Abstract
Background
Parkinson's disease (PD) is the second most prevalent neurodegenerative disorder with broad manifestations of motor and non-motor symptoms. While significant progress has been made in assessing motor dysfunction through wearable sensors, less attention has been directed towards bulbar issues like swallowing difficulties.
Objective
We introduce a digital health solution leveraging advanced acousto-mechanic (ADAM) sensors capable of comprehensively evaluating motor and bulbar dysfunction in PD that additionally offers artificial intelligence-driven haptic biofeedback to enhance swallowing frequency.
Methods
The swallow detection algorithm developed with data from n = 58 healthy subjects yielded an F1 score of 0.89 for swallow event detection.
Results
In a pilot study with PD patients (n = 20) experiencing mild (60%) or moderate (40%) dysphagia, the use of ADAM sensors with biofeedback significantly increased swallow frequency by 45%, from 0.77 to 1.10 swallows per minute (p < 0.0001). The sensors demonstrated high sensitivity (89%) and a strong correlation with visual observations by speech language pathologists (r = 0.92, p < 0.05), with 100% agreement on respiratory-swallow phase patterning. Feedback from patients and caregivers underscored the utility and comfort of the technology.
Conclusions
This tailored digital health solution not only monitors PD symptoms but also holds potential as an assistive device, marking a significant step in improving the quality of life for PD patients.
Plain language summary
Parkinson's disease (PD) is a common neurodegenerative disorder that causes various symptoms, affecting both movement and non-movement functions, like swallowing. While wearable devices have improved tracking of movement issues, less attention has been given to monitoring and assisting swallowing difficulties in PD. This study aimed to test a new wearable sensor system, called ADAM, designed to monitor and assist swallowing in people with PD by giving gentle feedback to encourage more frequent swallowing. Our team developed ADAM sensors that detect swallowing events and provide feedback to improve swallowing patterns. We tested the accuracy of the sensors with 58 healthy individuals and then conducted a pilot study with 20 people with PD who have mild or moderate swallowing issues. The ADAM sensors accurately detected swallowing, with a high accuracy score of 0.89 in tests with healthy subjects. In PD patients, the feedback feature of the device increased swallowing rate by 45%, from 0.77 to 1.10 swallows per minute. The device's detection results matched closely with expert observations, and both patients and caregivers found it comfortable and helpful. This wearable device may provide valuable support for people with PD by both monitoring and assisting swallowing. This solution has the potential to improve quality of life for those with swallowing difficulties in PD.
Keywords
Introduction
Parkinson's disease (PD) affects approximately 6.1 million individuals worldwide, with a concerning rise in both incidence and prevalence in the past two decades.1,2 The diagnosis of PD is clinical, with diagnostic criteria based on the presence of classic motor symptoms including bradykinesia, tremor, and rigidity. Beyond the classic motor symptoms, bulbar dysfunction is also common in PD, with profound negative health impact that has received substantially less attention in the literature.3,4 Up to 80% of patients with PD exhibit dysphagia, 5 which can lead to dehydration, malnutrition, and difficulty with oral medication intake, and aspiration pneumonia, a leading cause of death in patients with PD.6,7 Impairment in the physiological coordination of breathing and swallowing in patients with PD further exacerbates the risk of aspiration and aspiration pneumonia. Decreased swallowing frequency has been shown as a sensitive index of dysphagia.8–11 Previous studies have reported that patients with PD exhibit substantially lower swallow frequency compared to age-matched controls.12–15 The negative consequences of decreased swallowing frequency are two-fold. First, over 70% of patients with PD experience sialorrhea (drooling), which negatively impacts quality of life.14,16,17 Furthermore, decreased swallowing frequency reduces the usage of the swallowing musculature, potentially predisposing patients to muscle atrophy over time. As such, decreased swallow frequency emerges as a significant concern and potential target for intervention toward maintaining or improving swallowing function in a progressive neurologic disorder such as PD. 18
Currently, structured clinical exams such as the Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) 19 have become a de facto way for neurologists to guide diagnosis, assess the efficacy of new therapies, and track disease progression. Unfortunately, tools such as the MDS-UPDRS are labor intensive for both clinicians and patients, require trained personnel to perform, and lack the ability to continuously track patients in their naturalistic environment. 20 To address these limitations, there is a sizable body of work combining commercially available smartphones with a custom mobile software application and wrist-worn sensors using inertial motion units (IMUs) with reasonable correlation to specific domains of the MDS-UPDRS.21–26 However, this approach is largely limited to motion assessments captured for classic motor dysfunction such as gait, bradykinesia, and tremor without an ability to objectively capture bulbar dysfunction in patients with PD. Emerging work that goes beyond wrist-mounted wearables has shown that novel skin-mounted wearables placed on the upper thorax or neck region of a user can yield accurate and continuous assessments of swallowing.27–31 These devices provide critical insights into swallowing mechanics by capturing high-frequency signals associated with swallowing events.
This manuscript presents a comprehensive digital health technology (DHT) with the ability to capture both classic motor and bulbar symptoms in PD using a network of advanced acousto-mechanic sensors mounted on the hand and suprasternal notch. The mechano-acoustic sensor, or “ADAM” sensor, 32 uses a novel approach to monitoring bulbar dysfunction in Parkinson's disease. Unlike conventional wearables focused on gross motor function, the ADAM sensor is specifically designed to capture the subtle physiological signals associated with swallowing and respiration. Its multi-modal design integrates inertial measurement units (IMUs), including accelerometers, with acoustic vibration data to provide a more comprehensive assessment of bulbar function than traditional IMU-based motion tracking devices. This targeted approach addresses a critical gap in current wearable technology for monitoring PD-related bulbar dysfunction.
The ADAM sensor offers distinct advantages over other wearable technologies for monitoring swallowing. While surface electromyography (sEMG)32,33 measures muscle activity and pressure sensors detect force, the MA sensor provides a broader perspective on swallowing events, respiratory phases, and their coordination.
A key strength of the MA sensor lies in its capacity for ambulatory monitoring. This allows for data collection in natural, daily settings, providing a more ecologically valid assessment of swallowing behavior than traditional in-lab assessments. The resulting data, reflecting the complexities of real-world environments, is invaluable for developing individualized treatment plans and monitoring patient progress over time.
The ADAM sensor offers capabilities that extend beyond simple monitoring. An important feature of the device is its ability to provide real-time feedback, which is particularly beneficial for therapeutic interventions. Unlike many other wearable sensors that primarily collect data for later analysis, the MA sensor delivers artificial intelligence (AI)-driven haptic cues in real-time. The ADAM sensors have a high-frequency IMU for sensing and an embedded vibratory motor that enables haptic biofeedback to be delivered when a pre-specified threshold of swallow frequency is not reached.
New validation data in n = 20 PD patients builds upon our previous work leveraging skin-mounted sensors with haptic biofeedback capabilities in related applications such as reducing vocal tract straining 34 and pilot studies of dysphagia in chronic obstructive pulmonary disease, head and neck cancer, and PD. 35 In addition, user feedback from n = 10 PD patients and their respective caregivers was collected via structured focus groups to ensure user acceptability. This work builds on our previous studies by introducing a novel digital health solution that not only evaluates motor dysfunction but also comprehensively monitors bulbar dysfunction in PD. Unlike earlier studies, this research integrates AI-driven haptic biofeedback to enhance swallowing frequency, providing a unique therapeutic approach. Overall, this DHT has the potential to detect small but meaningful changes, defined as statistically significant variations in swallowing frequency, motor function, and bulbar symptoms that correlate with clinically relevant improvements in patient outcomes, such as reduced aspiration risk and improved quality of life. The DHT can track a broad range of symptoms, continuously monitor the benefit of new therapeutic drugs, devices, and behavioral interventions in the setting of clinical trials, and serve as a non-invasive assistive technology for improving swallowing problems in patients with PD.
Methods
Advanced acousto-mechanic sensor (ADAM) development
Figure 1(a) shows the key components of the ADAM sensor encapsulated with waterproof, medical-grade silicone enabling gentle attachment to the curved skin on the dorsal hand 36 (Figure 1(a)). The key hardware innovation in the ADAM sensor is the eccentric rotating mass (ERM) motor, which enables haptic biofeedback. The motor, preferentially selected for its small size (8.0 mm in diameter) and common application in small mobile and portable devices, rotates at 10,000 rotations per minute (RPM) generating 1.4 G of vibratory feedback, sufficient to alert patients, even during sleep. 37 The ERM motor is powered via a switched voltage regulator controlled with a General Purpose Input/Output (GPIO) pin from the microcontroller unit (MCU) on the ADAM sensor. The MCU uses pulse-width modulation (PWM) output on the GPIO pin to create variable strength and duration of feedback. PWM utilizes pulses of varying widths to create a duty cycle which modulates the rotation speed of the motor. Using this configuration, many complex haptic profiles can be created including gentle or intense vibrations, ramping effects, and short bursts. Prior publications describe the detailed electronics and encapsulation of the ADAM sensor.27,37
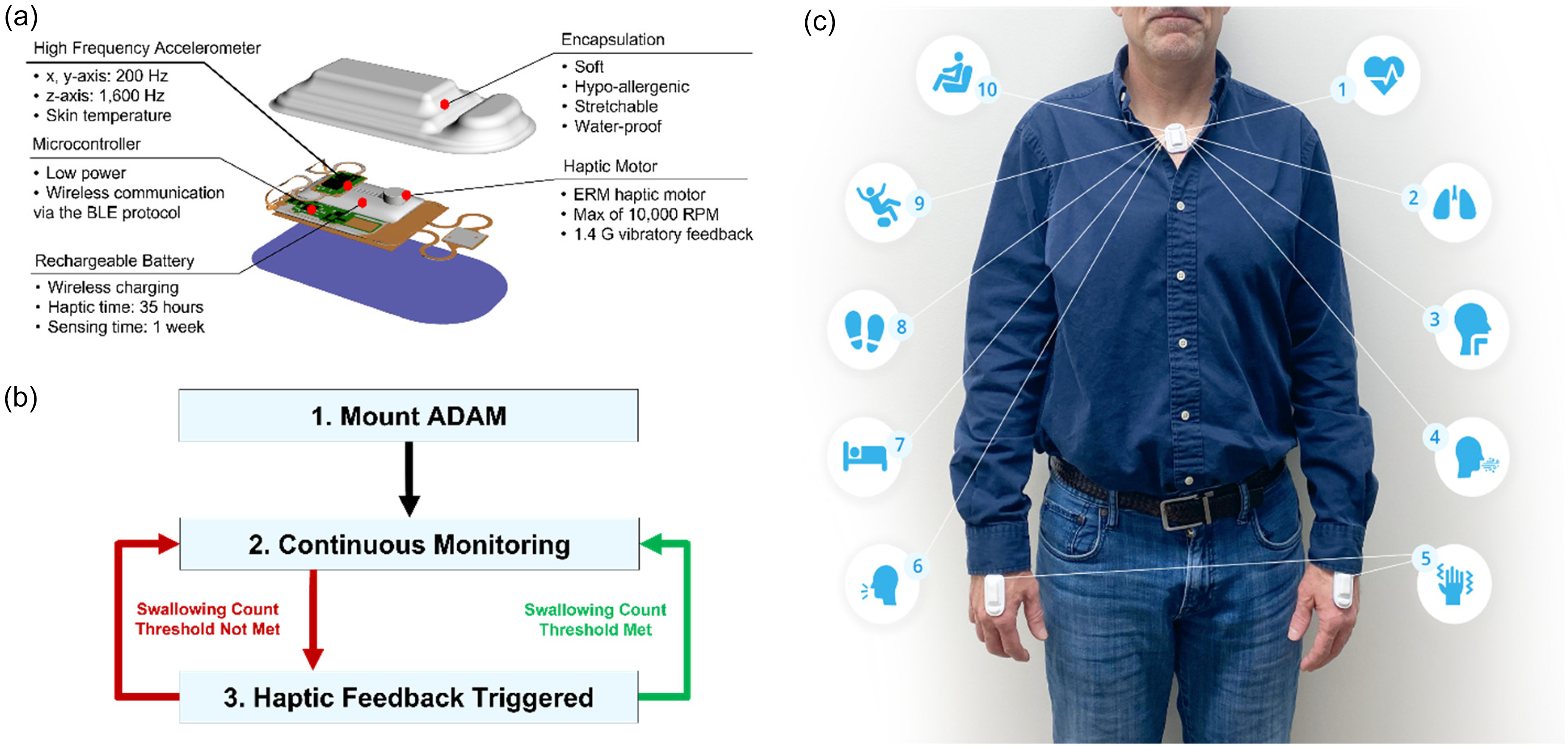
(a) Exploded view of the ADAM sensing and haptic technology. The electronics include a microcontroller, wirelessly rechargeable battery, accelerometer, and haptic motor. These components are contained within a soft, hypoallergenic, stretchable, and waterproof encapsulation. (b) Mechanism of action of the ADAM device: the device is worn by the patient and continuously monitors his/her swallowing events over time. When a pre-configured threshold of swallowing events per hour is not met, the device sends haptic cues via the motor to signal the patient to swallow, thus increasing his/her swallowing frequency. (c) Three ADAM sensors positioned on the suprasternal notch, left hand and right hand to capture (1) heart rate, (2) respiratory rate, (3) swallow count, (4) cough count, (5) tremor, (6) talk time, (7) sleep, (8) step count, (9) fall count, and (10) body position via the high frequency 3-axis accelerometer. (d) Time frequency plot of the Fourier results and the band-passed filtered signal demonstrating what processed data looks like.
Eligibility criteria for clinical studies
Participants were included in the study if they had PD of any severity, were at least 21 years of age, and presented with mild to moderate oropharyngeal dysphagia as determined by an SLP in a bedside evaluation. Healthy subjects were selected based on the following criteria: age between 18–65, no history of dysphagia or other swallowing disorders, and no neurological conditions. A total of 58 subjects participated, with a balanced representation of genders and age groups. Participants were excluded if they could not safely swallow at least one bolus of liquid without risk of aspiration or they experienced end-stage dementia.
Patient recruitment
The clinical research protocol was reviewed and approved by the Northwestern University Institutional Review Board (STU: STU00212981) and registered in ClinicalTrials.gov (06/12/2020, NCT04664634). Subjects were recruited from Northwestern Memorial Hospital. A subject search was conducted using Northwestern's Enterprise Database Warehouse (EDW), an integrated database system that includes 2.9 million patients searchable by ICD9/ICD10 code. For instance, we can search for all subjects in the Northwestern system with an associated G20 19 AND R13.10 (dysphagia). This generates a prospective patient population to recruit. In addition, authors and collaborators of this study actively work with PD patient rehabilitation.
Ethics statement
All methods were carried out in accordance with relevant guidelines and regulations. The study protocol was reviewed and approved by the Northwestern University Institutional Review Board (STU: STU00212981), and all participants provided written informed consent prior to participation. For participants with Parkinson's Disease, informed consent was obtained from both the patients and/or their legal guardians where applicable.
Clinical study protocol
In-clinic evaluations were conducted by certified SLPs together with a clinical research assistant. First, the SLP conducted a clinical swallowing assessment for evidence of oropharyngeal dysphagia to determine eligibility. In this standardized assessment, the SLP asked the participants questions related to history of swallowing problems, troublesome foods, and the frequency, severity, and onset of symptoms. The SLP examined the appearance and function of the patient's teeth, lips, jaws, tongue, cheeks, and soft palate. A series of food substances ranging from water to thicker liquids, pureed foods, soft foods, and regular foods were then given to the participants to determine functional oral intake. Additionally, patients were asked to complete the EAT-10 questionnaire, a dysphagia specific quality of life patient reported outcome, to establish a baseline of how dysphagia affects their quality of life.
Patients were seated in a comfortable chair and the ADAM sensor was placed on the suprasternal notch via a disposable adhesive to record breathing and swallowing movements. Respiratory movements of the rib cage (RC) and abdomen (AB) were simultaneously recorded using respiratory inductance plethysmography (RIP) (Inductotrace System; Ambulatory Monitoring, Inc., Ardsley, New York) coupled to a data acquisition system (BIOPAC Systems, Inc., Santa Barbara, CA, Model MP150). 6 The RC band was placed around the rib cage at the midsternal level, and the AB band was placed around the abdomen just below the lowest rib. In addition, a nasal cannula was placed to record nasal airflow.
Baseline data were collected to assess the monitoring performance of the ADAM sensor and swallow algorithm. To this end, patients were asked to breathe quietly and swallow spontaneously for 15 min while the ADAM device was affixed to the suprasternal notch. Following breathing tasks, patients were asked to consume an average of 12 small amounts of liquids and pudding, with consistencies deemed safe by the clinical swallow evaluation. The ADAM device monitoring mode was used to record breathing and swallowing movements. After the study, this respiratory and swallowing data were reviewed and labeled by three expert SLPs (with consensus scoring) to mark respiratory phase (i.e., inspiration and expiration) and swallowing events to compare against ADAM swallowing and breathing cycle motion capture mode. The gold standard for evaluation was the consensus labels from three expert SLPs. RIP data was used to cross-validate the respiratory phase and swallowing events detected by the ADAM sensor. Discrepancies in labeling were resolved through discussion and re-evaluation until consensus was achieved.
Then, the patients were randomly assigned into one of two arms in the interventional phase of the study: the experimental condition arm, where haptic biofeedback was delivered in a time interval randomized every 20–40 s; the control condition arm, where the ADAM sensor was set to monitoring mode only without haptic biofeedback delivery. Participants were observed for 15 min before crossing over to the alternate condition for an additional 15 min. Based on the current arm of the study, the clinical researcher activated or deactivated the haptic biofeedback via the mobile application. Participants were blinded to study outcomes during the investigation for the duration of the study. The ADAM device was removed from the patients’ suprasternal notch at the conclusion of the session. Photographs of the neck area were taken immediately after removal and 5 min after removal. These photographs were graded by a board-certified dermatologist on the Likert scale (none, mild, moderate, severe) for skin redness, ulceration, and dermatitis.
PD focus groups
A total of n = 10 patients with PD (mean age 69 years) and n = 2 PD caregivers were asked to participate in a structured focus group facilitated by a trained moderator. In a semi-structured interview using a standardized coding schema, the moderator guided patients on the operation of the device, potential interference with daily activities, and preferred methods for training. Participants were asked their preferences for a wearable sensor including physical form factors, software operation, charging/battery life, and desired data output for themselves and caregivers. In addition, the moderator asked participants about their perceptions on the benefits and drawbacks of the DHT. All interviews were transcribed and subsequently categorized according to the coding schema.
Swallow algorithm development
Data pre-processing
The modeling used the z-axis data from accelerometers, sampled at 1666 Hz. To preprocess this data, we first removed baseline values by subtracting a five-second moving average from the signal. Following this, we computed the spectrogram of the baseline-corrected signal. This was done using a two-second window with a 0.5-s step size, applying a Hanning window before performing the Fourier transform on each segment. The absolute value of the logarithm of the Fourier transform was then taken, yielding a 1666-dimension vector of spectral features for each window. Each of these vectors underwent z-score normalization, resulting in a vector with zero mean and a variance of 1.
Separately, we also processed the baseline-removed signal with a 10th order Butterworth bandpass filter, setting the lower and upper cutoff frequencies at 84 Hz and 512 Hz, respectively. The bandpass-filtered signal was then segmented resulting in windows of 3332 samples. Each of these windows was also z-score normalized. The bandpass filter parameters were based on exploratory work with the sensor. The lower cutoff threshold of 84 Hz was found to remove larger body motion well, while maintaining the signal within the frequency range of swallows.
We combined 1666 frequency domain features from the spectrogram with 3332 time domain samples from the band-pass filtered signal for each two-second window, resulting in a total feature vector of 4998 samples. This process resulted in a comprehensive feature vector of length 4998 for each window, encompassing both spectral and time-domain characteristics of the accelerometer data.
Model architecture
We utilized a convolutional neural network (CNN) model. 38 Two sequential steps of 1D convolution (each with an output dimension of 32 filters) followed by batch normalization were used to extract features. The first 1D convolutional layer has a kernel size of 5, while the subsequent layer has a kernel size of 13. Batch normalization follows each convolutional layer to stabilize learning. 39 The network then flattens the output for processing through two dense layers, the first with 32 neurons using ReLU activation and the second being the output layer with a single neuron and sigmoid activation. We trained the model using Adagrad 40 with a learning rate of 0.0015, and binary cross-entropy as the loss function. The model is trained to classify swallowing/not swallowing, outputting the probability of swallowing for each segment of accelerometer data. A CNN network was selected over other machine learning models (such as a random forest) for the CNN’s greater flexibility in learning relevant features, and the lower implementation complexity within an embedded environment. The model was developed using Tensorflow in Python, with initial signal processing steps performed using Scipy. Due to patient privacy, the model code and data are not publicly available. See Supplemental Figure 1.
Model training
The model training was conducted using a leave-one-subject-out cross-validation (LOSO-CV) approach with the n = 58 healthy subjects. The model was only trained on healthy subjects, and PD patient data was evaluated on a frozen model. In each iteration, data from one subject was reserved for test time evaluation, while the remaining subjects’ data were split for training and validation purposes: 80% for training and 20% for validation. The training was executed over 20 epochs, employing a batch size of 64 samples. After completing these 20 epochs in each iteration, we assessed the model's performance on the test subject that was initially held out. We define an epoch as one full pass through the entire training dataset. The full pass was repeated 20 times for each leave one out cross validation fold. To address the imbalance in the training data, we adjusted the class weights to ensure equal representation of both swallow and non-swallow classes. The accurate performance of the algorithm was demonstrated in this preliminary assessment, with the model achieving an F1 score of 0.89, a sensitivity of 95%, and a specificity of 99%.
Edge computing
Edge computing allows computational analysis to occur closer to the original data source, saving bandwidth, improving response times, and eliminating additional mobile devices and computational resources. In this work, we bridge the gap between data collection and offline analysis by enabling real-time scratch detection on the ADAM device itself through edge AI using the sensor's onboard low-power microcontroller. The original convolutional neural network (CNN) model processed data by frames. To replicate frames on the ADAM sensor, a fixed size queue of 512 data points (307 milliseconds with 1666 Hz sampling rate) was defined. As new data was generated, the queue was filled. Once the queue was full, the data contained in the queue was passed to the signal processing block where a short-time Fourier transform (STFT) was performed. Then, the STFT, along with the corresponding time domain signal, was passed to the CNN model. The model was replicated on the ADAM sensor by inputting the weights of the optimized offline CNN model. For every new incoming data point, the oldest data point was removed, and the new data point was added to the queue via a first in, first out (FIFO) scheme. This enables the sensor to operate in a fully closed-loop fashion for swallow events without the need for an additional mobile device.
Rational selection of haptic biofeedback profiles
In order to select the most appropriate haptic biofeedback profile, we recruited n = 20 healthy non dysphagic adults to evaluate 19 distinct vibratory profiles (Supplemental Figure 2). These profiles provided variations in strength, length, and ramp. Volunteers were asked to describe their response to each of these profiles with a set of standardized terms (e.g., favorable, unfavorable, gentle, pleasant, annoyed). Based on the responses, profile 4 characterized by a soft buzz was selected due to its pleasant nature while also captivating the wearer. While initial haptic profile selection was based on feedback from healthy non-dysphagic adults, future studies will prioritize feedback from PD patients to ensure the profiles are optimized for the target population.
Real world applicability
The approach we take with the swallow model is that individual baselining is not necessary; the output of the swallow model is causal and only depends on the input data over the receptive window of the neural network. We tested the real world applicability and generalizability on a Parkinson's disease population, completely separate from the healthy normal training population dataset. Further therapeutic applications of this technology is outside the scope of this work, but is currently a topic under active investigation.
Results
Skin-integrated wireless device for monitoring motor and bulbar function in patients with PD
The DHT reported here leverages either a single or network of time-synchronized high-frequency ADAM sensors that can be mounted in areas of anatomical importance. Figure 1(a) is an exploded view of the ADAM sensor with vibratory biofeedback functionality. The core sensing element is a high-frequency accelerometer (1600 Hz) that captures both gross limb and trunk motion and acoustic range signals relevant for assessing complex physiological processes like swallowing and speech. An embedded vibratory motor enables haptic biofeedback when triggered. The onboard microcontroller unit runs all necessary algorithm processing. In a pure sensing mode, a single ADAM sensor offers one week of operating time. With haptic biofeedback activated, the ADAM sensor provides 35 h of operating time. The sensor is rechargeable and communicates with a wide range of mobile devices via BLE. The charging time of the ADAM device is 3 h while placed on the wireless charger included. Figure 1(b) describes the operating logic of the vibratory biofeedback function of the sensor. The ADAM sensor is programmed to trigger haptic biofeedback when a threshold of swallows per unit of time is not reached. The biofeedback process involves an AI-driven algorithm that detects swallowing frequency in real-time. When the swallow frequency falls below a pre-set threshold, the device triggers haptic biofeedback to prompt the user to swallow. This closed-loop system uses edge computing to perform real-time analysis and biofeedback without requiring external devices. The threshold for triggering haptic biofeedback is defined based on clinical guidelines for normal swallowing frequency and empirical data collected during initial trials. In this work, real-time swallow detection via an AI-algorithm is performed on the microcontroller of the ADAM device itself via edge computing. This reduces response time and mitigates the need for additional mobile devices for operation. The AI algorithm was trained on data collected during both swallowing and various non-swallowing activities, such as finger tapping and hand movements. This comprehensive training ensures that the sensor can accurately differentiate between swallowing events and other movements, enhancing its real-life applicability. In Figure 1(c), three ADAM sensors are mounted (one on each hand and one on the suprasternal notch) to capture the full spectrum of motor symptoms in PD. In this configuration, the suprasternal mounted ADAM sensor can capture core vital signs such as heart rate, respiratory rate, respiratory cycling, swallowing, speech, sleep parameters, step count, and core body position including fall count. 28 The upper extremity mounted sensors offer additional capabilities to capture tremor, bradykinesia, gait, and fine motor movements of both limbs.
In an illustrative subject, we show the raw data outputs of each ADAM sensor mounted on either the suprasternal notch or upper extremities for a wide range of physiological processes relevant to PD and the MDS-UPDRS. In Figure 2(a), we show the raw data of the sensor and demonstrate the sensor's ability to capture swallowing events across different body postures and activities, including walking, sitting, and lying, ensuring comprehensive monitoring in real-life scenarios. In Figure 2(b), we show the raw data of the suprasternal-mounted ADAM sensor capturing bulbar functions such as speech, coughing, and swallowing differently compared to the upper extremity mounted ADAM sensors. This further leverages the high-frequency signal generated by the ADAM sensor (1600 Hz) that is not captured by traditional wrist-mounted actigraphy systems. 28 In Figure 2(c), the ADAM sensor mounted on the suprasternal notch further captures core vital signs such as respiratory rate, respiratory phase patterning (i.e., inspiration or expiration), and heart rate. Data were collected over a period of 15 min per subject. Each subject's data included approximately 12 swallow events. During non-swallowing moments, subjects engaged in activities such as talking, resting, and light physical activity to provide a diverse set of data for the model. Depending on the use case, either a single ADAM sensor or 3 or more can be deployed. In Figure 2(d), the time frequency plot of the Fourier results and the band-passed filtered signal are shown demonstrating what processed data looks like.
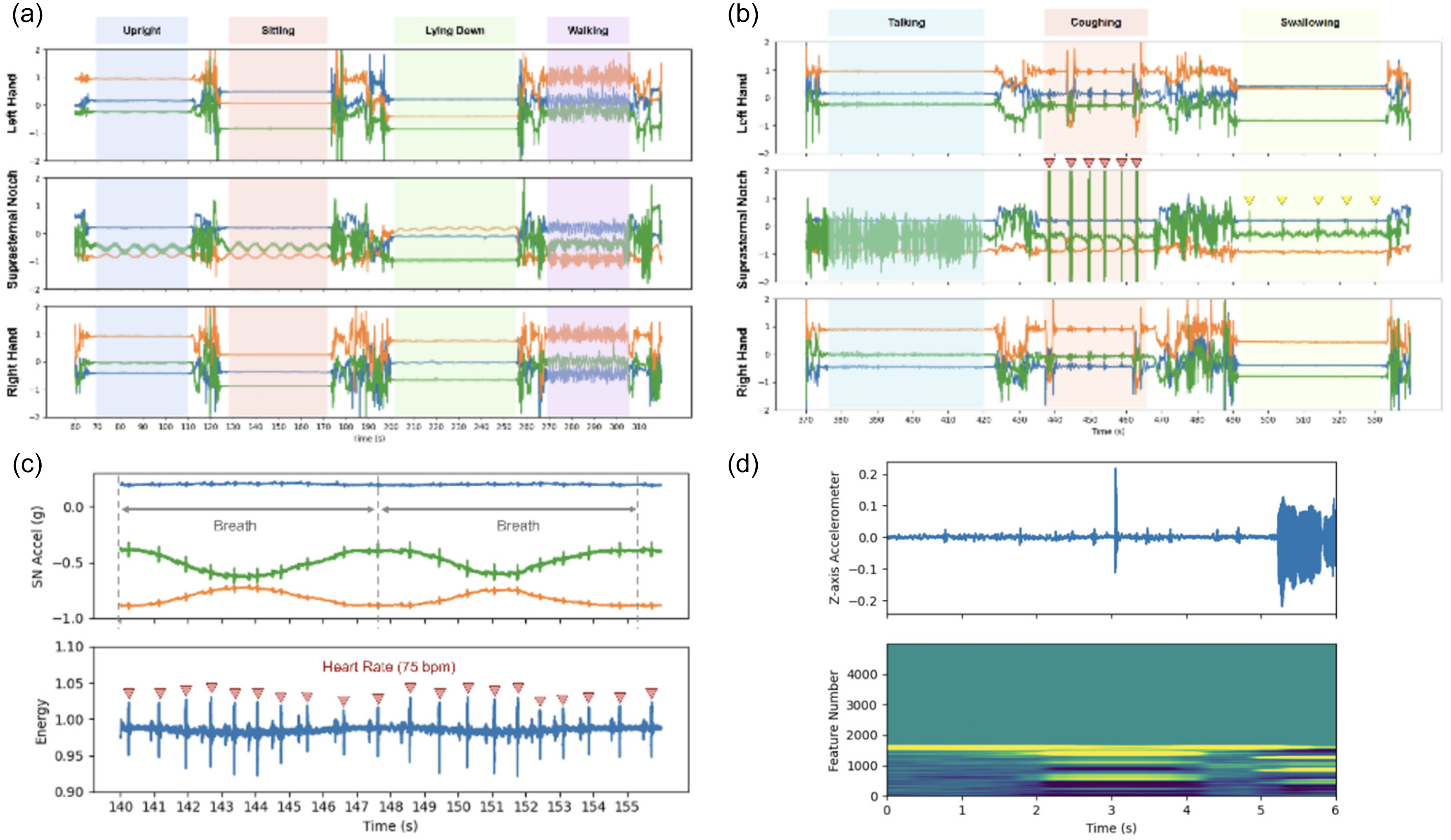
(a) Macromotion data. High-frequency 3-axis accelerometer signals in gravity units (g) from each of the ADAM sensors positioned in the left hand, right hand (RH), and suprasternal notch for the following positions and activities: standing upright (blue segment), sitting (orange segment), lying down (green segment), and walking (purple segment). In the hands (RH and LH), the color of each accelerometer axis corresponds to: palmar-dorsal vector (green), proximal-distal vector (orange), and medial-lateral vector (blue). In the suprasternal notch (SN), the color of each accelerometer axis corresponds to: frontal plane normal vector (green), transverse plane normal vector (orange), sagittal plane normal vector (blue). (b) Bulbar function data. High-frequency 3-axis accelerometer signals in gravity units (g) from each of the ADAM sensors positioned in the left hand, right hand (RH), and suprasternal notch for the following bulbar functions: talking (blue segment), coughing (orange segment), and swallowing (yellow segment). Coughing events are marked by orange triangles and swallowing events are marked by yellow triangles. (c) Cardiopulmonary data. High-frequency 3-axis accelerometer signals in gravity units (g) from an ADAM sensor positioned in the suprasternal notch can be used to capture the subject's respiratory rate and heart rate vital signs. The top graph shows raw accelerometer data from each of the three axes: frontal plane normal vector (green), transverse plane normal vector (orange), sagittal plane normal vector (blue). The movements from exhalation and inhalation captured by the sensor are used to measure the breathing cycle. The bottom graph combines data from the three axes to display movement associated with each heartbeat. Identification of heart beats is then used to determine the subject's heart rate (seismocardiogram).
Figure 3 showcases the predictive values over time for one subject compared to the true values.
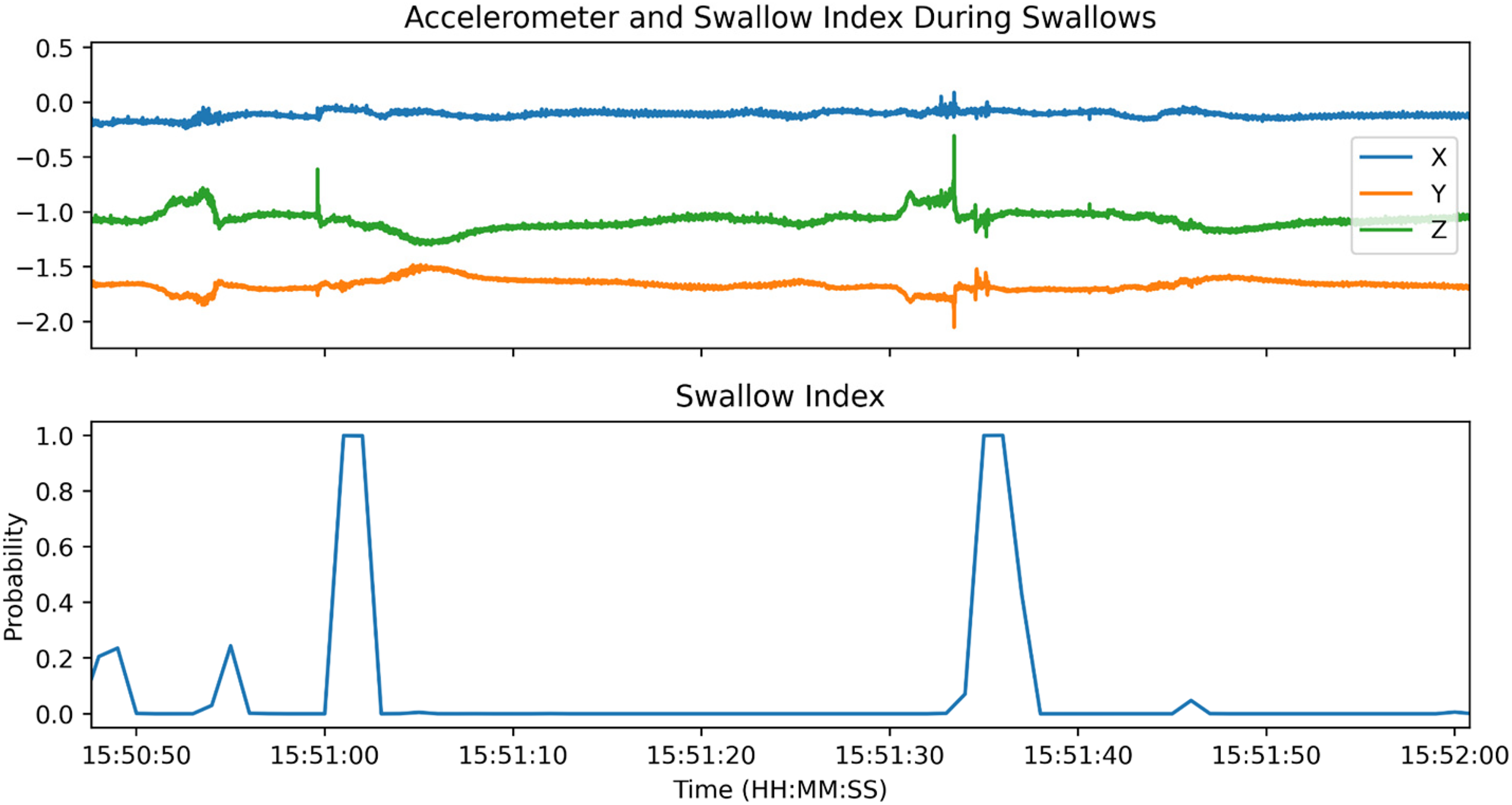
A case example showcasing the performance and predictive values over time compared to the true values. This dynamic representation helps interpret the model's performance over time. The accelerometry data can be seen at the top with the corresponding swallow index at the bottom in real time.
Figure 4 illustrates the sensor's ability to capture motor functions as defined by the UPDRS. This provides a comprehensive assessment of the sensor's capabilities, ensuring that it can accurately monitor both motor and bulbar symptoms in patients with Parkinson's disease. Specifically, we show in Figure 4(a) the ability to capture finger tapping, hand opening, and hand pronation-supination with either upper extremity. The hand to nose motion and tremor can be captured in both extremities with outputs from each ADAM sensor. In Figure 4(b), we show opening and closing of the fist for both the right and left hand. In Figure 4(c), finger tapping for each hand is shown. Finally, in Figure 4(d), we show a zoomed in view of pronation-supination for each hand. In all these upper extremity activities, the suprasternal notch-mounted ADAM sensor captures heart beats, breaths, and vocalizations continuously when the subject is at rest.
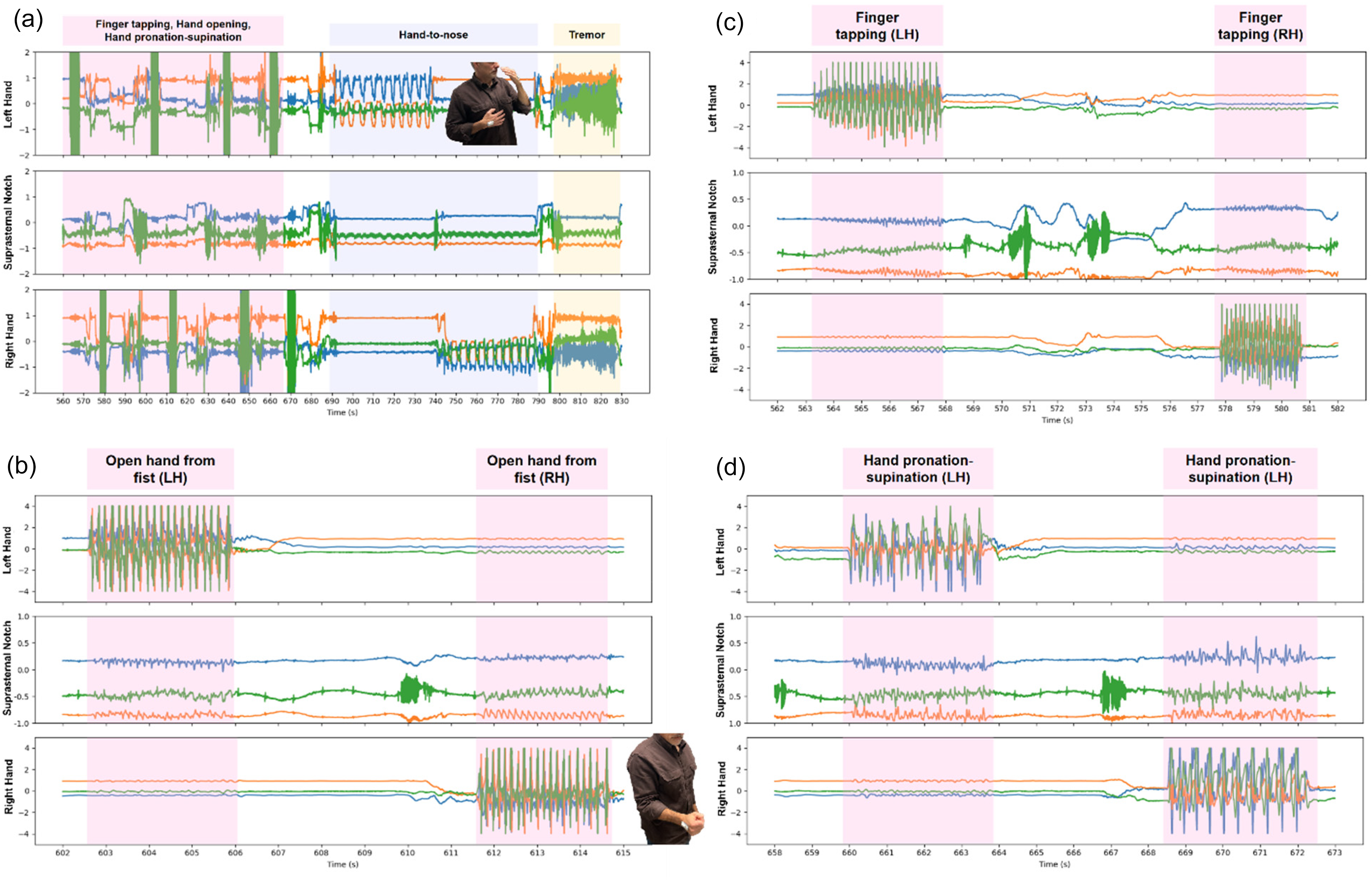
(a) High-frequency 3-axis accelerometer signals in gravity units (g) from each of the ADAM sensors positioned in the left hand, right hand (RH), and suprasternal notch for the following Parkinson's-related diagnostic motions and maneuvers: finger tapping, hand opening, hand pronation-supination (all in pink segment), hand-to-nose (blue segment), and hand shaking/tremor (yellow segment). In the hands (RH and LH), the color of each accelerometer axis corresponds to: palmar-dorsal vector (green), proximal-distal vector (orange), and medial-lateral vector (blue). In the suprasternal notch (SN), the color of each accelerometer axis corresponds to: frontal plane normal vector (green), transverse plane normal vector (orange), sagittal plane normal vector (blue). (b) Expanded time axis view of the Movement Disorders Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) finger tapping test segment for the left40 and right (RH) hands. Fast, individual taps from the index finger to the thumb can be discerned clearly in both hands. (c) Expanded time axis view of the MDS-UPDRS fist opening test segment for the left and right (RH) hands. The subject's fast opening of the fist can be measured accurately in both hands. (d) Expanded time axis view of the MDS-UPDRS hand pronation-supination test segment for the left and right (RH) hands. Fast changes in hand orientation can be measured accurately in both hands.
To train and develop an AI algorithm for swallow events, a total of n = 58 healthy normal subjects were recruited (Table 1). Healthy subjects were included to establish a baseline for normal swallowing patterns and to ensure the AI algorithm could accurately detect swallow events in a controlled environment. This baseline data is crucial for developing a robust model before applying it to the PD population. In this group, a structured protocol of swallowing events of different foods and material consistencies, and confounders (e.g., coughing, motion, tapping the sensor) was conducted. Model training was conducted using a leave-one-subject-out cross-validation (LOSO-CV). The use of out of sample validation is critical for the success of any machine learning model; to that end we performed the leave one subject out cross validation during the training process on healthy normal volunteers. Additionally, we validated the model's performance on a Parkinson's disease population with frozen model parameters. While the test protocol was similar, the patient population was quite different from the original training population, thereby demonstrating the generalizability of the deep learning model. The performance reported for the PD population used the same swallow detection model that was trained using LOCO-CV for the 58 healthy subjects. Overall, this yielded a global sensitivity of 95% and specificity of 99% (F1 score: 0.89) for swallow event detection. Supplemental Figure 1 describes the final model architecture. For the classification results, true positives were defined as a high probability (>50%) swallow model estimate occurring within 10 s of the true swallow event. A true negative is defined as when the swallow model outputs all low swallow probability (<50%) for non-overlapping 10 s windows, during which no swallow event occurred. We have included additional details on the predictive performance of our model, including a confusion matrix (Table 2) and per-subject analysis to better interpret the sensitivity, specificity, and predictive values. The confusion matrices (Figure 5) provide a detailed view of the model's performance in distinguishing between swallowing and non-swallowing events, and the per-subject analysis highlights individual variability in model performance.
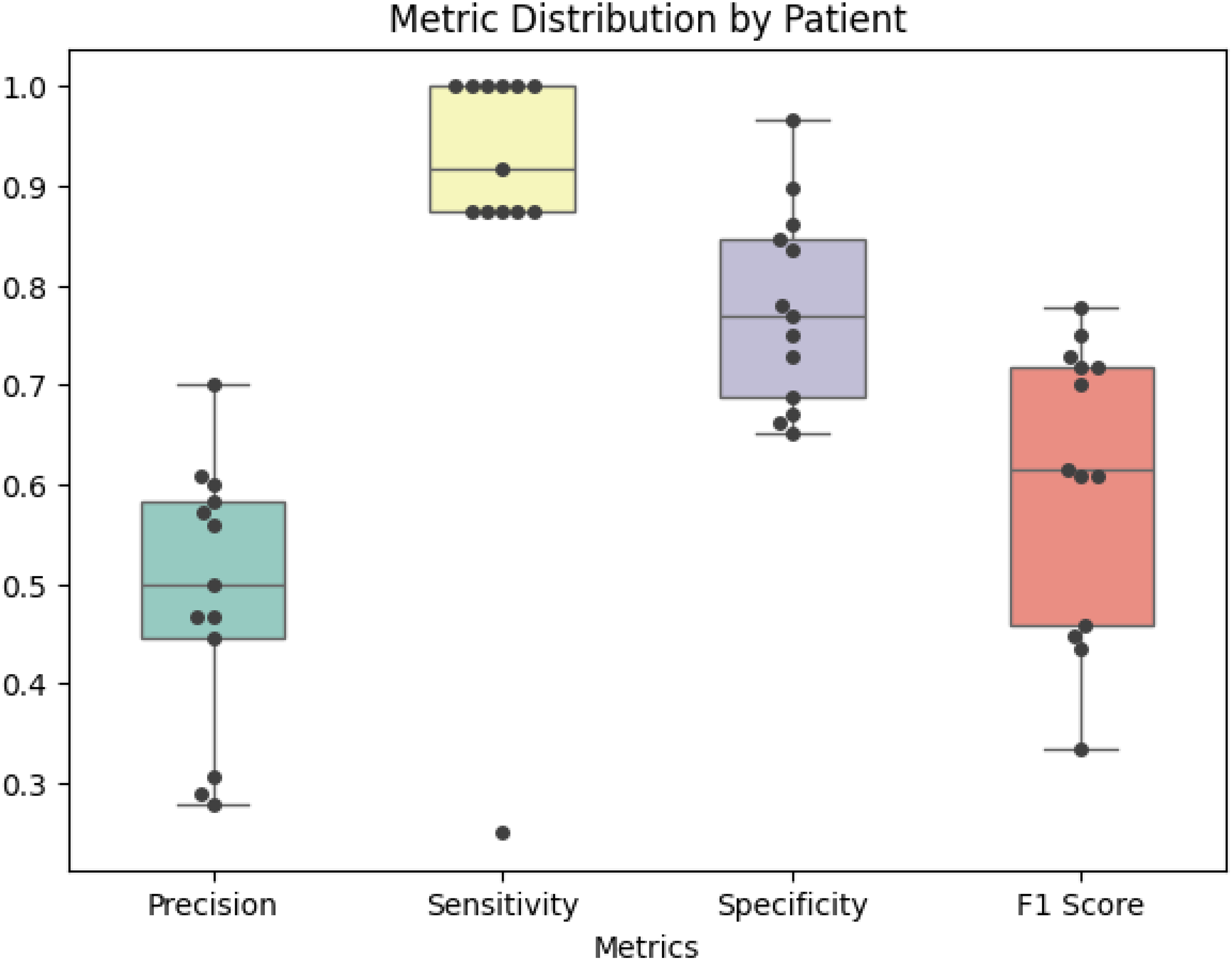
The chart shows classification metrics for events by patient, where each dot represents one patient's total performance.
Performance of AI algorithm for swallow detection. This table summarizes the overall performance metrics of the swallow detection algorithm of the ADAM device when classifying saliva swallowing and swallowing of various liquid types and delivery routes.
Overall performance of classifier prediction vs. actual value shown in a confusion matrix for PD patients.

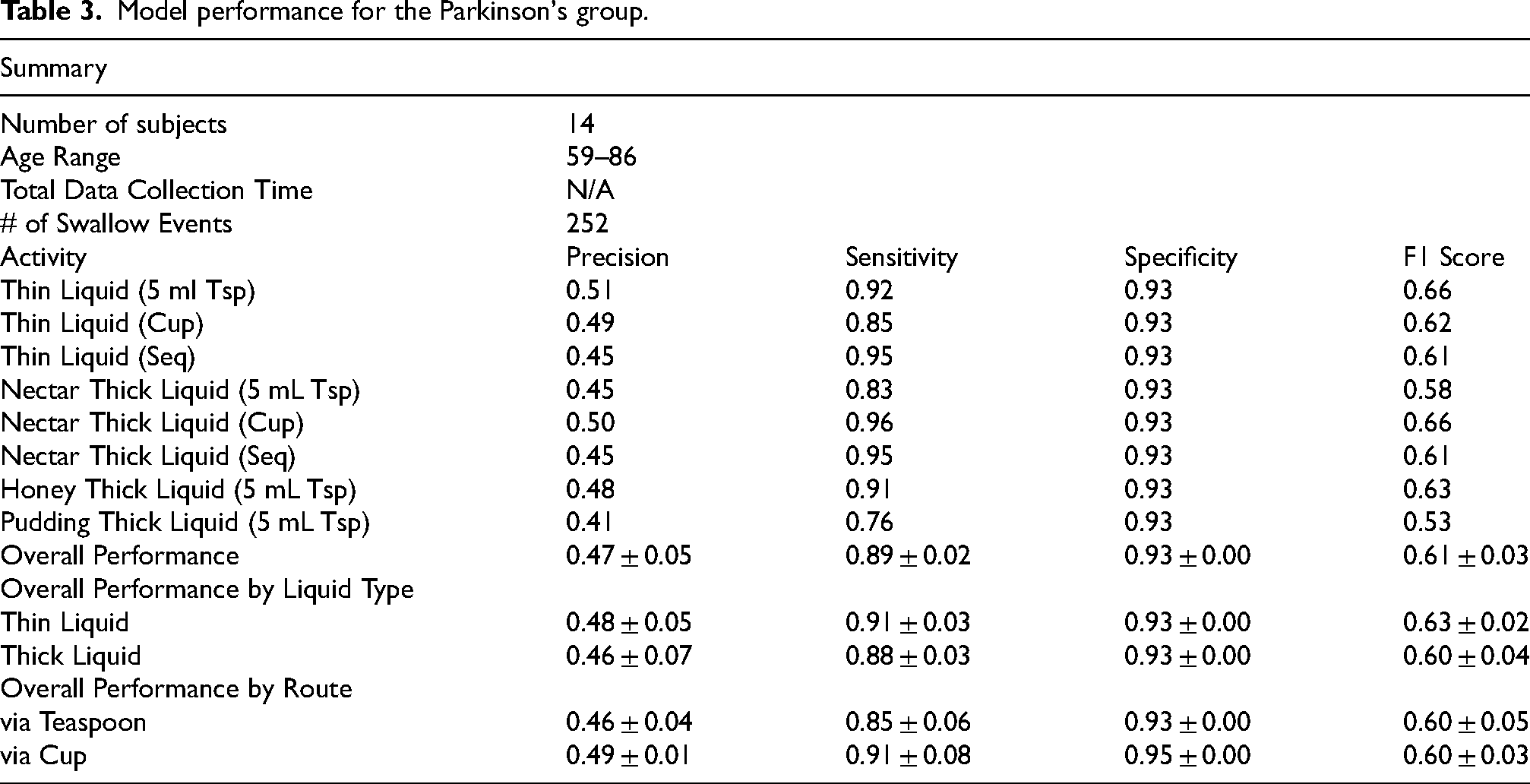
The model performance for the Parkinson's group (Table 3) demonstrates a high sensitivity for swallows labeled by three expert SLPs. However, the positive predictive value is lower than what is expected given the performance on the healthy normal group. There are several possibilities for this discrepancy. The first is that swallows outside of the protocol (including natural swallows) were not annotated, thereby penalizing the model performance as false positives when true swallows may have occurred. Additionally, we expect that higher levels of movement in the PD subjects relative to the healthy normal subjects may have increased the false positive rate as well. While this model performs well for the detection of swallows, we expect that further false positive reduction can be achieved by enhanced signal streams (such as audio signals) in the future.
Model performance for the Parkinson's group.
Clinical studies with patients with PD
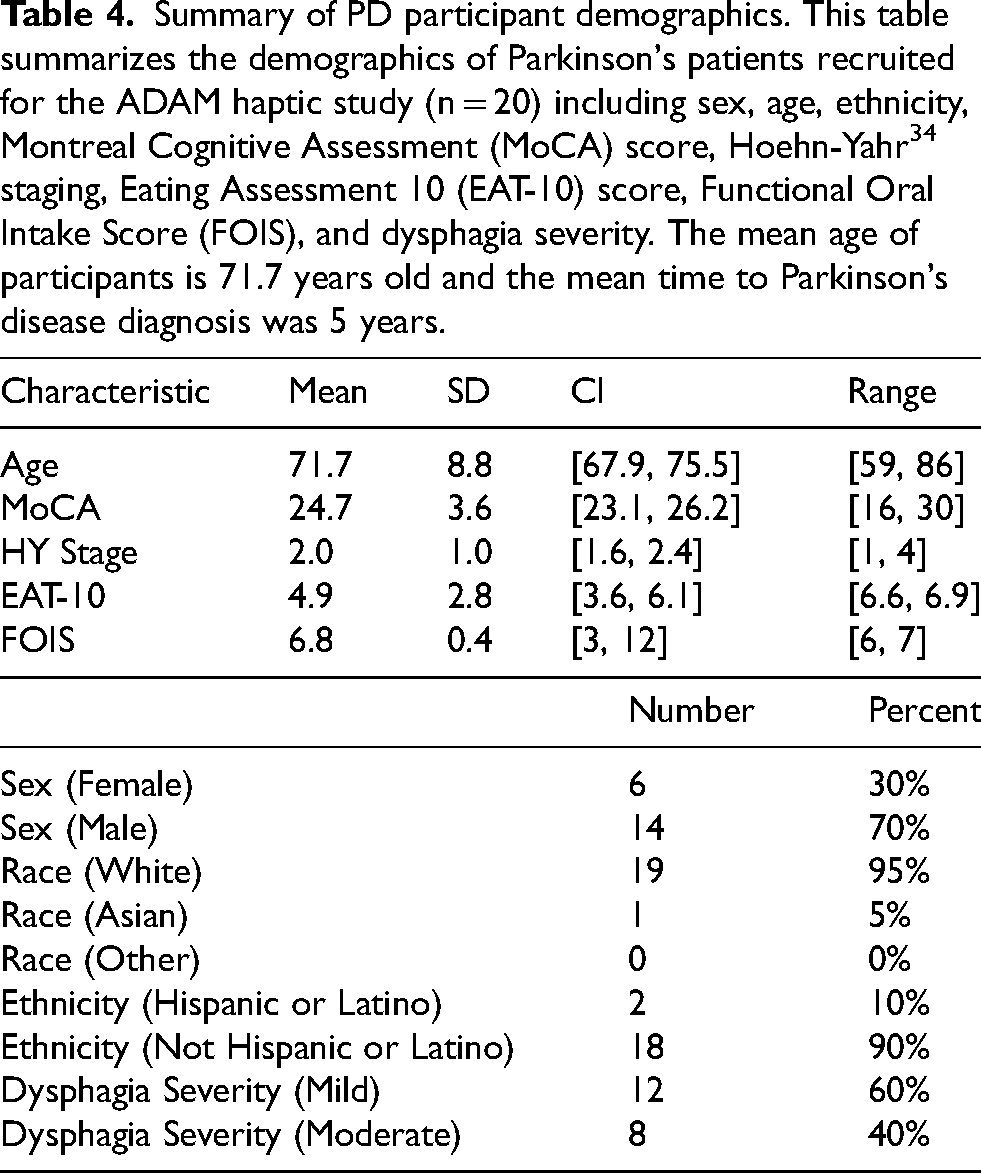
To evaluate the performance of the DHT in the context of swallowing and respiratory events in patients with PD, we mounted the ADAM sensor on the suprasternal notch to compare against the reference standard monitoring systems41,42 (e.g., respiratory inductance plethysmography and a pressure-transducer linked nasal cannula) (Figure 6(a)—(c)). A certified speech language pathologist (SLP) visually observed and labeled distinct swallowing events. In a study of n = 20 patients with PD, all with mild to moderate dysphagia (mean age 72; mean HY stage 2) (Table 4), the sensitivity of swallow detection with ADAM was 89% (r = 0.92, p < 0.05) compared to expert visual labeling by a certified SLP. PD subjects performed similar protocols and tasks as the healthy subjects, including swallowing different consistencies of liquids and pudding. This consistency ensures the AI algorithm is trained on comparable data sets, allowing for accurate detection and analysis of swallow events across both groups. Furthermore, respiratory phase identification in this dataset was 100% (K-weighted = 1.00) when comparing ADAM motion breathing cycle signals against the reference standard. With activation of haptic biofeedback (i.e., soft buzz), subjects exhibited a 45% mean increase in swallow events per minute (p < 0.0001) (Figure 7) with all subjects exhibiting an increase in swallowing frequency with haptic biofeedback. After 5 min of sensor removal, no observable skin adverse events such as redness, irritation, erosions, or pain were observed. Patients reported a high degree of satisfaction with the haptic biofeedback in terms of strength and sensation. Supplemental Video 1 demonstrates the operation of the ADAM sensor mounted on the suprasternal notch for heart rate, respiratory phase, and swallow events.
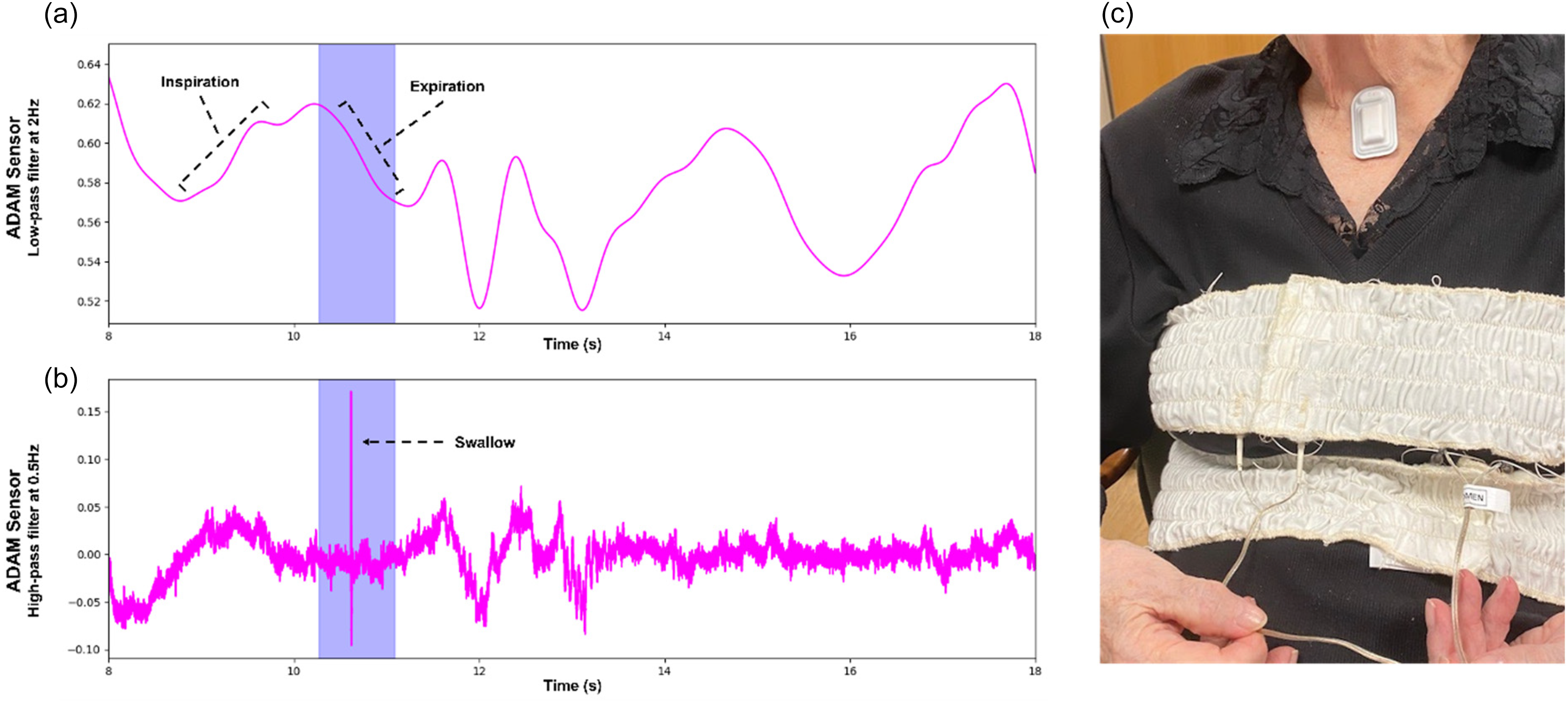
Panel (a) presents respiratory rate output from a patient with Parkinson's disease detected by the ADAM sensor. The respiratory signal is obtained by applying a 5th-order Butterworth low-pass filter with 2 Hz cutoff frequency. This signal is overlaid with Panel (b) demonstrating swallow initiation during expiration. The swallowing signal is obtained by applying a 5th-order Butterworth high-pass filter with 0.5 Hz cutoff frequency. In our study, patients with PD were found to initiate swallowing during inspiration 30% of the time and occurred at higher lung volumes compared to healthy normal adults. Panel (c) shows a Parkinson's patient wearing the ADAM device on the suprasternal notch and two reference respiratory measurement bands in the chest and abdomen.
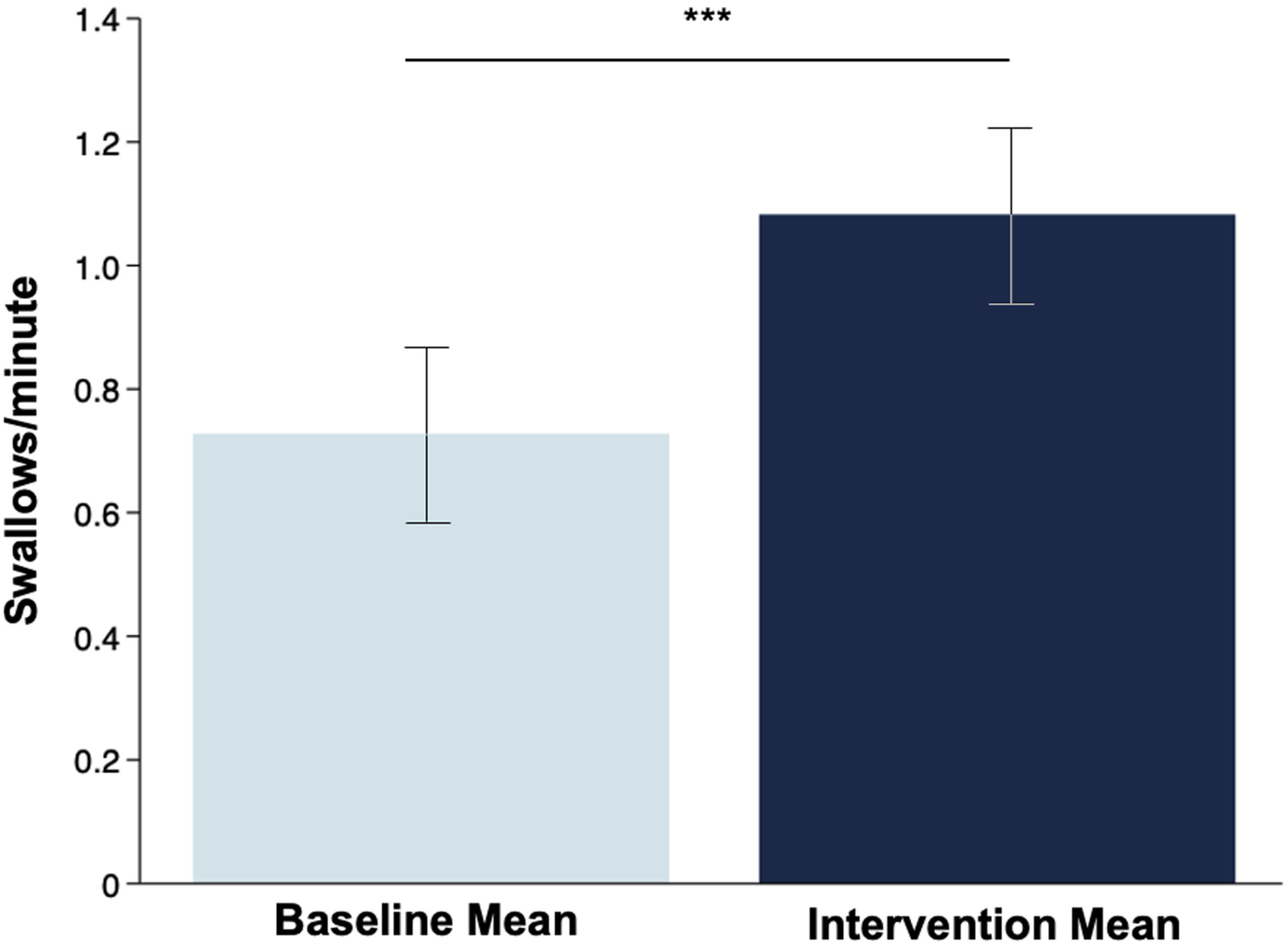
Comparison of the cumulative baseline mean swallow count per minute against the mean swallow count per minute following the intervention, demonstrating a statistically significant difference (***p < 0.001). Apart from a statistically significant difference between groups, every individual participant (n = 20) showed an increase in swallow count per minute during the intervention.
Summary of PD participant demographics. This table summarizes the demographics of Parkinson's patients recruited for the ADAM haptic study (n = 20) including sex, age, ethnicity, Montreal Cognitive Assessment (MoCA) score, Hoehn-Yahr 34 staging, Eating Assessment 10 (EAT-10) score, Functional Oral Intake Score (FOIS), and dysphagia severity. The mean age of participants is 71.7 years old and the mean time to Parkinson's disease diagnosis was 5 years.
During structured focus groups involving n = 10 patients with PD and their caregivers (n = 2), trained moderators identified positive and negative aspects of the DHT. Overall, participants expressed positive impressions of the wearable sensor. They appreciated its comfortable design, highlighting its softness, flexibility, and small form factor. Many of the participants reported minimal to no discomfort wearing the sensor, with one noting, “I don't even know it's there. There is really nothing there that I can really feel.” The battery life was also cited as a positive feature.
While participants appreciated the sensor's comfort and design, both the participants and the caregivers also provided constructive feedback. Specifically, they noted the adhesive patch was initially difficult to remove from its paper lining and apply to the sensor. For example, one caregiver noted, “Yea. So, I’m having trouble getting it off.” This feedback resulted in refinements to the adhesive patch, improving its application and removal. Additionally, participants desired a more discreet appearance, recommending skin-tone color options to minimize visibility in public settings. One participant suggested, “Maybe if we change the color of it like match it with the skin tone it would be pretty discrete to wear it outside.”
Furthermore, participants expressed interest in additional functionalities such as guided swallowing exercises and remote monitoring of swallowing, with one highlighting the value of tracking it over time: “If you could track over time … see if you’re improving. Or not improving … or staying the same.” To minimize disruptions to daily routines, participants also suggested less frequent recharging and automatic data transfer, recognizing the challenge of adding another task to an already busy schedule: “You’re already trying to figure out how to remember to take your pills at the exact right moment 4–5 times a day. You just don’t need one more thing.”
Discussion
PD remains a major financial burden for patients, caregivers, and health systems 43 with direct and indirect costs reaching nearly $52 billion annually in the U.S. alone. 44 The progressive degenerative nature of PD spans over decades and affects nearly every bodily function given its pathogenesis of dopaminergic neuronal death. 1 Concurrently, broader advancements in DHTs that include wearable sensors and AI45,46 have found a natural application in the multifaceted challenges posed by PD. The majority of work that intersects DHTs and PD involve using either smartphones or wrist-mounted actigraphy to reproduce or augment clinical rating scales such as the MDS-UPDRS20,47,48 with an emphasis on assessing classic motor symptoms such as tremor, gait, and bradykinesia. Even though bulbar symptoms such as swallowing problems and drooling have a profound impact on quality of life and overall mortality for PD,49,50 there is considerably less work and available DHTs that can assess these parameters in a continuous manner over prolonged periods. Given the strong motivation by both regulators and the broader medical community to elevate the voice of patients in medical innovation,36,51 new DHTs for PD should focus on assessing the most meaningful aspects of health for patients—not necessarily what data is most practical to collect. The DHT reported here offers the ability to capture both classic motor and bulbar symptoms in a single integrated solution by leveraging a network for skin-mounted patches, representing a significant step toward more comprehensive and patient-centric management of PD.
Beyond continuous monitoring, DHTs that offer assistive technology and therapeutic features hold promise for significantly enhancing adoption, engagement, and overall clinical utility. In the context of dysphagia, SLP-led rehabilitative swallow therapy is the standard of care.52,53 However, this therapy typically must be done in-clinic and requires specialized equipment. A key goal of swallowing rehabilitation is for patients to maintain or improve the acquired skills during rehabilitation once discharged from care. Thus, carryover of functional improvements with rehabilitation is impeded by the absence of available in-home training technologies that are practical, affordable, and match the in-clinic training paradigm. The DHT technology presented herein offers a potential solution to durability of the swallowing therapy outcome.
While the overall sample size is small (n = 20), the average swallow frequency per minute captured by the ADAM sensor at baseline (mean 0.77 swallows per minute) is consistent with data reported in the literature for patients with PD (0.6 to 0.8 swallows per minute) and is lower than the swallowing frequency of healthy normal adults (>1 swallow per minute).14,16,17 Following activation of haptic biofeedback from a single, wireless, wearable patch, our study demonstrated that subjects reached the typical swallowing frequency levels produced by healthy adults without dysphagia. We hypothesize that, in the future, increases in swallow frequency will lead to improvements in swallowing and reductions in drooling.
Based on feedback received from patients and caregivers during the focus group interviews, future development of the MA sensor will prioritize patient-centered design improvements. We will refine the adhesive patch for easier application and removal, offer skin-tone color options for increased discretion, and explore functionalities such as guided swallowing exercises and remote monitoring with automatic data transfer. These improvements aim to maximize user comfort, convenience, and therapeutic benefit while minimizing disruption to daily routines.
There are several important study limitations to note. First, this study was small and only included n = 20 subjects with PD. The heterogeneity of the disease warrants larger studies that capture the entire spectrum of PD severity and disease duration in a prospective investigation. While increasing swallow frequency may decrease skeletal muscle atrophy by encouraging swallowing muscle use, patients with PD are also known to have altered respiratory-swallow phase patterning that increases the risk of dysphagia and aspiration. 6 Healthy adults typically initiate swallowing during the expiratory phase of the respiratory cycle. 54 This coordinative pattern is important for airway protection, facilitates physiological events beneficial to swallowing safety and efficiency, and aids in bolus clearance.54–56 Patients with dysphagia initiate swallowing during the inspiratory phase of the respiratory cycle, putting them at risk for aspiration and more severe swallowing impairments.7,54,55 Therefore, future work should focus on not only stimulating more frequent swallow events but also encourage patients to initiate swallows during the expiratory phase of the respiratory cycle with unique haptic biofeedback profiles or visual alerts.
While there are new advancements that hope to offer new therapeutic options in the near future, 57 the timely diagnosis and accurate symptom tracking of PD remain major unmet clinical needs. Misdiagnosis of PD is common by non-professionals or when atypical symptoms present.58,59 Moreover, the progression of PD can occur over decades with a highly variable prodromal period and variable symptom presentation across all symptom domains.60,61 By offering comprehensive monitoring for parameters that matter most to patients, DHTs can support the early diagnosis of PD patients and detect meaningful changes in symptomatology before overt clinical signals. We further see opportunities for these technologies to also support new therapeutics—the ADAM sensor described herein has recently been accepted into the FDA's Drug Development Tool program as a novel clinical outcome assessment. 62 Finally, therapeutic functions of the ADAM sensor likely represent the most exciting opportunity for adoption and patient impact.
Supplemental Material
Supplemental Material
Supplemental material
Supplemental material
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Ethical considerations
This study was conducted in accordance with the ethical guidelines outlined by the Declaration of Helsinki and was approved by the Institutional Review Board at Northwestern University. All research activities complied with institutional and national ethical standards for studies involving human participants.
Consent to participate
Written informed consent was obtained from all participants prior to their inclusion in the study.
Consent for publication
All participants provided consent for the publication of anonymized data collected during the study. No identifiable personal information is included in this manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Therapeutic Pipeline Program Applied Technology grant (ID: 17777) by the Michael J. Fox Foundation.
Conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Several authors are employees of Sibel Health, the company developing and commercializing the ADAM sensor used in this study. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from Sibel Health, Inc. but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Sibel Health, Inc. For data access, please contact Dylan Richards (Head of Data Science) at dylan.richards@sibelhealth.com.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
