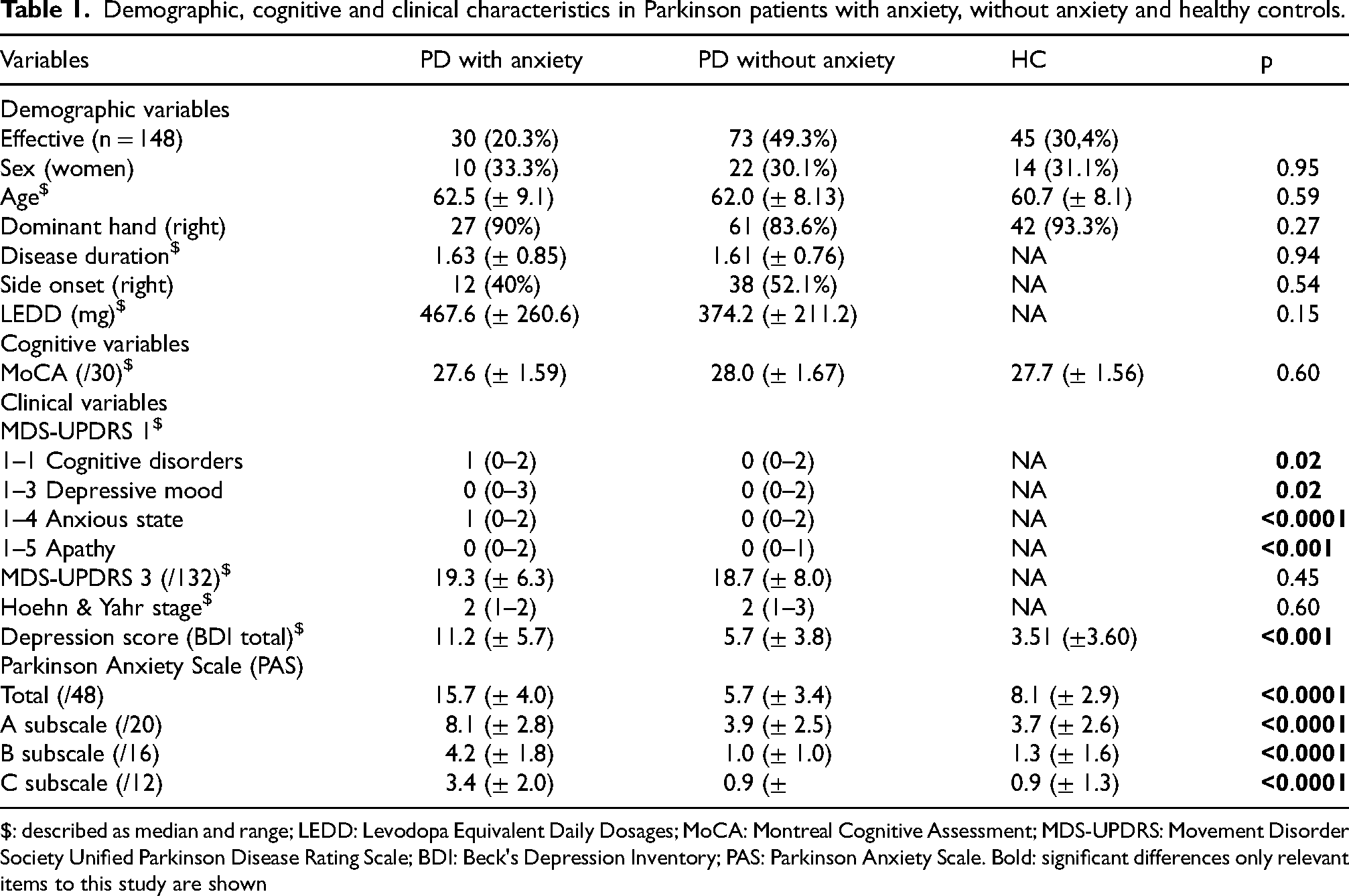
$: described as median and range; LEDD: Levodopa Equivalent Daily Dosages; MoCA: Montreal Cognitive Assessment; MDS-UPDRS: Movement Disorder Society Unified Parkinson Disease Rating Scale; BDI: Beck's Depression Inventory; PAS: Parkinson Anxiety Scale. Bold: significant differences only relevant items to this study are shown
ROI-based analyses
There was no significant difference in the mean cortical thickness of each ROI using Z-scores between the three groups (Supplemental Table 1).
In the group of PD patients (both anxious and non-anxious), there were significant weak negative associations between the PAS-total score and the cortical thickness of the left temporal cortex (fusiform gyrus, lingual gyrus, middle and superior temporal gyrus), the left medial orbitofrontal cortex, and the left rostral middle frontal gyrus (R between 0.27 and 0.39; Supplemental Table 2) There was no association of anxiety with the ROIs of the right hemisphere (Supplemental Table 2).
There were significant weak negative associations between the PAS-A sub-score (persistent anxiety) and the cortical thickness in the superior frontal cortex, the rostral middle frontal cortex, the middle and inferior temporal cortex and the lingual cortex, in the left hemisphere (R between 0.34 and 0.40; Supplemental Table 3).
In the group of PD patients with anxiety, there was a significant strong negative association (R = −0.71, p = 0.001) between the cortical thickness of the left lingual gyrus and the severity of anxiety according to the PAS-total score (Table 2 and Figure 1). Additionally, there were significant strong negative associations between the PAS-B sub-score (episodic anxiety) and the cortical thickness in the superior and inferior frontal cortex, pars orbitalis and pars triangularis, the paracentral cortex, the isthmus and posterior cingulate cortex and the inferior temporal cortex in the left hemisphere; and the pericalcarine cortex in the right hemisphere (R ranging from −0.57 to - 0.65; Table 2 and Figure 2).
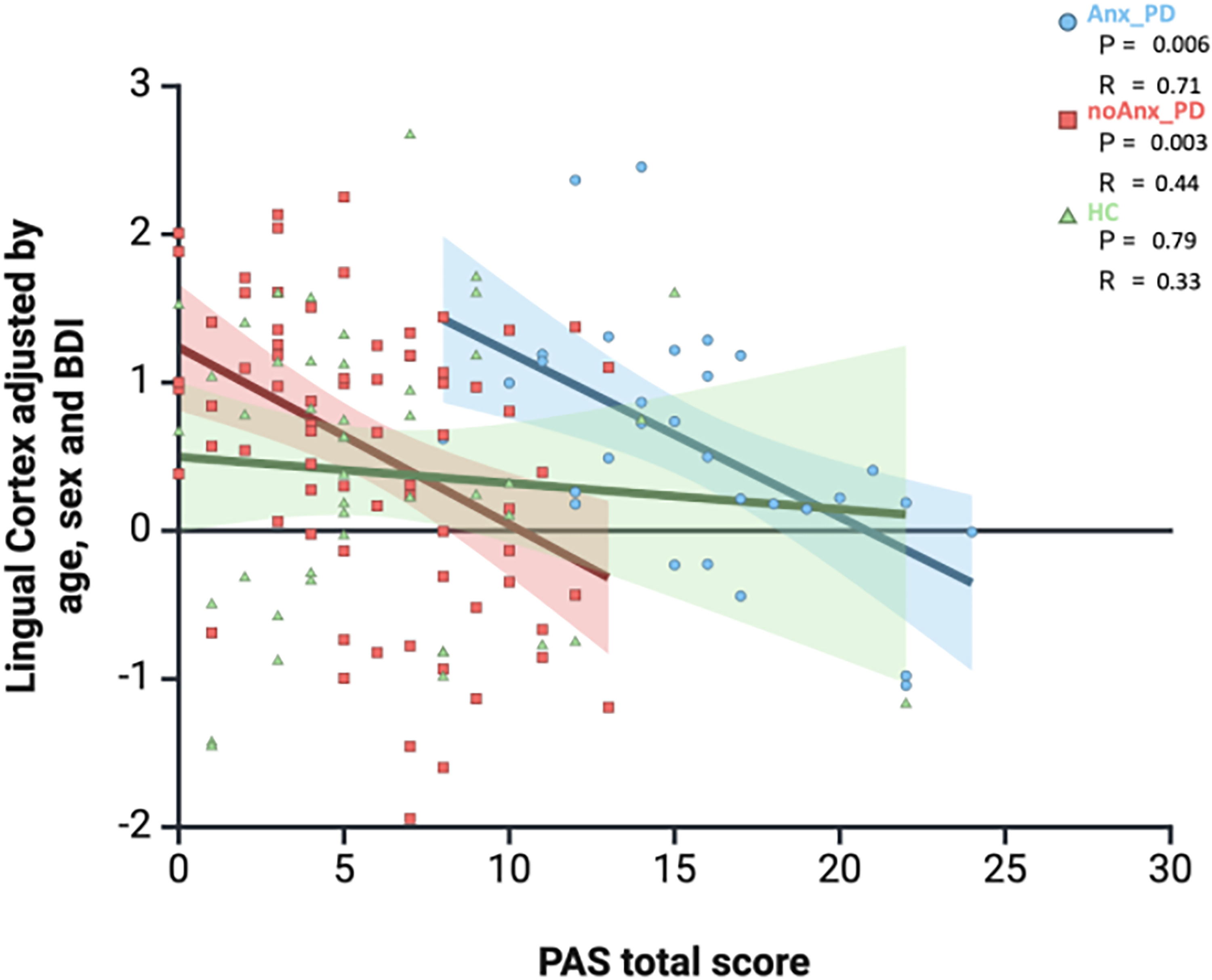
Regression analysis between cortical thickness and severity of anxiety symptoms according to PAS-total score in Parkinson's disease (PD) patients and healthy controls: Visualization of the negative association between the cortical thickness (Z-score) of the left lingual gyrus and PAS-total score in anxious PD patients. Adjusted by sex, age, and BDI-score.
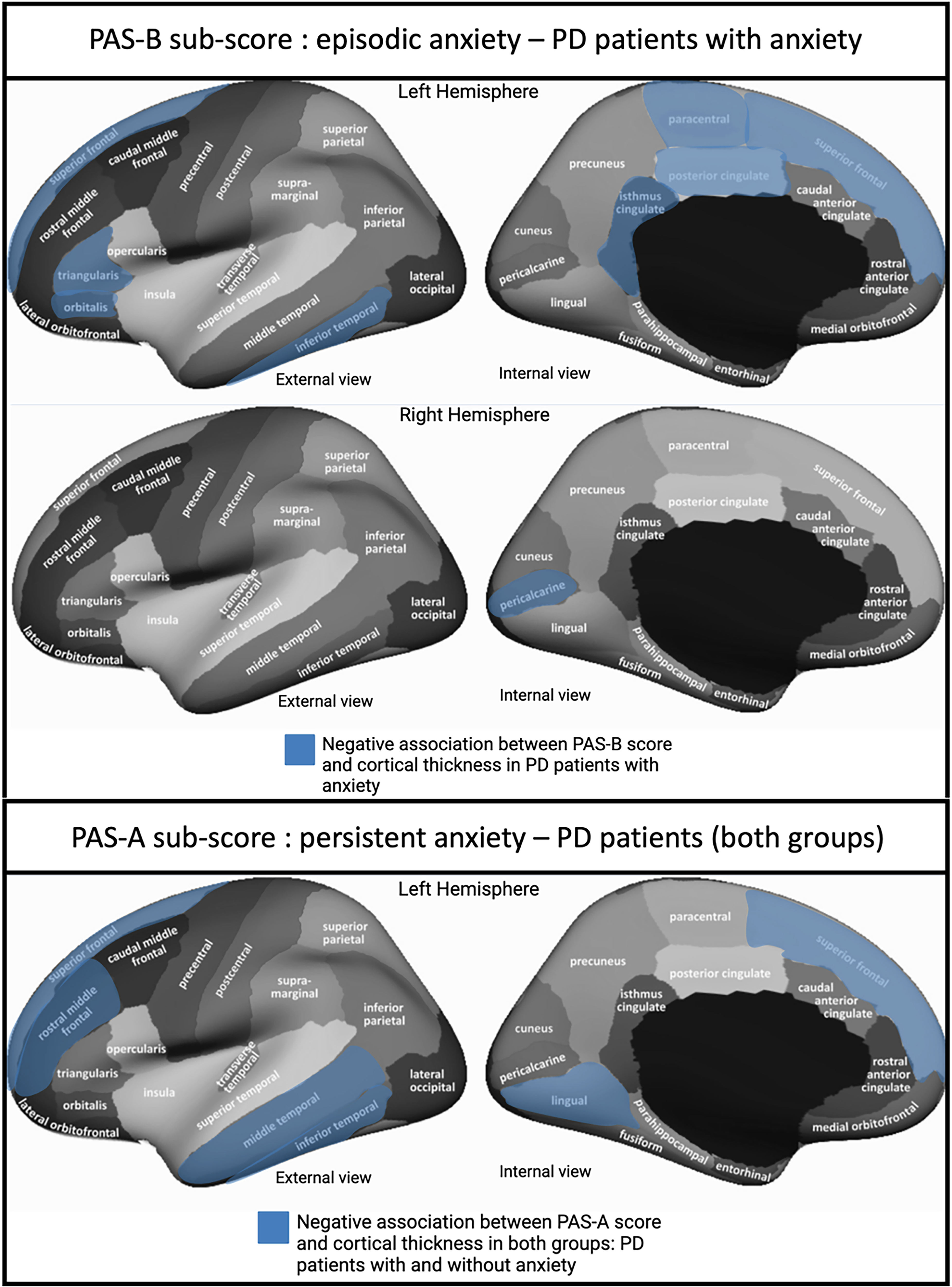
Representation of the multiple linear regression analysis in PD-patients with anxiety (upper image) and in PD-patients both with and without anxiety groups (lower image).
Regression analysis between the cortical thickness in brain region of interest (ROI) and PAS-total score (A) and PAS-B subscore (B) in Parkinson's disease PD patients with anxiety subgroup.
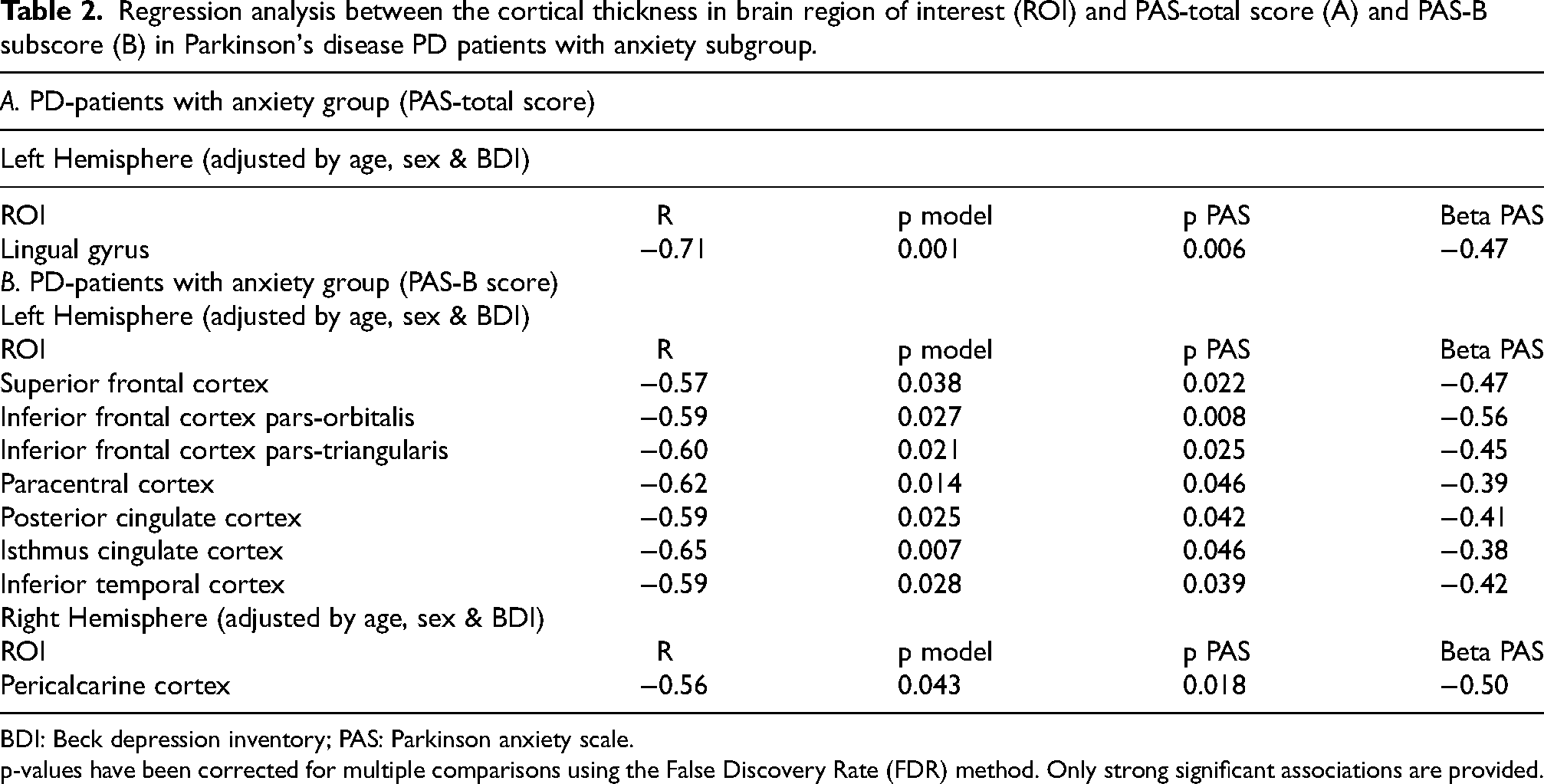
BDI: Beck depression inventory; PAS: Parkinson anxiety scale.
p-values have been corrected for multiple comparisons using the False Discovery Rate (FDR) method. Only strong significant associations are provided.
There was no significant association between the cortical thickness and the PAS (sub)scores, in the HC group.
Whole-brain vertex-wise cortical thickness analysis
In the three groups, there was a significant cluster in PAS-total score, as in PAS-A subscale, which shows a weak negative association with cortical thickness in the left dorsolateral prefrontal cortex (DLPFC), mostly in the anterior part of the middle frontal gyrus (R = −0.25, p = 0.00201) (Figure 3). There were no significant associations with PAS-B and PAS-C sub-scores.
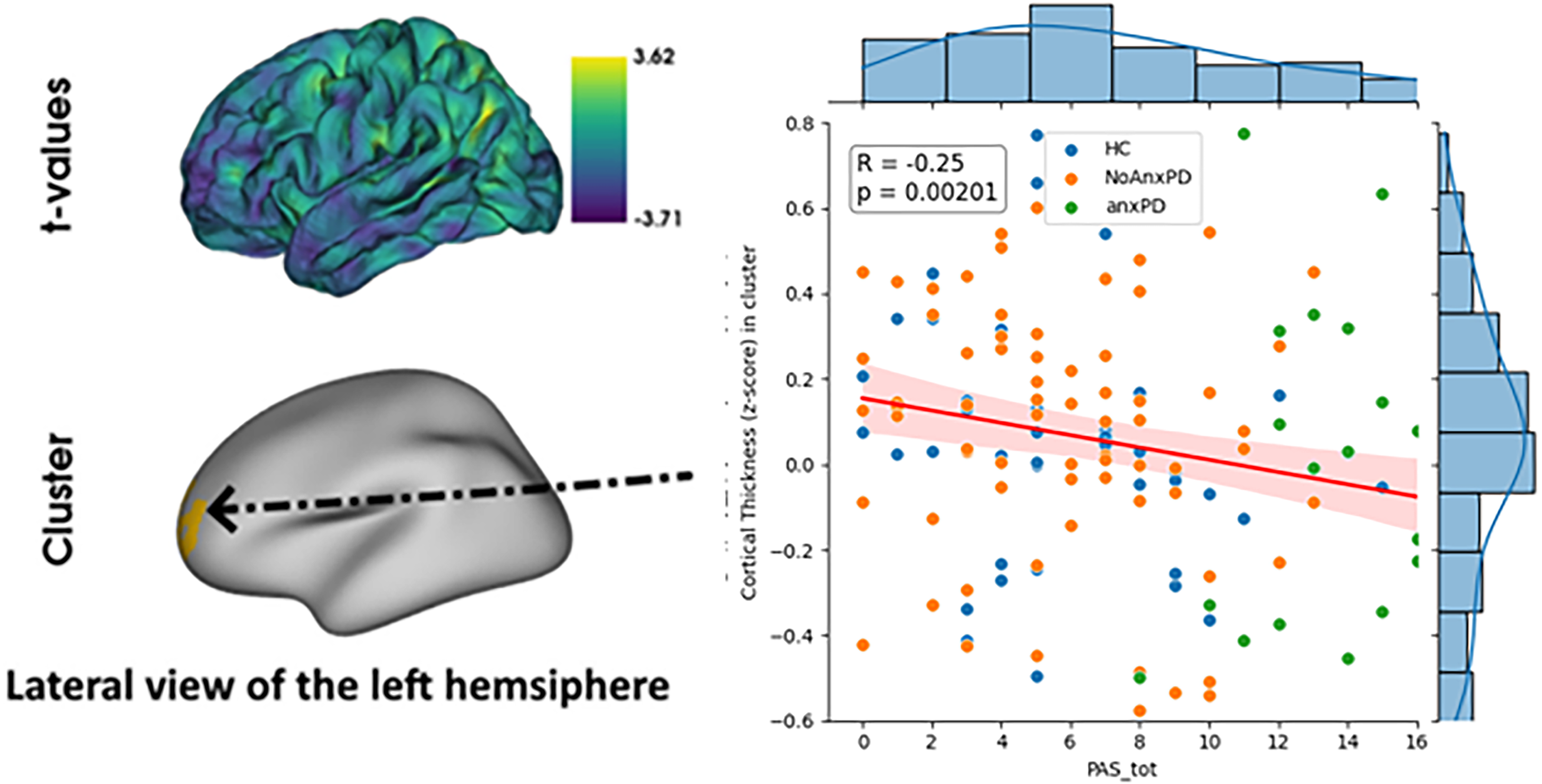
Whole-brain vertex-wise cortical thickness analysis. PAS-total score shows a significant negative association with cortical thickness in the middle frontal gyrus in Parkinson patients with anxiety (anxPD), compared to Parkinson's Disease patients without anxiety (noAnxPD) and healthy controls (HC). Adjusted for age, sex and BDI-score. PAS: Parkinson Anxiety Scale.
Discussion
In this study, we examined the differences in cortical thickness between PD patients with anxiety, PD patients without clinically relevant anxiety and healthy controls. We conducted both ROI-based and whole-brain analyses. The ROI-based analyses revealed no between-group differences but did show strong negative associations between the PAS-total score and the cortical thickness of the left lingual gyrus in both anxious and non-anxious PD patients and between the PAS-B score and the cortical thickness of the left fronto-cingulate, left temporal and right pericalcarine cortex in the group of PD patients with anxiety. Furthermore, there were weak negative associations between PAS (sub)scores and parts of the left frontal and temporal cortex in the group of PD patients (with and without anxiety). In the whole-brain analyses, there was a weak association between the cortical thickness of the left DLPFC and the PAS-total and PAS-A scores. Those results are partly in line with previous findings showing reduced thickness of the left fronto-cingulate cortex in PD-related anxiety, but they also show that underlying mechanisms could differ depending on the subtype of anxiety.7,8,15 These results also suggest a potential implication of the lingual cortex.
PD related anxiety is associated with cortical thinning in visual and prefrontal regions
While most recent studies on cortical thickness in anxiety used 3 T MRI,3,4,6,8 the application of ultra-high field 7 T MRI in an early PD population enabled higher spatial resolution and potentially more accurate cortical thickness measurements. 16 The ROI-based analyses revealed a strong negative association between the cortical thickness of the lingual gyrus in the temporo-occipital lobe and anxiety severity in PD patients. This is a novel finding that may reflect the potential enhanced sensitivity of 7 T MRI, which could explain why this area was not reported more often with regards to PD anxiety in 3 T MRI studies. The whole brain analyses show a weak negative association between anxiety severity and cortical thickness of the dlPFC, which is mentioned more often with regards to anxiety in literature.5,7
The lingual gyrus is part of a large network that encodes, analyzes, and gives emotional meaning to visual information and is connected to the amygdala through the ventral visual stream.17–19 Existing literature on anxiety mentions that the lingual gyrus was thicker in patients with social anxiety disorder. 20 While this contrasts with our finding of cortical thinning, it may suggest that anxiety patients are particularly sensitive to interactions with the environment. Structural thinning of the lingual gyrus could disrupt the integration of visual and emotional information, potentially exacerbating anxiety symptoms in PD patients. 21 Future research should investigate the potential of the lingual gyrus being a potential imaging marker for anxiety progression in PD.
The PFC is a key structure of both the fear and limbic circuits and thinning in this area has previously been observed in anxious individuals and anxious PD patients.7,22 The observed negative association between anxiety severity and cortical thickness in the dlPFC may reflect the vulnerability of structures within these circuits for cortical thinning in anxiety.23,24 The present finding aligns with prefrontal regions responsible for cognitive control over fear and emotion showing cortical thinning in anxiety disorders.5,7
Episodic anxiety in PD patients is associated with cortical thickness of the fronto-cingulate regions
The ROI-based analyses showed a strong negative association between episodic anxiety severity and cortical thickness of structures within the left fronto-cingulate region. The fronto-cingulate regions play a key role in mediating top-down regulation of emotions through the inhibition of limbic structures such as the amygdala, which are involved in fear signaling.7,25 Through this inhibitory regulation, the structures within this region can reduce negative emotional activity and intrusive thoughts.
In non-PD patients suffering from anxiety, the strength of the functional connectivity between the posterior cingulate cortex and amygdala showed a negative association with the severity of anxiety. 26 Cortical thinning in the frontal and cingulate regions within the fear circuit could alter the activation of these structures, reducing top-down regulation. Consequently, this could lead to overactivation of the amygdala, resulting in an excessive response to perceived threats.2,7
In terms of cortical thickness, our findings in the fronto-cingulate region are in line with previous findings in anxious PD patients.2,8,15 While cortical thickness in this region is associated with episodic anxiety, rather than persistent anxiety, it highlights the vulnerability of these fear-processing areas in PD patients. Possible therapeutic options, such as cognitive behavioral therapy (CBT), have shown to induce functional connectivity changes between brain structures in PD. 23 Similarly, neurofeedback therapy proved to improve symptoms in anxiety. 27 Future studies could investigate the effect of such therapies on the longitudinal changes in cortical thickness in these areas.
Subtypes of anxiety in PD are associated with different patterns of cortical thinning
Our findings reveal that the patterns of cortical thickness can vary significantly between PAS-total and PAS sub-scores. Specifically, PAS-A (persistent anxiety) and PAS-B (episodic anxiety) demonstrate significant negative associations with cortical thickness, while PAS-C (avoidance behavior) shows no significant results.
This pattern of alterations per anxiety subtype suggests that different forms of anxiety exhibit unique patterns of cortical thinning.5,28 While the results support our hypothesis of cortical alterations in an overactive fear circuit and underactive limbic circuit in anxiety, the underlying mechanisms that cause these alterations could be more extensive than those usually associated with PD related anxiety. In this population, the severity of anxiety is likely due to a combination of factors and is not exclusively due to the neurobiological changes associated with PD. For instance, trait anxiety presents before the onset and diagnosis of the disease as well as the individual coping strategies in response to the emotional stress caused by PD and its symptoms. These findings highlight that research into anxiety must consider the various clinical subtypes, which may have different underlying mechanisms and result in different clinical trajectories.
Additionally, both the ROI and whole brain analyses suggest that cortical thinning is confined to the left side of the brain. While hemispheric lateralization is well-known for language and problem-solving, its role in emotional processing is less clear. 29 The observed pattern of cortical thinning lateralizing to the left hemisphere may indicate that anxiety could be hemisphere specific, possibly aligning with some functions being hemisphere specific.
Strengths and limitations
This study has several strengths as we included a large and well-characterized sample of PD patients and healthy controls. The PAS-score was used as a reliable measure of anxiety symptoms in PD. Imaging was performed with an ultra-high field 7-Tesla MRI scanner. However, this study also has some limitations. First, TRACK-PD is a large cohort study designed to assess the structural characteristics of PD patients. 9 While anxiety assessment was performed for patient characterization, the study design was not focused specifically on anxiety. Secondly, since alterations in cortical thickness were the prime focus in this analysis, we did not analyze subcortical structures such as the basal ganglia and amygdala. Therefore, further analyses including subcortical structures and functional connectivity are needed. The use of a 7-Tesla MRI scanner will help to study these structures with high precision in further analyses. Thirdly, we excluded patients that were diagnosed with MDD. As there is a high comorbidity between anxiety and depression in PD, 30 the generalizability of this study could be limited. However, with this approach we aimed to reduce the confounding effects of depression to allow clearer interpretation of anxiety-related findings. Finally, we focused on the baseline data of an on-going cohort. PD patients were then at a relatively early stage of the disease. Using the follow-up data once they are available is necessary to have a more comprehensive understanding of the mechanisms of PD-related anxiety according to the patients’ clinical trajectory.
Conclusion
The main finding of our study is that anxiety in early PD is associated with cortical thinning of left-hemisphere regions, with the fronto-cingulate region and the left lingual gyrus in particular. Most notably, the strong negative association between PAS-total scores and the left lingual gyrus thickness, identified with 7 T MRI, highlights a novel finding in PD anxiety. While the exact implication of both results in PD anxiety remains unclear, the observed association with anxiety symptoms is strong and significant and warrants further investigation. Our findings aid in the understanding of the underlying pathophysiology of anxiety symptoms in PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251367302 - Supplemental material for Cortical thickness alterations in Parkinson's disease related anxiety: A cross-sectional 7 T MRI study
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251367302 for Cortical thickness alterations in Parkinson's disease related anxiety: A cross-sectional 7 T MRI study by Michiel van Lier, Guillaume Carey, Romain Viard, Mark L Kuijf, Amée F Wolters, Kathy Dujardin and Albert FG Leentjens in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors thank all participants of the study for their patience and cooperation.
ORCID iDs
Ethical considerations
Ethical approval was provided by the local medical ethical committee, and the study has been registered (Dutch Trial Register, NL7558).
Consent to participate
Written informed consent was obtained from all participants.
Consent for publication
Not applicable
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MRI data included in this study were acquired at Scannexus (Maastricht, The Netherlands). This study is funded by a grant from the Weijerhorst Foundation (Stichting de Weijerhorst). This Dutch foundation supports and promotes activities in the public interest, in particular scientific research. The sponsor has no role in the design of the study and collection, analysis, and interpretation of data. Also, the sponsor was not involved in writing this manuscript.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.F.G. Leentjens received consultancy fees from Lilly. G. Carey participated to advisory boards for ORKYN’ and Abbvie and gave consultancies to Abbvie. He was employed by University of Reims (French Government) and earned a grant from France Parkinson.
Mark L. Kuijf, Stijn Michielse and Albert F.G. Leentjens were employed by Maastricht University (Dutch government). Amée Wolters was employed by Catharina Hospital Eindhoven. Kathy Dujardin was employed by University of Lille (French Government).
Data availability statement
This study uses data from the dataset TRACK-PD. The data that supports the findings of this study are available and can be requested from M.L. Kuijf. The data are not publicly available due to privacy considerations.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
