Abstract
Background
Sexual health is influenced by a complex interplay of biological, psychological, and social factors, all of which can be impacted by Parkinson's disease (PD). Female sexual dysfunction includes reduced sexual desire and/or arousal, pain during sexual activity, or difficulty achieving orgasm. Despite its impact on quality of life, sexual health in women with PD remains poorly understood.
Objective
To investigate the impact of motor, non-motor, and social PD aspects on sexual health of women with PD.
Methods
We conducted a cross-sectional study with 100 women with PD (Hoehn and Yahr stages 1–3) who reported an active sex life in the last six months. Data were collected via remote interviews and included demographic and clinical features, cognitive capacity, motor and non-motor experiences, fatigue, self-esteem, sleep disorders, couple relationship quality, depressive symptoms, and sexual health assessments using the Female Sexual Function Index (FSFI) and Sexual Quotient-Female (SQ-F). Multiple regression models were used to identify predictors of FSFI and SQ-F scores.
Results
Results indicated that while several motor, non-motor, and social factors correlated with sexual health, only couple relationship quality and sleep quality significantly predicted both short-term (FSFI) and long-term (SQ-F) sexual health. No significant associations were observed with age, disease onset, postmenopausal status, or daily medication dosage.
Conclusions
The present study's evidence identifies multiple key areas, such as couple's relationship quality and sleep quality that could be targeted for intervention to improve sexual health in women with PD.
Plain language summary
Sexual problems are common in people with Parkinson's disease. Many factors, including biological, psychological, and social aspects affected by Parkinson's disease, can influence sexual health. However, sexual problems in women with Parkinson's disease are not well understood and have not been thoroughly researched. This study aims to address this gap by focusing specifically on women with Parkinson's disease. This study looked at how motor, non-motor, and social factors impact the short- and long-term sexual health of women with Parkinson's disease. The study included 100 women diagnosed with Parkinson's disease, with an average age of 54 years. These women had mild to moderate Parkinson's disease and had an active sex life in the past six months.
The researchers collected data through remote assessments, covering demographic and clinical characteristics, short-term and long-term sexual health, cognitive capacity, motor and non-motor daily living difficulties, fatigue, self-esteem, sleep quality, couple relationship quality and signs of depression. The results showed that the couple's relationship and sleep quality were significant predictors of both short-term and long-term sexual health in women with Parkinson's disease. Age, disease onset, postmenopausal period, and daily medication dosage did not affect sexual health.
The findings from this study offer valuable insights that can guide the development of interventions aimed at enhancing sexual health in women with Parkinson's disease.
Introduction
Sexual health is a multifaceted phenomenon influenced by organic, environmental, and psychological factors affected by Parkinson's disease (PD). PD is the second most prevalent neurodegenerative disease, 1 affecting over 10 million people globally. 2 Clinically, PD is characterized by a range of heterogeneous motor symptoms. 3 In addition to motor dysfunction, people with PD experience several non-motor symptoms, including sexual dysfunction.4,5 However, existing data on sexual health in PD are incomplete and often contradictory. 6 Despite being poorly understood and infrequently studied, 5 sexual dysfunction is identified as one of the most bothersome symptoms of PD, according to patients. 7 Sexual dysfunction significantly contributes to the reduction in quality of life for people with PD, increasing medical, social, and economic burdens for patients, their sexual partners, and caregivers. 8 Multiple studies have demonstrated the negative impact of sexual dysfunction on the quality of life.9,10
In the absence of disease, aging plays a significant role in the development of sexual dysfunction, 11 with age being an independent predictive factor for erectile dysfunction in men and sexual dysfunction in women. 12 The impact of aging on sexual health in PD is complex, with several studies indicating that sexual dysfunction and sexual satisfaction deteriorate with advancing age.13–16 The age of PD onset also appears to be a crucial factor; people with early-onset PD (EOPD) report higher incidences of sexual dysfunction,17–20,21 decreased libido, and increased difficulty in sexual activities compared to those with late-onset PD (LOPD). 22
The prevalence of sexual dysfunction in women with PD ranges from 36% to 87.5%.14,21,23–25 The manifestation of sexual dysfunction in PD varies by gender. Women report greater challenges with arousal, achieving orgasm, low sexual desire, and overall sexual dissatisfaction, while men report erectile dysfunction, sexual dissatisfaction, premature ejaculation, and difficulties in reaching orgasm.14,26 Both men and women with PD experience reduced libido; 27 however, women with PD are more significantly affected than men. 21 Additionally, hypersexuality, defined as increased libido and repetitive behavior seeking sexual gratification beyond socially and personally accepted boundaries, is more prevalent in men with PD (5.2%) than in women with PD (0.5%) and is closely associated with the use of dopamine agonists. 28
Although certain motor symptoms can increase the difficulty of engaging in sexual activities, 29 the relationship between disease severity and sexual dysfunction varies across studies. Some studies indicate that sexual dysfunction worsens with disease progression,18,22,27 while others show no such correlation.16,19,30,31 Sexual dysfunction in men with PD was observed to increase over a 7-year follow-up period, whereas no changes were found for women with PD. 32 Disease stage and the duration of treatment have been correlated with significant changes in libido and sexual activity, particularly in women with PD. 21 The level of disability is inversely related to sexual activity 10 and sexual satisfaction in men with PD, but not in women with PD. 33 While some studies have found a significant association between disease severity and sexual dysfunction21–23,34 others have not.13,16,17,25,27
Some studies have found no correlation between depression and sexual dysfunction19,31,35 or cessation of sexual activity. 14 In contrast, other studies have identified depression as the sole factor associated with sexual and marital satisfaction 9 and the unique common factor explaining sexual dissatisfaction in people with PD. 33 The severity of depression was the main predictive factor for loss of libido in this population. 23 A 2-year longitudinal prospective study 10 found that lower levels of depression were associated with higher sexual activity in men with PD, but not in women with PD. Depression was also correlated with relationship dissatisfaction between patients and their partners in EOPD. 36
Although cognitive decline was associated with libido loss in men with PD, 23 it was not correlated with sexual activity in people with PD. 10 Other non-motor symptoms such as fatigue, lack of motivation, anxiety, apathy, and sadness were associated with sexual dysfunction, particularly in women with PD.31,37 Autonomic dysfunction, especially urinary dysfunction, 20 was correlated with sexual dysfunction, 38 and a higher dose of L-dopa was associated with decreased desire in women with PD. 14 However, the daily dosage of levodopa was unrelated to overall sexual dysfunction. 25
Among social factors, several studies have shown that sexual dysfunction affects both the patient and the sexual partner.9,26 The couple's relationship quality is bidirectionally related to sexual health. Brown et al. found that satisfaction with marital relationships was closely associated with satisfaction with sexual life in couples where one partner had PD. 9 However, satisfaction with marital relationships varied between genders. Although men with PD rejected sex less frequently and reported stronger desire, 39 they were more dissatisfied with their sex life and marital relationship compared to women with PD. 14
To the best of our knowledge, only two studies have focused exclusively on sexual dysfunction in women with PD. Welsh et al. found that women with PD reported greater dissatisfaction with the quality of their sexual experiences and were less satisfied with their sexual relationships and partners compared to controls. 30 More recently, Varanda et al. found a higher prevalence of sexual dysfunction in women with PD than in controls. 16 While age and depression severity were predictors of sexual dysfunction, disease duration, disability level, disease progression, and antiparkinsonian medication were not. In a recent study, Bronner et al. recommended that more research focus on women with PD, as they experience sexual dysfunction from the early stages of PD and often have difficulty expressing their sexual concerns. 40
Given the extensive range of factors that may affect sexual health and the limited number of studies that have examined the impact of these factors on sexual health in women with PD, this study aimed to investigate motor, non-motor, and social PD aspects influencing the sexual health of women with PD.
Methods
Study design and participants
We conducted a cross-sectional, exploratory study involving 100 women with PD. The eligibility criteria were (a) women; (b) willingness to participate in the study (c) confirmed diagnosis of idiopathic PD according to the UK Parkinson's Disease Society Brain Bank diagnostic criteria; 41 (d) age above 21 years; (e) reported sexual activity, not necessarily with penetration, within the last six months; and (f) access to telephone or internet. The exclusion criteria were the presence of neurological disorders other than PD, dementia, and speech or hearing disorders that could impair participation in a remote interview.
Recruitment
A convenience sample of participants was recruited from the AMPARO Network (http://www.amparo.numec.prp.usp.br). The AMPARO Network is part of the Research, Innovation and Dissemination Center for Neuromathematics (RIDC NeuroMat), and its main objective is to enhance the quality of life of people with PD in Brazil. This network comprises people with PD, their family members, caregivers, students, health professionals and researchers with an interest in PD. Initial eligibility was determined through outreach via calls and/or telephone messages. Following this, detailed information about the study procedure was provided, and participants were invited to provide their written consent for involvement.
This study was approved by the Ethics Committee of the Federal University of Amapa, Macapa, Brazil (#CAAE 39971420.0.0000.0003) and conducted in accordance with the Helsinki Declaration.
Study procedures
The study flow is illustrated in Figure 1. Evaluations were carried out in two sessions, each approximately 30–35 min in duration, conducted by the same researcher with an interval of less than 7 days between sessions. Upon consenting to participate, individuals were asked to indicate the two most suitable days and times for the remote assessment, ensuring it occurred during the ON period of their dopaminergic medication (40–120 min post-last intake). To minimize discomfort when addressing sensitive questions, only female researchers performed the assessments, thereby enhancing participant comfort and convenience.
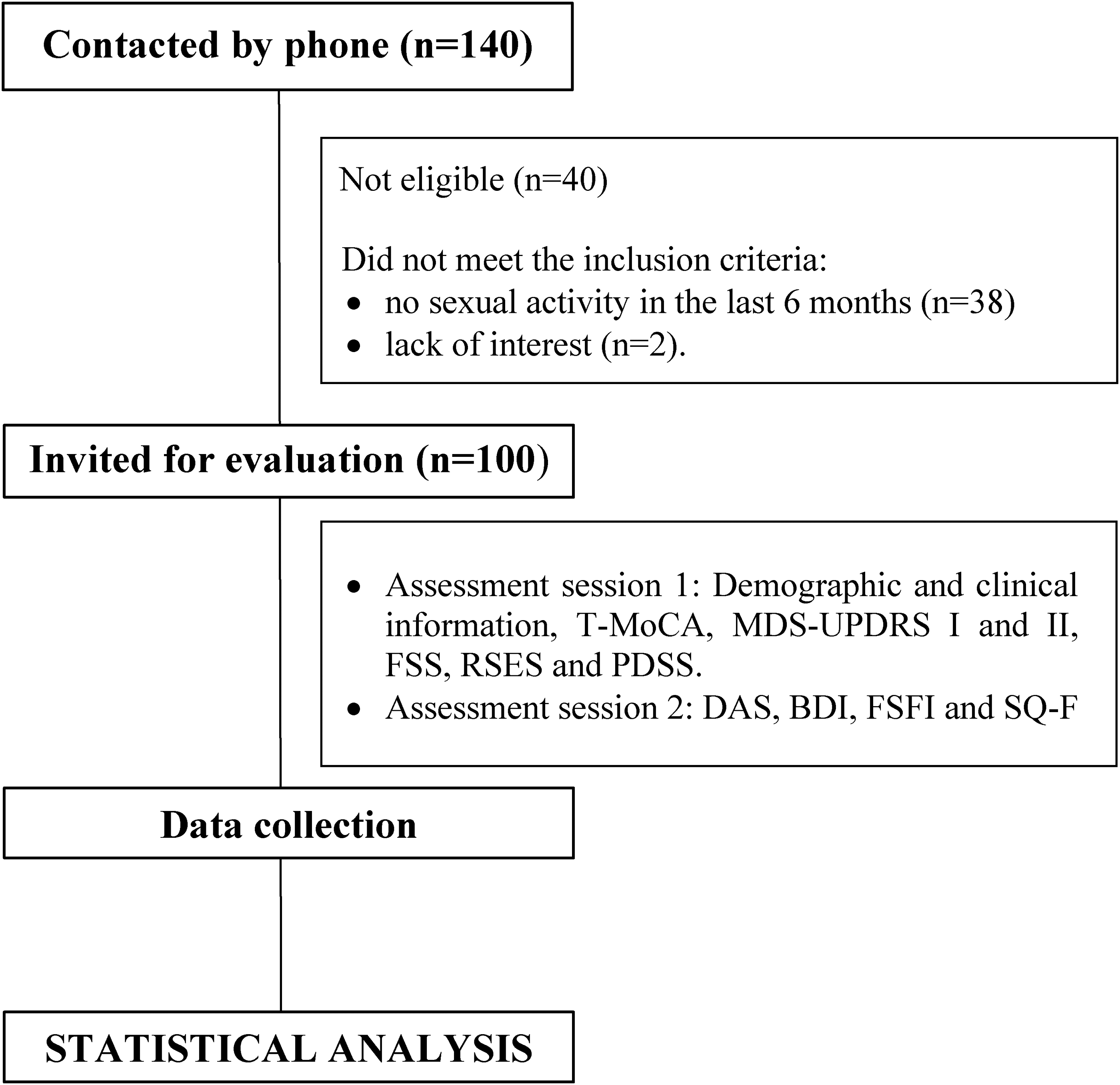
The schematic study design. T-MoCA: Telephone - Montreal Cognitive Assessment); MDS-UPDRS: Movement Disorder Society – Unified Parkinson's Disease Rating Scale; FSS: Fatigue Severity Scale; RSES: Rosenberg's Self-esteem Scale; PDSS: Parkinson's Disease Sleep Scale; DAS: Dyadic Adjustment Scale; BDI: Beck Depression Inventory; FSFI: Female Sexual Function Index; SQ-F: Sexual Quotient - Female.
The primary outcomes were short and long-term sexual health, assessed by Female Sexual Function Index (FSFI) and Sexual Quotient – Female (SQ-F), respectively.
Assessments were conducted by physiotherapists specialized in movement disorders, who underwent a three-phase training process led by an experienced researcher: (1) familiarization with each instrument, (2) practical training using simulations with non-patients, and (3) supervised evaluations with actual patients as part of a pilot study.
The clinical information, such as confirmed diagnosis, disease duration, comorbidities, previous surgeries, medications, and the most recent Hoehn and Yahr (H&Y) classification, was obtained from the healthcare system records where patients receive care for PD. The records were no older than six months to ensure that the information accurately reflected the participants’ current condition and to avoid outdated information. This information was subsequently verified with participants and their families. Details of dopaminergic treatment, including drug type and daily dose, were recorded to calculate the levodopa equivalent daily dose (LEDD). 42
Assessments tools
Female sexual function index (FSFI)
The FSFI was utilized to assess the short-term sexual health of women with PD. This comprehensive self-report questionnaire evaluates women's sexual function over the preceding four weeks through 19 questions across six domains: sexual desire, arousal, lubrication, orgasm, satisfaction, and pain. Each question is rated on a 5-point scale (1–5), with higher scores indicating better sexual functioning. 43 Domain scores are calculated by summing the scores of the respective questions and then multiplying by a specific factor (0.6 for desire, 0.3 for arousal, 0.3 for lubrication, 0.4 for orgasm, 0.4 for satisfaction, and 0.4 for pain). The total score is obtained by summing the scores of all six domains, with a total score of 26.5 or less indicating sexual dysfunction. Additionally, the desire domain score can be used independently, with scores of 5 or below meeting the diagnostic criteria for Hypoactive Sexual Desire Disorder. 44
Sexual quotient – female (SQ-F)
The SQ-F was used to assess the long-term sexual health of women with PD. It is a questionnaire designed to evaluate emotional and functional aspects related to women's sexual performance and satisfaction over the past six months. This self-report questionnaire consists of 10 questions across five domains: sexual desire and interest (libido level), foreplay (sexual activity that precedes intercourse), personal excitement and harmony with the partner (interest and satisfaction with the sexual partner), comfort (vagina's ability to relax and absence of pain), and orgasm and satisfaction (final satisfaction with intercourse). Each question is rated on a 5-point scale (1–5), where higher scores indicate better sexual functioning. For question 7, which concerns pain during intercourse, the final score is determined by subtracting the response value (ranging from 0 to 5) from 5. After obtaining the scores for all questions, the sum is multiplied by two, resulting in a total score ranging from 0 to 100. A score equal to or less than 60 indicates sexual dysfunction. 45
Socioeconomic condition (SEC)
SEC was assessed using the Brazilian Economic Classification Criterion, a socio-economic classification standard based on household, family education, and income. The score ranges from zero to 100 according to the following Brazilian Criteria Thresholds: A (45–100 points); B1 (38–44 points); B2 (29–37 points); C1 (23–28 points); C2 (17–22 points); and DE (0–16 points). Higher scores indicate better SEC conditions. 46
Telephone - Montreal cognitive assessment (T-MoCA)
The T-MoCA was used to assess the global cognitive capacity. This test is a modified version of the MoCA 30 that can be conducted over the phone. It effectively identifies cognitive impairment and is closely correlated to the MoCA test. 47 The T-MoCA has a maximum score of 22, with sub-scores for attention (6 points), language (3 points), abstraction (2 points), delayed recall (5 points), and orientation (6 points). High scores indicate good overall cognitive function.
Movement disorder society - unified Parkinson's disease rating scale parts I (MDS-UPDRS I) and II (MDS-UPDRS II)
The MDS-UPDRS I and MDS-UPDRS II were used to assess non-motor and motor aspects of daily life, respectively. This scale is used to assess the severity and progression of PD based on the difficulties experienced in the past seven days. It consists of four parts, but for this study only Part I (non-motor experiences of daily living) and Part II (motor experiences of daily living) were used. Part I comprises 13 questions that evaluate non-motor aspects of the patient's daily life, such as cognitive impairment, hallucinations, depression, anxiety, apathy, dopamine dysregulation syndrome, sleep problems, urinary and constipation issues, lightheadedness, and fatigue. The scores obtained in item 1.6 are utilized to evaluate the presence and severity of dopamine dysregulation syndrome, as it is linked to impulse control disorders and hypersexuality [28]. Part II also comprises 13 questions that assess motor aspects of daily life experiences, including speech, swallowing, dressing, walking, and balance. Each question is scored from 0 to 4, with higher scores indicating more impact on non-motor and motor symptoms. 48
Fatigue severity scale (FSS)
The FSS was used to measure fatigue. It is a nonspecific rating scale often used in PD. It comprises nine items rated on a scale from 1 (strongly disagree) to 7 (strongly agree). The total score ranges from 9 to 63, with scores of 28 or higher indicating the presence of fatigue. Specifically, scores between 28 to 39 reflect mild fatigue, scores between 40 to 51 indicate moderate fatigue, and scores from 52 to 63 denote severe fatigue. 49
Rosenberg self-esteem scale (RSES)
The RSES was utilized to measure self-esteem. It consists of ten questions assessing overall self-esteem, five of which focus on positive aspects and five on negative aspects related to self-esteem perception. Each statement in the scale offers four possible responses: 1 - completely agree, 2 - agree, 3 - disagree, and 4 - completely disagree. For questions 1, 2, 4, 6, and 7, which assess self-confidence, the scoring is as follows: completely disagree = 1, disagree = 2, agree = 3, and completely agree = 4. For questions 3, 5, 8, 9, and 10, which refer to self-deprecation, the scoring is as follows: completely agree = 1, agree = 2, disagree = 3, and completely disagree = 4. The total score, reached by the sum of all questions, ranges between 10 and 40, with a higher total score indicating a higher level of self-esteem. 50
Parkinson's disease sleep scale (PDSS)
The PDSS was used for evaluating sleep disorders. It is a specific scale designed to assess sleep disorders in people with PD. 51 PDSS consists of 15 questions focusing on sleep disturbances experienced over the last week. The first question assesses the overall quality of nighttime sleep. Questions 2–14 pertain to specific nocturnal sleep issues such as difficulty falling and staying asleep, restlessness at night, nocturnal hallucinations, nocturia, experiencing motor symptoms at night, and feeling refreshed after sleep, while question 15 addresses daytime sleepiness. Each question is scored from 0 (severe and always experienced) to 10 (never experienced), except for question 1, which is rated from awful (0) to excellent (10). The highest possible total score is 150, which indicates absence of all symptoms and best sleep quality. 52
Dyadic adjustment scale (DAS)
The DAS consists of 32 questions that evaluate marital adjustment across four dimensions: dyadic consensus, dyadic satisfaction, dyadic cohesion, and expression of affection. Respondents answer using a Likert scale of 5, 6, or 7 points, with higher values indicating more frequent occurrences. Two questions (29 and 30) have "yes" or "no" options. The total score, ranging from 0 to 151, is calculated by summing the scores from the four dimensions: consensus (0–65 points), satisfaction (0–50 points), cohesion (0–24 points), and expression of affection (0–12 points). A total score below 101 indicates dissatisfaction. 53
Beck depression inventory (BDI)
The BDI is widely used for screening and assessing the severity of depressive symptoms and is considered a valid instrument for evaluating depressive symptoms in individuals with PD. It consists of 21 items that evaluate the severity of depressive symptoms experienced over the past two weeks, with a total score ranging from 0 and 63. A score of 0 to 9 suggests the absence of depression, 10 to 18 indicates mild to moderate depression, 19 to 29 suggests moderate to severe depression, and 30 to 63 indicates severe depression. 54
Statistical analyses
Descriptive statistical analysis was conducted on demographic and clinical data. Spearman rank-order correlation was employed to evaluate the correlations among variables, including age, SEC, LEDD, dopamine dysregulation syndrome, disease duration, disease onset, H&Y stage, T-MoCA, MDS-UPDRS I, MDS-UPDRS II, FSS, RSES, PDSS, DAS, and BDI with FSFI and SQ-F scores, as well as between FSFI and SQ-F scores.
Additionally, two multiple regression models to predict the primary outcomes (FSFI and SQ-F scores) were developed, including all factors with a statistically significant moderate correlation (R > 0.30) with the total FSFI and SQ-F scores as predictor variables.
The Kolmogorov-Smirnov test was used to compare FSFI and SQ-F scores between postmenopausal women (defined as the period after 12 consecutive months without menstrual periods) and premenopausal women (the reproductive period), 55 as well as between women with EOPD (diagnosed before age 50) and LOPD (diagnosed after age 50). 56
Statistical significance was set at p < 0.05. All statistical analyses were performed using Statistica Version 13 (TIBCO Software Inc. USA).
Results
Demographic and clinical characteristics
The participants’ demographic and clinical characteristics are shown in Table 1. According to the H&Y classification, 45% of the participants were in stage 1, 40% in stage 2, and 15% in stage 3. Additionally, 36% of the participants had EOPD. Of the participants, 67% were postmenopausal, and only two reported mild to moderate dopamine dysregulation syndrome as assessed by item 1.6 of MDS-UPDRS, with none reporting hypersexuality. All participants identified as women both biologically and socially and reported being heterosexual. Finally, 83% of the participants reported being in long-term stable couple relationships, while 17% reported being in short-term couple relationships.
Demographic and clinical features of the participants.
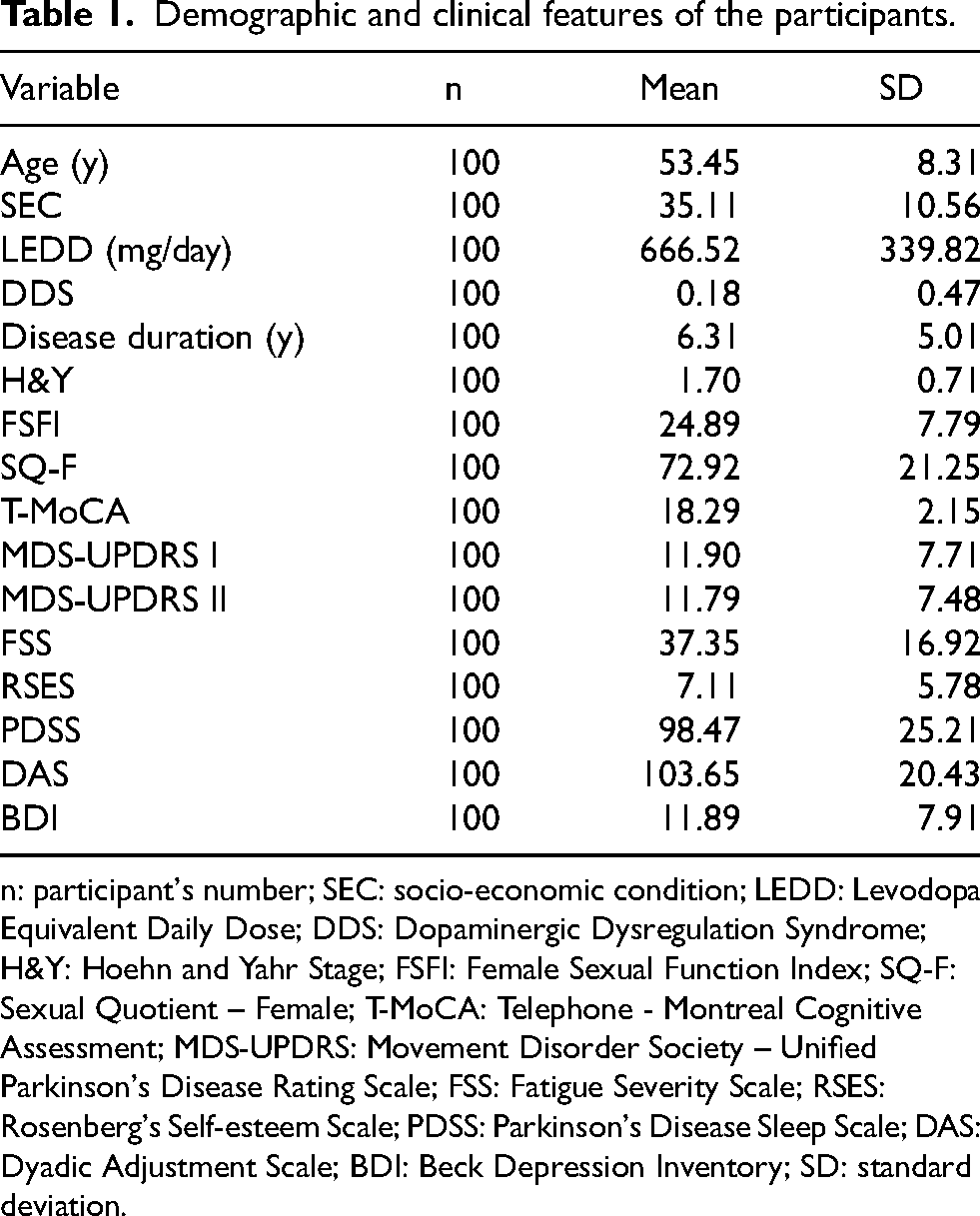
n: participant's number; SEC: socio-economic condition; LEDD: Levodopa Equivalent Daily Dose; DDS: Dopaminergic Dysregulation Syndrome; H&Y: Hoehn and Yahr Stage; FSFI: Female Sexual Function Index; SQ-F: Sexual Quotient – Female; T-MoCA: Telephone - Montreal Cognitive Assessment; MDS-UPDRS: Movement Disorder Society – Unified Parkinson's Disease Rating Scale; FSS: Fatigue Severity Scale; RSES: Rosenberg's Self-esteem Scale; PDSS: Parkinson's Disease Sleep Scale; DAS: Dyadic Adjustment Scale; BDI: Beck Depression Inventory; SD: standard deviation.
The mean FSFI score was 24.89 ± 7.79, which is below the threshold score of 26.5 used to identify sexual dysfunction, with 46% of participants meeting the criteria for sexual dysfunction based on this measurement. The mean score in the Desire domain was 3.6, which is below the cutoff score of 5 for diagnosing Hypoactive Sexual Desire Disorder. 44 The mean scores for each FSFI domain can be observed in Figure 2.
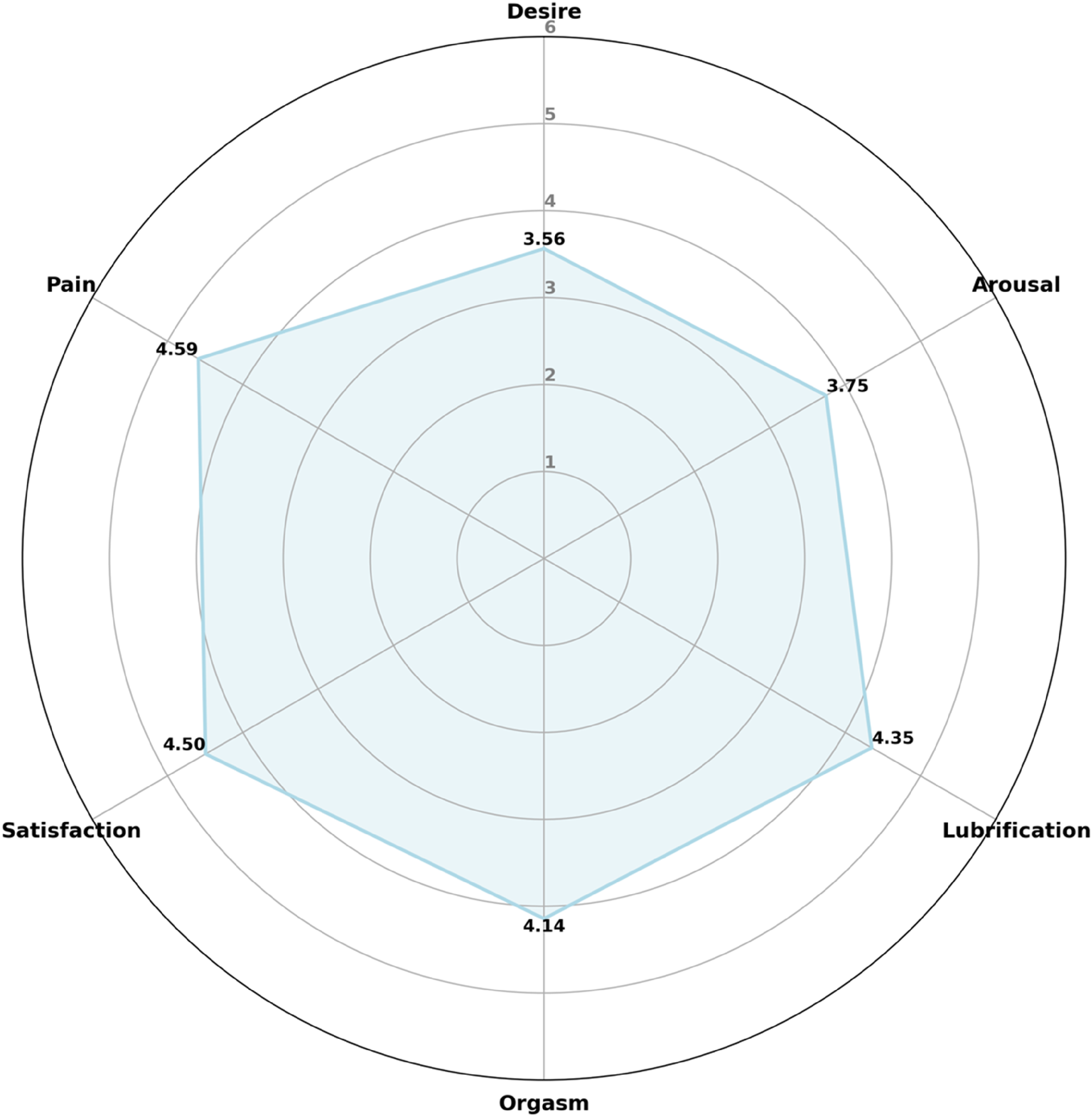
Mean scores across different domains of the FSFI. The radar chart illustrates the mean scores across different domains of the FSFI (Female Sexual Function Index) questionnaire, with a maximum score of 6. Each axis represents a domain of female sexual function, and the blue line indicates the average score for each domain. Higher scores, closer to 6, indicate better sexual function in that domain. The six domains represented are: Desire, Arousal, Lubrication, Orgasm, Satisfaction, and Pain. The purpose of this figure is to provide a visual analysis comparing the most and least affected domains, highlighting areas of strength and those with potential for improvement. Calculation: For each domain, the scores (range 1–5), for relevant questions are summed and then multiplied by a factor specific to that domain to adjust for the varying number of questions in each. Here's how each domain is calculated: Desire: Sum the scores of questions 1 and 2, then multiply by 0.6. Arousal: Sum the scores of questions 3 to 6, then multiply by 0.3. Lubrication: Sum the scores of questions 7 to 10, then multiply by 0.3. Orgasm: Sum the scores of questions 11 to 13, then multiply by 0.4. Satisfaction: Sum the scores of questions 14 to 16, then multiply by 0.4. Pain: Sum the scores of questions 17 to 19, then multiply by 0.4.
For the SQ-F, the median score was 72.92 ± 21.25, exceeding the cutoff score of 60 used to identify sexual dysfunction, with 22% of participants meeting the criteria for sexual dysfunction according to this instrument. The mean scores for each SQ-F domain can be observed in Figure 3.
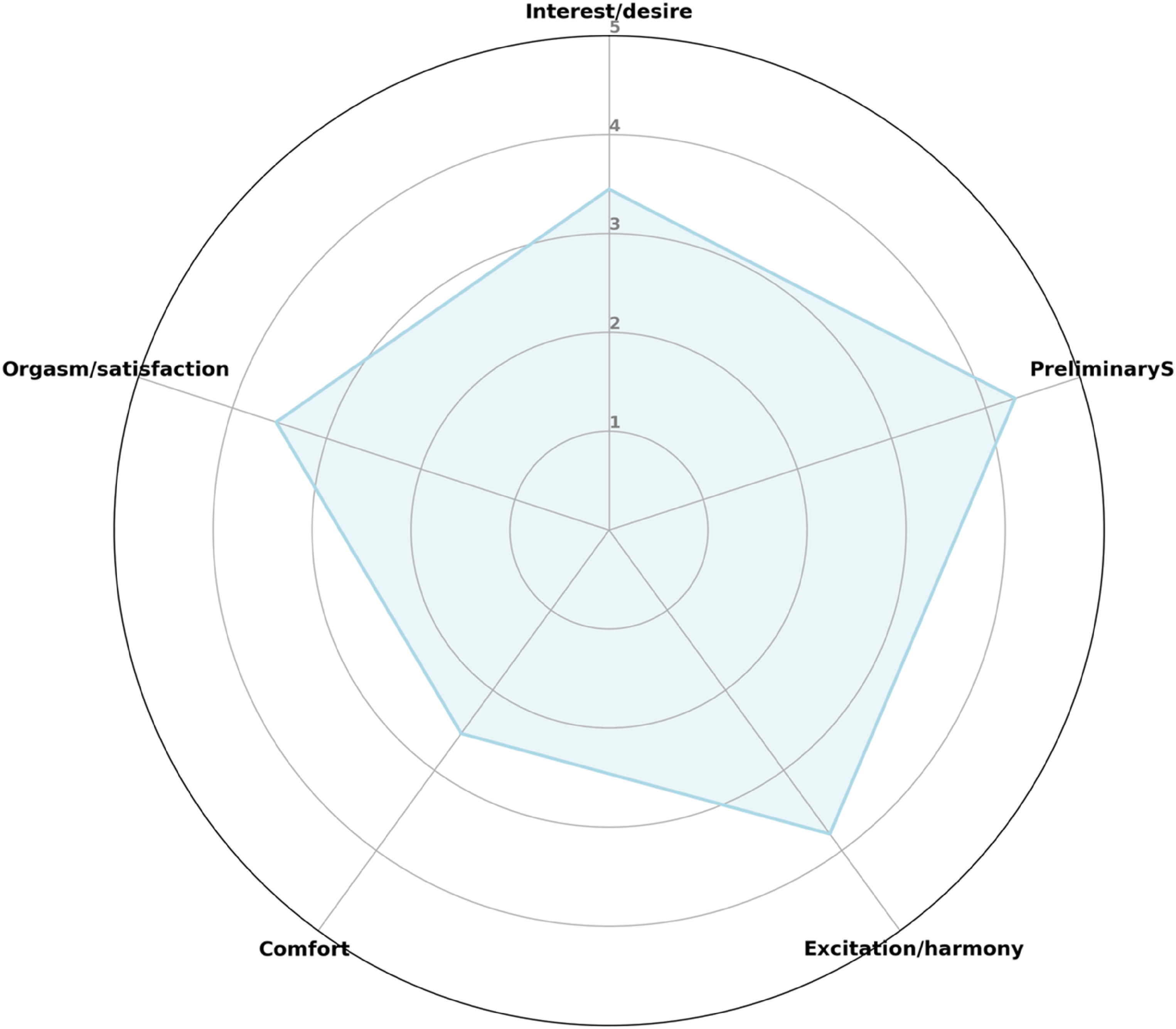
Mean scores across different domains of the SQ-F. The radar chart illustrates the average scores across different domains of the SQ-F (Sexual Quotient – Female) questionnaire, with a maximum score of 5. Each axis represents a domain of female sexual function, and the blue line indicates the average score for each domain. Higher scores, closer to 5, indicate better sexual function in that domain. The five domains represented are: Interest/desire, Preliminary stimulation, Excitation/harmony, Comfort, and Orgasm/satisfaction. The purpose of this figure is to provide a visual analysis comparing the most and least affected domains, highlighting areas of strength and those with potential for improvement.
No statistically significant differences were found in FSFI and SQ-F scores between participants with EOPD and those with LOPD (p > 0.10), or between postmenopausal and premenopausal women (p > 0.10).
A strong positive correlation was found between FSFI and SQ-F scores (R = 0.75; p < 0.000001).
Short-term sexual health
The total FSFI scores demonstrated a weak positive statistically significant correlation with SEC and T-MoCA scores, and a weak negative statistically significant correlation with MDS-UPDRS I, FSS, and RSES scores. Additionally, FSFI scores showed a moderately positive correlation with PDSS and DAS scores, and a moderately negative correlation with MDS-UPDRS II and BDI scores (Table 2).
Correlation between demographic and clinical variables and FSFI and SQ-F.
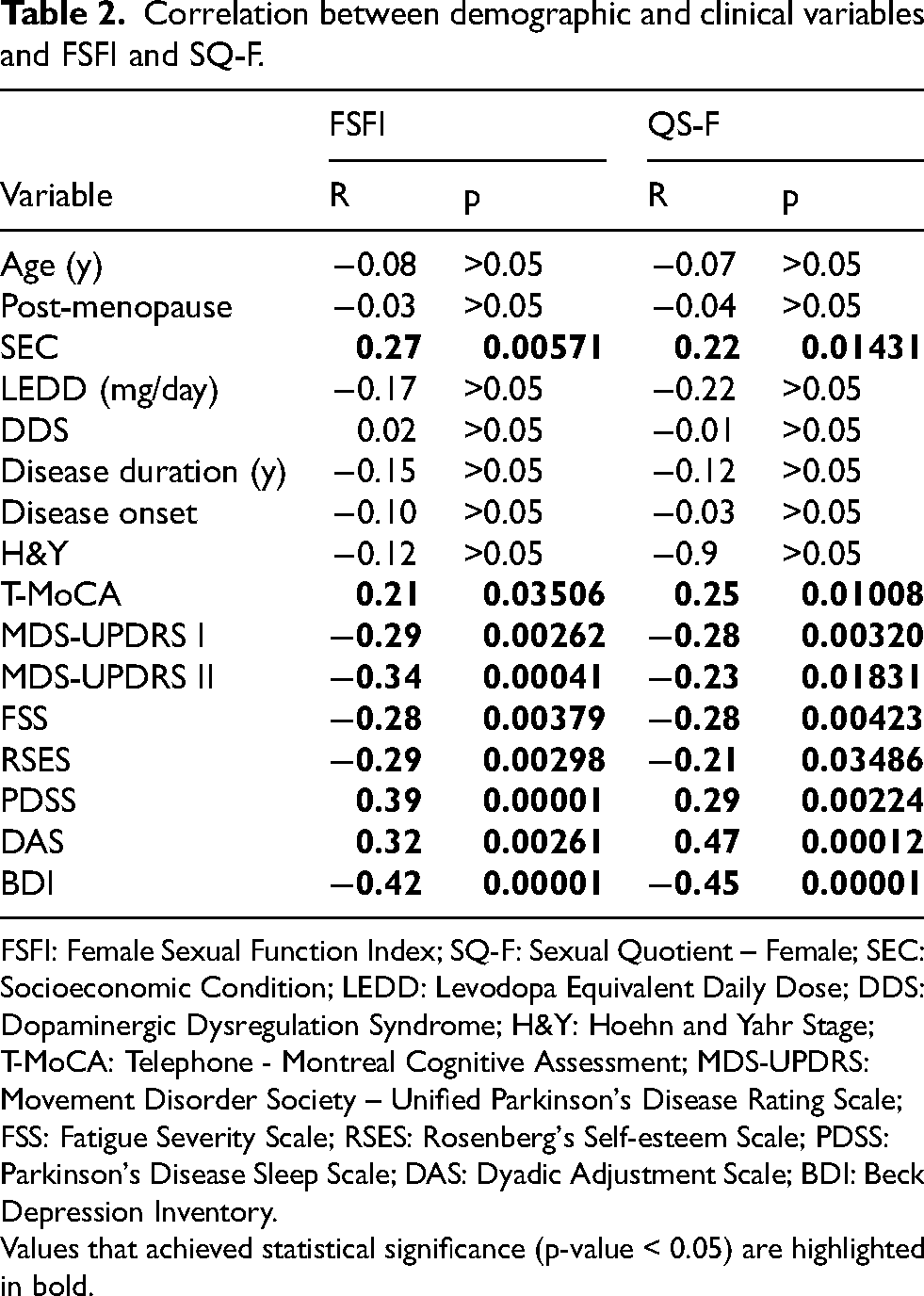
FSFI: Female Sexual Function Index; SQ-F: Sexual Quotient – Female; SEC: Socioeconomic Condition; LEDD: Levodopa Equivalent Daily Dose; DDS: Dopaminergic Dysregulation Syndrome; H&Y: Hoehn and Yahr Stage; T-MoCA: Telephone - Montreal Cognitive Assessment; MDS-UPDRS: Movement Disorder Society – Unified Parkinson's Disease Rating Scale; FSS: Fatigue Severity Scale; RSES: Rosenberg's Self-esteem Scale; PDSS: Parkinson's Disease Sleep Scale; DAS: Dyadic Adjustment Scale; BDI: Beck Depression Inventory.
Values that achieved statistical significance (p-value < 0.05) are highlighted in bold.
In the multiple regression analysis, which included variables with a significant moderate correlation with FSFI (MDS-UPDRS II, PDSS, DAS, and BDI), only PDSS and DAS remained as independent predictors of FSFI total scores. The final model had an adjusted R2 = 0.36 and F(3,80) = 16.57 with a p-value < 0.000001.
Long-term sexual health
The SQ-F total score showed a weak positive correlation with SEC and T-MoCA, and a weak negative correlation with MDS-UPDRS I, MDS-UPDRS II, FSS, and RSES. Additionally, there was a moderate positive correlation with DAS and PDSS, and a moderate negative correlation with BDI (Table 2).
The multiple regression analysis, which included variables with a significant moderate correlation with SQ-F (PDSS, DAS, and BDI), found that only DAS and PDSS remained as independent predictors of SQ-F total scores. The final model had an adjusted R2 = 0.30 and F(2,81) = 18.29 with a p-value < 0.000001.
Discussion
Although sexual health is a fundamental aspect of the overall health and well-being for individuals, families, and societies, 57 few studies have focused on sexual health in people with PD, particularly women. To the best of our knowledge, this study is the first to investigate the impact of motor, non-motor, and social PD aspects on the short-term sexual health and long-term sexual health of women with PD.
Our findings reveal that while several motor, non-motor, and social factors were correlated with short-term and long-term sexual health, only two factors—couple's relationship quality and sleep quality—were significant predictors of both short-term and long-term sexual health outcomes. Interestingly, variables such as age, postmenopausal status, disease onset, disease duration, disease stage, LEDD, dopamine dysregulation syndrome, and levels of daily life motor and non-motor disabilities did not predict sexual health.
Based on the results, several considerations can be made.
Firstly, the use of both the FSFI and the SQ-F in this study provides a comprehensive evaluation of sexual health in women with PD. The FSFI assesses sexual health over the past four weeks, while the SQ-F provides a qualitative assessment over the past six months. The high correlation between the results from both instruments suggests that they are influenced by similar factors. However, there are domain-specific differences. For instance, FSFI includes a specific domain for pain, whereas SQ-F includes a domain for foreplay, which involves preliminary intimate activities that enhance arousal and sexual pleasure. Although most previous studies on sexual health in women with PD have utilized the FSFI12,16,20,27,33,40,58 incorporating the SQ-F offers a more comprehensive understanding of the disease's impact on sexual health.
Secondly, our findings indicate that more than twice as many women met the criteria for sexual dysfunction using the FSFI compared to the SQ-F. Previous studies have reported varying prevalence rates of sexual dysfunction in women with PD ranging from 53–86.9%.12,16,27,33 The lower prevalence of sexual dysfunction in the present study may be partly explained by the inclusion criteria regarding reported sexual activities. The high number of women with PD who did not meet this inclusion criteria supports this interpretation (Figure 1). Further studies are needed to investigate the reasons for the interruption in sexual activities within this population. Interestingly, more than 20% of participants reported experiencing short-term sexual dysfunction over the past four weeks but not long-term sexual dysfunction over the past six months, suggesting a progressive decline in sexual activity. However, further longitudinal studies are required to fully understand this issue.
Thirdly, our study found no correlation between sexual dysfunction and age or clinical measures. While other studies have shown that sexual dysfunction and sexual satisfaction in people with PD worsen with age and disease progression13–16,30,31 and are affected by dopaminergic medication, 14 the lack of a significant correlation between sexual dysfunction and clinical measures such as disease onset, duration, stage, LEDD, and dopamine dysregulation syndrome suggests that sexual dysfunction may be present throughout the disease course. Despite more than 30% of women in our study meeting criteria to EOPD, no correlation was found between age, disease onset, and sexual health (SH). Previous studies have indicated that sexual dysfunction is a common non-motor symptom in EOPD and may be underestimated.18,20 Additionally, there was no correlation between postmenopausal status and sexual dysfunction, despite the increased prevalence of sexual dysfunction in postmenopausal women. 59 These findings highlight the importance of sexual health in women with PD regardless of age and underscore the need for monitoring, prevention, and management of sexual health changes from the onset of the disease.
Fourthly, our study found that women with PD who reported experiencing short- and long-term sexual dysfunction exhibited lower SEC, self-esteem and sleep quality, along with higher motor disability levels and depressive symptoms, compared to those without sexual dysfunction. These findings suggest that sexual health in PD can be influenced by various social, motor, and non-motor factors. While few studies have investigated social or non-motor factors such as fatigue, self-esteem, and sleep quality, several studies have explored the association between depression and sexual dysfunction. Depression severity has been associated with loss of sexual libido, 23 sexual and marital satisfaction,9,33 and decreased intimacy in women. Previous studies, including those focused exclusively on women with PD, have confirmed the negative impact of depression on SH. Welsh showed that women with PD exhibited higher levels of depression compared to controls, which was associated with decreased libido. 30 Varanda et al. demonstrated that depression severity could predict sexual health in women with PD. 16 Although our study found that depression was highly correlated with short- and long-term sexual dysfunction, it did not retain predictive value. However, given that 54.9% of women with PD report depressive symptoms and 15.7% experience major depressive symptoms, 60 further investigation into the relationship between depression and sexual dysfunction in people with PD is critical for future research.
It is important to note the correlation between SEC and SH. Individuals living in lower SEC reported poorer sexual health compared to those in better conditions. There is a paucity of evidence regarding the relationship between social support, access to healthcare services, and sexual health, even among healthy women. 61 Those who reported constrained socioeconomic conditions, particularly in terms of income, education, and occupation, also reported worse sexual health. 62 Economic hardship within the family was positively associated with sexual dysfunction, especially among women. 63 Additionally, low SEC is strongly associated with lower educational levels. Older people with higher education levels were more likely to be sexually active. 64 Factors such as reduced impact of negative stereotypes on sexual expression, better access to healthcare, health education, and activities that maintain and improve overall physical and mental health are more likely to be associated with greater sexual desire and activity. 65 Conversely, individuals with lower SEC may experience increased stress, leading to reduced time and desire for sexual activity. 66 Improving healthcare support, including access to social work and partner care, may enhance sexual health in people living in low SEC. Clinicians and health professionals should consider this important aspect of human function when evaluating PD patients to ensure appropriate referrals are made.
Regarding motor factors, our results showed that motor disability level was correlated with both short- and long-term SH. Motor disabilities can significantly impact various motor activities linked to foreplay and intercourse, which are essential components of sexual activity. 29 Raciti et al. found that PD patients with sexual dysfunction reported higher motor disability in daily living than those without sexual dysfunction. 27 In contrast, Kotkova et al. found that motor disability was a predictor of sexual health for men with PD but not for women with PD, possibly because other factors such as depression and anxiety have a greater impact on women than men. 33 Our current findings indicate that, despite the influence of other factors, motor disability impacts the sexual health of women with PD. This suggests that interventions aimed at preventing or improving motor disability associated with PD could positively affect sexual health. However, further studies are required to confirm this.
Finally, and most importantly, in the present study, among the various investigated factors, only couple's relationship quality and sleep quality emerged as predictors of both short-term and long-term sexual health. Couple adjustment is a complex phenomenon involving how individuals perceive their relationships, whether their expectations are met, their emotional satisfaction, and sexual enjoyment. 67 Research suggests that sexual and relationship well-being are closely linked in individuals unaffected by PD, 68 regardless of age and relationship status. 69 For middle-aged women, sexual functioning predicted relationship satisfaction. 70 However, few studies have explored the relationship between couple's relationship quality and sexual health in people with PD. Brown et al. showed that most women with PD and their partners had scores within the normal range for marital satisfaction, while men with PD and their partners exhibited significant marital dissatisfaction. Higher marital satisfaction was associated with better sexual dysfunction for both women and men with PD. 9 Similarly, Wielinski et al. showed that sexual dissatisfaction is correlated with relationship dissatisfaction in people with PD. 36 These findings reinforce the importance of providing intervention aimed at improving or maintaining couple's relationship quality for couples facing chronic and disabling diseases, rather than focusing solely on the patient.
Regarding sleep quality, the relationship between sleep and sexual function in women without PD has been previously studied. 71 Previous studies have revealed connections between insomnia, sleep duration, and sexual dysfunction in this population. 72 Similarly, other studies found that the presence and severity of Obstructive Sleep Apnea 73 and poor sleep quality 74 were linked to lower FSFI scores, indicating poorer sexual function. However, this study is the first to demonstrate the adverse effect of poor sleep quality on sexual health in women with PD. Sleep disturbances are prevalent among people with PD and encompass the entire spectrum of sleep disorders. The connection between these sleep disorders and motor function remains uncertain. 75 Specifically, rapid-eye-movement behavior disorder has been linked to a higher risk of cognitive decline, anxiety, and depression in PD patients. 76 These motor and non-motor alterations associated with sleep disorders may have indirectly impacted the SH. Further research should explore this relationship in greater depth.
While this study did not compare men and women with PD, a recent study employing similar methodologies identified distinct predictors of sexual health for men. 77 For men with PD, depression and motor disability were the key predictors for sexual health. These gender-specific differences might be attributed to varying social roles, educational backgrounds, and values. The sexual health of women appears to be more affected by the quality of their intimate relationships, 78 whereas men's sexual health is more closely associated with their mental and physical health. 29 To gain a comprehensive understanding of this issue, future studies should directly compare the impact of these factors on the sexual health of women and men with PD.
Lastly, it is important to acknowledge that sexual health is a significant concern for women with PD. Our study revealed that 67% of participants considered sexual health to be a “very important health issue.” Despite 85% of participants feeling comfortable discussing sexual issues with healthcare professionals, only 1% reported being asked about their sexual health by neurologists or other healthcare providers. This lack of attention is particularly troubling, as none of the participants received guidance on preventing or managing potential sexual dysfunction.
The present study has several strengths, including the use of multidimensional generic instruments to assess both short and long-term sexual health, couple's relationship quality, depression, and cognition. Moreover, disease-specific instruments were employed to comprehensively evaluate motor and non-motor dysfunction in PD.
However, the study has some limitations. A principal limitation is its cross-sectional design, which provides information at a single point in time. This limitation was partially mitigated by using various tools to assess short-term sexual health and long-term sexual health. Another limitation is the absence of motor examinations to measure disease severity, although self-reported motor disability levels provided some insight. The sample size may also be insufficient to generalize findings to the entire PD population. Nevertheless, including participants from five different geographical areas with significant sociocultural diversity within a large country helped address this limitation. Lastly, the lack of women with advanced PD stages who reported an active sexual life could be considered a limitation of the study.
The study did not aim to compare sexual health between women with and without PD. Instead, we focused on identifying factors that negatively affect sexual health in women with PD, including motor, non-motor, and social factors. Future studies should compare sexual health between women with and without PD.
Conclusion
Numerous factors, including social, motor, and non-motor domains, can negatively impact the sexual health of women with PD. Among these, the quality of their relationships and sleep are particularly significant.
The findings from this study offer valuable insights that can guide the development of interventions aimed at enhancing sexual health in women with PD. By identifying critical factors such as the quality of relationships and sleep disturbances, which play a significant role in affecting sexual health, the study provides a foundation for more tailored and effective therapeutic approaches.
Footnotes
Acknowledgments
This article was produced as part of the activities of the FAPESP Research, Innovation, and Dissemination Center for Neuromathematics (grant #
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by FAPESP Research, Innovation and Dissemination Center for Neuromathematics (Grant number: #2013/07699-0, São Paulo Research Foundation). This article was funded in part by the Coordination for the Improvement of Higher Education Personnel, CAPES, Brazil and National Council of Technological and Scientific Development, CNPq, Brazil (Grant number: 303359/2022 303359/2022 6).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available upon request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions
