Abstract
Background
Despite its relevance, the clinical progression of motor- and non-motor symptoms associated with Parkinson's disease (PD) is poorly described and understood, particularly in relation to sex-specific differences in clinical progression.
Objective
Identification of differential aspects in disease progression in men and women with PD.
Methods
Linear mixed-model analyses of 802 people with typical PD from the Luxembourg Parkinson's study's prospective cohort (median time of follow-up = three years). We estimated the effect of time and its moderation by sex (alpha ≤ 0.05), including confidence intervals, for the following outcomes: MDS-UPDRS I-IV, Starkstein Apathy Scale, Beck Depression Inventory, Montreal Cognitive Assessment (MoCA), Sniffin’ sticks, bodily discomfort, rapid eye movement sleep behavior disorder questionnaire, PD Sleep Scale (PDSS), Munich Dysphagia Test-PD, Functional Mobility Composite Score, and the MDS-based tremor and postural instability and gait disturbances scale. In addition, the marginal means illustrated the symptoms’ trajectories in men and women. Men and women had similar age.
Results
Overall, we observed a slower progression (interaction effect) in women compared to men, especially for MoCA (−0.159, 95%CI [−0.272, −0.046], p = 0.006), PDSS (−0.716, 95%CI [−1.229, −0.203], p = 0.006), PIGD (0.133, 95%CI [0.025 0.241], p = 0.016), and MDS-UPDRS II (0.346, 95%CI [0.120, 0.572], p = 0.003). The finding for MDS-UPDRS II was significant (FWER of 5%) after adjustment for multiple comparisons (Bonferroni-Holm).
Conclusions
Next to the further exploration of sex-specific progression, interventions, proactive monitoring and communication strategies tailored to the symptoms progression and needs of men and women need to be developed.
Plain language summary
Although Parkinson's disease is 1.4 times more frequent in men compared to women, it is not clear how the symptoms develop over time in men and women. Consequently, we aimed to describe the changes of different symptoms over time in both sexes and to find out if women with Parkinson's disease showed a different progression than men. By analyzing repeated measurements of 802 individuals, we found an overall slower worsening of the symptoms in women compared to men, particularly for memory, sleep, walking and balance as well as motor symptoms in general. Consequently, compared to women, men not only experience Parkinson's disease more frequently but also experience a faster worsening of symptoms. More research is needed to fully understand why. To improve the well-being of men and women with Parkinson's disease, we suggest taking into account the important differences between them when doing research and providing health care.
Keywords
Introduction
In the 2016 Global Burden of Disease Study, the age-standardized prevalence of Parkinson's disease (PD) was 1.4 times higher in men than in women. 1 This male preponderance might be explained by a protective effect of female sex hormones, a different genetic mechanism or different exposures to environmental risk factors in men and women. 2 Consequently, sex-specific factors in PD merit further study. However, most research has focused on biological differences between men and women, neglecting to place these in the psychosocial context that impacts clinical care and quality of life of men and women with PD.3–5 Therefore, the effect of sex and/or gender should be considered in designing future studies in PD. 6
Moreover, previous longitudinal studies addressed the sex-specific progression of some symptoms. Thus, the association of sex with patient-reported and clinician-assessed motor symptoms, the phenotype, activities of daily living and medication with progression was investigated. 7 Another study 8 explored the role of sex in the progression of patient-reported motor symptoms, cognition, dyskinesia, wearing off, depression, rapid eye movement (REM) sleep behavior disorder and some non-motor symptoms. However, most often single studies3–5 have mainly reported cross-sectional sex differences of selected symptoms in men and women with PD while a comprehensive empirical description and illustration of the motor- and non-motor symptoms associated with PD progression has not been reported in the literature. Aiming to provide an overview of symptom and general disease progression of PD in men and women that can be easily interpreted by health professionals, we describe the progression of motor- and non-motor symptoms in men and women and quantify the effect moderation by sex in people with typical PD participating in a large monocentric longitudinal cohort.
Methods
Study design, setting, participants, and study size
This retrospective analysis is part of the Luxembourg Parkinson's study, a nationwide, monocentric, observational, longitudinal-prospective and dynamic cohort.9,10 The completed STROBE reporting guideline checklist 11 is included in Supplemental Table 3.
All participants underwent diagnostic evaluation and were assigned a clinical diagnosis of typical PD or Parkinson's disease dementia (PDD) by a neurologist based on established United Kingdom Parkinson's Disease Society Brain Bank Clinical Diagnostic Criteria. 12 The diagnosis was not required before participation as the Luxembourg Parkinson's study also included converters. The participants were recruited from Luxembourg and the Greater Region (geographically close areas of the surrounding countries, namely Belgium, France, and Germany). In addition to the referral by medical doctors, a communication campaign (advertisement on radio and television, dedicated webpage, social media campaign, multilingual flyers and posters, fact sheets and bi-annual print newsletter, collaboration with patient associations) informed the population about the option to enroll themselves. Recruitment started in 2015 with annual follow-ups. The Luxembourg Parkinson's Study aims at stratification and differential diagnosis of PD.9,10
Variables, data sources, and measurement
The outcomes of interest were progression (i.e., change per additional year since diagnosis) of motor and non-motor symptoms. Patient-reported outcomes included: apathy, measured by the Starkstein Apathy Scale (SAS); depression, assessed using the Beck Depression Inventory (BDI-I); dysphagia, assessed by the Munich Dysphagia Test for PD (MDT-PD); functional mobility, evaluated by the PDQ-39 based functional mobility composite score (FMCS); non-motor symptoms, captured by the Movement Disorders Society-Unified Parkinson's Disease Rating Scale Part I (MDS-UPDRS I); motor symptoms, assessed by MDS-UPDRS II; bodily discomfort, measured using the respective subscale of the Parkinson's Disease Questionnaire-39 (PDQ-39); quality of sleep, evaluated with the Parkinson's Disease Sleep Scale (PDSS); rapid eye movement (REM) sleep behavior disorder, screened using the REM sleep Behavior Disorder (RBD) Screening Questionnaire (RBDSQ). Clinician-assessed outcomes and performance tests included global cognition, assessed using the Montreal Cognitive Assessment (MoCA); motor symptoms evaluated by the MDS-UPDRS III; motor complications, measured by MDS-UPDRS IV; olfaction, tested using the Sniffin’ Sticks Identification Test; Postural Instability and Gait Disturbances (PIGD), assessed using the MDS-UPDRS-based PIGD score; tremor, evaluated using the MDS-UPDRS-based tremor scale. Table 1 describes the characteristics of the outcomes and provides sources of data and details of the assessment methods. All outcomes were numerical and assessed during annual follow-ups varying by a maximum of three months to minimize seasonal influences. The progression could be distinguished from cohort or period effects as people with PD were included at different time points 13 due to the dynamic cohort study design. People with PD with complete data for time since diagnosis were included in the longitudinal analysis.
Instrument, assessment types and variable name of the included constructs.
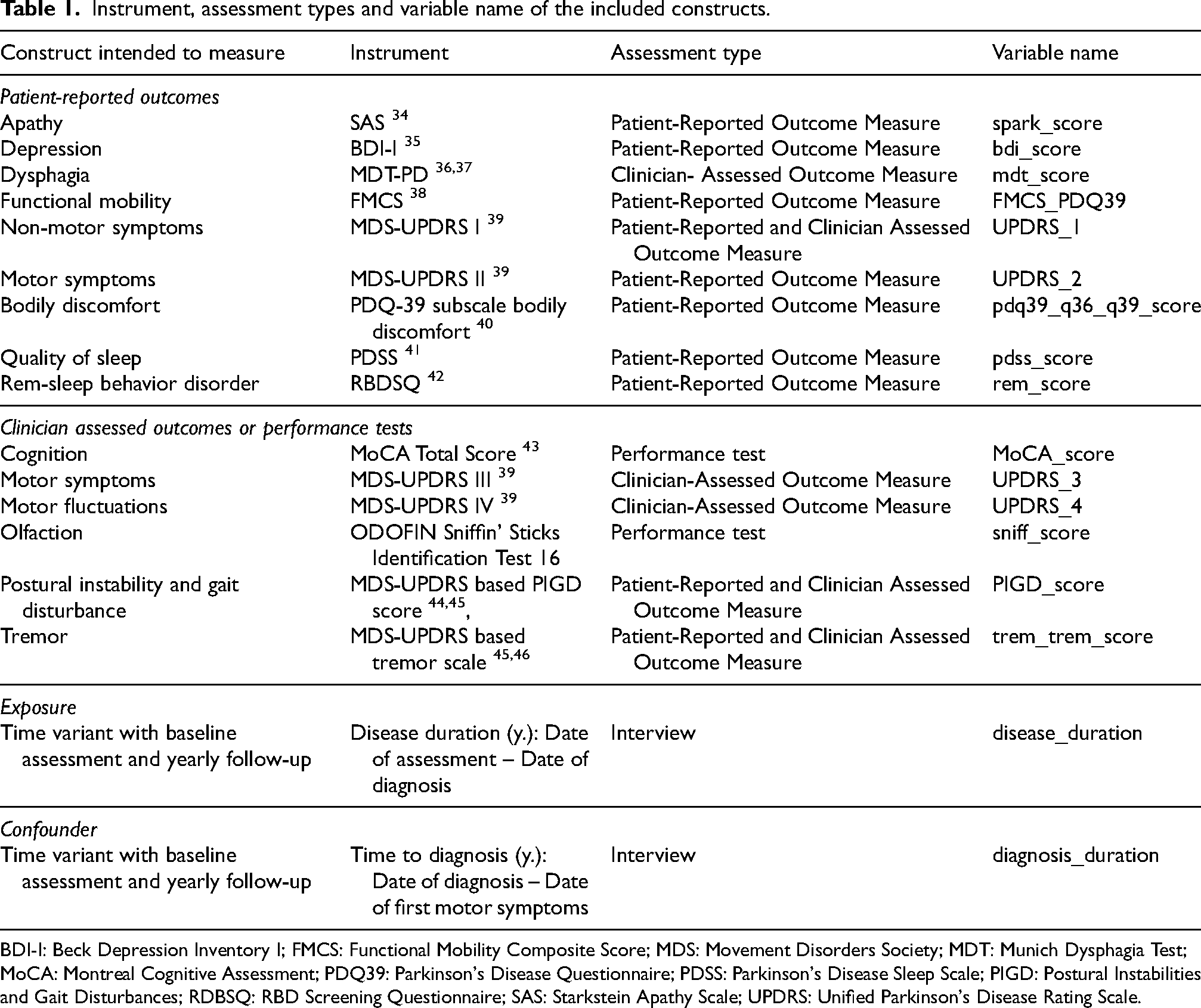
BDI-I: Beck Depression Inventory I; FMCS: Functional Mobility Composite Score; MDS: Movement Disorders Society; MDT: Munich Dysphagia Test; MoCA: Montreal Cognitive Assessment; PDQ39: Parkinson's Disease Questionnaire; PDSS: Parkinson's Disease Sleep Scale; PIGD: Postural Instabilities and Gait Disturbances; RDBSQ: RBD Screening Questionnaire; SAS: Starkstein Apathy Scale; UPDRS: Unified Parkinson's Disease Rating Scale.
Statistical methods
Data analysis was carried out in R, version 4.3.1. 14 We used the two-sided Wilcoxon rank-sum test for discrete variables and the chi-squared (χ2) test for categorical variables compared baseline characteristics between men and women (using the “stats” package 14 ). In addition to the Bonferroni-adjusted p-values (p-value * 30 variables ≤ 0.05) we provided the unadjusted p-values (p-value ≤ 0.05).
To describe the progression of different motor- and non-motor symptoms and the effect moderation by sex, we created one model per outcome (using “lmer”-function of the “lme4”-package
15
). Consequently, we performed longitudinal two-level mixed models analyses with fixed effects for years since diagnosis and sex, a random intercept on participant level and a random slope for years since diagnosis. In addition to the linear effect we tested whether adding a quadratic effect of time significantly improved the fit of the model. If this was the case, we additionally tested the cubic effect of time. Difference in progression between men and women was described by a significant interaction effect for sex and time on the symptoms. We estimated the linear mixed models using the maximum likelihood method while statistical significance and confidence intervals for the mixed models were obtained with the Kenward-Roger approximation for degrees of freedom. We adjusted the p-values using the Bonferroni-Holm procedure to maintain the Family-Wise Error Rate (FWER) at 5%. To enhance clinical interpretation, we provided estimated marginal means, i.e., estimated means of motor- and non-motor symptoms given 0, 10, and 20 years since diagnosis. Thus, we examined the range of the years since diagnosis from its minimal observed value to its maximal observed value, then fixed the covariates (diagnosis duration) at their mean to finally look at the estimated values for the different symptoms for the whole range of values of years since diagnosis (using “ggpredict”-function of the “ggeffects”-package
16
). Those estimated means for the different symptoms (y-axis) given years since diagnosis (x-axis) and the mean value for the covariates were illustrated as an interaction plot (using the “plot_model”- function of the “sjPlot”-package
17
). As women's ratings of disability differed between self-reported and physician-reported,
18
we categorized the results in patient-reported or clinician-assessed outcomes / performance tests. Time, in this case modelled as years since diagnosis, was included in the mixed models to describe progression of the different outcomes (significance tested via t-test). Degree of disability as illustrated in Figure 1 was calculated by the following formula: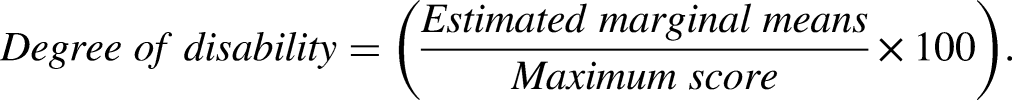
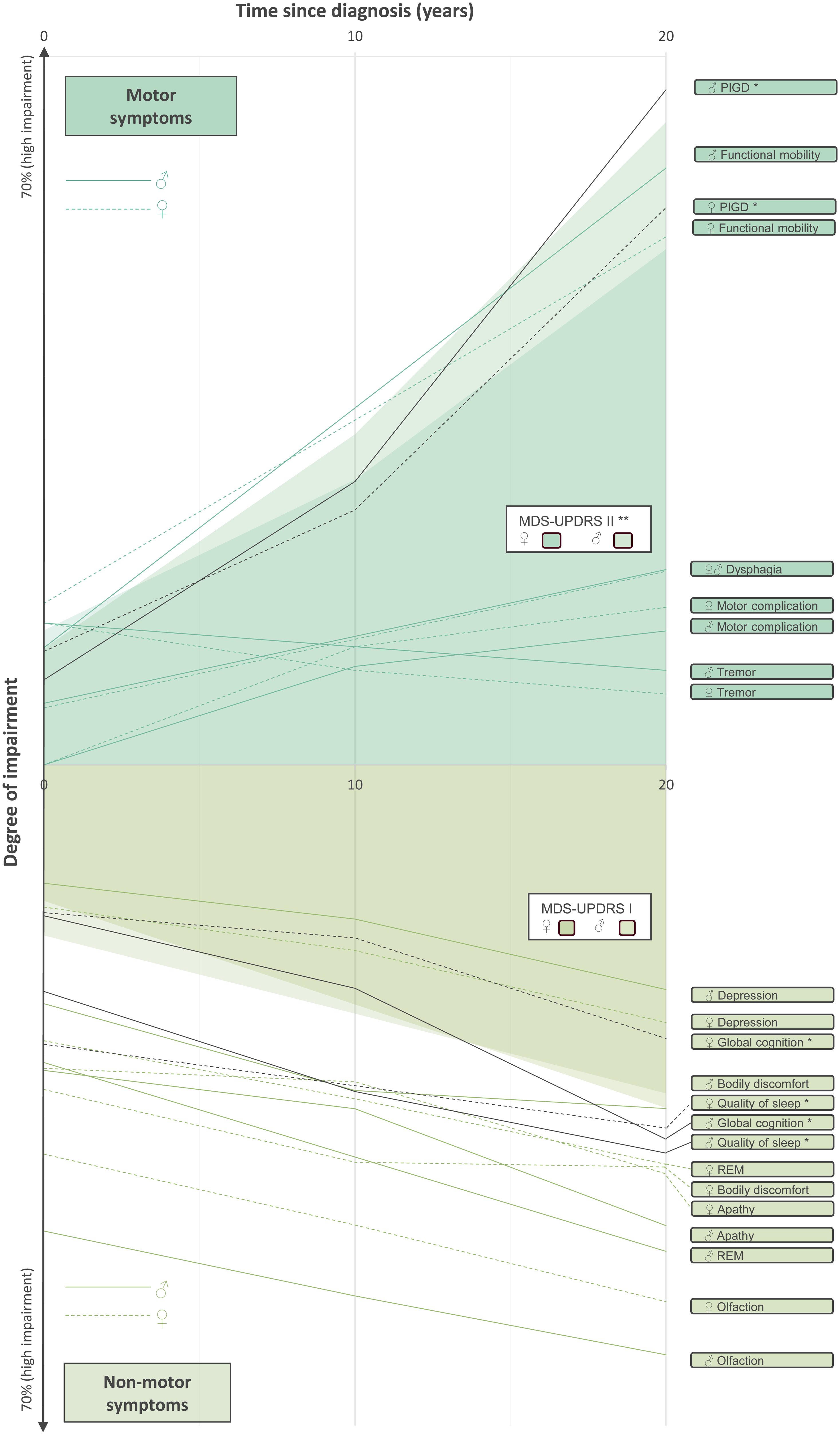
Progression of motor- and non-motor symptoms in men and women with typical PD 0-20 years after diagnosis. Degree of impairment = 0–100% (greater = worse). * = nominally significant, ** significant after adjustment for FWER 5% (Bonferroni-Holm), lines of significant results are highlighted in black. PD: Parkinson's disease; PIGD: postural instabilities and gait disturbances; RBD: rapid eye movement (REM) behavior disorder.
For illustrative purposes in Figure 1, the following scores were inverted to the higher, the worse: functional mobility (FMCS), quality of sleep (PDSS), global cognition (MoCA), and olfaction (Sniffin’ Sticks). Also, we focused in the figure from data the first twenty years after diagnosis as most observations were collected in that period.
Results
As illustrated in Figure 2, 957 persons participated in the Luxembourg Parkinson's Study up to the date of data export (2023-06-22). After the exclusion of people with atypical PD, we included 802 people with typical PD with a baseline assessment between 04.03.2015 and 22.06.2023.
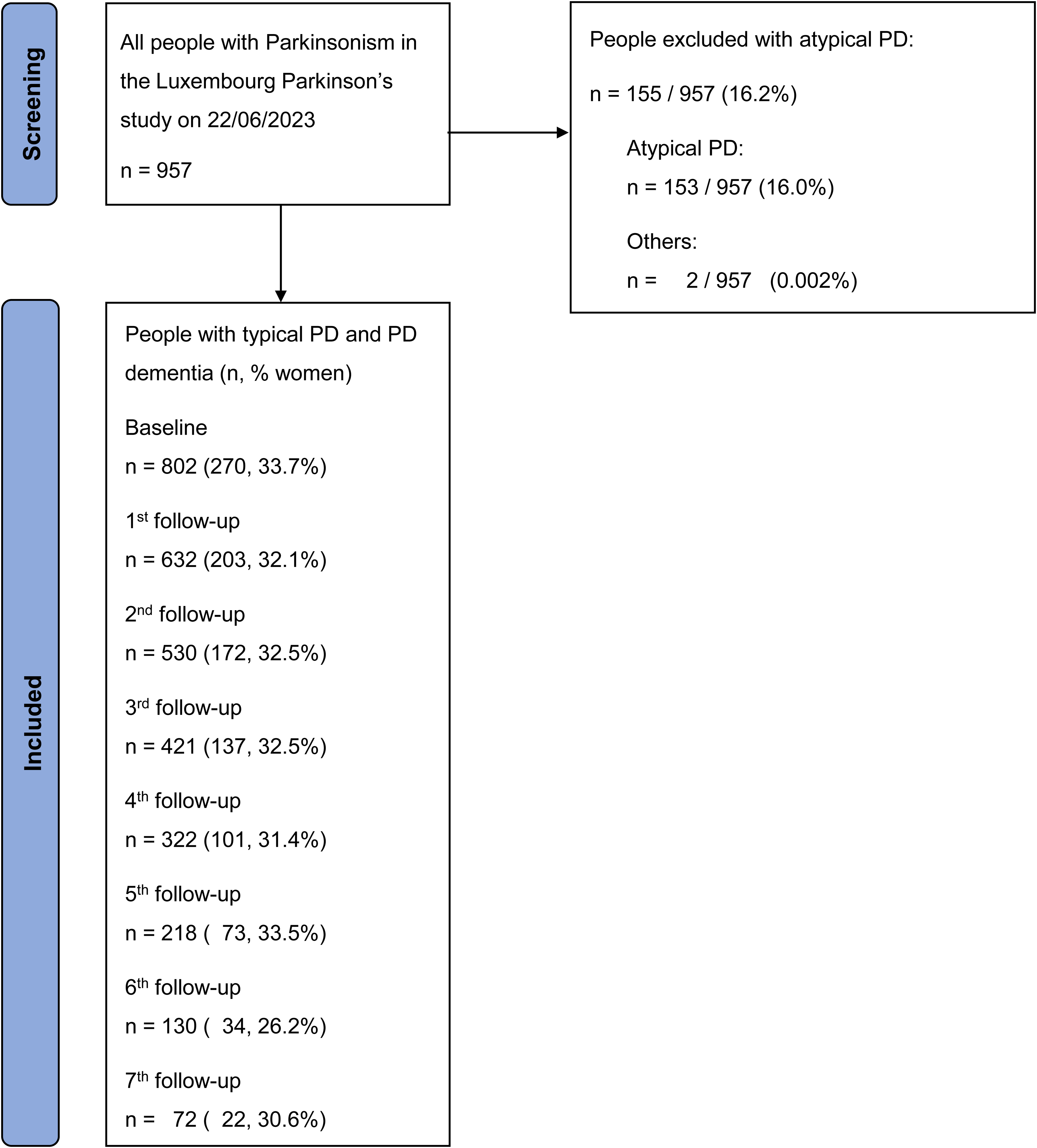
Flow diagram of recruitment.
Table 2 summarizes key study characteristics to understand the potential applicability, and thus generalizability of the findings, while Supplemental Table 1 provides a detailed description of the study participants and missing data. The clinical and demographic characteristics of study participants at baseline by sex are presented in Table 3. Testing for differences at baseline in 30 characteristics at a Bonferroni-adjusted 5% significance level, women had worse scores for depression (Beck Depression Inventory I - BDI-I) than men, with a median difference of 1.99 (95% CI [0.99, 2.99]), W = 74343, p = 0.006. Also, women reported worse scores for bodily discomfort (PDQ-39 subscale bodily discomfort) than men, with a median difference of 8.3 (95% CI [0.000006, 8.33]), W = 74468, p = 0.004. However, women had better olfaction scores (Sniffin’ sticks) than men with a median difference of 1.0 (95% CI [0.99, 1.99]), W = 74969, p < 0.001. Finally, women had a higher Levodopa Equivalent Daily Dose (LEDD) per kg of body weight (mg/ kg) than men with a median difference of 1.2 mg/kg (95% CI [0.5, 1.9]), W = 57869, p = 0.02 and a lower body mass index (BMI) than men with a median difference of 2.0 kg/m2 (95% CI [−2.7, −1.3]), W = 50270, p < 0.001. Also, women had less years of education (median difference = 1 (95% CI [−1.9, −0.9]), W = 58475, p = 0.004) and experienced a bereavement more often than men, χ2 (df = 2, N = 797) = 48.81, p < 0.001. We did not identify any statistically significant differences for age, years since diagnosis or time to diagnosis at baseline between men and women with typical PD. Missing data patterns were visually inspected for sociodemographic characteristics and the different outcomes; most variables had missing data for less than 5% of the male and female samples. Rates for missing data were higher for Munich Dysphagia Test-assessed dysphagia (51% and 55% for men and women, respectively).
Key characteristics.

Categorical variables: counts (%), numerical variables: Median (IQR). PD: Parkinson's disease; BDI-I: Beck Depression Inventory; FMCS: Functional Mobility Composite Score; LEDD: Levodopa Equivalent Daily Dose; MDS: Movement Disorders Society; MDT: Munich Dysphagia Test; MoCA: Montreal Cognitive Assessment; PDQ39: Parkinson's Disease Questionnaire; PDSS: Parkinson's Disease Sleep Scale; PIGD: Postural Instabilities and Gait Disturbances; RBDSQ: RBD Screening Questionnaire; SAS: Starkstein Apathy Scale; UPDRS: Unified Parkinson's Disease Rating Scale.
Characteristics of men and women.
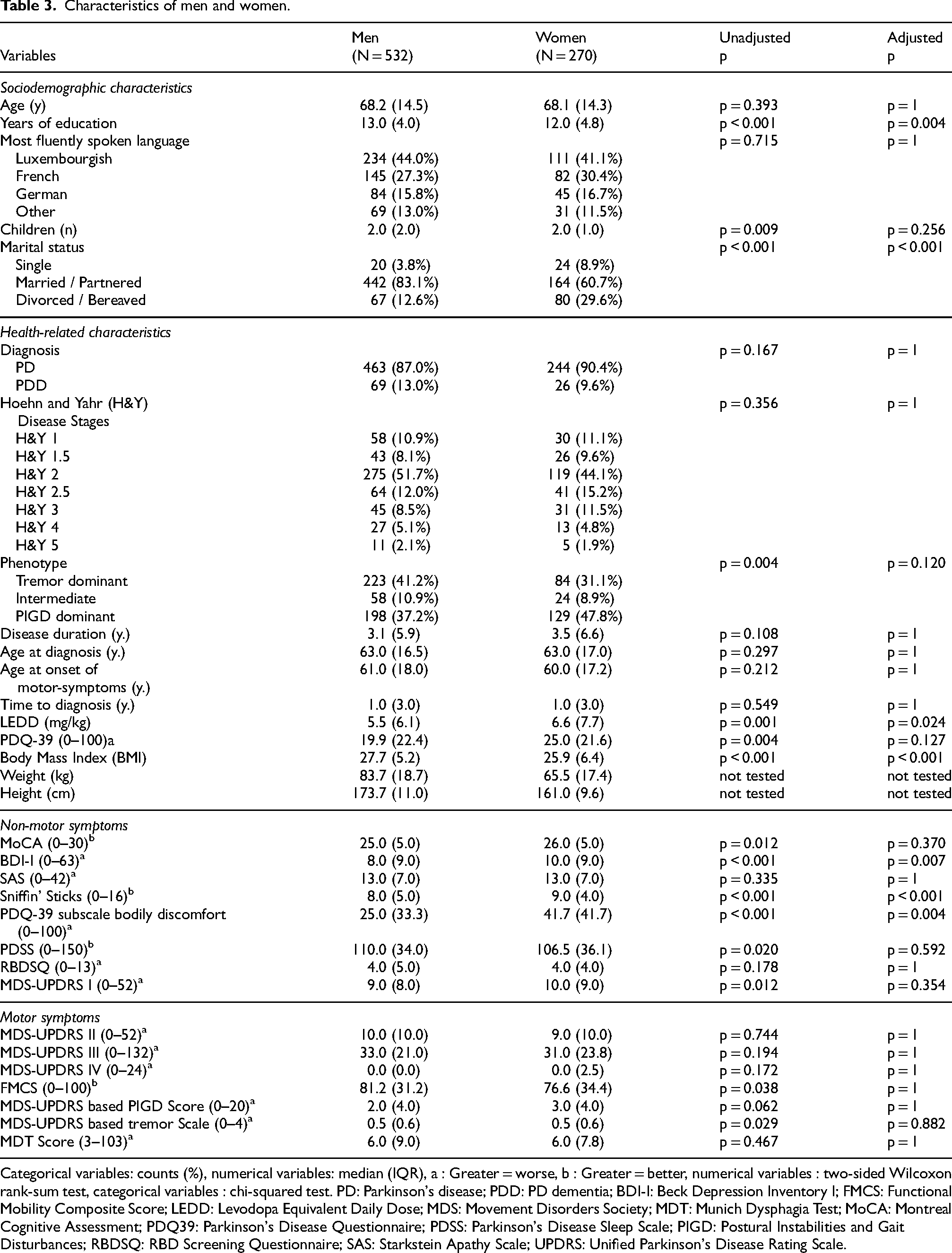
Categorical variables: counts (%), numerical variables: median (IQR), a : Greater = worse, b : Greater = better, numerical variables : two-sided Wilcoxon rank-sum test, categorical variables : chi-squared test. PD: Parkinson's disease; PDD: PD dementia; BDI-I: Beck Depression Inventory I; FMCS: Functional Mobility Composite Score; LEDD: Levodopa Equivalent Daily Dose; MDS: Movement Disorders Society; MDT: Munich Dysphagia Test; MoCA: Montreal Cognitive Assessment; PDQ39: Parkinson's Disease Questionnaire; PDSS: Parkinson's Disease Sleep Scale; PIGD: Postural Instabilities and Gait Disturbances; RBDSQ: RBD Screening Questionnaire; SAS: Starkstein Apathy Scale; UPDRS: Unified Parkinson's Disease Rating Scale.
While many outcomes showed a linear trajectory, this was not the case for apathy (SAS), global cognition (MoCA), depression (BDI-I), bodily discomfort (PDQ-39 subscale bodily discomfort), patient-reported motors symptoms (MDS-UPDRS II), motor complications (MDS-UPDRS IV), postural instability and gait disturbances (MDS-UPDRS based PIGD score) where adding the quadratic effect significantly improved the fit of the model. We described the model statistics and the detailed fixed and random effects in Tables 4 and 5 and illustrated the progression (estimated marginal means converted to % impairment) of men and women in Figure 1. Supplemental Figures 1–3 detail the interaction plots for clinical interpretation.
Fixed effects of non-motor symptoms in men and women and the interaction of sex with progression.
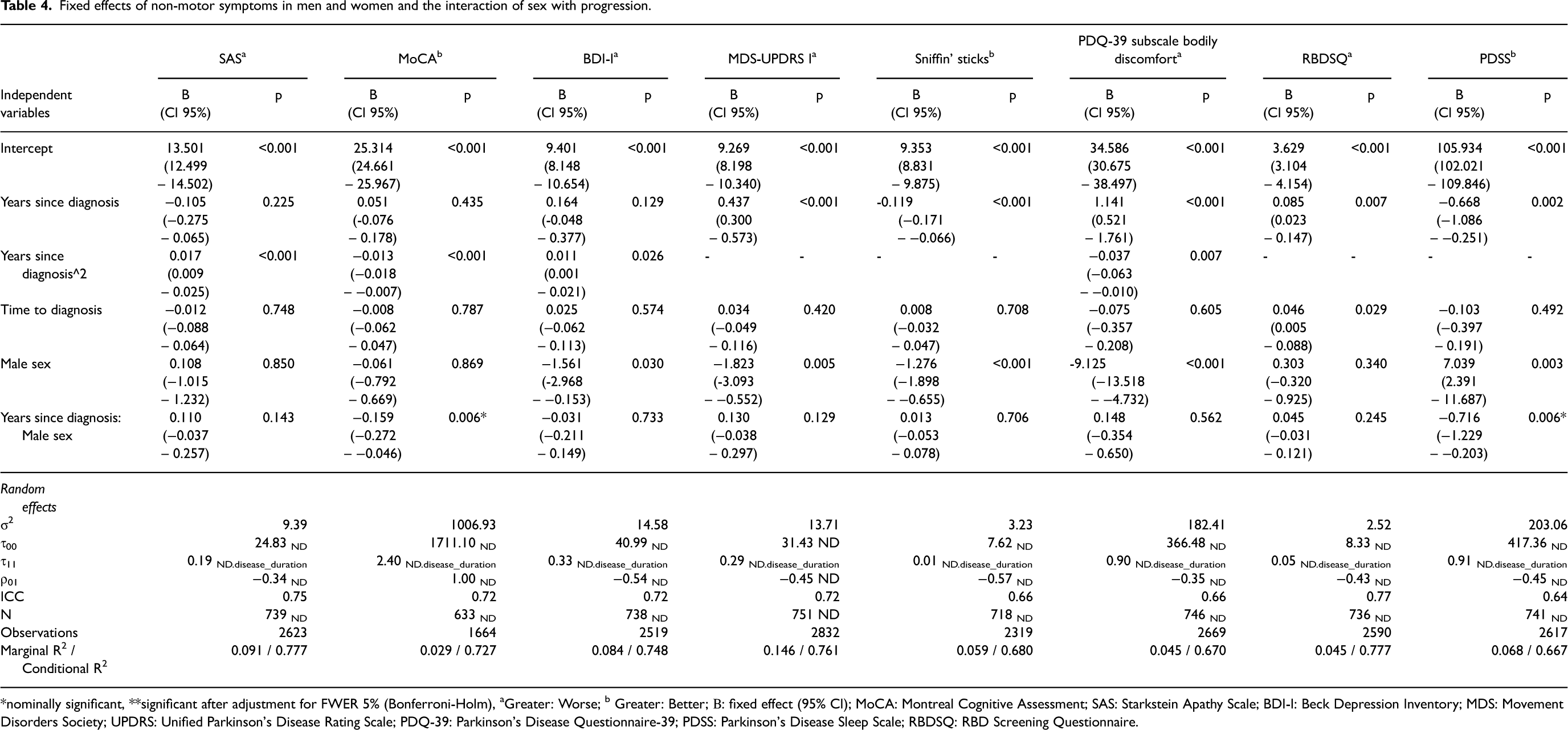
*nominally significant, **significant after adjustment for FWER 5% (Bonferroni-Holm), aGreater: Worse; b Greater: Better; Β: fixed effect (95% CI); MoCA: Montreal Cognitive Assessment; SAS: Starkstein Apathy Scale; BDI-I: Beck Depression Inventory; MDS: Movement Disorders Society; UPDRS: Unified Parkinson's Disease Rating Scale; PDQ-39: Parkinson's Disease Questionnaire-39; PDSS: Parkinson's Disease Sleep Scale; RBDSQ: RBD Screening Questionnaire.
Fixed effects of motor symptoms in men and women and the interaction of sex with progression.
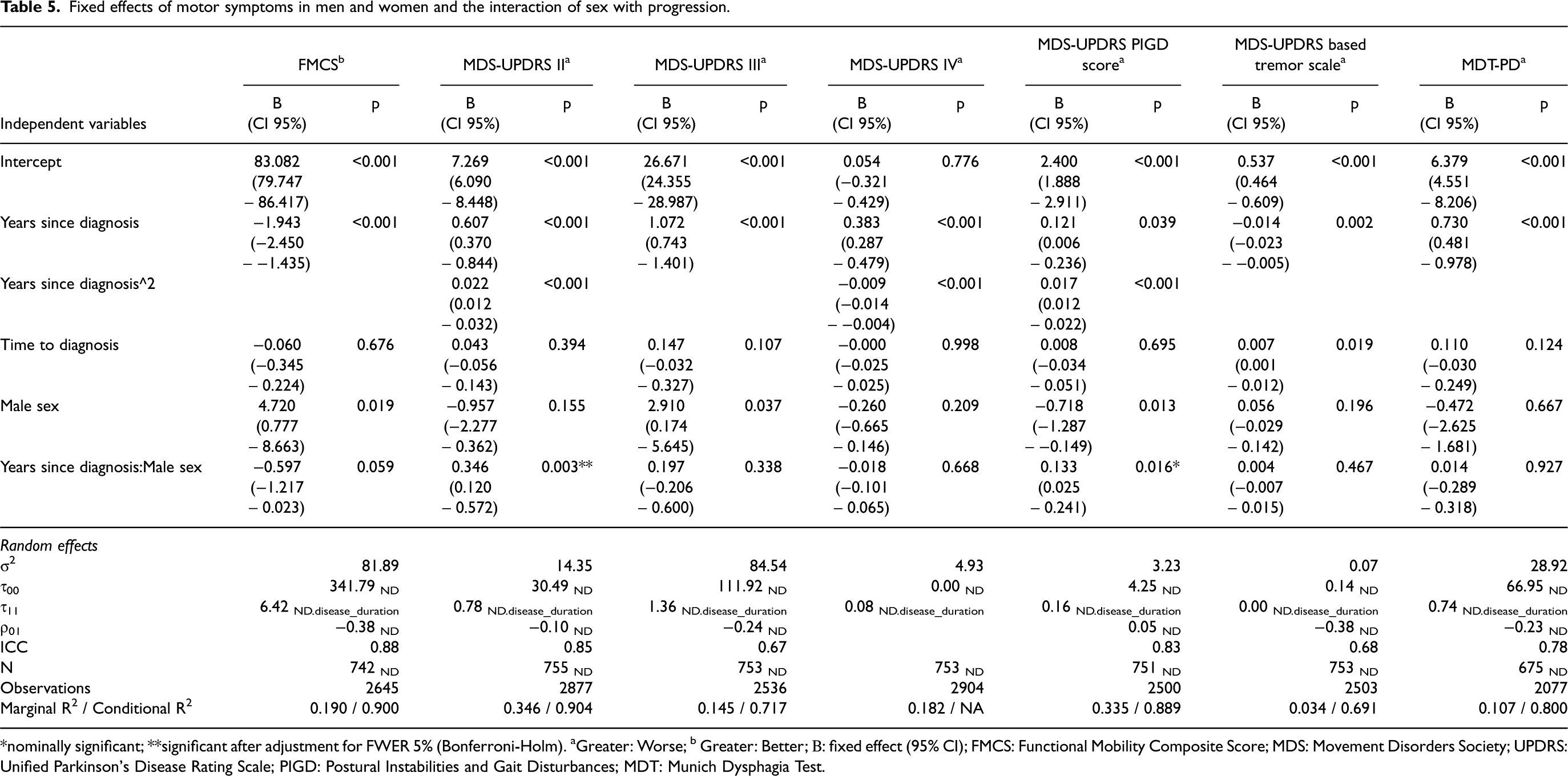
*nominally significant; **significant after adjustment for FWER 5% (Bonferroni-Holm). aGreater: Worse; b Greater: Better; Β: fixed effect (95% CI); FMCS: Functional Mobility Composite Score; MDS: Movement Disorders Society; UPDRS: Unified Parkinson's Disease Rating Scale; PIGD: Postural Instabilities and Gait Disturbances; MDT: Munich Dysphagia Test.
Women overall demonstrated a slower progression than men. More specifically, men had a significantly faster progression in global cognition (MoCA) (r = −0.159, 95% CI [−0.272, −0.046], p = 0.006, Table 4), quality of sleep (PDSS) (r = −0.716, 95% CI [−1.229, −0.203], p = 0.006, Table 4) and postural instabilities and gait disturbance (MDS-based PIGD score) (r = 0.133, 95% CI [0.025, 0.241], p = 0.016, Table 5) on an unadjusted significance level and in patient-reported motor symptoms (MDS-UPDRS II) (r = 0.346, 95% CI [0.120, 0.572], p = 0.003, Table 5). The findings for patient-reported motor symptoms were significant after adjustment for FWER 5%. After controlling for age in the model analyzing the progression in global cognition, the p-value for the interaction effect between sex and time on global cognition decreased from 0.006 to 0.004 while we did not identify any statistically significant differences for age, years since diagnosis or time to diagnosis at baseline between men and women with typical PD. Finally, the frequency of missing data at follow-up in women was not significantly higher than in men.
Discussion
The present study described and illustrated the progression of motor- and non-motor symptoms in men and women with typical PD. Both men and women showed a progression (i.e., deterioration) in all symptoms. Comparing symptoms progression between men and women, women experienced a slower progression in global cognition (MoCA), quality of sleep (PDSS), postural instabilities and gait disturbances (MDS-UPDRS based PIGD score) and patient-reported motor symptoms (MDS-UPDRS II). Finally, we observed similar trajectories for patient-reported outcomes compared to clinician-assessed outcomes in both men and women.
Non-motor symptoms
Previous reviews3–5 discussed the heterogeneous findings of sex-specific progression of PD. According to our findings, women tended to have a generally slower disease progression than men. As women had a slower progression of PIGD and patient-reported motor symptoms, the worse bodily discomfort at baseline in women compared to men (similar to previous findings18–21) might be experienced in relation to something else, e.g., different symptom expressions, such as the restless legs syndrome being more common and severe in women, 21 while this sex-specific effect moderation was not identified in the longitudinal data. Similarly, in women, depression (BDI-I) was worse at baseline while no sex-specific effect moderation was identified in the longitudinal data, similar to previous research. 7
Our study confirmed that after similar scores at baseline, women's cognitive performance (MoCA) declined slower over time. 8 Moreover, we observed a similar progression of apathy (SAS), a feature of PD dementia. 22 Interestingly, women had a similar age and frequency of PD dementia and had even less years of education than men. Consequently, future research should consider examining reserves (e.g., hormones, different genetic mechanism or different exposures to environmental risk factors) protecting women from a decline. Finally, while women had a worse quality of sleep (PDSS) at baseline, we detected a faster progression in men than compared to women.
Motor symptoms
Our results support previous longitudinal findings7,8 of women having higher disability scores at baseline, but men progressing faster. We did not detect any significant differences in the motor complications (MDS-UPDRS IV) while in women the mg/kg LEDD dose was significantly higher compared to men at baseline. As we included the LEDD (mg per kg body weight), i.e., taking into account the systematic differences in weight and height between men and women, our findings might not be comparable to previous findings. 7 Our results also confirm previous findings 6 that the PIGD dominant phenotype is more frequent in women. However, we cannot confirm previous findings 23 describing impairments in global cognition were associated with more severe PIGD symptoms, as despite the more frequent PIGD dominant phenotype at baseline, women had slower cognitive decline (MoCA). As only half of the phenotypes remain stable over three years 24 and postural instabilities and gait disturbances (PIGD) progressed slower in women, this finding needs to be further explored. We used assessment instruments as recommended by the Movement Disorders Society.25–27 While the sex-specific effect moderation was not significant for the clinician-assessed motor symptoms (MDS-UPDRS III), we found a significant sex-specific effect moderation for patient-reported motor symptoms (MDS-UPDRS II), warranting further investigation into the underlying reasons for this divergence (e.g., sex-specific validity, differences in how women and men experience or report motor symptoms). In addition to patient-reported motor symptoms (MDS-UPDRS II), PIGD and quality of sleep (PDSS), female sex was also associated with a slower progression of global cognition (MoCA). As global cognition could be associated with the experience of patient-reported outcomes, the role of global cognition as a covariate needs to be taken into account by future studies interested in the direct effect of sex on disease progression. Finally, our study confirms recent findings 28 of tremor severity decreasing over time. Further analysis about the difference between resting, postural and kinetic tremor are required.
Strengths and limitations
This study has some strengths and limitations. For instance, we enhanced the generalizability of our findings by analyzing data of all participants of the Luxembourg Parkinson's study including people with PD or PDD from Luxembourg and the Greater Region, who were treated and lived in varying settings and environments. More specifically, the range of people with PD was broad, including men and women from 22 to 92 years with 1 to 30 years of education, living from 0 to 32 years with the disease and speaking different languages. 68.7% of the people with PD were in disease stages H&Y 1–2, the disease stages ranged from H&Y 1 to H&Y 5. Recruitment started in 2015 when the estimated prevalence of PD in Luxembourg was 565–1356.9,10 As 486 of the participants lived in Luxembourg, this corresponds to an estimated coverage of 35.8 to 86.0% of the people with PD living in Luxembourg.
Moreover, we used advanced statistical methodology to estimate changes over time in our longitudinal dataset with mixed models taking into account the dependence between repeated measures. Although our analysis is observational, our longitudinal study provided a comprehensive description of the individual progression of symptoms in PD while previous studies were mainly cross-sectional analyses with some exceptions.7,29–31 Most observations refer to the first twenty years since diagnosis. Consequently, our results should not be interpreted beyond this period. Future research interested in the detailed symptoms trajectories could explore alternative non-linear models, such as asymptotic functions, which might better detail the long-term trajectories of motor complications. Also, the COVID-19 pandemic and deaths since baseline assessment (101, 12.6%) led to missing data. For the Munich Dysphagia Test (MDT) score, we noted higher rates of missing values, as it was added later during the study explaining the nature of the missing values. Consequently, the analyses on this outcome should be considered exploratory. Despite the potential sampling bias for the analyses involving the onsite test MDS-UPDRS III, the frequency of missing data at follow-up was similar in both groups. Data collection standards have been developed to minimize missing data and information bias.
Our research described the progression since the diagnosis and is applicable to the progression in the first twenty years. Future research should use data of risk and prodromal cohorts to describe the biological progression before the diagnosis of PD. 32 Our study focused on describing sex-specific progression. In the current study, women were characterized by fewer years of education, a higher likelihood of being divorced or widowed, a lower BMI, a higher LEDD (mg/kg), more symptoms of depression (BDI-I) and bodily discomfort (PDQ-39 subscale) but better olfaction (Sniffin’ sticks). Consequently, future research investigating the direct effects of sex could consider these variables along with relevant comorbidities or other factors (e.g., hearing loss, hormones, different genetic mechanisms or exposures to environmental risk factors) as covariates. Moreover, the biological plausibility for the sex-specific progression in PD and the sociocultural and behavioral protective factors in women need to be further investigated. In addition, health professionals should adapt their communication and non-pharmacological disease management strategies to the different needs of men and women (e.g., sex-specific communication strategies, flyers, and exercises). Specifically, in men during the first ten years after diagnosis, communication and interventions should focus on the promotion of global cognition, patient-reported motor symptoms (MDS-UPDRS II), quality of sleep (PDSS) and postural instabilities and gait disturbances (PIGD) 33 while information and interventions targeting women could focus on promoting bodily comfort (e.g., prevent muscle cramps, joint pain, unpleasant hot/cold sensation) and mental health.
In conclusion, our study provided a comprehensive data-based description and illustration of the clinical progression of motor- and non-motor symptoms associated with PD for men and women. Moreover, the detailed interaction plots should aid interpretation by health professionals. Factors explaining the resilience in women with PD especially in global cognition, quality of sleep, patient-reported motor symptoms and postural instability and gait disturbances need to be explored further. To enhance well-being and personalized treatment in PD, interventions, proactive monitoring and communication strategies tailored to the symptoms progression in men and women need to be developed.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251339201 - Supplemental material for Sex-specific progression of Parkinson's disease: A longitudinal mixed-models analysis
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251339201 for Sex-specific progression of Parkinson's disease: A longitudinal mixed-models analysis by Anne-Marie Hanff, Christopher McCrum, Armin Rauschenberger, Gloria A Aguayo, Claire Pauly, Sonja R Jónsdóttir, Olena Tsurkalenko, Maurice P Zeegers, Anja K Leist, Rejko Krüger and in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We would like to thank all participants of the Luxembourg Parkinson's Study for their important support of our research. Furthermore, we acknowledge the joint effort of the National Centre of Excellence in Research on Parkinson's Disease (NCER-PD) Consortium members from the partner institutions Luxembourg Centre for Systems Biomedicine, Luxembourg Institute of Health, Centre Hospitalier de Luxembourg, and Laboratoire National de Santé generally contributing to the Luxembourg Parkinson's Study as listed below:
Geeta ACHARYA 2, Gloria AGUAYO 2, Myriam ALEXANDRE 2, Muhammad ALI 1, Wim AMMERLANN 2, Giuseppe ARENA 1, Michele BASSIS 1, Roxane BATUTU 3, Katy BEAUMONT 2, Sibylle BÉCHET 3, Guy BERCHEM 3, Alexandre BISDORFF 5, Ibrahim BOUSSAAD 1, David BOUVIER 4, Lorieza CASTILLO 2, Gessica CONTESOTTO 2, Nancy DE BREMAEKER 3, Brian DEWITT 2, Nico DIEDERICH 3, Rene DONDELINGER 5, Nancy E. RAMIA 1, Angelo FERRARI 2, Katrin FRAUENKNECHT 4, Joëlle FRITZ 2, Carlos GAMIO 2, Manon GANTENBEIN 2, Piotr GAWRON 1, Laura GEORGES 2, Soumyabrata GHOSH 1, Marijus GIRAITIS 2,3, Enrico GLAAB 1, Martine GOERGEN 3, Elisa GÓMEZ DE LOPE 1, Jérôme GRAAS 2, Mariella GRAZIANO 7, Valentin GROUES 1, Anne GRÜNEWALD 1, Gaël HAMMOT 2, Anne-Marie HANFF 2, 10, 11, Linda HANSEN 3, Michael HENEKA 1, Estelle HENRY 2, Margaux HENRY 2, Sylvia HERBRINK 3, Sascha HERZINGER 1, Alexander HUNDT 2, Nadine JACOBY 8, Sonja JÓNSDÓTTIR 2,3, Jochen KLUCKEN 1,2,3, Olga KOFANOVA 2, Rejko KRÜGER 1,2,3, Pauline LAMBERT 2, Zied LANDOULSI 1, Roseline LENTZ 6, Ana Festas LOPES 2, Victoria LORENTZ 2, Tainá M. MARQUES 2, Guilherme MARQUES 2, Patricia MARTINS CONDE 1, Patrick MAY 1, Deborah MCINTYRE 2, Chouaib MEDIOUNI 2, Francoise MEISCH 1, Alexia MENDIBIDE 2, Myriam MENSTER 2, Maura MINELLI 2, Michel MITTELBRONN 1, 2, 4, 10, 12, 13, Saïda MTIMET 2, Maeva MUNSCH 2, Romain NATI 3, Ulf NEHRBASS 2, Sarah NICKELS 1, Beatrice NICOLAI 3, Jean-Paul NICOLAY 9, Maria Fernanda NIÑO URIBE 1,3, Fozia NOOR 2, Clarissa P. C. GOMES 1, Sinthuja PACHCHEK 1, Claire PAULY 2,3, Laure PAULY 2, 10, Lukas PAVELKA 2,3, Magali PERQUIN 2, Achilleas PEXARAS 2, Armin RAUSCHENBERGER 1, Rajesh RAWAL 1, Dheeraj REDDY BOBBILI 1, Lucie REMARK 2, Ilsé RICHARD 2, Olivia ROLAND 2, Kirsten ROOMP 1, Eduardo ROSALES 2, Stefano SAPIENZA 1, Venkata SATAGOPAM 1, Sabine SCHMITZ 1, Reinhard SCHNEIDER 1, Jens SCHWAMBORN 1, Raquel SEVERINO 2, Amir SHARIFY 2, Ruxandra SOARE 1, Ekaterina SOBOLEVA 1,3, Kate SOKOLOWSKA 2, Maud THERESINE 2, Hermann THIEN 2, Elodie THIRY 3, Rebecca TING JIIN LOO 1, Johanna TROUET 2, Olena TSURKALENKO 2, Michel VAILLANT 2, Carlos VEGA 2, Liliana VILAS BOAS 3, Paul WILMES 1, Evi WOLLSCHEID-LENGELING 1, Gelani ZELIMKHANOV 2,3
Luxembourg Centre for Systems Biomedicine, University of Luxembourg, Esch-sur-Alzette, Luxembourg Luxembourg Institute of Health, Strassen, Luxembourg Centre Hospitalier de Luxembourg, Strassen, Luxembourg Laboratoire National de Santé, Dudelange, Luxembourg Centre Hospitalier Emile Mayrisch, Esch-sur-Alzette, Luxembourg Parkinson Luxembourg Association, Leudelange, Luxembourg Association of Physiotherapists in Parkinson's Disease Europe, Esch-sur-Alzette, Luxembourg Private practice, Ettelbruck, Luxembourg Private practice, Luxembourg, Luxembourg Faculty of Science, Technology and Medicine, University of Luxembourg, Esch-sur-Alzette, Luxembourg Department of Epidemiology, CAPHRI School for Public Health and Primary Care, Maastricht University Medical Centre+, Maastricht, the Netherlands Luxembourg Center of Neuropathology, Dudelange, Luxembourg Department of Life Sciences and Medicine, University of Luxembourg, Esch-sur-Alzette, Luxembourg
ORCID iDs
Ethical considerations
The studies involving human participants were reviewed and obtained a positive opinion from the National Ethics Board (CNER Ref: 201407/13). We confirm that we have read the Journal's position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
Consent to participate
The patients/participants provided their written informed consent to participate in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Centre of Excellence in Research on Parkinson's Disease (NCER-PD) is funded by the Luxembourg National Research Fund (FNR/NCER13/BM/11264123). The work was further supported by the PEARL program (FNR/P13/6682797 to RK).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The LUXPARK database used in this study was obtained from the National Centre of Excellence in Research on Parkinson's disease (NCER-PD). NCER-PD database are not publicly available as they are linked to the Luxembourg Parkinson's study and its internal regulations. The NCER-PD Consortium is willing to share its available data. Its access policy was devised based on the study ethics documents, including the informed consent form approved by the national ethics committee. Requests for access to datasets should be directed to the Data and Sample Access Committee by email at request.ncer-pd@uni.lu. The R-code is available on the OSF Page: DOI 10.17605/OSF.IO/4VAWS
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
