Abstract
Mutations in the leucine-rich repeat kinase 2 (LRRK2) gene associate with familial and sporadic Parkinson's disease (PD). While various LRRK2 allelic variants have been studied, characteristics of R1441C carriers remain underexplored. We compared PD patients carrying the R1441C mutation (90% Israeli Arabs) to those carrying the G2019S (70% Ashkenazi Jews) and R1441G (42% Basque) variants. R1441C carriers exhibited a distinct clinical phenotype characterized by severe motor and non-motor symptoms and worse scores on the Montreal Cognitive Assessment. These findings highlight the importance of ethnic diversity and genetic stratification in PD research. These results need confirmation in larger, more diverse samples.
Plain Language Summary
Parkinson's disease (PD) is a brain disorder that affects movement and thinking. Some cases are caused by mutations in the LRRK2 gene, including G2019S, R1441G, and R1441C. While G2019S and R1441G are well studied, R1441C remains less understood. This study compared PD patients with the R1441C mutation to those with G2019S and R1441G. R1441C carriers had more severe motor and non-motor symptoms, including worse cognitive function. These findings highlight the need to study different genetic backgrounds to better understand PD and improve patient care.
Introduction
Mutations in the leucine-rich repeat kinase 2 (LRRK2) gene are among the most common genetic causes of both familial and sporadic Parkinson's disease (PD).1–5 Various allelic variants exhibit distinct prevalence patterns across geographic regions and ethnic groups.5–13 The LRRK2 Gly2019Ser (G2019S) mutation was found in up to 2.4% of North American PD patients and in 14% high-risk ancestry (Ashkenazi Jews, North African Berber or Spanish Basque) PD patients.6,11,13–15 In Israel, the LRRK2 G2019S mutation is prevalent among Ashkenazi Jews with PD, occurring in 26% of familial cases and in 10.6% of sporadic cases.
The LRRK2 R1441 (including R1441C, R1441G, and R1441H) mutation is the second most common allelic variant of LRRK2 mutation after G2019S. The LRRK2- Arg1441Gly (R1441G) was first detected in familial cases originating from the Basque region, 16 where approximately 13% of PD patients are heterozygous carriers. 9 The LRRK2-Arg1441Cys (R1441C) variant has been documented in both sporadic and familial PD cases in regions such as Germany, Italy, Belgium, the United States, Egypt and Iran.4,7,8,17–21 Additionally, asymptomatic heterozygous individuals of Palestinian Arab descent have been found to carry the R1441C mutation, with an allele frequency of 0.003, 22 although it has not been reported among Israeli Arab PD patients. Consequently, the clinical characteristics of the R1441C variant in this population remain underexplored. Here, we present the clinical characteristics of R1441C carriers and compare them with individuals carrying other LRRK2 variants.
Methods
This study included PD patients with a LRRK2 mutation (R1441C, R1441G, or G2019S); 9 Israeli Arab patients (all R1441C carriers) evaluated at the Movement Disorders Institute, Rambam Health Care Campus, between June 2021 and June 2024, along with 301 PD patients (274 G2019S carriers, 26 R1441G carriers and one R1441C carrier) enrolled in the Parkinson's Progression Markers Initiative (PPMI) study between March 2014 and August 2022. 23 We compared the clinical and demographic characteristics of R1441C carriers (n = 10) to G2019S carriers (n = 274) and to R1441G carriers (n = 26). To ensure group matching by disease duration, PPMI PD patients were evaluated at their three-year follow-up visit.
Participants
The R1441C group (n = 10; nine Israeli Arabs) included seven members from the same extended Israeli Arab family. Two members from this family underwent genetic testing through Invitae Hereditary PD and Parkinsonism Panel (ATP13A2, ATP7B, CHCHD2, DCTN1, DNAJC6, FBXO7, GCH1, LRRK2, MAPT, PARK7, PINK1, PRKN, PRKRA, SLC6A3, SNCA, SPR, TH, and VPS35). One was homozygous and the other heterozygous for LRRK2-R1441C mutation. Subsequently, five additional family members and two unrelated Israeli Arabs were screened for the R1441C mutation using Sanger sequencing and were found to be carriers. Thereafter, among 301 LRRK2 carriers of the PPMI cohort, we identified a single carrier of the R1441C mutation (0.3%), who was included in the analysis.
Clinical and cognitive evaluation
Diagnosis of PD was made by a movement disorder specialist employing the United Kingdom PD Brain Bank criteria. 24 Information regarding disease characteristics as well as clinical and family history, were obtained from both the participants and their caregivers. All participants were rated by the Movement Disorders Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS). 25 The Montreal Cognitive Assessment (MoCA) 26 was used for cognitive assessment.
Statistical approach
Descriptive statistics, demographic, and clinical characteristics were evaluated using ANOVA with Bonferroni post hoc test to identify differences among the independent groups for continuous variables, and we applied a Bonferroni correction to the alpha level, rather than adjusting the p-values, based on the number of pairwise comparisons within each variable. Chi-squared tests were utilized to examine associations between categorical variables and Fisher's Exact Test for variables where any expected count was ≤ 10. To compare the effects of the three LRRK2 mutations (R1441C, R1441G, and G2019S) on clinical and demographic outcomes, statistical analyses were conducted using Analysis of Covariance (ANCOVA); the model was corrected for relevant covariates, including age, sex, disease duration, and years of education. Assumptions for ANCOVA, such as homogeneity of regression slopes, normality, and homoscedasticity, were tested and met. Post-hoc pairwise comparisons were performed using the Bonferroni corrected p-values, adjusting for multiple comparisons within variables. The significance level of p < 0.05 was used. Statistical analyses were conducted using JASP version 0.18.3.
Results
Demographic and patient characteristics
This study included 310 PD patients with LRRK2 mutations (R1441C, R1441G, G2019S) (Table 1). The mean (SD) age of PD symptoms onset was 55.26 (11.3) years for R1441C carriers, 59.68 (12.8) years for R1441G carriers, and 58.32 (9.8) years for G2019S carriers. The mean age was 61.63 (8.8) years for R1441C carriers (80% females), 63.91(9.7) years for R1441G carriers (61% female), and 65.82 (8.2) years for G2019S carriers (56% female). The R1441C, R1441G and G2019S groups were similar in age (p = 0.18), sex (p = 0.31), age of PD symptoms onset (p = 0.58), and disease duration (p = 0.84).
Demographics and clinical characteristics of Parkinson's disease patients with LRRK2-R1441C compared to LRRK2-R1441G and LRRK2-G2019S mutations.
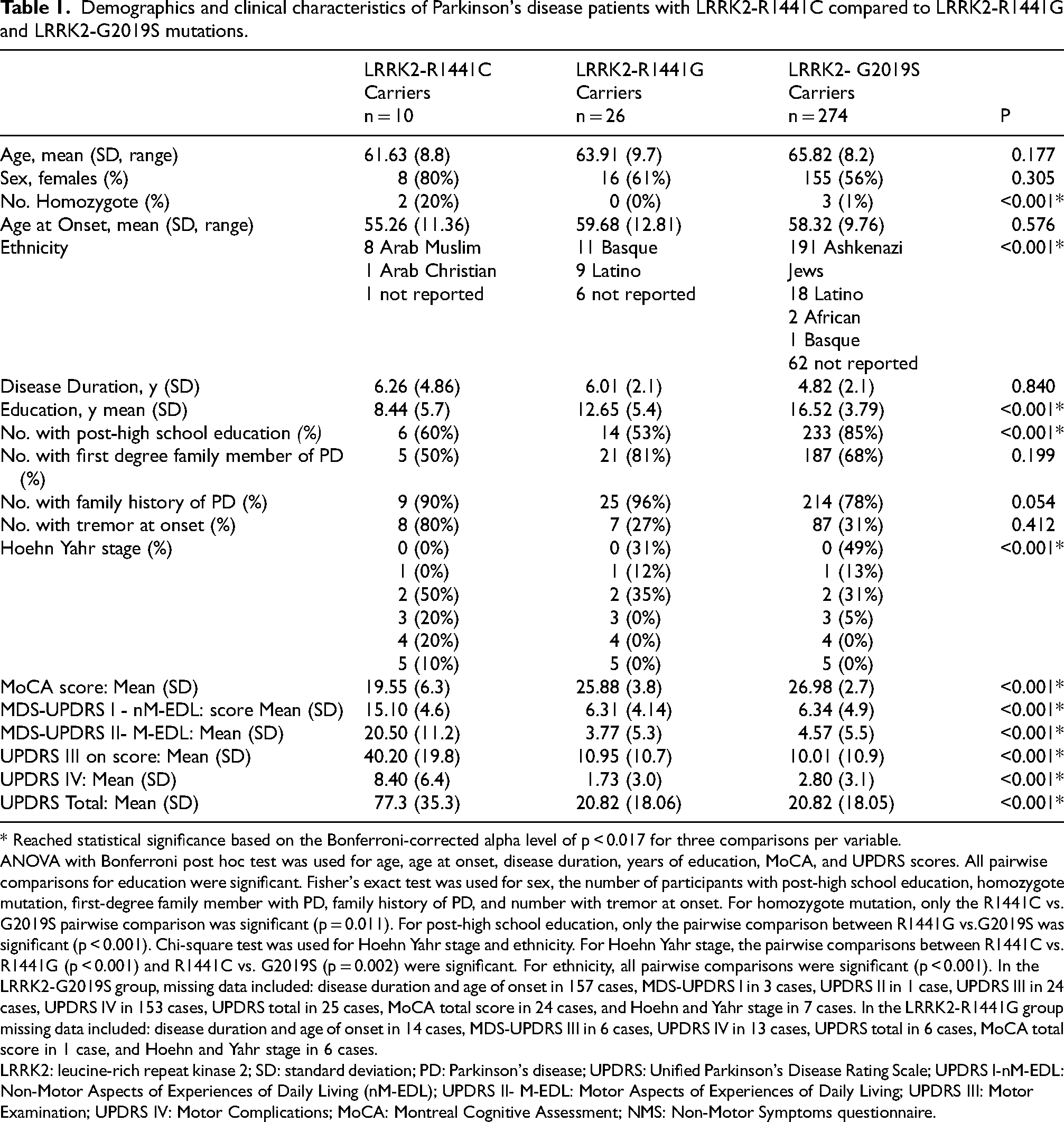
* Reached statistical significance based on the Bonferroni-corrected alpha level of p < 0.017 for three comparisons per variable.
ANOVA with Bonferroni post hoc test was used for age, age at onset, disease duration, years of education, MoCA, and UPDRS scores. All pairwise comparisons for education were significant. Fisher's exact test was used for sex, the number of participants with post-high school education, homozygote mutation, first-degree family member with PD, family history of PD, and number with tremor at onset. For homozygote mutation, only the R1441C vs. G2019S pairwise comparison was significant (p = 0.011). For post-high school education, only the pairwise comparison between R1441G vs.G2019S was significant (p < 0.001). Chi-square test was used for Hoehn Yahr stage and ethnicity. For Hoehn Yahr stage, the pairwise comparisons between R1441C vs. R1441G (p < 0.001) and R1441C vs. G2019S (p = 0.002) were significant. For ethnicity, all pairwise comparisons were significant (p < 0.001). In the LRRK2-G2019S group, missing data included: disease duration and age of onset in 157 cases, MDS-UPDRS I in 3 cases, UPDRS II in 1 case, UPDRS III in 24 cases, UPDRS IV in 153 cases, UPDRS total in 25 cases, MoCA total score in 24 cases, and Hoehn and Yahr stage in 7 cases. In the LRRK2-R1441G group missing data included: disease duration and age of onset in 14 cases, MDS-UPDRS III in 6 cases, UPDRS IV in 13 cases, UPDRS total in 6 cases, MoCA total score in 1 case, and Hoehn and Yahr stage in 6 cases.
LRRK2: leucine-rich repeat kinase 2; SD: standard deviation; PD: Parkinson's disease; UPDRS: Unified Parkinson's Disease Rating Scale; UPDRS I-nM-EDL: Non-Motor Aspects of Experiences of Daily Living (nM-EDL); UPDRS II- M-EDL: Motor Aspects of Experiences of Daily Living; UPDRS III: Motor Examination; UPDRS IV: Motor Complications; MoCA: Montreal Cognitive Assessment; NMS: Non-Motor Symptoms questionnaire.
Two R1441C carriers (20%) and three G2019S (1%) carriers had a homozygous mutation, while none of the R1441G carriers had a homozygous mutation. Family history of a member diagnosed with PD was reported by nine out of ten (90%) R1441C carriers, 25 out of 26 (96%) R1441G carriers and 214 out of 274 (78%) of G2019S carriers. A first-degree relative with PD was reported in 5 (50%) R1441C carriers, 21 (81%) R1441G carriers, and 187 (68%) G2019S carriers.
Clinical characteristics
R1441C carriers, compared to both R1441G and G2019S carriers, had significantly worse motor and non-motor symptoms (based on MDS-UPDRS subscales, p < 0.001) and greater cognitive impairment (lower MoCA scores, p < 0.001) (Table 1).
Post-hoc analyses with Bonferroni corrected p-values, adjusted for age, sex, education, and disease duration, revealed that the R1441C carriers compared to R1441G carriers, had significantly higher total MDS-UPDRS scores (p < 0.001) and motor examination scores (UPDRS 3, p = 0.005). R1441C carriers reported significantly worse non-motor aspects of experiences of daily living (UPDRS 1, p = 0.003), motor aspects of experiences of daily living (UPDRS 2, p < 0.001) and motor complications (UPDRS 4, p = 0.004). Similarly, compared to G2019S, R1441C carriers had significantly higher total UPDRS scores (p < 0.001) and significantly higher scores on all MDS-UPDRS subscales (UPDRS 1, p = 0.001; UPDRS 2, p < 0.001; UPDRS 3, p < 0.001; UPDRS 4, p = 0.021). R1441C carriers had lower MoCA scores compared to R1441G (p = 0.003) and G2019S carriers (p < 0.001). R1441G and G2019S carriers shared similar clinical characteristics with no significant differences across MoCA scores or UPDRS subscales (all p > 0.05) (Table 2).
Post-Hoc tables of the clinical feature adjusted for age, sex, education and disease duration using ANCOVA.
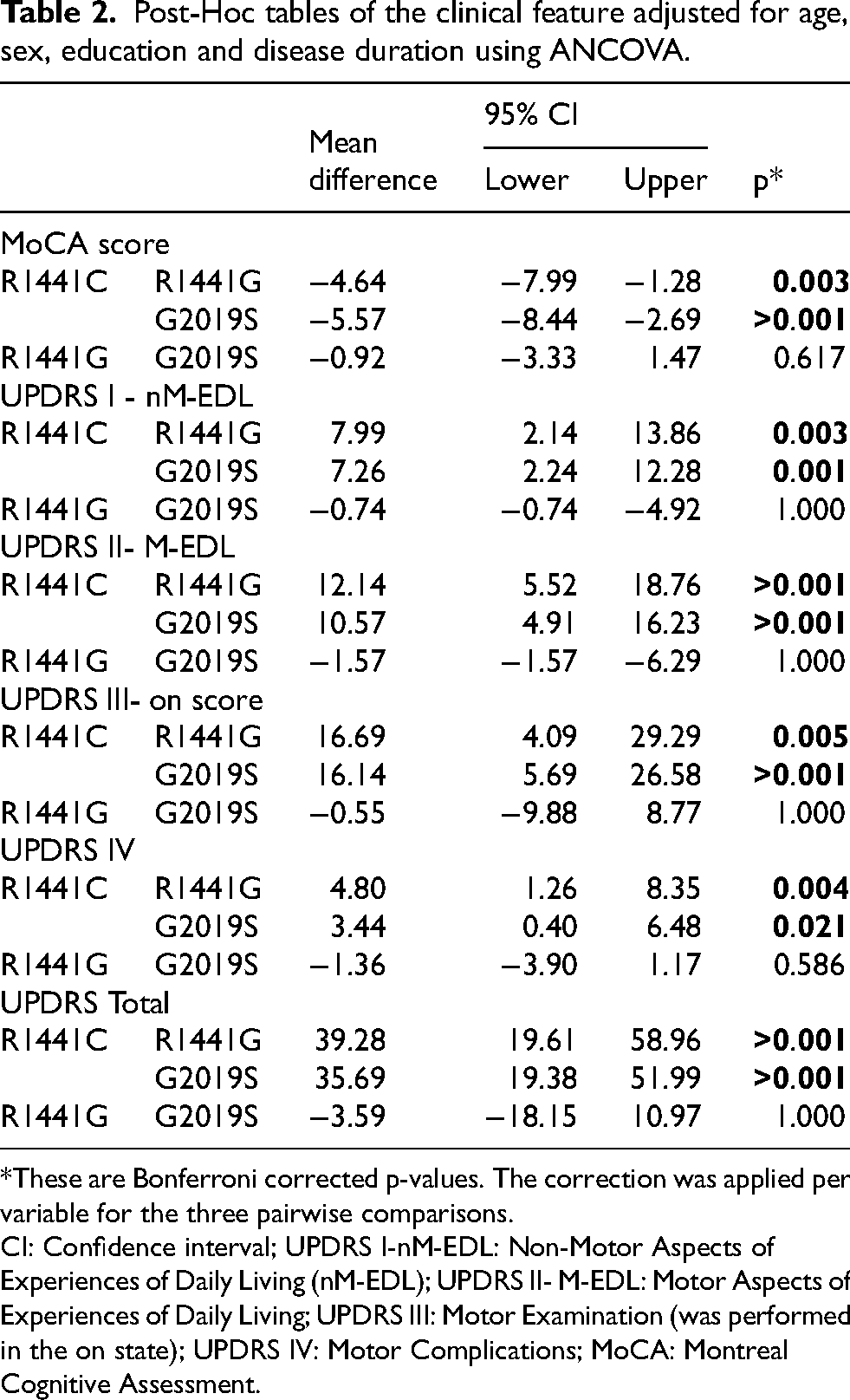
*These are Bonferroni corrected p-values. The correction was applied per variable for the three pairwise comparisons.
CI: Confidence interval; UPDRS I-nM-EDL: Non-Motor Aspects of Experiences of Daily Living (nM-EDL); UPDRS II- M-EDL: Motor Aspects of Experiences of Daily Living; UPDRS III: Motor Examination (was performed in the on state); UPDRS IV: Motor Complications; MoCA: Montreal Cognitive Assessment.
Discussion
This study highlights a distinct clinical phenotype for LRRK2-R1441C carriers, characterized by more severe motor and non-motor symptoms compared to R1441G and G2019S carriers. R1441C carriers had higher MDS-UPDRS scores across all subscales and lower MoCA scores, underscoring the mutation's unique clinical features.
Previous studies examining LRRK2 mutations predominantly focused on G2019S and R1441G carriers, with limited inclusion of R1441C carriers, yielding inconsistent findings regarding the clinical phenotype and cognitive changes associated with these mutations. While prior studies suggested that LRRK2 carriers (most of them G2019S and R1441G carriers) share similar clinical features to non-carriers, subtle differences, such as lower frequency of dementia, better performance on certain cognitive tests, and a higher prevalence of psychiatric symptoms, were reported.27–30 However, the unique clinical phenotype we found in R1441C carriers has not been previously described.
Our findings raise questions about the potential influence of genetic and ethnic factors. Most R1441C carriers in this study were Israeli Arabs, and it remains unclear whether these findings are generalizable to other R1441C carriers or are specific to the Israeli Arab ethnic background predominant among the participants in this study. Notably, the single R1441C carrier from the PPMI cohort exhibited similar clinical features to our Israeli Arab patients, suggesting the possibility of shared mutation-specific characteristics.
Recent studies have shown that various mutations in the LRRK2 gene result in distinct effects on the protein's function, contributing to the heterogeneity of clinical manifestations of PD and response to therapies. Understanding these mutation-specific effects is crucial for the development of targeted therapies and for tailoring patient management strategies based on their genetic profile.31–34
Our study has some limitations. First, the cohort of LRRK2-R1441C carriers was relatively small, and their education level was lower compared to the R1441G and G2019S carriers. To mitigate this, we adjusted our models to account for years of education. Second, the comparison groups were derived from the PPMI data, which differed in study settings and ethnic backgrounds. Most R1441C carriers were related, possibly violating the assumption of independent observations. Therefore, the current analyses represent the most feasible option given the sample constraints. This highlights the need for confirmation in larger, more diverse, and unrelated samples.
In conclusion, this study describes the clinical phenotype of LRRK2-R1441C carriers, most of whom were Israeli Arabs, highlighting the association of R1441C with more severe PD symptoms and cognitive impairment. These findings underscore the importance of examining each LRRK2 mutation independently, as each may present distinct clinical features. The present work underscores the importance of ethnic variations and genetic stratification in understanding clinical heterogeneities in PD.
Footnotes
Acknowledgements
We thank Bruce Miller for editing the manuscript. Data used in the preparation of this article were obtained on May 19, 2024 from the Parkinson's Progression Markers Initiative (PPMI) database (https://www.ppmi-info.org/access-data-specimens/download-data), RRID:SCR_006431. For up-to-date information on the study, visit ![]() . PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, BioArctic, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Capsida Biotherapeutics, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Jazz Pharmaceuticals, Johnson & Johnson Innovative Medicine, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Neuron23, Neuropore, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
. PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, BioArctic, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Capsida Biotherapeutics, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Jazz Pharmaceuticals, Johnson & Johnson Innovative Medicine, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Neuron23, Neuropore, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
ORCID iDs
Ethical considerations
The study received approval from the Israeli Supreme Committee for Clinical Trials in Humans. The PPMI study was conducted following approval from the ethics committees at each participating institution and is registered at ClinicalTrials.gov under identifier NCT01141023.
Consent to participate
All participants provided written informed consent. For the PPMI cohort, informed consent was obtained from all participants at each site in accordance with local IRB requirements. For the Israeli cohort, all participants signed consent forms approved by the local ethics board.
Consent for publication
This manuscript was reviewed and approved for publication by the PPMI Publication Committee. All data used in the study were collected with appropriate participant consent for publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study did not receive external funding. It was supported by institutional resources from the Movements Disorders Institute, Rambam Health Care Campus, Haifa, Israel.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Roy N. Alcalay has served as a consultant for Biogen, Biohaven, Capsida, Gain Therapeutics, Genzyme/Sanofi, Janssen, Servier, SK Biopharmaceuticals, Takeda, and Vanqua Bio. However, these relationships are not relevant to the research presented in this manuscript. Roy N. Alcalay is an Editorial Board Member of the Journal of Parkinson’s Disease but was not involved in the peer-review process of this article nor had access to any information regarding its peer review.The corresponding author and all other authors have no financial disclosures or conflicts of interest related to the research covered in this manuscript.
Data availability statement
Data used in this study are partially available. Public archiving of anonymized data is not approved by the study's institutional review board approval; however, request can be submitted to the Institutional Review Board (IRB). Additional data used in the analysis are sourced from the Parkinson's Progression Markers Initiative (PPMI) and can be accessed via “![]() ”.
”.
