Abstract
Parkinson's disease represents a major healthcare burden with over 1 million confirmed cases and economic costs of care exceeding $50 billion in the United States alone. A challenge in the evaluation of potentially disease delaying, preventing, or reversing agents is identification of patients in the very early, prodromal (asymptomatic, non-motor, or early motor) stages, which can span decades. Currently, diagnosis is based almost exclusively on clinical history and physical examination conducted by trained experts. Unfortunately for many patients, access to neurology and movement disorder specialists is geographically limited and coupled with the absence of coordinated disease awareness campaigns can contribute to substantial delays in diagnosis. As with any campaign, a key success factor is having multiple approaches which can appeal to a wide range of patient archetypes. Herein we elaborate on some emerging opportunities, which if fully developed could invigorate the approach to early detection and, critically, engage patients in this process. They include 1) Use of personal digital health technologies and community based point-of-care diagnostic instrumentation, 2) The potential to leverage annual testing services at non-traditional venues including optometry, dental and pharmacy, and 3) A consumer focused campaign to raise awareness of the importance of early detection, paralleling efforts in other diseases. There is every hope that combining traditional and non-traditional approaches to enhance early diagnosis rates can have an overall positive impact on patient outcomes and contribute substantively to efforts to develop new interventions. We urge the community to embrace these possibilities and begin active dialog on how to thoughtfully implement the approaches into routine care.
Plain language summary
There are approximately 8 million people living worldwide with Parkinson's disease. Our understanding of its causes continues to increase, but there are still no prescription drugs that can delay, prevent, or reverse the disease and its many symptoms. The benefits of new drug therapies may be higher for patients who are in the very earliest stages of the disease, but it is often difficult to find sufficient numbers of patients with early disease to participate in clinical drug trials. There are several limiting factors. Early stage symptoms are often subtle and highly variable, and it can sometimes take many years before a patient is formally diagnosed. Another factor is the shortage of trained medical specialists worldwide who are equipped to make these diagnoses. We outline in this opinion piece how consumer-oriented approaches might help with earlier diagnosis of the disease. For example, at-home and mail-in diagnostic tests, pharmacy based screening, and routine dental and optical examinations each offer the potential to identify early warning signals of disease. We anticipate that information from these new testing approaches will increase the likelihood of people seeking a focused checkup to evaluate their disease risk through their traditional care providers, such as primary care physicians and neurologists. However, these alternative testing paths would need to be coupled to an awareness campaign where ethical considerations are carefully balanced to be sensitive to consumer anxiety and also the potential for false positive results. Such concerns, though, may be outweighed by the benefits of earlier patient diagnosis. In particular, because eligibility to enroll in clinical drug trials is often restricted to early stage patients, new awareness and testing campaigns to diagnose disease should expand the pool of patients who can participate in clinical drug trials. Expanded recruitment, may, in turn, accelerate the development and ultimate approval of new disease modifying therapies and greater potential to slow, halt or reverse the disease process.
With annual diagnosis rates now exceeding 100,000 patients per year, 1 Parkinson's disease (PD) represents a major healthcare burden with over 1 million confirmed cases and economic costs of care exceeding $50 billion in the United States alone. 2 In contrast to the pathophysiology of some other major diseases, e.g., cardiovascular, that of PD has not yet been shown to be reversed or reduced by any intervention, from modifiable lifestyle and dietary factors to drugs, making their development a critical unmet need. A challenge however in the quest to evaluate potentially disease delaying, preventing, or reversing agents is identification of patients in the very early, asymptomatic prodromal stages, which can span multiple decades. 3 Confirmatory tests, typically ordered or performed by expert neurologists may involve quantitation of misfolded alpha-synuclein protein (a presumed culprit) through seed amplification assays using patient cerebrospinal fluid (CSF) extracted through lumbar puncture. 4 Additional methods include nuclear imaging for dopamine receptors (DAT scan), 5 skin biopsy for phosphorylated alpha-synuclein through skin punctures, 6 and olfactory testing. 7
The use of these molecular diagnostics coupled with genetic diagnostics that can identify heritable forms of the disease, 8 is largely limited to specialty PD care currently while their availability, cost, invasiveness, and therapeutic applications evolve. Currently, whether DAT imaging, an alpha-synuclein diagnostic, both, or (still most commonly) neither is employed in a diagnostic workup is highly dependent on the particular patient, clinician, and practice environment. However, rapid progress suggests that most if not all of these limitations could be overcome in the next decade, transforming their use from movement disorders to general neurologists, on to general practitioners, and possibly directly to healthcare consumers.
Beyond supporting early diagnosis, the therapeutic utility of these tests will of course require carefully designed studies that rely on a well-defined PD population, and demonstrate the efficacy of an intervention in substantially maintaining/ improving the lives of people with clinically diagnosed PD or in preventing future disability in those at high risk. Only a few disease-modification trials in PD have thus far enriched for PD populations by incorporating DAT deficit on neuroimaging as an eligibility criterion,9,10 and the first trials that will enrich for a positive synucleinopathy test are in development. 11
For most patients today, the diagnosis of PD is based almost exclusively on clinical history and physical exam with only the occasional inclusion of biomarkers. This strategy proves sufficient for many patients, particularly those able to be evaluated by a trained movement disorders specialist soon after symptoms develop. Unfortunately, for most patients their being evaluated by a sub-specialist may not always be possible or practical, given the relatively limited number and geographically concentrated locations of movement disorders specialists. In the United States, it was recently reported that 40% of people with PD are not receiving care from a neurologist and only 9% are receiving care from a movement disorder specialist. 12 The lack of specialist care can lead to delays in diagnosis for many, with consequential delays in therapies, both pharmacologic and non-pharmacologic, as well as delays in social support through PD focused support groups and education.
Even within the setting of highly specialized clinics the diagnosis is not always accurate in a significant percentage of patients at clinical presentation. Therefore earlier, more accurate diagnosis, prior to development of characteristic symptoms, is not possible without strong support from biomarkers and diagnostics. Some argue that earlier diagnosis is not necessary because it would not lead to changes in therapy; however, this approach leaves the field at an impasse. Clinical trials to progress preventative therapies cannot move forward without patients identified at the very earliest stages of disease prior to symptom onset, but currently patients at early stages are not prioritized for diagnosis due to the lack of effective therapies and availability of more precise, widely available markers and tools.
Many patients experience years of symptoms before the disease itself becomes clinically manifest. Patients struggle with understanding their change in health, often seeking an answer to explain these symptoms. As suggested above, the patient journey from recognition of symptoms to receiving a diagnosis can be long as there is not only a dearth of specialist providers but also inexact methods available for diagnosis of the disease. 13 Here, patients feel empowered and experience improved perceptions of health by knowing more about their disease. As has been shown for genetic testing in PD, patients find personal utility in knowing more about their personal health even though treatment options may not yet be available.14,15
As with any disease awareness campaign, a key success factor is having multiple approaches which can appeal to a wide range of patient archetypes, which range from motivated and health conscious all the way to ambivalent and skeptical. One must also consider the dimension of how patients engage in preventative medicine which can be purely episodic and healthcare provider (HCP)-centric, through to proactive self-monitoring. Herein we elaborate on some key emerging opportunities, which if fully developed could invigorate the approach to early detection of PD and, critically, engage patients in this process.
Early assessments of research participation appetite among individuals who are at risk for PD (either genetically or based on prodromal features like REM sleep behavior disorder) indicate a strong interest, including for prevention studies. The motivation of those at-risk genetically may be boosted by personal experience seeing the challenges of PD in a parent or sibling with PD, 15 whereas the motivation of those at-risk due to prodromal features may be amplified from their personal experience confronting symptoms that indicate disease already manifesting in their bodies.
As outlined in Figure 1, at the patient level there are many functions in consumer grade devices (smartphones, smart watches) which can identify and track signals which may be indicative of detecting early-stage PD. For example, motion sensing both gait 16 and asymmetry of walking, 17 two hallmark PD characteristics, are routinely monitored by the Apple Health app on watches and smartphones. Similarly, ballistographic and inertial measurement (IMU) sensors are capable of detecting tremor like events which are hallmarks of later stages of the disease but could be insightful for generation of early warning signals in some patients. 18 Likewise, keystroke analysis has been shown to be effective in identifying symptomatic from healthy control subjects, 19 and in terms of keyed content there is even a commercial test now available which purports to provide PD diagnosis with instant turnaround and with up to 90% accuracy identifying early stage Parkinson's disease based on results from over 40,000 tests. 20 Both the microphone and front camera of smartphones can provide rich information on PD associated acoustic / linguistic features, 21 and signature oculomotor events, 22 and gamified approaches (often used in clinical tests with confirmed PD patients) 23 could be envisioned to drive even richer signals.
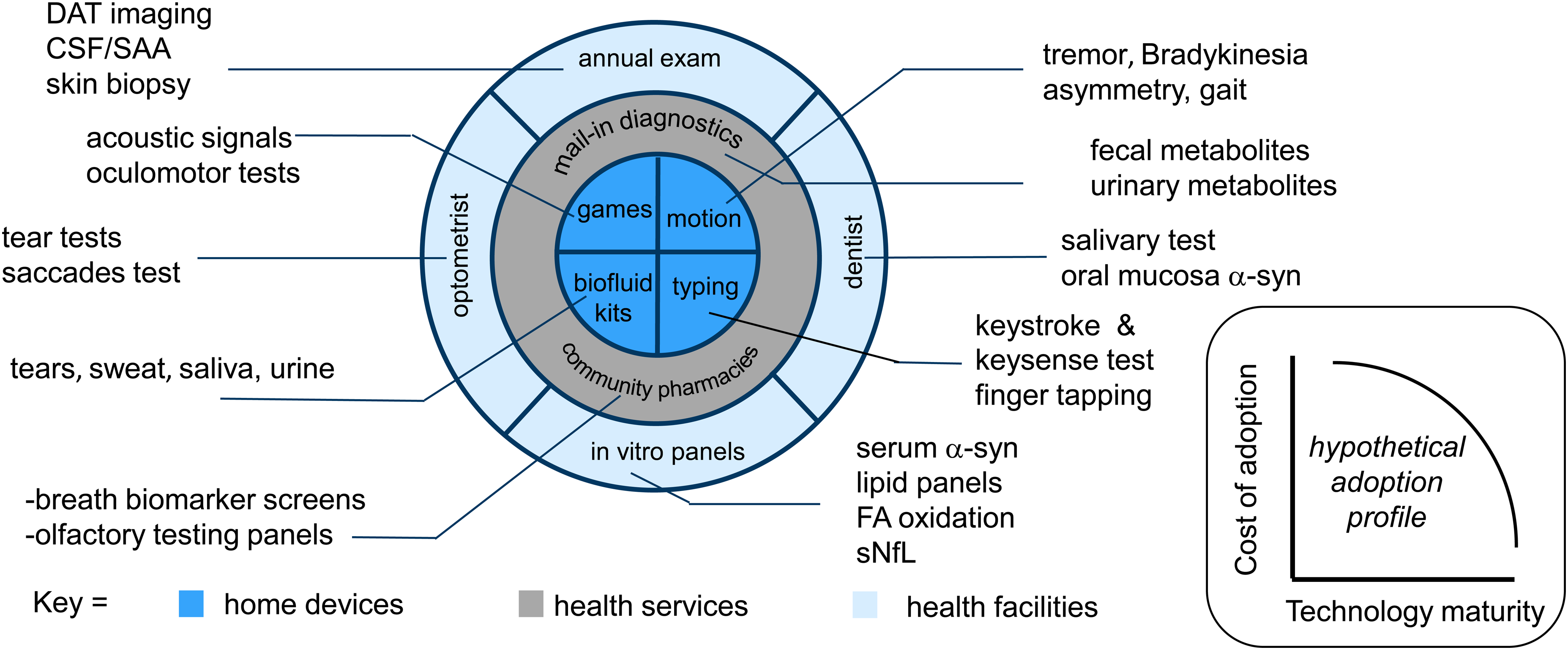
Diagnostic opportunities for Parkinson's disease across the health services continuum. CSF: cerebrospinal fluid; SAA: seed amplification assay; α-syn: α-synuclein; FA: fatty acids; sNfL: serum neurofilament light chain.
Several recent studies in PD have demonstrated the willingness of patients to consider engaging in clinical activities at home, from the very simple to the more complex. Anosmia is well-regarded prodromal symptom of PD. As part of the Parkinson's Progression Marker Initiative (PPMI) study, individuals both with and without PD over the age of 60 are consented to receive an UPSIT smell test via the mail and record their results online. 24 Genetic testing in PD is underutilized but can provide a clear link and diagnostic confirmation of PD. The PD GENEration study combines both at-home and in-person participation of sample collection (buccal swab or peripheral blood) and genetic counseling to drive recruitment with currently over 18,000 participants.8,25 More involved is the Trial Of Parkinson's And Zoledronate (TOPAZ), a decentralized randomized control study that brings study nurses to the homes of people with Parkinson's to receive infusions of zoledronic acid as potential modifier of fractures this largely elderly population and has over 2300 participants. 26 Each of these studies, with their high levels of participation, underscores the interest and willingness of those living with PD to contribute to research and to learn more about their disease. Moreover, the ability to bring testing to where PD patients is especially critical as the majority of those living with PD are elderly and PD itself is a disorder of movement, thereby making travel to sites of specialized care or testing to be problematic for this population.
Another longitudinal observational study of people with early to mid-stage disease, AT-HOME PD has shown good engagement across multiple decentralized monitoring modalities (video visits from home with a modified Unified Parkinson's Disease Rating Scale (UPDRS) examination by a movement disorders clinician), smartphone PD metrics via the mPower app, and online survey completion through Fox Insight. 27 Also interesting is emerging evidence through use of ‘invisible’ Wi-Fi-based technology monitoring changes in sleep patterns, 28 nocturnal breathing, gait/ gait speed, balance among at-home movement monitoring methods. 29 Such approaches offer benefits of contactless background monitoring while also raising some questions regarding ensuring privacy.
There are numerous challenges that need resolved prior to implementing any at-home diagnostics for PD. First and foremost is understanding and educating around test accuracy, particularly within this context-of-use. As the possibility of at-home testing emerges data on the accuracy of any test needs to be evaluated in the context of people taking the test with a low pre-test probability of having PD. Indeed, most biomarker data is collected within the context of people with either some level of disease-related symptoms or some level of disease-related risk, leading to a moderate to high pre-test probability that they have PD. This reduces the false positive rate and elevates the subsequent positive predictive value (i.e., if the test is positive what is the likelihood that the person has the disease) of the test. However, any test at scale in a wider population that includes people who are less likely to have the disease, such as those with just constipation or just change in gait, needs to be assessed for specificity within this context-of-use. While screening tests are typically skewed toward high sensitivity to avoid a false negative, this does increase false positives. Education around interpreting a positive result would be critical in this setting to avoid unintentional harm that would occur if a test outcome led a person to believe they have a diagnosis of PD, when they do not. Further, clear guidelines on what to do after a positive at-home test would be essential, as well as sufficient resources to support people who have taken an at-home test and require a follow-up evaluation for confirmation testing. This infrastructure requires engagement with communities and gate-keeper medical personnel, with particular attention to lower resource areas or those without the medical insurance needed to pay for follow-up confirmatory evaluations.
Moving from at-home to remote testing possibilities, there are a number of mail-in diagnostic services which include urine and fecal analysis where sample kits are purchased and have pre-paid residential collection by carriers. In the case of PD, there are associations with lipids and metabolites which may be detected in urine, 30 and there is considerable interest in fecal microbiome and possible causal relationships with PD. 31 The latter is particularly interesting, as it may offer a patient led option to alter the gut-brain microbiome axis through dietary modification. 32 A second, non-residential option for diagnostics is through community pharmacies, many of which now offer mini-clinic and phlebotomy services. One potential offering could be olfactory analysis. It is known that PD patients’ sense of smell is impaired 7 and a standardized testing service, calibrated using analytical controls could be an informative patient challenge. 33 There may also be potential for the use of breath biopsy and analysis of the volatilome in early stage diagnostics. 34
Finally, at the level of managed health care, there are of course myriad new opportunities for PD diagnostics. In the case of HCP-based annual and follow-on checkups, there is potential for a physician to refer the patient to a neurologist for examination, and after consultation and assessment on the MDS-UPDRS (or Hoehn-Yahr) scale, recommend confirmatory DAT scan, 5 CSF seed assay, 4 or skin punch biopsy. 6 However, in some cases, e.g., in rural areas where availability of neurologists is more limited, additional in vitro assays could be prescribed, over and above those typically arranged, e.g., lipid panels, A1C etc. Emerging markers, including serum-based alpha-synuclein, 35 fatty acid metabolites, 36 and neurofilament fragments 37 may be useful for early PD detection despite known limitations relating to specificity for PD and related conditions.
The final categories are particularly interesting in that they are non-traditional yet potentially powerful. It is known that a large proportion of oral cancers are first identified by a dental examination, 38 and in this same vein it is known that the oral mucosa can contain alpha-synuclein which could be diagnostic. 39 Could such a salivary/mucosal screening test be explored for early PD in the United States and more broadly in other parts of the world? Our familiarity of rapid testing using buccal swabs was strengthened during the COVID-19 pandemic and could become a powerful and low friction screening tool for PD.
Other opportunities exist through optometry. A significant number of people above 50 years of age regularly visit ophthalmologists for annual examinations. It is common during these visits for persons to be offered an (elective) glaucoma test and a battery of PD relevant tests could be envisioned. For example, analysis of saccades can now be performed using benchtop systems and can be highly informative for identification of neuromuscular anomalies associated with PD.40,41 Similarly, tear analysis for qualitative assessment of alpha-synuclein may become a reality, and has evident potential if approached as part of holistic healthcare. 42
For all of the potential diagnostic approaches which may be considered it will be important to refine for sensitivity and selectivity in order to offer meaningful outputs. Advances in analytical methodologies, instrumentation and data processing algorithms will serve as enablers, as evidenced in skin-based α-synuclein assays, where pooled 43 and composite 44 assays have achieved up to 95% sensitivity and 97% specificity, comparing favorably with gold standard CSF derived methods (98% and 95% respectively). 45 It can also be expected that as awareness, interest, and ultimately commercial demand for diagnostic tests increased economies of scale will lower associated costs. This has been demonstrated in the case of salivary diagnostics, 46 and underscored dramatically during the COVID-19 pandemic. 47 Similarly for infrastructure intense diagnostics such as SPECT based DAT scans the majority of the costs (which average approximately $2750) stem from the radiotracer itself, suggesting scale might reduce this burden through clustering patient visits (many are even offered free of charge if under the terms of an existing research study). 48 There is also growing momentum in the development of digital diagnostics using apps deployed on consumer products, 49 benefitting from the learnings of recent research studies 50 and being obvious candidates for scaling to population level. With these and related consumer-centered approaches, there naturally needs to be appropriate consideration for medical ethics and availability of targeted counselling from medical professionals. This is particularly relevant in the absence of effective disease modifying therapies; however, numerous reports point to improved outcomes for early diagnosed patients who initiate lifestyle interventions.51,52
In summary, there exists a growing need for early identification of prodromal PD patients, which will require appropriate screening tools coupled with effective incentivized engagement models. Recent advances with personal devices, remote diagnostics, and managed healthcare suggest breakthroughs may be possible at population level in the near future. In order to accelerate this potential we suggest the following possible actions:
Initiatives to help develop point-of-care diagnostic instrumentation for early PD detection. This will require active collaboration between developers, health providers, health authorities and payers among others. The economic benefits of such tools through their role in improving early diagnosis rates will need to incentivize developers and help develop a market for such, and active dialog among stakeholders is needed. Re-examination of care provision to facilitate annual testing at non-traditional venues including optometry, dental and pharmacy. Engagement of professional bodies such as the American Dental Association, American Optometric Association and American Pharmacists Association will be necessary in order that clear standards and guidelines can be developed for subsequent implementation. A coordinated campaign for raise awareness of the importance of early detection of PD. One possibility is to develop an early warning risk assessment-based acronym to raise awareness for PD screening, analogous to the F.A.S.T. campaign in stroke and Crucial Catch campaign for prostate cancer.
53
Such an alert could be comprised of a composite of signals including select motor, cognitive and sensory. Such an awareness campaign may also help stimulate demand for community-based screens and self-testing services, all of which can benefit by identifying patients as early as possible. Research foundations and non-profits could play an enabling role in such a campaign as their patient centric and advocacy initiatives could help ensure maximum outreach.
Obviously, the intent of these efforts is to identify and enable more potential early-stage patients to be afforded access to expert diagnosis and care from trained neurologists, and the opportunity to benefit from clinical trials and emerging treatments. Although access to such interventions will vary globally and costs (e.g., for DAT imaging, neurofilament light chain screening) may be a barrier, over the long-term this has the potential to reduce overall healthcare expenditures and improve quality of life particularly if introduced in a manner which tempers undue anxiety and potential ethical challenges associated with health information overload.54,55 Such efforts to increase the number of activated patients through awareness campaigns have proven effective in other disease areas. There is every hope they can do likewise for currently undiagnosed PD patients in cost effective approaches that could fit into routine clinical visits and/ or low burden at home testing and we urge the community to embrace this possibility.
Footnotes
Acknowledgments
The authors thank Mr John Poma for editing and providing patient perspective to the plain language summary. Views expressed are those of the author group and Novartis Pharmaceuticals had no influence on the content of the submitted manuscript. References cited were selected using search engines (SciFinder, Google Scholar) on the basis of scientific relevance, with priority afforded to most recent exemplars. Generative AI was not used in any portion of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AK and GJ are employees of Novartis AG and may hold stock in the company. JB is employed by the Parkinson's Foundation and receives funding for grants through their institution from NIH and ASAP. KP has been on the Scientific Advisory Board for Amprion, where she receives stock options, has been a consultant for Novartis, Biohaven, Curasen and Neuron23, where she receives consulting fees, and has received grant funding from the NIH, Michael J Fox Foundation for Parkinson's Research, the Knight Initiative for Brain Resilience, the Wu Tsai Neuroscience Institute, Lewy Body Dementia Association, Alzheimer's Drug Discovery Foundation and the Sue Berghoff LBD Research Fellowship. MS received consulting fees through the Parkinson Study Group (PSG) for its advisory services (including to Biogen (LUMA/LIGHTHOUSE trials global SC) and to UCB (ORHCESTRA trial PSG SC), and consulting fees and/or travel coverage for steering committee service from Sutter Health (TOPAZ trial, NIA), Northwestern University (SPARX3 trial, NINDS), Parkinson's Foundation (PD-GENEraton study, and Cure Parkinson's (International Linked Clinical Trial Committee).
