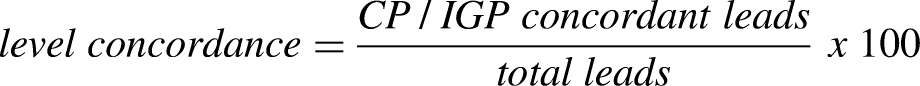Abstract
Background
Advances in STN-DBS technology, among which directional stimulation, improved Parkinson's disease (PD) treatment efficacy, while increasing the clinical programming complexity. Lead localization software may aid the stimulation contact selection process.
Objective
We aimed to assess the concordance between imaging-suggested (IGP) and conventional-programming (CP) selected stimulation contacts one year after surgery and its impact on motor outcomes.
Methods
Sixty-four PD patients with bilateral STN-DBS were enrolled. Lead localization was reconstructed with BrainlabTM software. For each electrode, the vertical contact level and, when applicable, the directionality predicted by the lead reconstruction software to be the most effective were established and compared to the stimulation parameters clinically activated one-year post-surgery. IGP/CP concordance ratio was calculated for both stimulation level and directional contacts. Post-operative modifications of PD motor symptoms severity were compared among groups of concordant and discordant IGP/CP programming.
Results
One-year post-surgery, IGP/CP concordance was 80% for active stimulation vertical contact level and 51% for directionality. No significant difference in motor outcomes was found between IGP/CP concordant and discordant patients for contact level activation, whereas patients with concordant IGP/CP active directional stimulation (c-Direction) showed superior motor outcomes at one-year follow-up than those discordant (d-Direction) (UPDRS-III stimulation-induced improvement: c-Direction = −25.66 ± 13.74 vs. d-Direction = −12.54 ± 11.86; p = 0.011).
Conclusions
Visual reconstruction software correctly predicted the most clinically effective stimulation contact levels in most patients. Imaging therefore facilitates classic STN-DBS clinical programming while assuring similar outcomes. Moreover, better motor outcomes were reached by patients with concordant IGP/CP directional parameters, suggesting that visualization can represent an added value in particular for directional stimulation programming.
Keywords
Introduction
Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is an established, highly effective therapy for Parkinson's disease (PD).1–3 Clinical outcomes strongly depend on pre-operative, intra-operative and post-operative factors. 4 Among these, a correct programming of the neurostimulator is crucial.
To date, conventional programming (CP), consisting of clinical evaluation of motor symptoms and side effects throughout monopolar review, is the gold standard for stimulation parameters selection. 4 However, this assessment is stressful and time consuming for both patients and clinicians and monopolar review demands specialized training and is affected by the subjective evaluation of fluctuating symptoms. Recent advances in DBS technology, such as directional stimulation systems, provided increasingly sophisticated programming tools to meticulously shape the target stimulation. Directional leads have the two middle rings divided into three segments. In contrast to conventional stimulation, which is delivered to the entire ring, selecting a segment creates an axially asymmetric electric field. While this patient-tailored configuration extends the therapeutic window by helping to avoid the stimulation of unwanted structures neighboring the STN, the consequent increase in potentially available combinations of parameters makes the clinical evaluation of each one of them virtually impossible. 5 In this light, visualization of the electrodes’ placement based on the patient's anatomy through 3D-reconstruction software can simplify the programming session, limiting the clinical assessment to the in-target contacts.6,7 Moreover, imaging guided programming (IGP) can help clinicians to avoid stimulation-induced side effects and was reported to yield comparable motor outcomes despite reduced programming time as compared to CP. 8 The extent of the agreement between the clinical-based and imaging-guided approaches to the choice of the stimulating contacts was reported in few previous works with limited sample size, however it remains to be further elucidated, along with its clinical relevance (characteristics and findings of such previous studies are summarized in Supplemental Table 4).9–12
Our study aimed to assess the concordance between the best stimulation contact selected through CP and IGP in PD patients with STN-DBS and its relation to the clinical outcome at 1-year follow-up.
Methods
In this retrospective observational cohort study, consecutive PD patients who underwent bilateral STN-DBS at a tertiary care university hospital (Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Rome, Italy) were screened to be enrolled in the study.
Inclusion criteria were diagnosis of PD according to the United Kingdom Parkinson's Disease Brain Bank criteria, 13 fulfillment of the inclusion and exclusion criteria proposed by the core assessment program for surgical interventional therapies in Parkinson's disease panel, 14 treatment with bilateral DBS of the STN, active follow-up after one year since DBS implant, and availability of lead placement reconstruction. Exclusion criteria were suboptimal pre-operative MRI/post-operative CT quality interfering with 3D reconstruction protocol and unavailability of stimulation parameters at 1-year follow-up.
After the intervention, all patients were followed up at Fondazione Policlinico Universitario Agostino Gemelli IRCCS, in collaboration with other Neurology Clinics based in Lazio region with expertise in the management of DBS for movement disorders.
The study was approved by the local Ethics Committee and all enrolled patients provided their informed consent.
Surgery
The leads were placed using the NexFrame system (Medtronic, Inc., Minneapolis, MN).15,16 Targeting was performed using the StealthStation S8 (Medtronic). Electrodes placement was monitored intraoperatively with microelectrode recordings and O-arm 3D fluoroscopic imaging system (Medtronic). The number of tracks performed and track placement depended on the electrophysiological findings from the previous track. Unit recordings were performed with a Leadpoint 4 system (Medtronic). Intraoperative microstimulations were used to assess the therapeutic as well as side effects thresholds. After targeting the correct location, DBS leads were implanted. All implanted leads had four electrode levels, either with all four full-ring shaped contacts (Medtronic 3389) or with two annular contacts at the proximal and distal level and two central electrode levels with three segmented contacts each (Boston Scientific Cartesia DB2202; Abbott/St Jude directed 6172; Medtronic Sensight B33015), except for one patient who had an eight-level non-directional electrode (Boston Scientific Standard DB2201). The implantable pulse generator placement was performed under general anesthesia after lead placement.
Lead localization visual reconstruction
All patients underwent pre-operative 1.5 Tesla magnetic resonance imaging (MRI) (1mm-sliced whole brain, T1-weighted sequences + T2-weighted sequences covering from the top of thalamus to base of substantia nigra). within a 3-month window before the procedure. Post-operative, thin sliced (as per lead localization software-specific instructions: acquisition slice thickness ≤1 mm with no gap between slices; reconstruction slice thickness ≤ 0.625 mm; soft tissue reconstruction kernel) computed tomographies (CT) were obtained at least 3 months after surgery to avoid brainshift artifacts. Anatomical segmentation of brain structures and leads placement were obtained through Brainlab Elements software (Brainlab AG, Munich, Germany). This software locates the electrodes’ position within the patient specific anatomical segmentation of the basal ganglia through three consecutive steps (total duration of the process: 40–50 min):
Image Fusion (Brainlab AG, Munich, Germany): automatically coregisters patient's preoperative MRI and postoperative CT; Elements Anatomical Mapping (Brainlab AG, Munich, Germany): visualizes patient specific anatomy and runs an MRI-based subcortical segmentation; Elements Lead Localization (Brainlab AG, Munich, Germany): detects the leads’ localization and, when applicable, orientation based on postoperative CT artifact and provides 3D visualization of lead contacts within the patient's own anatomy of target structures.
All steps were visually inspected by our team and minor corrections to the automated process output were performed when needed. For directional systems, orientation of the marker was defined by Brainlab according to CT artifact and was further investigated with two orthogonal projections skull X-ray in case of automatic orientation failure. Nonetheless, directional electrodes whose orientation could not be unequivocally established (i.e., not clearly visible marker artifact) were excluded from the directionality concordance ratio calculation with stimulation parameters at the 1-year follow-up, and concordance was only assessed for the contact level. The electrode level localized into the STN by the 3D reconstruction was considered the best according to IGP (Figure 1); if two levels were localized inside the STN, they were both considered IGP-optimal, as in a previous work.
10
For directional systems, the contact expected to yield the best motor outcome was visually identified as that aiming to the dorsolateral portion of the STN, whose stimulation allows for the greatest motor improvement.17,18 Two independent raters (L.R and F.B.) visually inspected the lead localization as reconstructed within the patient's-specific subcortical anatomy by BrainlabTM and identified the IGP-suggested contact as that residing inside the STN; uncertainties or disagreements were solved by discussion with a senior Author (C.P.). For patients with bipolar or double monopolar configurations activated at the one-year follow-up, we considered the electrode to be CP/IGP concordant when >50% of the VTA, as simulated by the BrainlabTM pipeline according to the ongoing stimulation parameters at the follow-up visit, resided within the STN. Similarly, in patients with active directional stimulation concordance between the CP-chosen and the IGP-predicted best contact was assessed. In all cases, stimulation settings were chosen in the follow-up by clinicians blinded to the software-based lead localization.
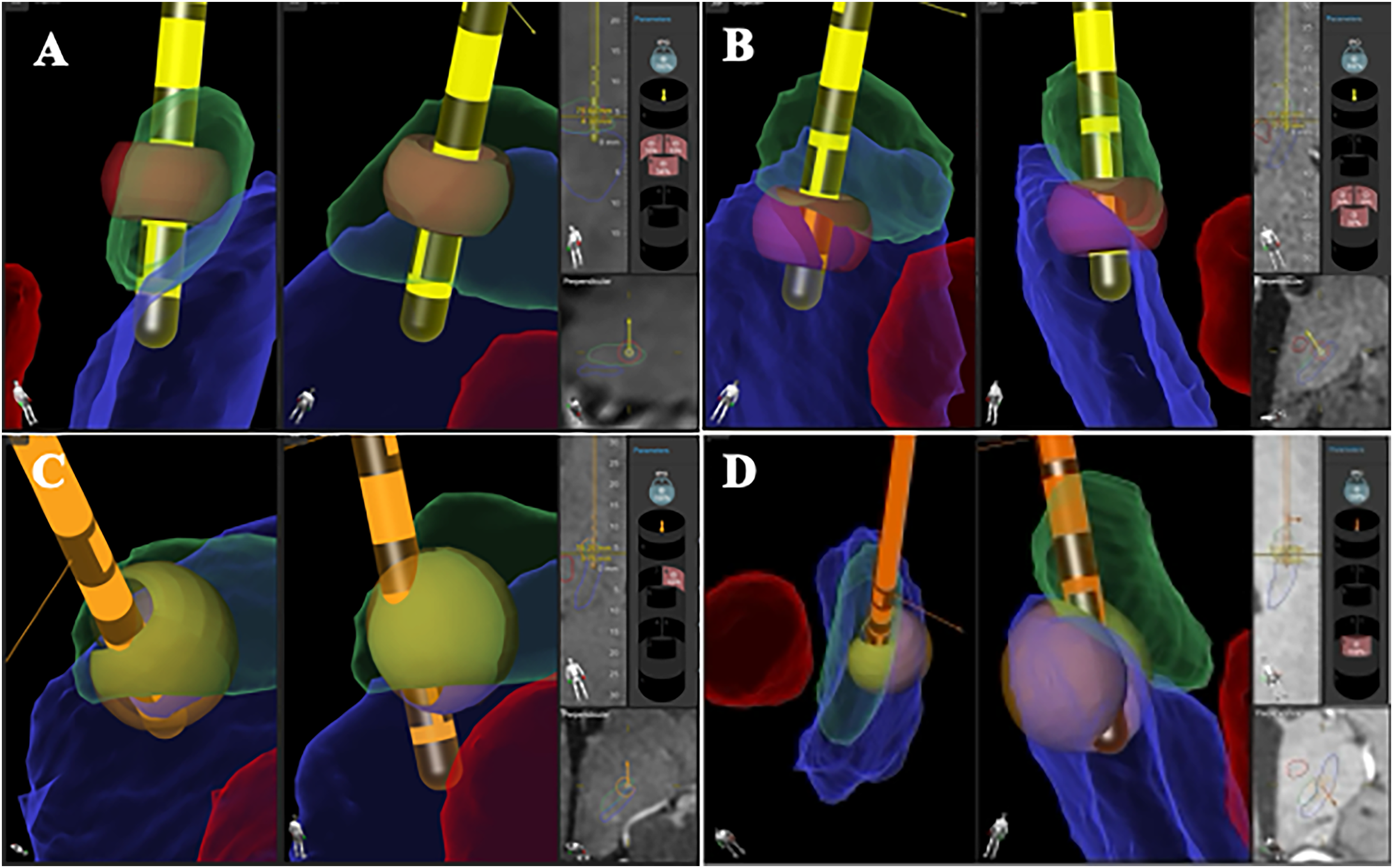
Examples of assessments of level contact and directionality CP/IGP concordance from our cohort. Lead and contact position in the 3D anatomical reconstruction of the basal ganglia anatomy and estimation of stimulated tissue as displayed in GuideTM XT software (Boston Scientific, Marlborough, MA). Green: subthalamic nucleus, red: red nucleus, blue: substantia nigra, yellow: stimulated tissue. (A) Concordance between CP-activated and IGP-suggested stimulation contact level; (B) Discordant CP-activated and IGP-suggested stimulation contact level; (C) Concordance between CP-activated and IGP-suggested stimulation directionality; (D) Discordant CP-activated and IGP-suggested stimulation directionality.
Clinical assessments
Initial programming was performed through monopolar review by movement disorders specialists, blinded to the 3D lead localization visual reconstruction, one to three weeks after surgery to minimize the impact of transient lesion effect on stimulation contact selection.
The following clinical data were collected both at baseline and one year after surgery: neurological examination, levodopa equivalent daily dose (LEDD) calculation, 19 Hoehn and Yahr (H&Y) score, 20 Unified Parkinson's Disease Rating Scale (UPDRS) 21 parts I-II-IV, UPDRS III and its axial sub-score (items 27–31) in on- and off-medication states. In addition to the aforementioned clinical variables, at the follow-up evaluation also data regarding the stimulation therapy were collected, namely the UPDRS III and its axial sub-score evaluated sequentially in each of the four possible conditions (off-medication/on-stimulation, off-medication/off-stimulation, on-medication/off-stimulation, and on-medication/off-stimulation) and the active stimulation parameters. All evaluations performed at both the baseline and follow-up visits took place on the same day. The off-medication states were assessed after prolonged dopaminergic therapy withdrawal (>12 h for levodopa-based formulations, >24 h for all other antiparkinsonian drugs). The on-medication state was assessed at the baseline evaluation after levodopa challenge (150% of the morning dopaminergic intake), and the same dose of levodopa was administered at the post-surgery visit to assess the on-medication conditions. At the post-DBS evaluation, on-stimulation conditions were assessed with the ongoing activated parameters while the off-stimulation states were assessed 30 min after switching off the DBS system.
Primary and secondary outcomes
Primary outcome of the study was to assess the rate of agreement between the activated contacts through CP at 1-year follow-up and those expected to be the best stimulation contacts according to visualization software reconstruction.
Secondary outcome was to assess the differences in motor outcomes between the two subgroups of patients with optimal vs. suboptimal stimulation settings according to 3D visual reconstruction, comparing clinical data at baseline and one year after surgery.
Statistical analysis
Contact level and directionality CP/IGP concordances were calculated as percentages through the following formulas:
Clinical variables at baseline and follow-up were compared with the paired-samples Wilcoxon-signed rank test. Between groups comparisons of the clinical variation between two time points (Δ variable = follow up value - baseline value) were carried out with the Student's t-test or Mann-Whitney test, as appropriate, while chi-square test was employed for categorical variables. All statistical computations were two-tailed, and a p-value < 0.05 was considered significant. The statistical analyses were performed using JASP (version 0.18.3, jasp-stats.org).
Results
Study population
Seventy-five PD patients implanted with bilateral STN-DBS were enrolled. Eleven patients were excluded due to suboptimal quality of neuroradiological assessments. The remaining 64 patients, accounting for 128 total leads, were included in the final analysis (Figure 2). Sixteen patients had a Medtronic 3389 non-directional system, whereas 47 subjects had a directional DBS implant: among those, Abbott directional, Boston Scientific Cartesia and Medtronic BA30035 leads were implanted in 8, 36, and 3 patients, respectively. One subject had a mixed stimulation system, with a directional Boston Scientific Cartesia lead and a Boston Scientific Standard octopolar non-directional lead in the right and left STN respectively. We reported one lead replacement due to infection; no intracranial hemorrhage or other severe adverse events were observed in this cohort. Clinical and demographic features of our study population at baseline and 1-year follow-up timepoints are summarized in Table 1. In our cohort, mean age at DBS implant was 57 years with a mean disease duration at time of surgery of 11 years; the male/female ratio was roughly 1:2. One year after DBS implant a significant stimulation-induced improvement of motor symptoms severity (mean ± SD, baseline UPDRS-III OFFmed 39.25 (± 12.92) vs. follow-up UPDRS-III ONstim-OFFmed 24.71 (± 10.55), p = <0.001; baseline H&Y 2.90 (± 0.74) vs. follow-up H&Y 2.31 (± 0.60), p = <0.001) and clinical fluctuations (mean ± SD, baseline UPDRS-IV 7.03 (± 3.41) vs. follow-up UPDRS-IV 3.21 (± 1.94), p = <0.001), as well as LEDD reduction (mean SD, baseline LEDD 1302.59 (± 482.93) vs. follow-up LEDD 832.51 (± 356.33), p = <0.001), was reported as compared to baseline (Table 1).
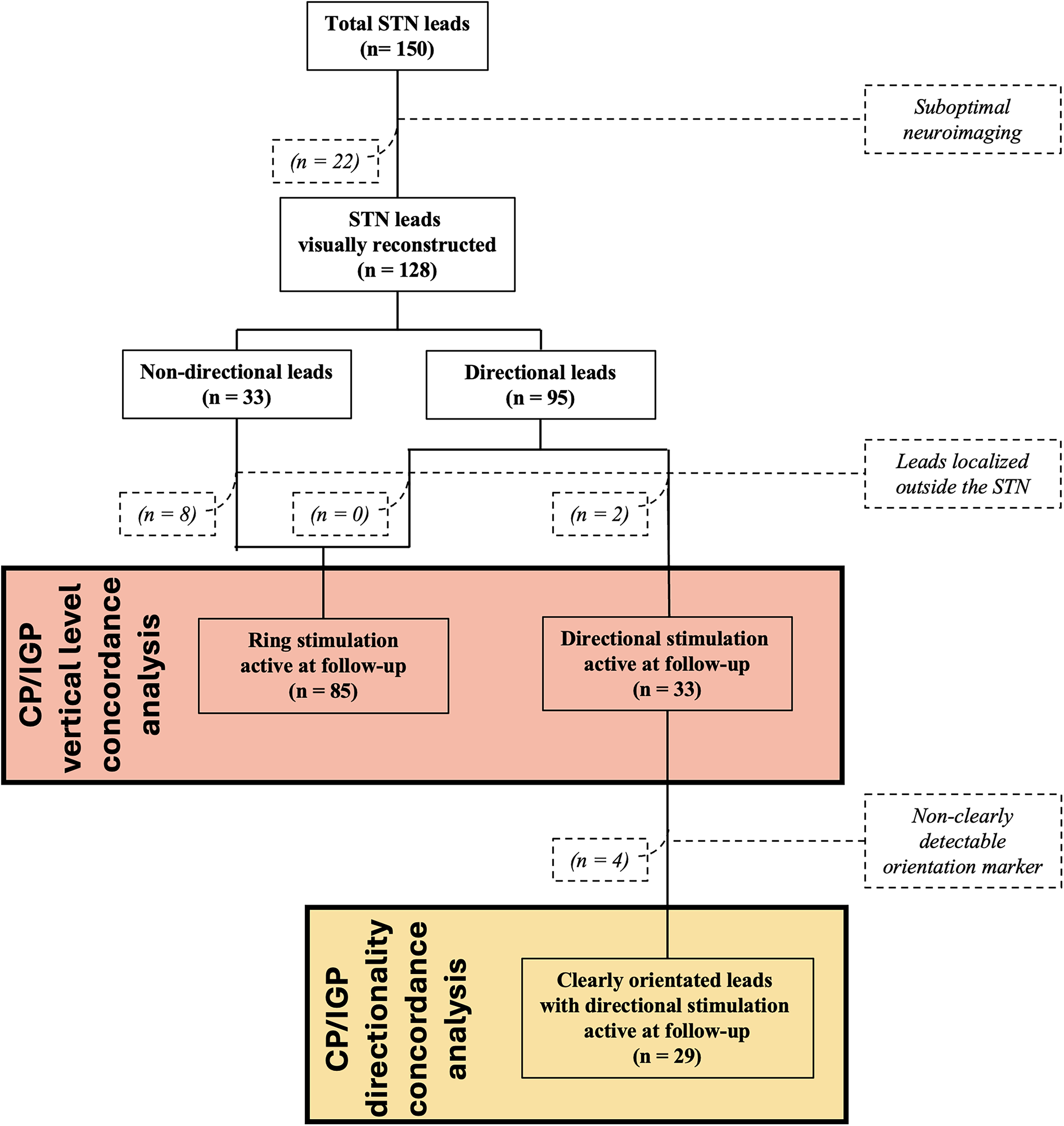
Flowchart of the study. STN: subthalamic nucleus; CP: clinical programming; IGP: imaging-guided programming.
Demographic and clinical features at baseline and one-year follow-up of the study population. TD: tremor dominant; AR: akinetic-rigid; MIX: mixed; UPDRS: Unified Parkinson's Disease Rating Scale; LEDD: Levodopa Equivalent Daily Dose.
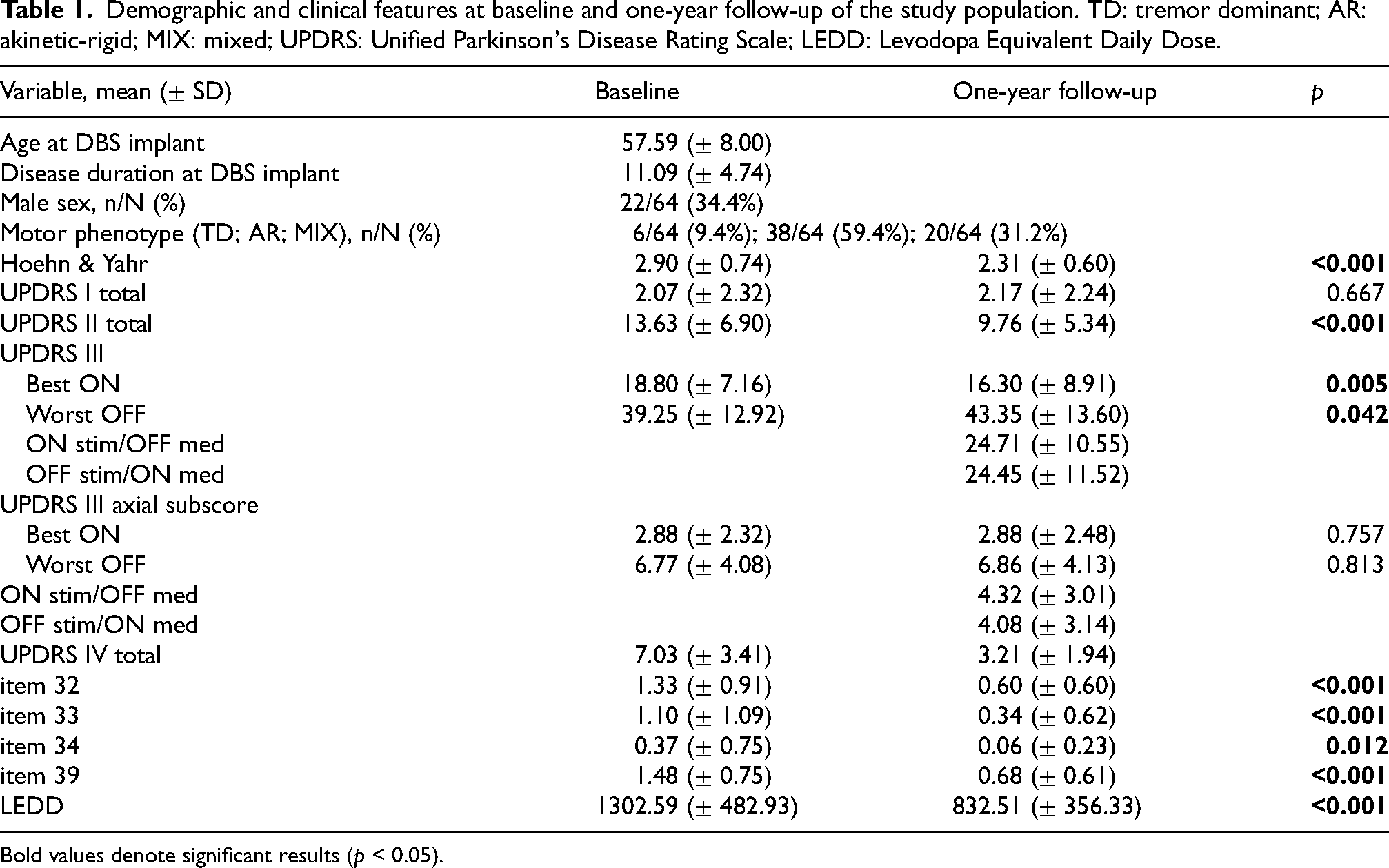
Bold values denote significant results (p < 0.05).
Stimulation parameters and visual reconstruction
Automated 3D-visual reconstruction of the lead placement was successfully carried out for all subjects. In three patients a manual refinement to the automatic STN segmentation was performed after visual inspection of the Elements Anatomical Mapping by a member of our team (LR) to better match the subject-specific nucleus anatomy. Such corrections were minor (<1 mm shift of nucleus border) and were performed with the specific BrainlbTM built-in tool (“Smart Brush”). No refinements to the other reconstruction steps (the Image Fusion and the Elements Lead Localization steps) were needed in our cohort. Visual reconstruction documented ten leads (10/128, 7.8%), belonging to total 8 subjects, localized outside the STN; such electrodes were excluded by the concordance ratio calculation and the patients were not considered in the motor outcomes analysis. Electrode levels localization could accurately be reconstructed in all cases and all subjects were included in the CP/IGP level concordance ratio analysis. In two cases disagreement between the two independent raters (LR and FB) on the determination of the IGP-identified best vertical contact level was settled by senior Author (CP) consultation.
One year after surgery, directional stimulation was used in 26 patients, accounting for 35 total electrodes. Among those, 6 leads, belonging to four patients, were excluded from the directionality concordance ratio analysis (2 electrodes localized outside the STN, 4 electrodes had non clearly detectable orientation markers); these subjects were not considered in the comparison of motor outcomes.
Active parameters at the 1-year follow-up evaluation are reported in Supplemental Table 1.
At the follow-up, bipolar, double monopolar or triple monopolar configurations were activated in 11, 21, and 2 electrodes, respectively (Supplemental Table 1). Among electrodes with these types of stimulation patterns, lead placement reconstruction visualized two contacts inside the STN in 24 leads; three electrodes resided outside the STN, while the remaining 7 leads showed only one contact inside the target nucleus.
Contact prediction concordance ratio
Among the leads considered in our cohort we observed an 80.5% (95/118) concordance between clinically-activated and reconstruction-suggested electrode level. According to the reconstruction software, two contacts resided within the STN in 69/128 electrodes as previously stated in the Methods section; in these cases both contacts were considered as IGP-suggested.
Thirteen patients presented suboptimal CP/IGP concordance (5 leads contralateral and 8 leads homolateral to the most impaired body side); the remaining 3 and 40 subjects had both electrodes either CP/IGP-discordant or -concordant respectively. As described in the Methods section, we assigned the patients with suboptimal concordance to either the CP-IGP concordant or discordant group considering the placement of the lead contralateral to the more symptomatic body side. In conclusion, according to the CP/IGP stimulation level concordance, we found 48 concordant (c-CP/IGP, 85.7%) and 8 discordant (d-CP/IGP, 14.3%) subjects (Figure 3A).
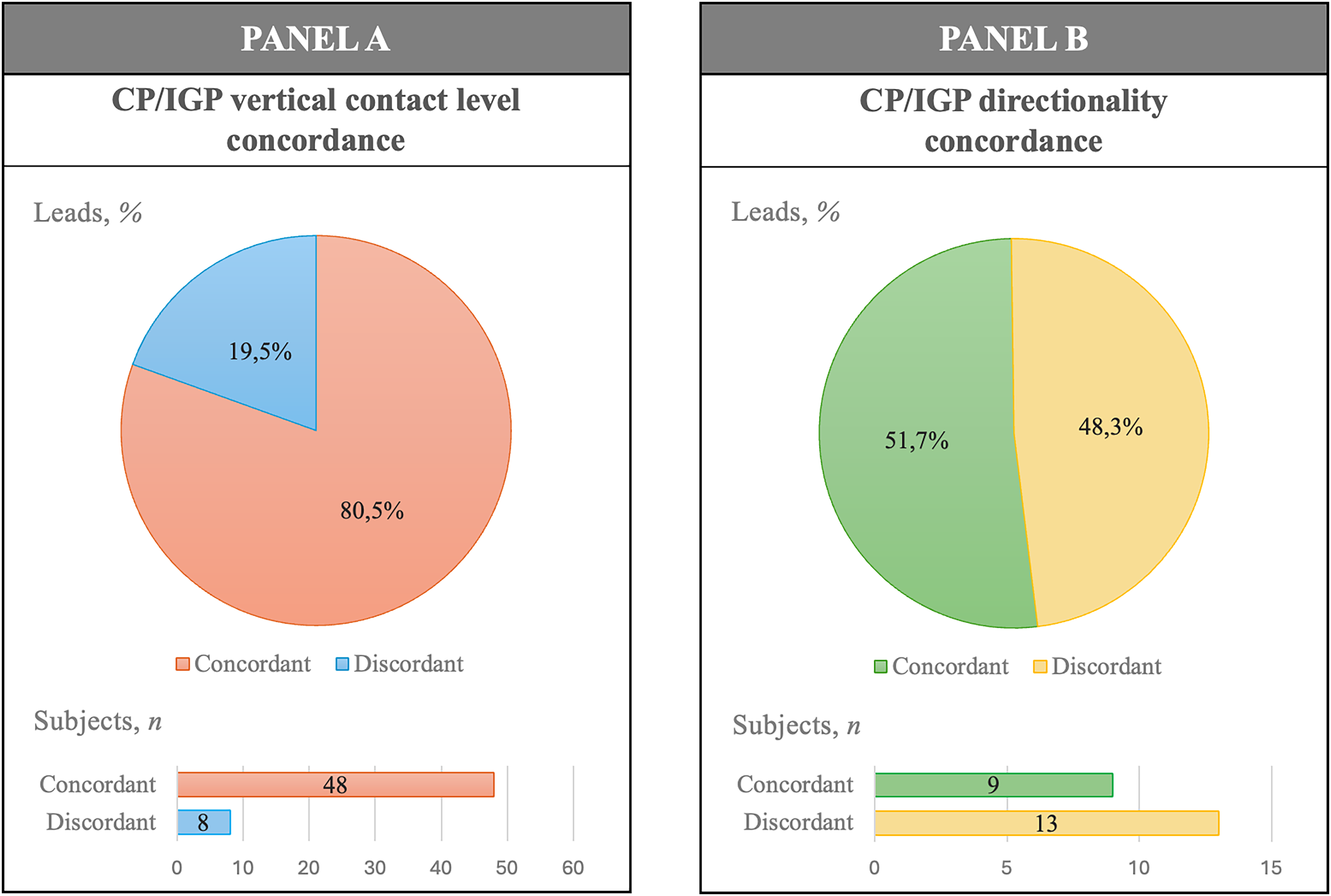
Cp/IGP concordance analysis for vertical contact level (A) and directionality (B). In each panel, CP/IGP concordance regarding leads is displayed on top while that regarding subjects is depicted on the bottom. CP: clinical programming; IGP imaging-guided programming.
Among the subset of leads with active directional stimulation parameters at the 1-year follow-up, CP/IGP agreement ratio on directionality was 51.7% (15/29). According to the reconstruction software, two directional segments pointed towards the dorsolateral STN in 10/29 electrodes: consistent to the methods used for the contact level assessment, in these cases both segments were considered as IGP-suggested. In the subset of patients with directional stimulation at the one-year follow-up, directionality was CP/IGP concordant (c-Direction) or discordant (d-Direction) in 9 and 13 subjects respectively (Figure 3B).
DBS outcomes comparison according to stimulation level concordance
No significant differences were found comparing the clinical and demographic features at baseline between the two groups of stimulation level concordance (c-CP/IGP and d-CP/IGP) (Supplemental Table 2).
No significant difference was found comparing the extent of motor improvement or LEDD reduction between the two groups (Table 2).
Comparison of modifications in clinical variables after DBS between groups of concordant and discordant patients for CP/IGP contact stimulation level. Improvement in clinical variables is expressed as the difference between post- and pre-DBS measurements (

DBS outcomes comparison according to directionality concordance
No significant difference was found comparing the clinical and demographic features at baseline between the two groups of directionality concordance (c-Direction and d-Direction) (Supplemental Table 3). We found a significantly higher stimulation-induced improvement on motor symptoms (Δ UPDRS III-OFF = UPDRS III ONstim/OFFmed – baseline UPDRS III OFFmed) in the c-Direction group as compared to the d-Direction (Δ UPDRS III-OFF = mean ± SD; c-Direction = -25.66 ± 13.74; d-Direction = -12.54 ± 11.86; p = 0.011, Figure 4). No other significant difference was found comparing the extent of motor improvement or LEDD reduction between the two groups (Table 3).
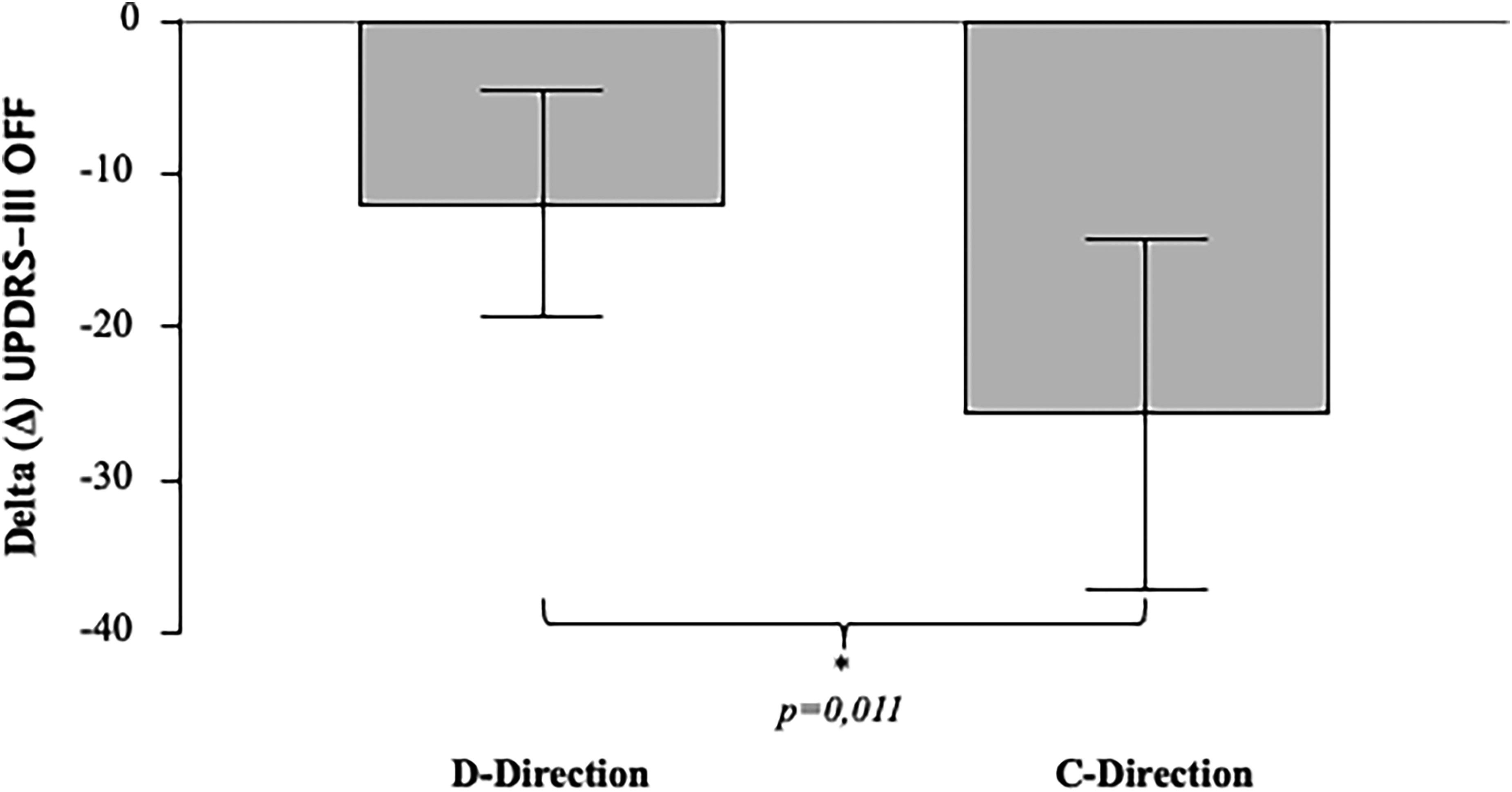
Motor outcome comparison per directionality concordance. Stimulation-induced motor improvement is expressed as Delta (Δ) UPDRS-III OFF = (post-operative UPDRS-III ONstim/OFFmed) - (preoperative UPDRS-III OFFmed). UPDRS: Unified Parkinson's Disease Rating Scale; d-Direction: discordant CP/IGP stimulation directionality; c-Direction: concordant CP/IGP stimulation directionality; * significant per Mann-Whitney test.
Comparison of modifications in clinical variables after DBS between groups concordant and discordant patients for CP/IGP stimulation directionality. Improvement in clinical variables is expressed as the difference between post- and pre-DBS measurements (Δ). CP: clinical programming; IGP: imaging guided programming; UPDRS: Unified Parkinson's Disease Rating Scale; LEDD: Levodopa Equivalent Daily Dose.
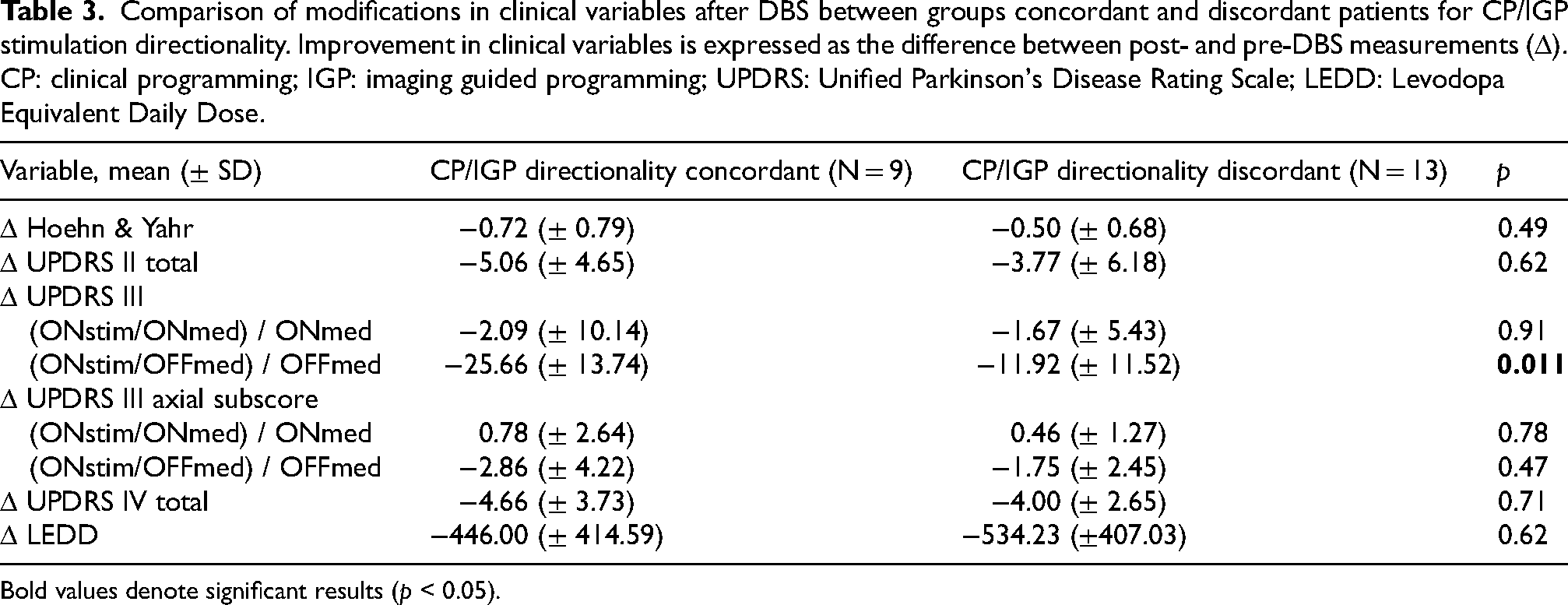
Bold values denote significant results (p < 0.05).
Discussion
To date, CP through monopolar review is the gold-standard for DBS programming in PD, despite being time consuming and stressful for patients. In this light, increasing interest lies in the development of technologies aimed to optimize the contact selection process and improve the stimulation outcomes. Among them, lead placement visual reconstruction software could be helpful in identifying the best stimulation contact, while limiting stimulation-induced side effects.8,10 However, only few studies, conducted on limited samples, previously investigated CP/IGP concordance.9,10,12,22,23 In our cohort, comprising of 128 STN-DBS leads, IGP correctly predicted the most clinically effective contact, selected by monopolar review and clinical refinement through one-year follow-up, in >80% of cases, confirming findings of the previous works. Possible explanation to the incomplete, although high, CP/IGP contact level concordance relies in the beneficial effects induced by the stimulation of structures neighboring the STN, such as the Zona Incerta and the hyperdirect cortico-subthalamic pathway, consistently reported in literature.24–27 In our cohort, a consistent proportion of CP/IGP discordant patients could be receiving a clinically effective stimulation of these structures, accounting for the decrease in concordance ratio.
Since visualization software suggested the activation of the same contact level as CP in most patients, IGP can be very helpful in clinical practice, increasing neurologists’ time effectiveness and patients’ comfort by reducing the number of clinical assessments and monopolar reviews needed to point out the most beneficial stimulation level. Moreover, IGP can be a valuable tool to reduce the impact of patients’ motor fluctuations and raters’ subjective evaluation on DBS programming.
On the other hand, in our analysis roughly half of the leads activated with directional settings at the one-year follow-up displayed agreement between the clinical- and imaging- based directionality selection. The limited concordance between clinically-established and imaging-suggested best directional stimulation contact can be partially explained by the limited number of reliably testable contacts in the outpatient clinic setting. The time burden of a full monopolar review (including directional contacts), the possible overlap of previously tested stimulation contacts due to incomplete effects washout, and the patient's discomfort in the OFF-Meds state make a fine evaluation of all possible stimulation contacts combination during routinary visits virtually impossible. In addition, in patients with electrodes correctly placed within the STN, all directional contacts might provide similar efficacy on motor symptoms improvement and clinical identification of best directional contact might rely more on side effects and patients’ stimulation tolerability rather than on steering stimulation towards the sensorimotor region of the STN.
In our study we did not find differences in motor outcomes between patients with and without IGP/CP contact level concordance. Although interpretation of this finding requires caution, the results suggest a comparable efficacy of IGP and CP. However, interesting trends of greater motor improvement were highlighted in our cohort in the CP/IGP contact level concordant group (Δ Hoehn & Yahr = mean ± SD;CP/IGP-concordant: −0.67 (± 0.70); CP/IGP-discordant: −0.19 (± 0.60); p = 0.05; Δ UPDRS III best ON = mean ± SD; CP/IGP-concordant: −3.65 (± 7.04); CP/IGP-discordant: 1.71 (± 4.31); p = 0.06). These findings suggest that better motor outcomes might be achieved by patients where CP and IGP agree on the contact selection. However, larger study samples and more balanced group sizes might be needed to confirm statistical significance. On the other hand, we reported a significantly greater stimulation-induced improvement of motor symptoms in patients with agreement between clinically-chosen and imaging-recommended directional stimulation contacts. Directional electrodes allow to expand the DBS therapeutic window by shaping the electric field in order to increase stimulation efficacy while avoiding stimulation-induced side effects.28,29 Our findings suggest that the concomitant use of directionality and visualization technologies can optimize stimulation settings in a patient-tailored manner, leading to further improvement of clinical outcomes. Previous works reported that the imaging-guided refinement of the active contact may provide some extent of further improvement in patients with suboptimal effects of DBS,8,10 and our finding suggests that visualization software might be useful particularly for patients with directional stimulation settings. The lack of significant differences between CP/IGP-concordant vs. -discordant directionality groups in the other outcome measures (e.g., UPDRS II and UPDRS IV) might be at least partially due to lower reliability of these scales. In fact, motor fluctuations and the patient's subjective perception of symptom burden have a greater impact on self-reported measures than on objectively measured scales (i.e., UPDRS III).
Globally, in our cohort safety and clinical effectiveness of STN-DBS at one-year follow-up fell within the anticipated ranges in literature,30–32 validating the accuracy of patient selection, targeting, and programming processes.
Main limitations of our study are the lack, owing to the retrospective design of the study, of an imaging-based clinical refinement in CP/IGP discordant patients. Moreover, initial activation was performed after monopolar review testing ring stimulation without evaluating all possible directional contacts, which were tested in the following months only in patients requiring directional stimulation due to clinical necessity (i.e., unsatisfactory DBS efficacy, particularly due to the onset of stimulation-induced side effects such as dysarthria or focal muscle contraction). Although possibly more representative of real-world outpatient clinic DBS programming, testing directional contacts only when clinically indicated might underestimate the further improvement sometimes achievable in a subset of patients with directional stimulation.
To overcome the previously reported limitation of visual reconstruction suboptimal quality due to brainshift and pneumocephalus artifacts, 10 all our patients performed the post-operative slim slices CT scan at least three months after surgery. Moreover, CP/IGP concordance analysis were carried out with the parameters chosen after one year of real-life repeated stimulation settings refinements through clinical follow-up. This reduces the impact of possible discrepancies between the most clinically effective contacts in the long term and those suggested by monopolar review performed in the outpatient clinic, as some stimulation induced side effects might have a delayed onset. 33 Moreover, this approach takes more extensively into account the chronic effects of brain stimulation. 34
In conclusion, our study highlights the remarkable utility of lead placement reconstruction in DBS clinical practice, in particular for directional stimulation, and its valuable supportive role to standard CP in aiming to maximize the clinical benefit while minimizing the care burden and patients’ discomfort.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241305725 - Supplemental material for Concordance between imaging and clinical based STN-DBS programming improves motor outcomes of directional stimulation in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241305725 for Concordance between imaging and clinical based STN-DBS programming improves motor outcomes of directional stimulation in Parkinson's disease by Leonardo Rigon, Francesco Bove, Alessandro Izzo, Nicola Montano, Livia Brusa, Rocco Cerroni, Alessandro De Biase, Lazzaro di Biase, Giorgio Quintino D’Alessandris, Danilo Genovese, Pasquale Maria Pecoraro, Antonella Peppe, Marina Rizzo, Alessandro Stefani, Antonio Suppa, Anna Rita Bentivoglio, Paolo Calabresi, Carla Piano and in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The original contributions presented in the study are included in the article/supplemental materials. Further inquiries can be directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
Appendix A
§ Lazio DBS Study Group:
Marco Ciavarro, CPsych: IRCCS Neuromed, IRCCS Neuromed, Via Atinense 18, Pozzilli (IS), Italy.
Francesca Cortese, MD, PhD: Neurology Unit, San Filippo Neri Hospital ASL Roma 1, Roma, Italy.
Antonio Daniele, MD, PhD: Neurology Unit, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy; Department of Neuroscience, Università Cattolica del Sacro Cuore, Rome, Italy.
Manuela D’Ercole, MD, PhD: Neurosurgery, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy.
Daniela Di Giuda, MD: Section of Nuclear Medicine, University Department of Radiological Sciences and Haematology, Università Cattolica del Sacro Cuore, Rome, Italy.
Isabella Imbimbo, CPsych: Neurology Unit, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy; Clinical Psychology Unit, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy.
Rosa Liperoti, MD, PhD: Geriatrics, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy.
Giuseppe Marano, PsyD: Institute of Psychiatry and Psychology, Department of Geriatrics, Neuroscience and Orthopedics, Fondazione Policlinico Universitario A. Gemelli IRCCS, Università Cattolica del Sacro Cuore, Rome, Italy.
Marianna Mazza, MD, PhD: Institute of Psychiatry and Psychology, Department of Geriatrics, Neuroscience and Orthopedics, Fondazione Policlinico Universitario A. Gemelli IRCCS, Università Cattolica del Sacro Cuore, Rome, Italy.
Nicola Modugno, MD, PhD: Department of Neurology, IRCCS Neuromed, Via Atinense 18, Pozzilli (IS), Italy.
Alessandra Monge, MD: Department of Neurology, St John the Baptist Hospital, ACISMOM, 00148 Rome, Italy.
Michela Orsini, CPsych: Clinical Psychology, Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy.
Mariangela Pierantozzi, MD, PhD: Neurology Unit, University Hospital “Tor Vergata”, Rome, Italy; Department of Systems Medicine, University of Rome Tor Vergata, Rome, Italy.
Camilla Rocchi, MD: Department of Systems Medicine, University of Rome Tor Vergata, Rome, Italy.
Pasquale Sanginario, MD: Department of Neuroscience, Università Cattolica del Sacro Cuore, Rome, Italy.
Fabio Viselli, MD: Department of Neurology, St John the Baptist Hospital, ACISMOM, 00148 Rome, Italy.
Alessandro Zampogna, MD: Department of Human Neurosciences, Sapienza University of Rome, Rome, Italy
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.