Abstract
Background
Motor dysfunction in Parkinson's disease (PD) is characterized by a loss of functioning neurons in the substantia nigra. Two options exist when encountering damaged neurons: replace or support. We implemented a strategy of using autologous peripheral nerve tissue, in a reparative state, to provide a collection of neurorestorative support to unhealthy neurons with the goal of modifying the motor progression of PD.
Objective
Report on two-year compliance feasibility, safety, and clinical experience of combining this delivery at the time of deep brain stimulation (DBS) surgery.
Methods
Participants with PD undergoing open-label peripheral nerve tissue implantation to the substantia nigra at the time of DBS surgery were followed from pre-surgery to two years after surgery through clinical evaluations.
Results
Seventeen of 18 participants who underwent the procedure completed the 2-year study visits. No study-related serious adverse events occurred.
Conclusions
The trial met its primary endpoints of feasibility and safety. We were able to practicably and safely implant participants and have participants comply with 2-year visits and exams. Adverse events related to study participation were deemed manageable by participants.
Plain language summary
Parkinson's disease (PD) causes the loss of cells within the substantia nigra, which is an area of the brain involved with movement. In this study, we used a piece of nerve from a participant's own ankle to provide proteins and factors to help sick or dying cells. The goal of the study was to understand if it would be too much of a burden to ask participants to attend study visits for 2 years and if the procedure was safe. We also did preliminary testing of whether the support provided by the nerve implant could slow or stop the disease from getting worse over time. Eighteen persons with PD who were already undergoing deep brain stimulation (DBS) surgery also received peripheral nerve implants to the substantia nigra. Seventeen out of 18 participants who received the implant completed the 2-year study visit, meaning that participants were willing to return for study visits and exams. The most common complaint that participants had was that the side of the foot or ankle where we took the nerve hurt or tingled, but after time, they did not feel the problem was very bothersome. Based on these results, we conclude that we could perform the surgery, the demands of the study were not too burdensome on participants, and that it was safe. The results from this study encourage us to perform more clinical trials on this procedure in the future.
Introduction
The continual degeneration of dopamine neurons in the substantia nigra and the resultant progression of symptoms and dysfunction continue to be a significant challenge to the effective treatment of the motor symptoms of Parkinson's disease (PD) over time. Dopaminergic medications and surgical interventions, such as deep brain stimulation (DBS), alleviate symptoms for a portion of time but do not significantly alter the course of the disease.1–3 The lack of any therapy that treats disease progression represents the greatest unmet need for patients with PD. 4
Taking on this important clinical challenge, clinicians and scientists have focused efforts on repairing damaged brain systems based on strategies that attempted to either replace lost neurons or implement interventions to support cell survival and function. Cell replacement approaches have involved the transplantation of several different cell sources including fetal cells, autologous cells, xenotransplants, and stem cells.5–16 Trials implementing cell support mechanisms have included the direct delivery of gene therapy products or neurotrophic factors.17–24 While both replacement and support strategies have theoretical and practical advantages and disadvantages, we have combined aspects of both methods as the basis of our line of investigation.
We are trialing the direct delivery of a cell therapy with the aim of providing support to the remaining neurons that are either diseased and dysfunctional or healthy and at risk for becoming diseased. For this purpose, our cell source is autologous peripheral nerve tissue (PNT). 25 PNT has a long history of use in non-clinical, preclinical, and clinical studies to provide a regenerative environment for the nervous system.8–10,26–31 Beyond its known capacity for repair in the peripheral nervous system, adult peripheral nerve tissue can survive implantation to the central nervous system and promote regenerative mechanisms. We first explored this concept in a one-year pilot study utilizing the DBS-Plus platform where 8 participants received the standard-of-care DBS plus the investigational cell therapy, in this case, PNT.32,33 Based on the overall positive results, we expanded our line of investigation to include a new set of 18 participants. This new group also received unilateral autologous PNT grafts utilizing the DBS-Plus approach for a two-year trial. We recently reported the one-year safety, feasibility, and clinical evaluations. 34 In this current report, we present the final two-year endpoint results including safety, feasibility, and clinical evaluations. Our primary goals were safety and feasibility. Our secondary and exploratory goals were clinical evaluation measures and advanced imaging measures designed to support or deter future studies that would determine the efficacy of this investigational cell-based therapy that is paired with DBS.
Methods
Study design, protocol approvals and patient consents
The study is an investigator-initiated, open-label, single-center, phase I trial (NCT02369003) designed to determine the safety and feasibility of PNT implantation in participants undergoing DBS surgery. The primary aim of the trial was to assess the feasibility and safety of combining PNT with the DBS surgical procedure while the evaluation of clinical experiences were secondary and exploratory outcomes. The study procedures were approved by the University of Kentucky Institutional Review Board, and all participants provided written informed consent before participating in the study. The design, operative procedure, and 12-month interim results were previously published 34 as was the gait analysis from a subgroup of participants. 35
Participant selection
Eighteen participants with PD underwent the DBS-Plus procedure (Figure 1). We used the following inclusion criteria: undergoing DBS of the subthalamic nucleus (STN) or globus pallidus internus, age range 40–75, able to give informed consent, a positive response to carbidopa-levodopa, no significant cognitive deficit per a formal neuropsychological exam, and able to tolerate the surgical procedure. The exclusion criteria included: previous PD surgery or intracranial surgery, or a female who was pregnant, lactating, or of child-bearing potential and unwilling to use an adequate birth control method during the period of the study.
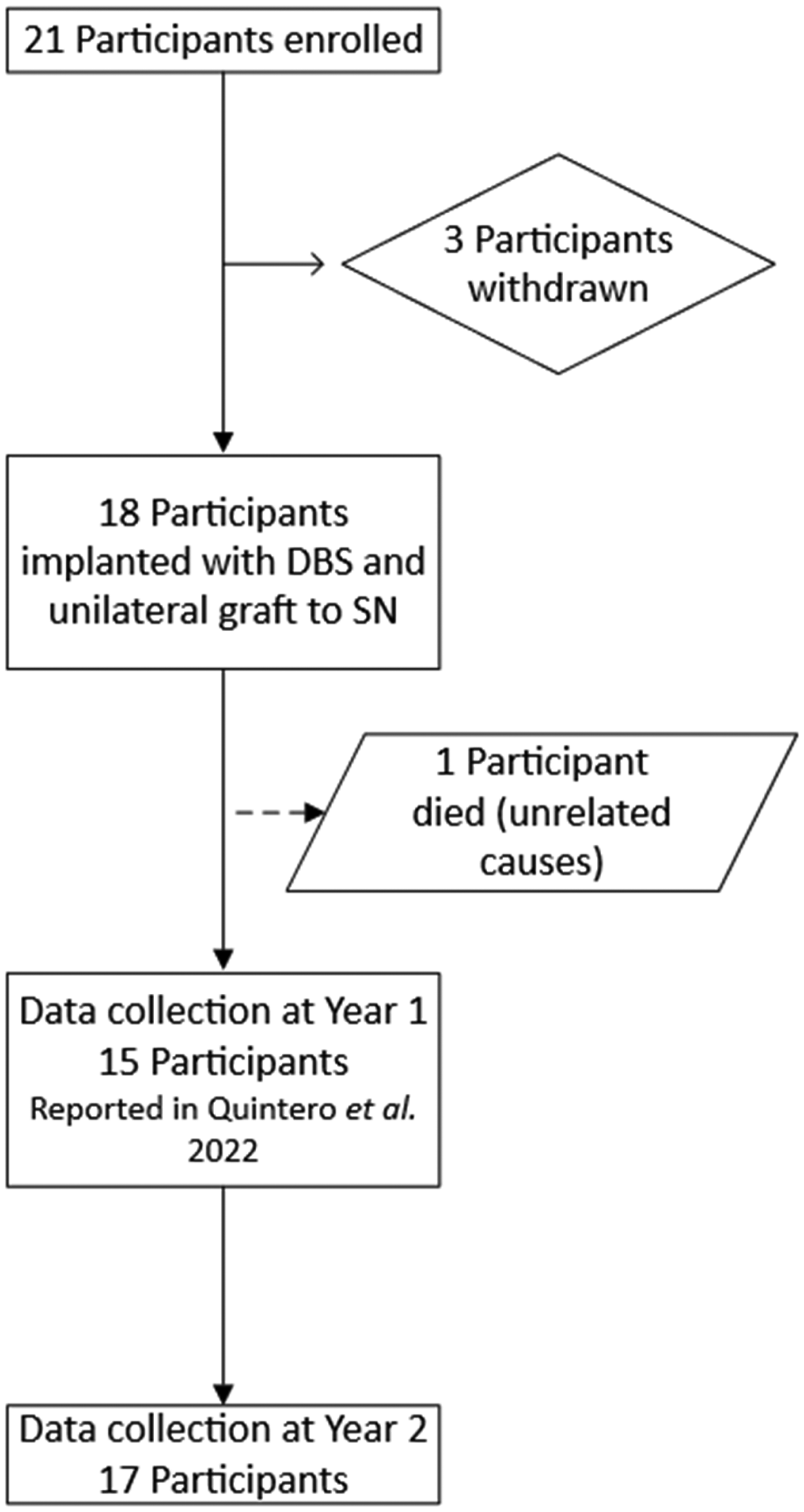
Clinical trial flow.
Surgical implantation
The surgical implantation techniques were described in our previous publications.32–34 In brief, the DBS-Plus surgery was carried out in two stages. During Stage I, the pulse generator and lead extensions were implanted as standard-of-care DBS treatment. Following hardware implantation, the sural nerve was transected at the ankle and left in situ to condition it for later implantation.
Stage II occurred 3 to 14 days after Stage I. 34 During Stage II, the DBS electrodes were implanted, secured, and connected to the extensions as standard of care. Following the DBS portion of the case, the distal portion of the transected sural nerve was harvested and PNT was implanted unilaterally to the substantia nigra contralateral to the most symptomatic side, based on Unified Parkinson's Disease Rating Scale (UPDRS) 36 lateralized scores. As such, the PNT was accessed and implanted during the DBS surgeries but separated temporally from the standard of care portion of the surgery.
Safety and feasibility monitoring
Participants were monitored for perioperative and long-term postoperative adverse events throughout the study for two years following DBS surgery. Adverse events from clinical reports, chart reviews, and clinical visits were recorded. All adverse events were submitted to and reviewed by the Data and Safety Monitoring Board (DSMB) at the University of Kentucky. Adverse events were classified as mild, moderate, or severe. Furthermore, adverse events were reported as possibly, probably, or definitely related to the graft or graft procedure and reported at the Primary Term Level based on Medical Dictionary for Regulatory Activities (MedDRA) 24.0.
Neurocognition assessments
Participants completed a clinical standard of care pre-surgical neurocognitive evaluation to determine pre-surgical neurocognitive status and eligibility for surgery. Neurocognitive evaluations included: Wechsler Memory Scale-Fourth Edition Logical Memory subtest (WMS-IV) 37 measures contextual verbal encoding, spontaneous recall after a delay, and recognition memory; Hopkins Verbal Learning Test – Revised (HVLT-R) 38 measures verbal list encoding, spontaneous recall after a delay, and recognition memory; Boston Naming Test – Second Edition (BNT-II) 39 measures expressive confrontation naming; Animal naming 40 measures the ability to generate the maximum number of words from a semantic category in a one minute time period; Phonemic verbal fluency (FAS) 41 measures the ability to generate the maximum number of words from phonemes in a one minute time period per phoneme; Wechsler Adult Intelligence Scale-Fourth Edition Digit Span subtest (WAIS-IV) 42 measures working memory, mental manipulation, and cognitive flexibility; Stroop Color and Word Test (Stroop) 43 measures verbally dependent processing speed and verbal inhibition.
Clinical assessments
As part of secondary and exploratory outcome assessments, participants were evaluated clinically using the UPDRS. Pre-operative motor tests were performed in the ON-state (ON PD medications) and in the practically-defined OFF-state (at least 12 h off their antiparkinsonian medications). Post-operative motor evaluations included ON-state (ON medication and ON DBS therapy) and practically-defined OFF-state (at least 12 h off both antiparkinsonian medications, if using medication, and DBS therapy). Participants received additional pre-operative and post-operative testing including the Parkinson's Disease Questionnaire–8 (PDQ-8), Non-Motor Symptoms Scale for PD (NMSS), and Schwab and England assessments.
Motor assessments
The UPDRS assessment was used to track participants across all four components of the UPDRS. Participants were scored by the same movement-disorder evaluators throughout the study and the evaluator was blind to the lateralized location of the implant. Participants were scored using the UPDRS rating scale at baseline, 6-, 12-, 18-, and 24-months post-operation. Data were stored in the REDCap database.
Non-motor assessments
The NMSS, PDQ-8, and Schwab and England assessments were performed at baseline and at 2 years.
Imaging
MRI data acquisition
Participants underwent MR post-operative imaging acutely (within 2 days 34 ) and 2 years after surgery. MPRAGE and SWI were evaluated for any abnormalities potentially related to the implant or the implant procedure. Images were obtained at 1.5-tesla due to MRI conditional device regulations.
123 I-FP-CIT/SPECT analysis
123I-FP-CIT/single-photon emission computed tomography (SPECT) striatal binding ratios (SBR) were analyzed for the anterior, posterior, and total putamen, caudate and total striatum of each participant's scan using DaTQUANT software (2.0, GE Healthcare, Boston,USA) to collect measurements based on a pre-defined 123I-FP-CIT VOI template. SBR was calculated based on the formula:
Analysis
Feasibility
Protocol feasibility evaluated whether participants completed an OFF-state UPDRS Part III exam at five different study visits (enrollment, 6-month, 12-month, 18-month, and 24-month).
Safety
The evaluation of the study intervention's safety was based on the go/no-go criterion that would trigger a halting of the study. Namely, three moderate-grade adverse events determined to be “probably/definitely related to the study intervention” would result in a pausing of the trial and an assessment, in conjunction with the DSMB and the bioethics committee, on whether to continue the trial.
UPDRS III (motor) scores
Mean motor scores were estimated using a linear mixed model (LMM) for a repeated measures design which assumed missing responses (7.8%) occurred at random and that the correlation among the responses at any two visits on the same participant met the compound symmetry assumption. Post hoc comparison of the estimated means (least squares means) was based on Fisher protected least significant differences procedure. The results were not changed when the data were re-analyzed using a mixed model that assumed an unstructured covariance matrix for the repeated measures (re-analysis not reported below). Mean differences and 95% confidence intervals (CI) were calculated on the 17 participants who completed the 24-month visit. Analyses were conducted on PC-SAS (9.4), IBM SPSS (New York), or JMP (14, SAS, Cary, NC) with significance determined at the 0.05 level.
Results
The primary goal of this study was safety and feasibility. Data are reported from several aspects of the study including rate of subject participation, data collection fidelity, complications, and adverse events. Secondary and exploratory goals include clinical and imaging evaluations. Clinical data were used to measure potential changes in motor performance and non-motor manifestations. 123I-FP-CIT /SPECT analysis was performed to evaluate potential changes in striatal signal intensities possibly associated to implantation.
Patient summary
Eighteen participants met the study criteria and were deemed eligible for surgery. Seventeen participants successfully completed the 2-year study protocol; one participant died after the 6-month assessment due to circumstances unrelated to the study (bowel obstruction). The average age at enrollment was Mean (Standard Deviation) 62 years old (10 years), and the mean disease duration was 10 (4) years. Participants included 13 males and 5 females. Seventeen participants received globus pallidus internus DBS and one received STN DBS.
Feasibility of protocol
We analyzed our protocol for overall feasibility with the flow of participants shown in Figure 1. We previously reported the feasibility of the surgical portion of the DBS-Plus procedure. All 18 participants who underwent surgery received bilateral DBS implantation and unilateral PNT implants. With respect to participant compliance and subject participation, 83 of 90 (92% completion) possible total assessment events were attended by all the participants (Table 1). Seventeen of the participants completed the final study visit within the designated 24-month period except for one who was not able to complete the final visit until 39 months (Table 2) due to medical and personal issues unrelated to the study.
Participant compliance.

Study visit timing.
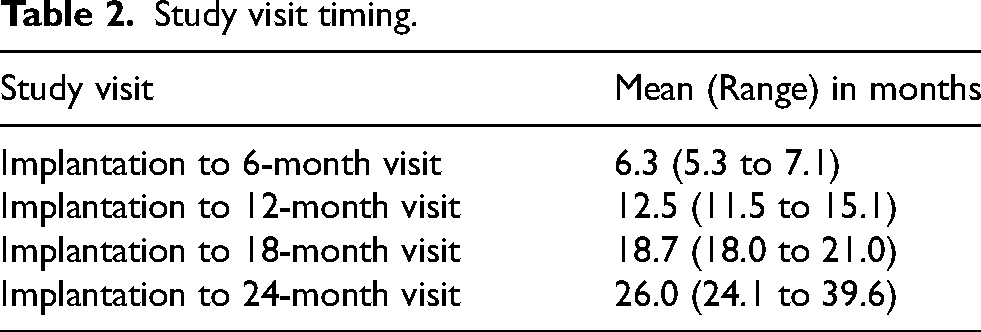
Safety and tolerability
MRI evaluation
Participants underwent MR imaging at 24 months. Three participants did not receive imaging: one participant declined due to a concern for potential side effects of the contrast agent and two developed high impedance values in an electrode contact thereby creating a contraindication for obtaining an MRI. Neuroradiologic evaluations of the images of the scanned participants did not detect any graft- or graft delivery-related abnormalities. Graft cannula trajectories were identified on both the immediate and 24-month images (Figure 2(A)).
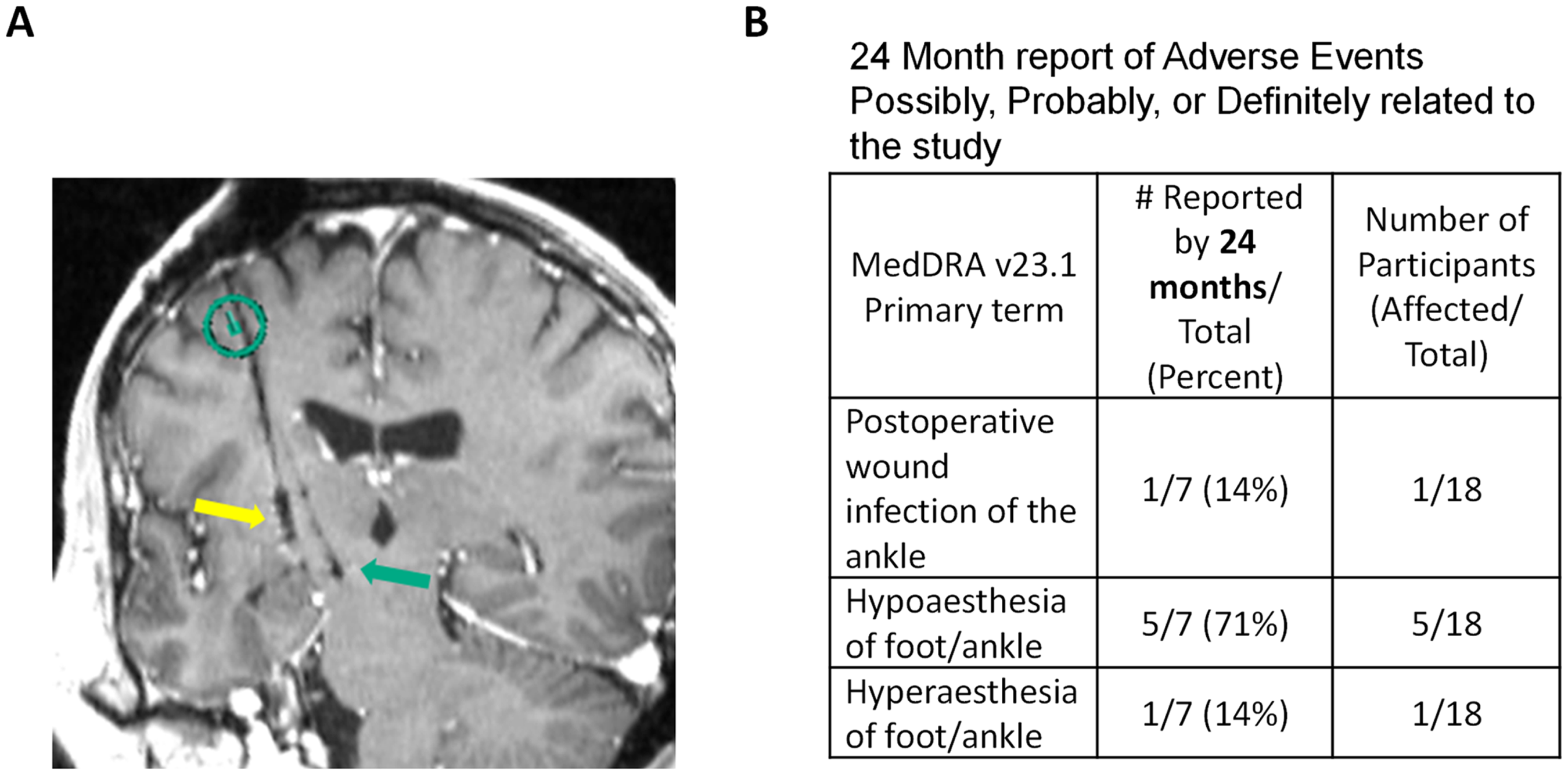
Safety-related outcomes. (A) MRI coronal section showing the PNT graft delivery trajectory (right arrow) and the DBS electrode (left arrow). The line in the circle depicts the entry of the graft cannula that was in a different plane. (B) Study-related adverse events.
Adverse events
We reported the cumulative data from study-related adverse events for the first 12 months of the study in our interim report. 34 The data presented in Figure 2(B) includes that data and any additional adverse events related to the grafting procedure through study closeout at 24 months post-implantation. Overall, the data appears the same due to the absence of any additional adverse events.
Neurocognition
Participants also completed a research neurocognitive evaluation at the end of the 2-year protocol (average of 24.9 months (SD = 2.9) post PNT implantation and bilateral GPi DBS). Results are displayed in Figure 3(A). Statistically significant reductions occurred in working memory (WMS-IV Digit Span; t(15) = 3.35, p < 0.01) and speeded color naming (Stroop Color; t(15) = 2.49, p < 0.05). Overall, these effect sizes were small to medium ranging from (d = 0.42–0.64) and all reductions were less than one standard deviation below pre-surgical values (Figure 3(B)). The one participant who received bilateral electrodes to the STN and unilateral PNT showed largely stable performance with an exception of clinically diminished verbal knowledge.
Clinical assessments
Analysis of all the motor scores (UPDRS III OFF) across study time points revealed a significant variation in the mean values (F = 6.40, d.f. = 4, 61; p < 0.002, Table 3, Supplemental Figure S1). Post hoc comparison of the least squares indicated that the mean response at each visit beyond baseline differed significantly from baseline (p < 0.004 in all cases). Further analysis revealed that there was no significant change in scores beyond the initial change from baseline at 6, 12, 18, or 24 months (p > 0.22 in all comparisons). Additional analyses of the clinical evaluations are reported in Table 3.
Clinical evaluations.
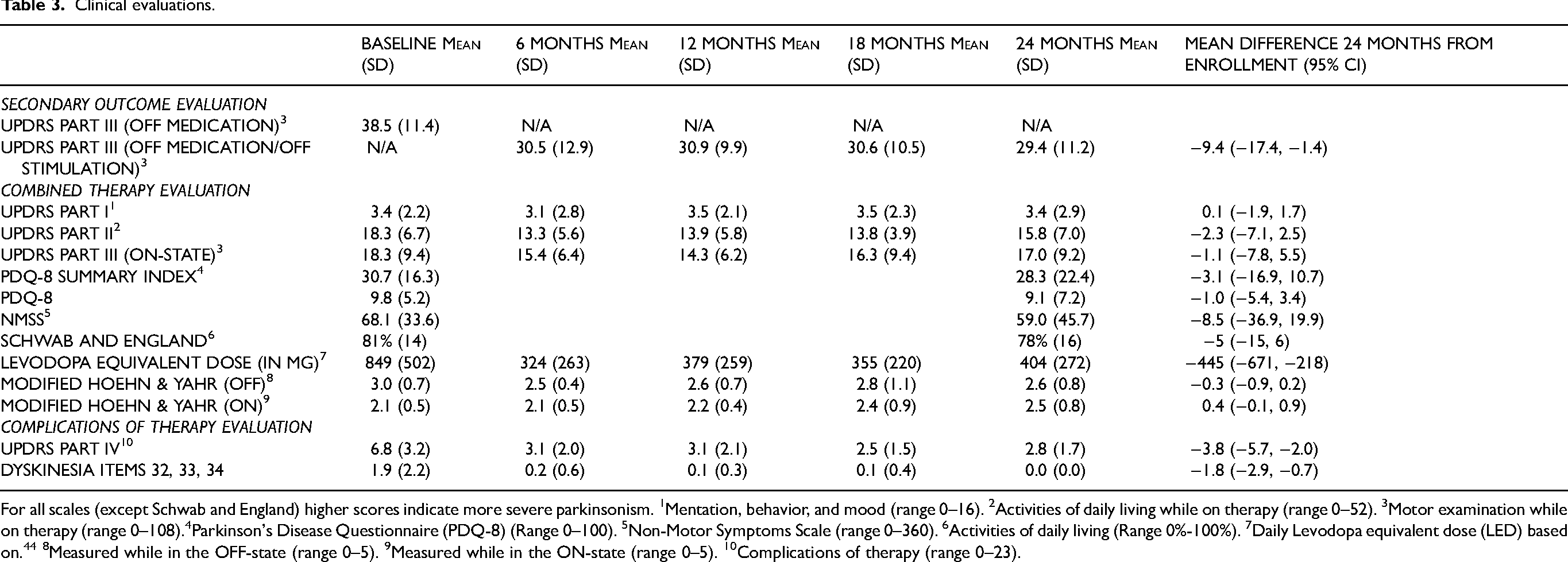
For all scales (except Schwab and England) higher scores indicate more severe parkinsonism. 1Mentation, behavior, and mood (range 0–16). 2Activities of daily living while on therapy (range 0–52). 3Motor examination while on therapy (range 0–108).4Parkinson's Disease Questionnaire (PDQ-8) (Range 0–100). 5Non-Motor Symptoms Scale (range 0–360). 6Activities of daily living (Range 0%-100%). 7Daily Levodopa equivalent dose (LED) based on. 44 8Measured while in the OFF-state (range 0–5). 9Measured while in the ON-state (range 0–5). 10Complications of therapy (range 0–23).
123 I-FP-CIT analysis
123I-FP-CIT SPECT imaging was performed at baseline and at 24 months to explore the utility of 123I-FP-CIT to monitor changes in dopamine transporter binding longitudinally to guide its use in future studies. Baseline scans confirmed that all participants who underwent grafting had imaging consistent with marked striatal dopamine depletion. We focused further analyses between baseline and 24-month images on the posterior putamen striatal binding ratio (SBR). 45 Several participants (9) had higher SBR ratios in the ipsilateral-to-the-graft posterior putamen at 24-months compared to baseline, and only four of these participants had higher SBR at 24-months on the contralateral side. While some individual participants showed a correlative change between SBR and lateralized UPDRS Part III scores over the course of the study (Figure 4(A)), other participants did not (Figure 4(B)) and as a group, there was not a significant correlation between lateralized posterior putamen SBR and UPDRS Part III changes. Mean differences between 24-month and baseline visits in the posterior putamen SBR (Figure 4(C)) were not different between the grafted (−0.032, 95%CI −0.13 to 0.07) and non-grafted side (−0.040, 95%CI −0.17 to 0.09).
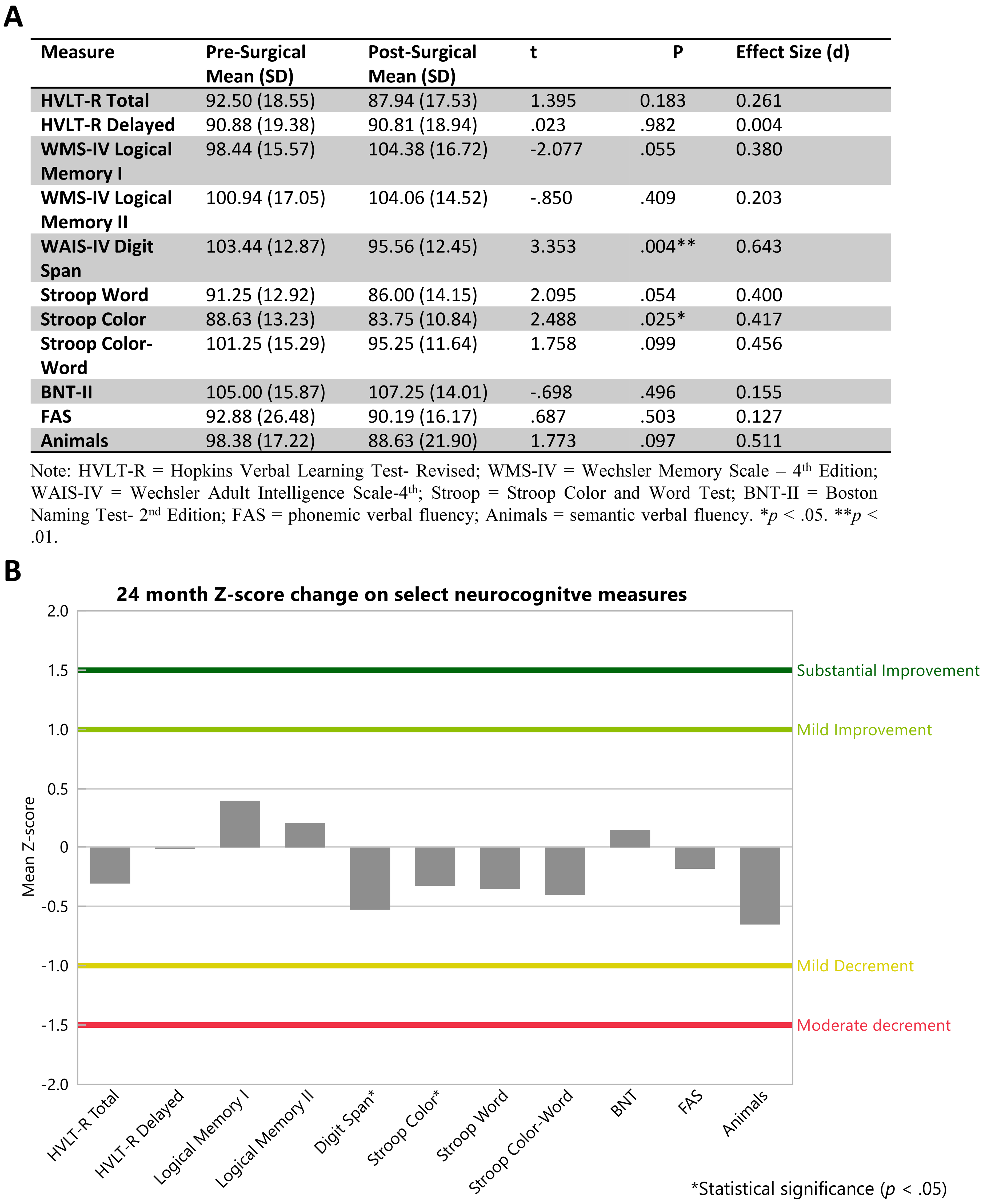
Safety-related neurocognitive outcomes. (A) Pre and post DBS-Plus PNT mean scores on select neurocognitive measures. (B) Pre and post DBS-Plus PNT Z-score changes on select neurocognitive measures.
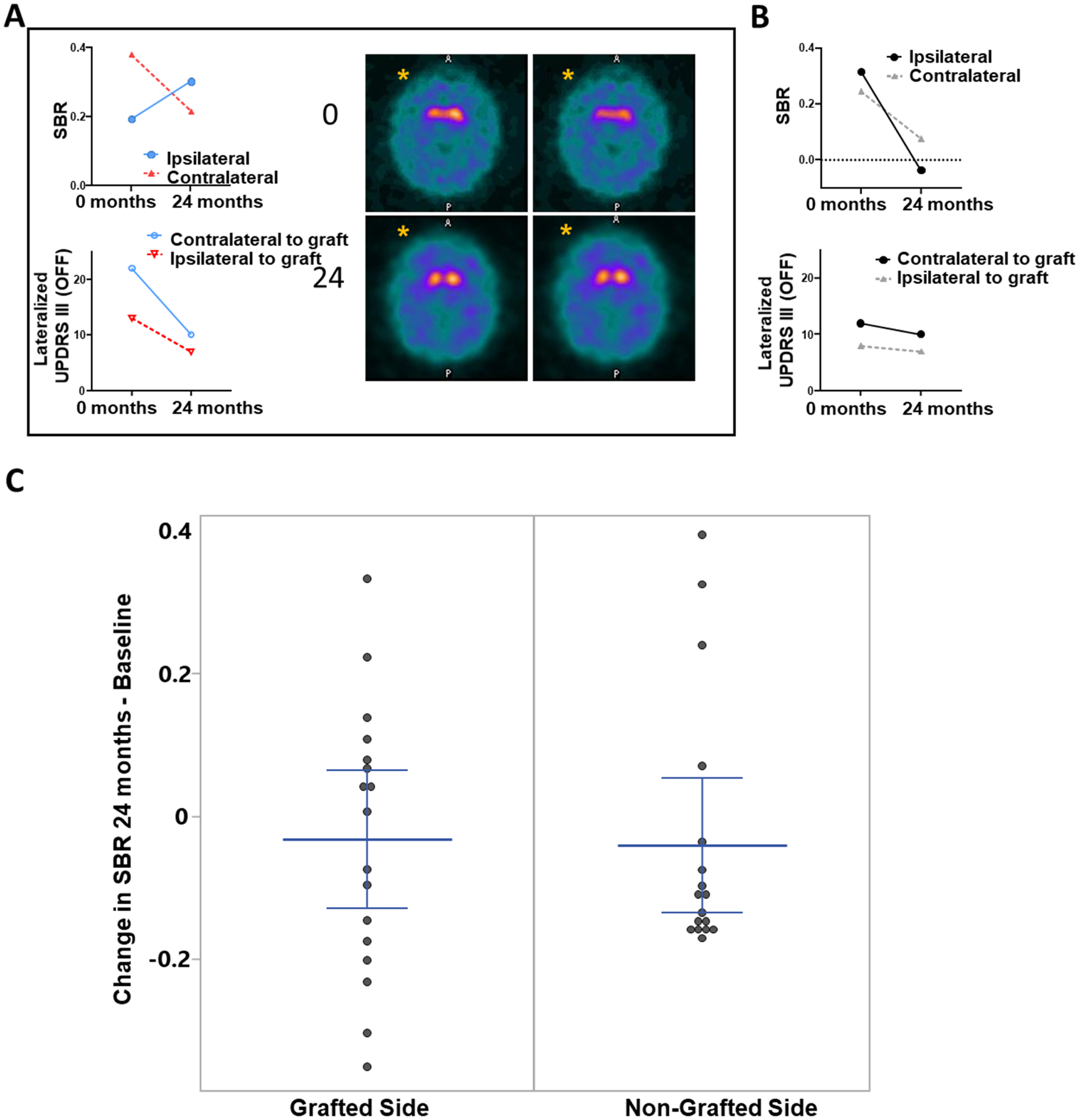
123I-FP-CIT analysis at 24 months. Exploratory outcome analysis of the changes in DAT binding at 24 months following PNT implant was inconclusive. (A) In one of several participants, the posterior putamen SBR increased at 24 months on the ipsilateral side (the graft side is indicated by an (*) in two corresponding SPECT image sections before and 24 months after surgery) while the contralateral posterior putamen SBR decreased. The lateralized scores from the UPDRS Part III showed a decrease after 24 months on the side contralateral to the PNT graft and ipsilateral to the PNT graft. (B) However, in other participants, we observed decreases in lateralized UPDRS Part III scores with decreased posterior putamen SBR as shown for one participant. (C) Mean differences (with 95%CI shown) in the posterior putamen. Nine participants showed an increase in posterior putamen SBR while four were observed on the non-grafted side.
Discussion
This follow-up report highlights the continued feasibility and safety of the DBS-Plus approach from enrollment to two years after PNT implantation and provides further analyses of feasibility, safety, and tolerability including adverse event tracking, non-motor scales, neurocognitive assessments, and MR and 123I-FP-CIT imaging. We also report the trial's clinical impact on changes in motor evaluation scores.
Feasibility of protocol
Our previous interim report 34 described the successful execution of the surgical component of the trial protocol, highlighting that the grafting procedure portion of the protocol did not interfere with the participants’ ability to receive the DBS device or the stimulation therapy. Here we present data focused on the long-term adherence to the post-procedure protocol related to clinical visits and testing. Participants completed 92% of all scheduled study visits and 17 out of 18 completed the final visit at 24 months. Participants were able to complete the UPDRS Part III assessments during these visits, despite the challenges associated with being without PD medication and stimulation for 12 h for the practically-defined OFF exam. The missed visits occurring during the interim period were related to participants relocating out of state or facing unrelated health issues. Eliciting feedback from patients highlighted challenges associated with participating in the trial. The most profound difficulty centered on travel and accommodations for our rural participants in the setting of being in the OFF-state required for testing. We have amended future trial protocols to include increased participant compensation to cover travel and accommodation expenses based on this feedback.
Safety and tolerability
No additional study-related adverse events were reported beyond the results presented in our one-year interim report. 34 MRI scans conducted at the two-year mark did not identify any indication of anomalous growth or signs of infection within the graft site, such as edema, swelling, or contrast enhancement. One limitation of the DBS-Plus paradigm is that, as was the case with two participants, problems with the DBS system can prevent the acquisition of future head MRIs used to track safety observations for cell/tissue implants. However, this is a known issue with DBS and is routinely managed. In addition, there were no new adverse events or complaints related to the graft harvest site. From a clinical standpoint, it is important to note that there were no instances of emerging OFF-state dyskinesias as has been reported in other clinical trials utilizing fetal tissue transplants. 46 To the contrary, the overall frequency of dyskinesia among our participants decreased.
Neurocognitive assessments
Despite statistically significant changes in working memory and speeded color naming reductions were small. Further, the reductions were generally reflective of typical post-surgical DBS cognitive changes observed in both subthalamic nucleus and globus pallidus interna DBS.47–53 For the one participant with DBS leads to the STN, the diminished verbal knowledge performance differred from previous findings and will warrant further investigation to determine clinical meaning. 32 When interpreted for clinical meaningfulness, none of the reductions were substantial enough for concern regarding noticeable change; in fact, all scores remained interpretable at the steadily low average to average range. Current results are thought to offer initial neurocognitive safety evidence for the DBS-Plus procedure. The DBS-Plus procedure did not appear to add incremental risk of cognitive changes beyond those expected following standard of care DBS.
Motor assessment
Overall, we found a significant reduction of the UPDRS III score in the OFF-state by 9.4 points beginning at 6 months and remaining stable throughout the end of the study. These findings are consistent with our previous study, which demonstrated a 7.4 reduction in the UPDRS III practical OFF scores at 12 months in 8 participants receiving unilateral PNT grafts and bilateral STN DBS. 32 We did not find any significant changes in the 123I-FP-CIT imaging quantification related to changes in the motor scores.
Non-motor assessment
We collected and analyzed a variety of non-motor evaluations. While most measures remained stable, critically, dyskinesias improved. This finding with dyskinesia is important in that previous studies implanting fetal tissue for PD observed a troublesome side effect in the form of a significant worsening of dyskinesia in some patients. 46
Limitations
The major limitation of this study is a byproduct of the DBS-Plus approach in measuring some of the clinical outcomes. The DBS therapy confounds any potential effect on clinical measures that cannot be obtained through the practical OFF-state testing protocol. Thus, while the UPDRS III OFF scores can be meaningfully measured in relation to disease progression, the other UPDRS measures and non-motor components cannot. Nevertheless, the additional measures are valuable as they represent the clinical state of the participants and thus can be used for monitoring safety. We did not find that including these measures was overly burdensome to the participants.
In addition to the limitation of the DBS-Plus approach, the present study is limited based on the open-label design of the trial. While raters were blinded to the lateralized location of the PNT graft, a more rigorous masking protocol will be used in future trials. Clinical trials in PD are notably prone to the influences of placebo effects.54,55 However, disease progression in the case of PD is relentless and proceeds. The current findings support sustained effects on the UPDRS OFF motor scores at the two-year time point despite the fact that normally, patients progress by 2–3 points in severity per year. 56 Thus, the findings of this study at the two-year time point encourage additional work and support the concept of future blinded clinical trials.
Summary
The trial meets the primary endpoints of safety and feasibility. Participants showed a worsening in some cognitive outcomes in line with the standard of care but no serious adverse events related to the study intervention. The surgical procedure and tissue graft implantation did not exacerbate clinical difficulties or worsen symptoms. Our findings do not show any adverse impact of the grafting protocol on the benefits of DBS therapy. With respect to feasibility, we have demonstrated that patients are willing to participate and complete the 2-year trial. The surgical procedure can be performed successfully without disrupting the ability of participants to receive DBS therapy.
Regarding the secondary evaluations, the clinical data showed a significant reduction in baseline UPDRS motor scores contralateral to graft placement. While potentially promising, it is important to keep in mind that this trial is an open-label, single-center, Phase I trial that is not designed to assess efficacy. Based on these findings, we plan to continue this line of work to investigate the potential impact of increasing the amount of tissue delivered and the potential effects of bilateral graft delivery. We successfully executed these protocols, which prepare us to determine the efficacy of cell implantation in later-stage clinical trials.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241312409 - Supplemental material for Two-year feasibility and safety of open-label autologous peripheral nerve tissue implantation during deep brain stimulation in patients with Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241312409 for Two-year feasibility and safety of open-label autologous peripheral nerve tissue implantation during deep brain stimulation in patients with Parkinson's disease by Jorge E Quintero, Monica J Chau, John T Slevin, Lisa Koehl, Julie A Gurwell, Elizabeth Wallace, Richard J Kryscio, Riham El Khouli, Amelia J Anderson-Mooney, Frederick A Schmitt, Greg A Gerhardt and Craig G van Horne in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We appreciate Ann Hanley's assistance with participant support and trial design. We thank Morgan Yazell for trial and data management support, Stephanie Morris and Renee Wagner for helping with trial execution, Jaimie Hixson and Star Manning with data processing, Lola Plum for comments on the manuscript, and Mike Hilvers for operative support.
ORCID iDs
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by gifts to the UK Neurorestoration Center; Ann Hanley Neuroscience Fund; Pro’s Players FORE Parkinson’s; the UK College of Medicine BRAIN Alliance; and the National Center for Advancing Translational Sciences, through NIH grant UL1TR001998.
Data availability
The data supporting the findings of this study can be available on request by qualified investigators from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

