Abstract
Background
Progressive supranuclear palsy (PSP) is a rare neurodegenerative movement disorder clinically characterized by falls, axial rigidity, vertical supranuclear gaze palsy, bradykinesia, and cognitive decline. There is a relative lack of studies on the functional neuroimaging correlates of cognitive impairment in PSP.
Objective
This study investigated the relationship between regional cerebral glucose metabolism as assessed by static 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) with global scaling and the profile of cognitive performance according to the Consortium to Establish a Registry for Alzheimer's Disease (CERAD) test battery in a sample of PSP patients representative of clinical practice.
Methods
22 PSP patients from three tertiary movement disorder centers with CERAD testing and FDG-PET in close proximity were included retrospectively. Neuropsychological test performance was assessed for correlation with FDG uptake on a voxel-by-voxel basis with cluster-level correction for multiple testing, separately for each subtest.
Results
In comparison to matched healthy controls, PSP patients showed reduced FDG uptake in the left inferior frontal gyrus and right angular gyrus. Reduced overall cognitive performance according to Montreal Cognitive Assessment was associated with reduced FDG uptake in the right frontal eye field. Word list learning correlated with FDG uptake in the left frontal eye field, while language fluency was linked to FDG uptake in the bilateral premotor and supplementary motor areas.
Conclusions
Reduction of FDG uptake in PSP primarily affects frontal brain regions and is linked to the performance in specific cognitive domains. These findings may have implications for the interpretation of FDG-PET to support the etiological diagnosis of PSP.
Plain language summary
Progressive supranuclear palsy (PSP) is a rare brain disorder that affects movement and can lead to problems like falling, stiffness, difficulty with eye movements, slow movements, and memory issues. Unlike Alzheimer's disease, there hasn't been much research on how brain scans relate to cognitive problems in PSP. In this study, we wanted to explore how different parts of the brain function and how this relates to thinking skills in people with PSP. We looked at 22 patients with PSP from three specialized centers in Germany. The patients took tests to measure their cognitive abilities and also underwent a brain scan using a method called FDG-PET, which shows how well different brain areas are working based on their metabolism of blood sugar. The results showed that PSP patients had lower brain activity in certain areas, particularly in the frontal parts of the brain, compared to healthy individuals. The patients struggled most with tasks that required flexible thinking, planning, and visual skills. Interestingly, better memory performance was linked to activity in the front part of the brain, and language skills were associated with areas involved in movement planning. Overall, the study found that PSP patients have a specific pattern of reduced brain activity mainly in the frontal regions. This pattern is related to specific cognitive difficulties experienced by the patients and may have relevance for diagnosing PSP.
Keywords
Introduction
Progressive supranuclear palsy (PSP) is the most frequent atypical neurodegenerative Parkinson syndrome. The underlying pathomechanism is a tauopathy leading to fall propensity, gait instability, apraxia and limitation of voluntary eye movements in affected subjects. 1 Another aspect of PSP is cognitive impairment, which is observed at early stages in up to half of the patients 2 and in the majority of patients over the course of the disease. 3 Recently, there has been an increased focus on cognitive dysfunction in PSP due to its significant impact on both the quality of life of both patients and their caregivers.4,5
PSP predominantly affects the executive domain.2,6 Furthermore, individuals with PSP exhibit a notable deterioration in verbal fluency that progressively worsens over the course of the disease. The decline in verbal fluency serves as a distinguishing factor between PSP patients and patients with Parkinson's disease, demonstrating high sensitivity and specificity three years after the clinical onset of the disease. 7 PSP patients also show impaired visuospatial functions as needed in construction testing. 3 In addition, approximately one third of individuals diagnosed with PSP experience a decline in memory, including disturbances in autobiographical memory. 8 Behavioral changes seen in PSP include apathy, which is prevalent in about 80% of patients3,9 and, on the other hand, impulsivity, which might in some cases be masked by severe motor impairment. 7 Inadequate inhibition can be observed beyond the social context, as demonstrated by the presence of the applause sign, which is more commonly present in PSP patients as compared to other Parkinson syndromes. 10
A PSP-typical finding in positron emission tomography (PET) with the glucose analog 18F-fluorodeoxyglucose (FDG) is reduced FDG uptake in the medial and dorsolateral frontal cortex as well as in the caudate, thalamus and in the upper brain stem. 11 Motor dysfunctions in PSP are linked to reduced FDG uptake in certain brain areas: postural imbalance was reported to be associated with thalamic reduction of FDG uptake, 12 freezing of gait with reduced FDG uptake in midbrain areas, 13 and vertical gaze palsy with reduced FDG uptake in the anterior cingulate gyrus. 14 Aphasia was linked to reduced FDG uptake in the frontal lobe and basal ganglia. 15 Concerning cognitive performance in PSP, an FDG PET study found reduction of FDG uptake in the frontal cortex to be associated with overall cognitive impairment according to the Mini-Mental State Examination. 16 However, there is a lack of studies on potential associations between specific cognitive functions and the cerebral FDG uptake pattern in PSP.
Against this background, the aim of this study was to test for correlations between the cognitive profile as characterized by the neuropsychological assessment battery of the Consortium to Establish a Registry of Alzheimer's Disease (CERAD) and regional FDG uptake in PSP. The study included a sample of well-characterized patients with PSP diagnosis according to the criteria of the Movement Disorder Society (MDS) 1 representative of the spectrum of PSP phenotypes typically encountered in clinical practice.
Methods
Subjects
The study retrospectively included 22 patients (13 women, 9 men) with probable (n = 17) or suggestive (n = 5) MDS PSP diagnosis from the tertiary movement disorder centers at the University hospitals of Hannover, Munich (LMU) and Hamburg, Germany (for demographics, see Table 1). PSP-subtypes included PSP-RS (n = 13), PSP-P (n = 3), PSP-CBS (n = 2), PSP-PI (n = 1), PSP-PFG (n = 1), PSP-F (n = 1), and PSP-OM (n = 1). All participants underwent neurocognitive testing using an extended CERAD-NAB test battery and a static resting state FDG-PET in clinical practice. The mean delay between imaging and cognitive testing was 6.5 days. Most of the patients had been included in a previous study on the diagnostic performance of FDG-PET in PSP as described in a recent article. 17 The patient sample comprised 15 right-handed subjects and 7 patients with unknown handedness. The retrospective analyses had been approved by the MHH ethics committee (vote 3558-2017). Most of the patients had been included in a previous study on the diagnostic performance of FDG-PET in PSP. 17
Demographics and clinical data.
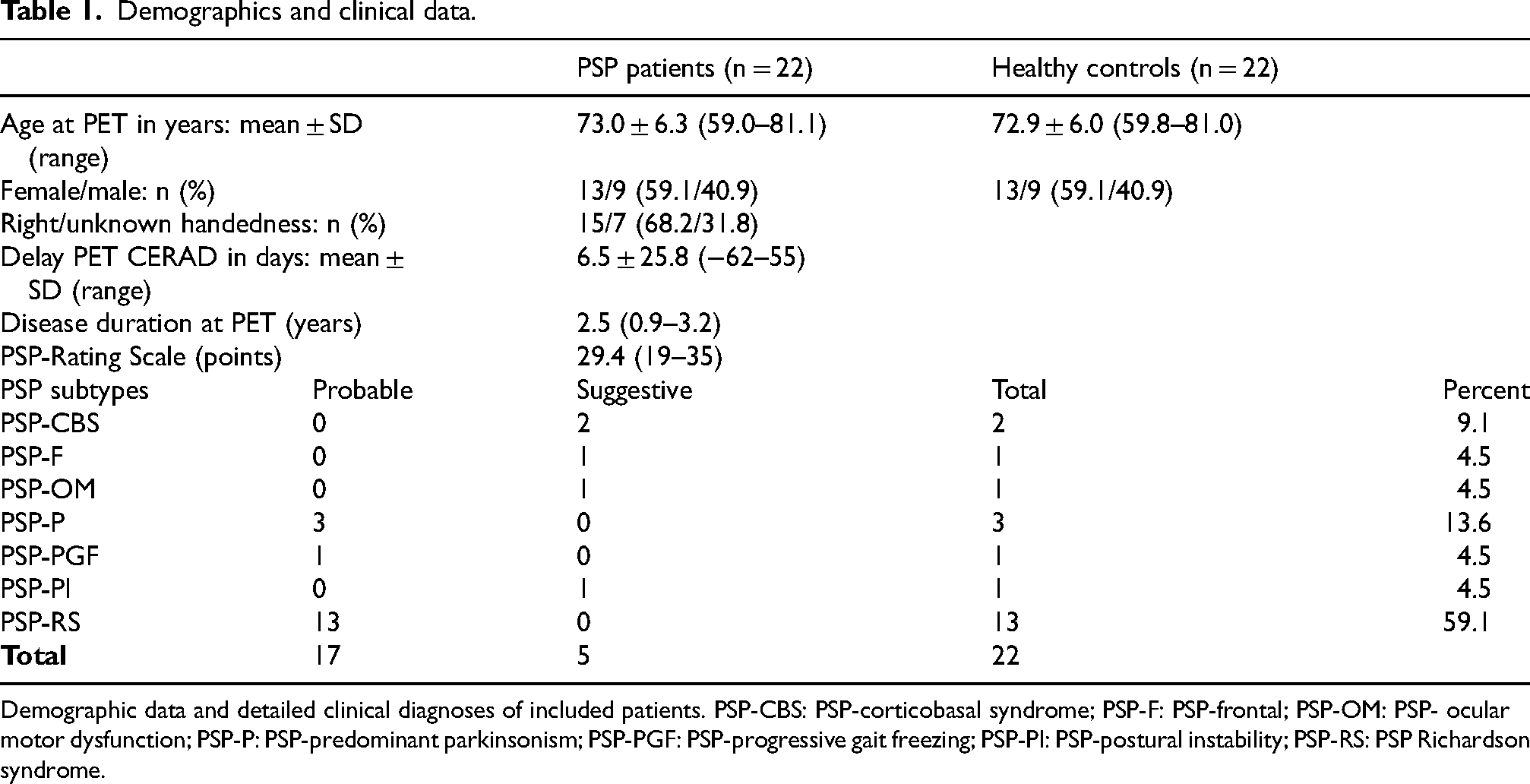
Demographic data and detailed clinical diagnoses of included patients. PSP-CBS: PSP-corticobasal syndrome; PSP-F: PSP-frontal; PSP-OM: PSP- ocular motor dysfunction; PSP-P: PSP-predominant parkinsonism; PSP-PGF: PSP-progressive gait freezing; PSP-PI: PSP-postural instability; PSP-RS: PSP Richardson syndrome.
In addition to the PSP patients, the study included brain FDG-PET images of 22 sex- and age- matched healthy control (HC) subjects from the Alzheimer's Disease Neuroimaging Initiative (ADNI). All HC subjects had been cognitively stable for at least 36 months after the FDG-PET scan. The ADNI was launched in 2003 as a public-private partnership, led by Principal Investigator Michael W. Weiner, MD. For up-to-date information, see www.adni-info.org.
Demographical and clinical characteristics of the PSP patients and the HC subjects are summarized in Table 1.
Cognitive testing
The cognitive status of the PSP patients had been evaluated as part of clinical care according to the local standard at each of the 3 participating sites. This included the extended German version of the Consortium to Establish a Registry for Alzheimer's Disease Neuropsychological Assessment Battery (CERAD-NAB18–20) at each site. The CERAD-NAB core comprises measures of semantic (“animal fluency”, naming of animals within 1 min), the abbreviated Boston Naming Test (BNT), word list learning (immediate recall of 10 words), word list recall (delayed recall of 10 words), word list recognition (recognition of 10 words on a 20 words list), constructional praxis (“figures drawing”, copying of geometric forms), and constructional recall (“figures recall”, delayed recall of geometric forms). The CERAD-NAB extension adds phonological verbal fluency (“letter fluency”, naming of words beginning with the same letter within 1 min), as well as the Trail Making Tests A and B (TMT-A/ TMT-B), assessing mental speed. The Montreal Cognitive Assessment Test (MoCA) score, 21 as measure of overall cognitive status, was available in 15 patients. The “Executive Function Composite” of the Modified Wisconsin Card Sorting Test (WCST) 22 was available in 7 patients.
Due to physical limitations (e.g., motor impairment), not all tests could be performed by all patients. This particularly affected the MoCA. The WCST was routinely administered at one of the three centers only.
The raw CERAD-NAB scores were transformed to age-, sex-, and education-adjusted z-scores (https://www.memoryclinic.ch/de/).
FDG-PET
In the PSP patients, brain FDG-PET images had been acquired with two different PET/CT systems (Hannover, Hamburg) and one PET/MRI system (Munich) after intravenous injection of 204 ± 38 MBq FDG (range 133–317 MBq), following the standard operating procedure at each site. To ensure consistent spatial resolution of 7 mm full-width-at-half-maximum across PSP patients and HC subjects, the PET images of the PSP patients were harmonized across the different PET/CT systems based on Hoffman 3D brain phantom measurements as described previously. 23
Statistical analysis
The Wilcoxon signed-rank test was used to test the CERAD z-scores for non-zero median. The significance threshold was set at two-sided p < 0.05 after Bonferroni correction for multiple testing (that is, p < 0.05/11 = 0.0045).
The PET images, now harmonized in spatial resolution, were stereotactically normalized to the anatomical reference space of the Montreal Neurological Institute using the Statistical Parametric Mapping (SPM) software package (version 12). Default parameter settings were utilized for this process. Stereotactically normalized PET images were smoothed by convolution with an isotropic Gaussian kernel with 14 mm full-width-at-half-maximum. To achieve intensity scaling, each smoothed and stereotactically normalized image was divided voxel-by-voxel by the individual mean intensity in the brain parenchyma. 24
Voxel-based nonparametric testing for reduced FDG uptake in the PSP patients compared to the HC subjects was performed with the “2 Groups (one scan per subject)” model implemented in the Statistical nonParametric Mapping (SnPM) toolbox for SPM (version 13.1.08, http://nisox.org/Software/SnPM13/). 25 The model parameters were set to their default values: 5000 iterations, isotropic variance smoothing with 14 mm full-width-at-half-maximum (= image smoothing during preprocessing, see above), cluster forming threshold 3.09. 26 The one-sided test for reduced FDG uptake in the PSP patients was restricted to cortical gray matter in the cerebrum (Supplemental Figure 1). In order to account for multiple testing, cluster-level inference from the resulting statistical nonparametric map was performed using the family-wise error (FWE) corrected p ≤ 0.05 threshold.
Voxel-based nonparametric testing for positive correlation between scaled FDG-uptake and CERAD z-scores was performed with the “Simple Regression” model implemented in SnPM, separately for each z-score. Model parameters, spatial masking and cluster-level inference at FWE-corrected one-sided p ≤ 0.05 were the same as in the PSP versus HC group comparison.
As secondary analyses, all voxel-based tests were repeated with voxel-level inference at FWE-corrected one-sided p ≤ 0.05.
The HC subjects were not included in the voxel-based testing for correlations between cognitive performance and FDG uptake in order to avoid mixing of group effects (PSP versus HC) and the relationship between FDG uptake and the severity of cognitive impairment in PSP.
Results
Cognitive function
The average MoCA score of the PSP patients was 22.5 ± 2.9 (n = 15) indicating mild cognitive impairment (MCI).
Among the CERAD subtests, the z-score was significantly reduced for animal fluency, letter fluency, word list learning and both TMT tests (Figure 1). All other subtests missed Bonferroni-corrected statistical significance. Close to normal z-scores were seen for the abbreviated Boston naming test and word list recognition (Figure 1).
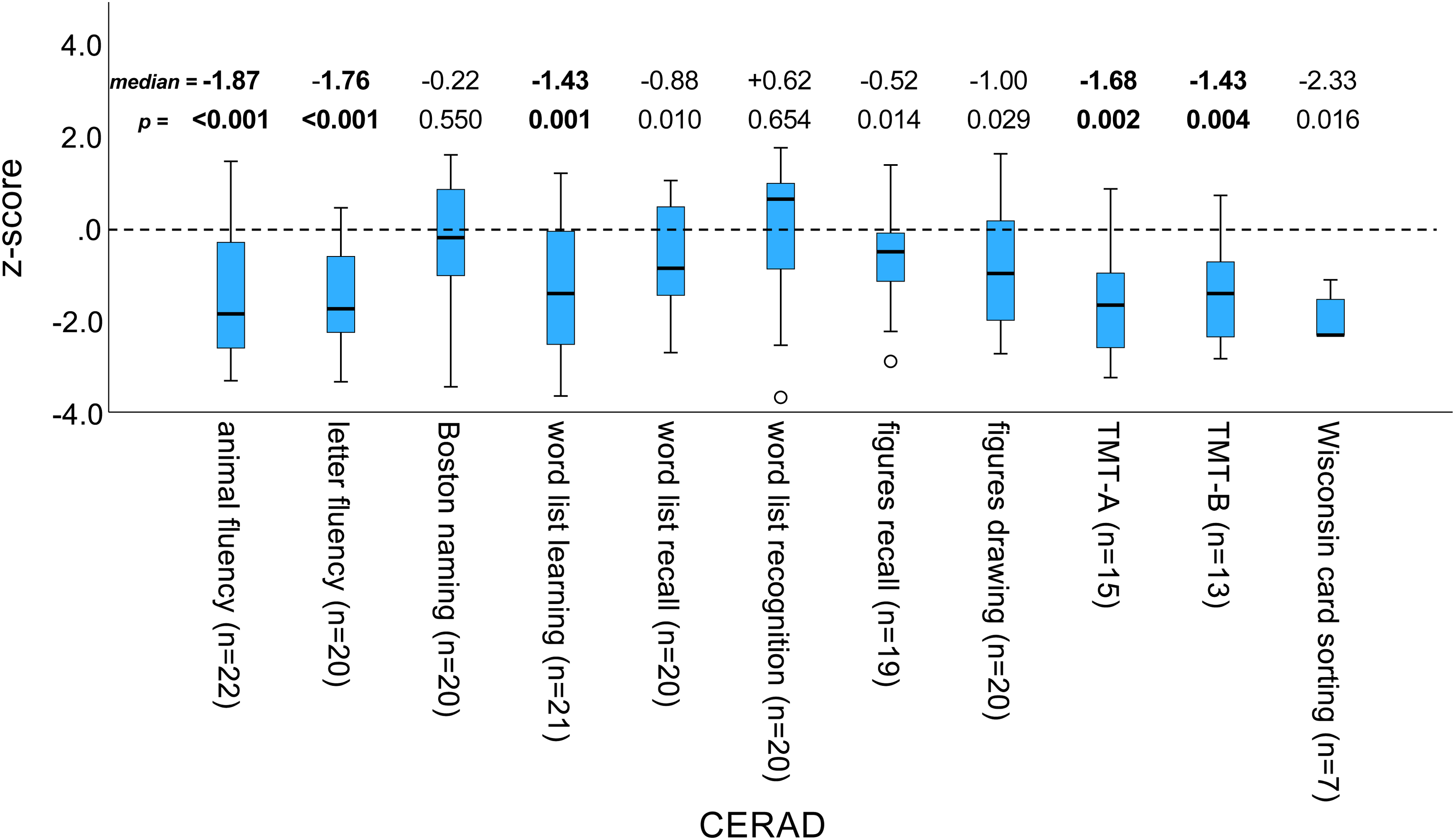
CERAD z-scores of PSP patients.
Box-and-whisker plot of the CERAD z-scores (corrected for age, sex and education) of PSP patients. The number (n) of PSP patients differed between the subtests and therefore is shown in brackets separately for each subtest. Median and two-sided p-value (from the Wilcoxon signed-rank test for zero median) are also shown separately for each subtest. Effects that are significant at the two-sided 5% significance level after Bonferroni correction for multiple testing (p < 0.05 / 11 = 0.0045) are shown in bold. (TMT-A/B = Trail making test A/B)
Reduction in FDG uptake
PSP patients showed reduced FDG uptake in the frontal cortex, particularly the left inferior frontal gyrus (n = 22, p < 0.001), and in the right angular gyrus (n = 22, p = 0.01) (Figure 2A, B).
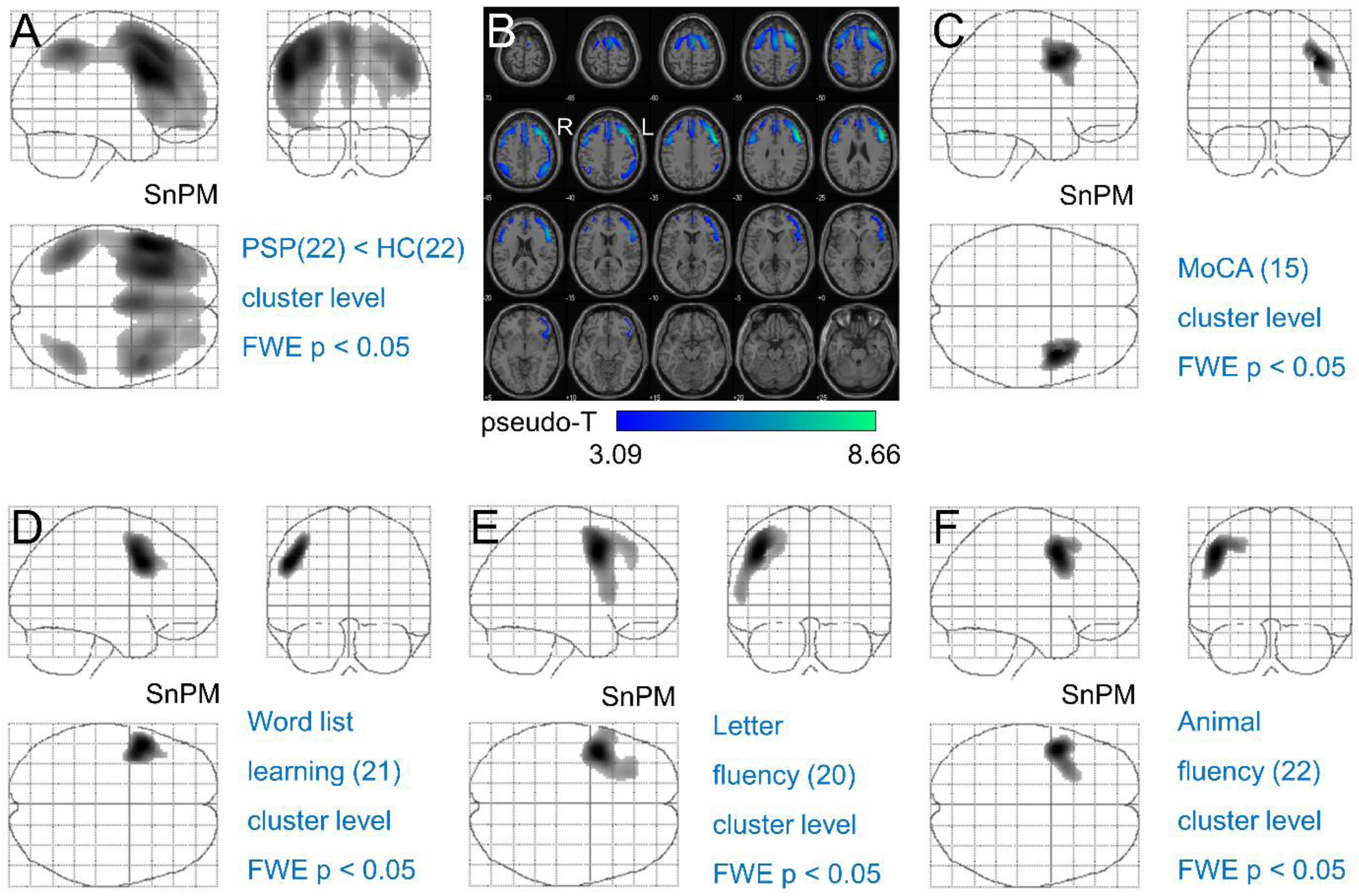
FDG-PET imaging of peak metabolic reductions in PSP patients compared to healthy controls.
Moca versus FDG uptake
The performance on the MoCA was associated with higher FDG uptake in the right frontal eye field (n = 15, p = 0.0274, Figure 2C).
Among the CERAD subtests, word list learning and the two verbal fluency tasks, semantic verbal fluency (animal fluency) and phonological S-words verbal fluency (letter fluency), showed a significant correlation with regional FDG uptake. None of the other subtests showed a significant correlation with regional FDG uptake.
Memory versus FDG uptake
Word list learning was correlated with FDG uptake in the left frontal eye field (n = 21; p = 0.01, Figure 2D). There was no significant correlation between word list recognition scores and FDG uptake.
Language versus FDG uptake
Letter fluency and animal fluency were both correlated with FDG uptake in the left premotor and supplementary motor cortices (n = 20, p = 0.004, Figure 2E; n = 22, p = 0.02, Figure 2F).
The results of the voxel-based testing with voxel-level correction for multiple testing are shown in Supplemental Figure 2.
18F-fluoro-2-deoxy-d-glucose positron emission tomography (FDG-PET) in patients with PSP. Glass brain representations of the results from the nonparametric voxel-wise testing that reached statistical significance on the cluster level with the family-wise error corrected p ≤ 0.05 threshold. (A) Reduced FDG-uptake in PSP patients (n = 22) compared to healthy controls (HV, n = 22). (C-F) Positive correlation between FDG-uptake and MoCA z-score (n = 15, C), world list learning z-score (n = 21, D), letter fluency z-score (n = 20, E), and animal fluency z-score (n = 22, F), respectively. There were no significant associations detected between FDG-uptake and the remaining CERAD subtest z-scores. Part B shows transaxial slices of the significantly reduced FDG-uptake in the PSP patients overlaid to SPM's “single subject” MR scan. The results with voxel-level correction for multiple testing are shown in Supplemental Figure 2.
Table 2
Correlations of FDG-uptake, group and cognitive testing.
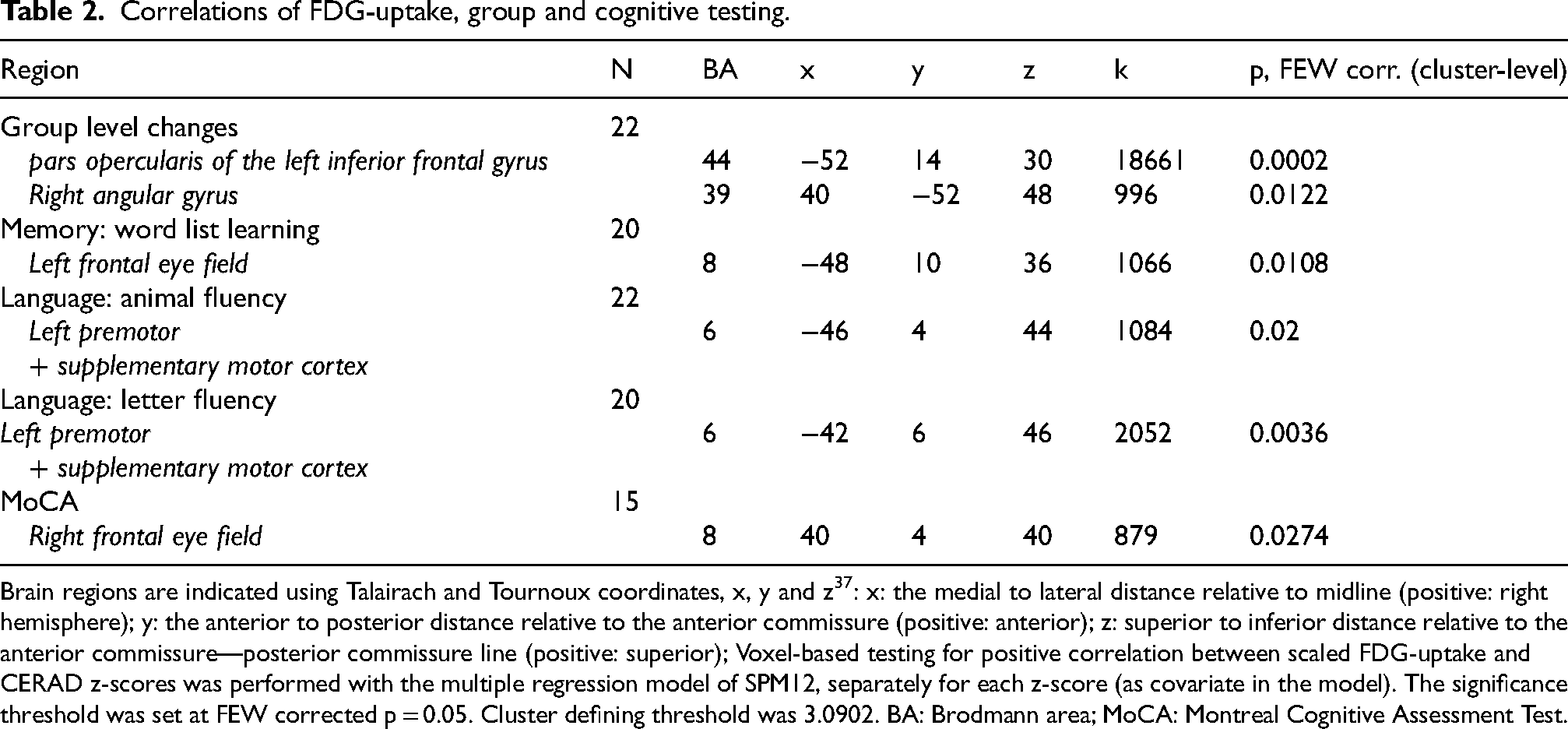
Brain regions are indicated using Talairach and Tournoux coordinates, x, y and z 37 : x: the medial to lateral distance relative to midline (positive: right hemisphere); y: the anterior to posterior distance relative to the anterior commissure (positive: anterior); z: superior to inferior distance relative to the anterior commissure—posterior commissure line (positive: superior); Voxel-based testing for positive correlation between scaled FDG-uptake and CERAD z-scores was performed with the multiple regression model of SPM12, separately for each z-score (as covariate in the model). The significance threshold was set at FEW corrected p = 0.05. Cluster defining threshold was 3.0902. BA: Brodmann area; MoCA: Montreal Cognitive Assessment Test.
Discussion
PSP patients showed reduced FDG uptake in the frontal cortex, statistically most significant in the left inferior frontal gyrus, which is consistent with previous studies showing a reduced FDG uptake in prefrontal cortex areas27–29 and a grey matter loss 30 in in prefrontal cortex areas in PSP. Moreover, FDG uptake was reduced in the right angular gyrus in the PSP patients. This brain region is particularly affected by the tau pathology underlying this disease. 31 Reduced FDG uptake in the angular gyrus in PSP patients has been reported to be linked with apathy. 31
Overall cognitive performance as characterized by the MoCA score was significantly associated with FDG uptake in the right frontal eye field. Interestingly, a study comparing connectivity patterns during memory tasks between HC subjects and MCI patients found connectivity of the right frontal eye field with other activated brain regions to be more prominent in MCI, indicating a more visual approach to memory tasks. 32 Given that the participants in the current study were also identified as having MCI, our observation of this FDG uptake pattern aligns with the findings from this previous research.
However, the primary aim of the study was to identify correlations between specific neuropsychological domains as tested by the CERAD-NAB and regional brain metabolic activity as characterized by FDG uptake in PSP. Statistically significant effects were observed for memory and language performance.
Memory
Word list learning was associated with FDG uptake in the left frontal eye field. While this is consistent with a functional MRI study that observed activation in the frontal eye field during a category learning task, 33 an experimental study in non-human primates actually found no impairment of memory after an inactivation of this brain area. 34 However, the frontal eye field has been shown to be crucial for visual attention and eye movement planning, which in general is required for memorizing visual stimuli. 34
Language
Verbal fluency performance, both semantic and phonological, was correlated with FDG uptake of the bilateral premotor and supplementary motor area. A study in Parkinson's disease patients found verbal fluency to be strongly correlated with motor impairment and thus hypothesized a cognition-motor interaction specifically with regard to verbal fluency. 35 Interestingly, a study in patients with Alzheimer's disease found a correlation between verbal fluency and FDG uptake in the temporo-parietal cortex. 36 This suggests that alterations of verbal fluency are caused by different mechanisms in PSP compared to Alzheimer's disease.
Strengths and limitations
A major strength of this study is the inclusion of several PSP subtypes other than PSP-RS, which makes it more representative of clinical practice than studies including only PSP-RS.
The limitations of this study include the following: First, the sample of well characterized patients with MDS-PSP diagnosis, CERAD testing and FDG-PET was rather small, despite the participation of 3 tertiary movement disorder centers, as PSP is a rare disease. The small sample size may limit the generalizability of the findings. It certainly limited the statistical power for the detection of correlations between specific cognitive domains and regional FDG uptake. This is particularly true for the cognitive domains for which test scores were available for a subset of the PSP patients only. In particular, the WCST was completed by only seven PSP patients. Although the WCST showed a tendency to perseveration, as expected in PSP, the correlation with reduced FDG uptake in the right inferior temporal gyrus and the left cerebellum, which we observed without correction for multiple testing (results not shown) was no longer significant after correction for multiple testing. Regarding other subtests, such as word list recognition and the Boston naming test, no significant correlations with FDG uptake were expected a priori, as the PSP patients did not show significant deterioration in these tests.
Second, apathy was not assessed in this study, although it is a frequent symptom in PSP and may affect the outcome in cognitive testing.
Third, the inability of some patients to perform certain cognitive tasks due to motor difficulties may have caused a selection bias.
Fourth, voxel-based statistical testing was restricted to cortical gray matter in the cerebrum. Thus, potential effects in subcortical structures including thalamus and mesencephalon could not be detected.
Finally, the handedness was not known in 7 of the included PSP patients.
Conclusion
PSP patients showed reduced FDG uptake in the frontal cortex, most significant in the left inferior frontal gyrus and in the right angular gyrus, consistent with previous studies. Reduced word list learning performance was linked to reduced FDG uptake in the left frontal eye field (involved in visual attention and eye movement planning), while reduced verbal fluency was associated with reduced FDG uptake in the bilateral premotor and supplementary motor areas. These findings may have implications for the interpretation of FDG-PET to support the etiological diagnosis of PSP.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251343080 - Supplemental material for Associations between neuropsychological profile and regional brain FDG uptake in progressive supranuclear palsy
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251343080 for Associations between neuropsychological profile and regional brain FDG uptake in progressive supranuclear palsy by Johanna Doll-Lee, Martin Klietz, Stephan Greten, Bruno Kopp, Georg Berding, Matthias Brendel, Ida Wilkens, Sabrina Katzdobler, Johannes Levin, Adrian Danek, Sophia Rogozinski, Günter Höglinger, Monika Pötter-Nerger, Carsten Buhmann, Ralph Buchert, Florian Wegner and in Journal of Parkinson's Disease
Footnotes
Acknowledgements
Data collection and sharing for this project was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer's Association; Alzheimer's Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer's Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer's Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
ORCID iDs
Ethical considerations
The retrospective analyses had been approved by the MHH ethics committee (vote 3558-2017).
Consent to participate
Informed consent was obtained from all study participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Matthias Brendel was supported by grants from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany's Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy – ID 390857198).
Günter Höglinger was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany's Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy – ID 390857198) ; Niedersächsisches Ministerium für Wissenschaft und Kunst / VolkswagenStiftung (Niedersächsisches Vorab); Petermax-Müller Foundation (Etiology and Therapy of Synucleinopathies and Tauopathies).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Statistical parametric maps are available from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
