Abstract
Background
Freezing of gait (FoG) is among the most disabling gait disorders of Parkinson's disease. The full understanding of its mechanisms requires a network study approach. So far, FoG was mainly studied using magnetic resonance imaging, and especially using the resting state functional sequence, which does not completely reflect the brain actual modifications.
Objective
This study aims to investigate metabolic networks using position emission tomography (PET) imaging. Exploration after a rest or gait session combined with a delayed tracer's uptake are assumed to reflect the actual metabolism modifications.
Methods
Twenty-six patients in the off-drug state underwent two PET imaging sessions using [18F]- fluorodeoxyglucose, the first after 30 min of rest (rest condition) and the second after 30 min of real gait (gait condition). Twelve patients presented real FoG during cerebral glucose uptake. Brain connectivity matrices were measured for each group and condition, and then compared.
Results
In the rest condition, the freezing group showed globally reduced metabolic connectivity between brain regions compared to the non-freezing group. During gait, enhanced connectivity was observed in the cerebellum, cerebello-cortical loops and parieto-frontal regions, with high recruitment of the visual cortex in the freezing group. However, connectivity inside cerebellar networks remained lower in the freezing group than in the non-freezing group, while occipito-frontal connectivity was higher in the freezing group.
Conclusions
Studying real freezing of gait in a vertical position emphasized the role of the visual cortex and cerebellum in gait problems.
Plain language summary
Freezing of gait is among the most disabling gait disorders associated with Parkinson's disease. It is characterized by a sudden episodic inability to produce effective stepping despite the intention to walk. Brain imaging, primarily using magnetic resonance imaging (MRI), has been widely utilized to study its mechanisms. However, this imaging modality does not allow for the exploration of actual gait aspects, as images are acquired while the patient is in a supine position within the machine. In this study, we propose using nuclear medicine imaging, which allows for the assessment of metabolism over the 20–30 min following the injection of a radiotracer. Thus, even though the acquisition is also performed in a supine position, this delay is expected to reflect the actual mechanisms. Twenty-six patients were examined twice using nuclear medicine imaging: in a resting condition and after a gait session. Among the patients, twelve exhibited freezing. Images were analyzed to measure the connectivity between cerebral regions. The results showed that patients with freezing had lower connectivity between brain regions. After the gait session, some connections were re-established, but overall, the connectivity remained lower than in patients without freezing. The affected connections mainly involved the cerebellum, cerebello-cortical loops, and parieto-frontal regions, with implications for the visual cortex. Thanks to the study conducted in a vertical position, this research highlighted the roles of the visual cortex and cerebellum in gait problems.
Keywords
Introduction
Freezing of gait (FoG) is a sudden episodic inability to produce effective stepping despite the intention to walk. 1 It is one of the most disabling gait disorders associated with Parkinson's disease (PD), with prevalence increasing with disease severity. The known underlying pathophysiology is derived from clinical studies, particularly magnetic resonance imaging (MRI) ones. FoG involves an interplay of motor, cognitive, and even neuropsychiatric symptoms, such as anxiety.2,3 Freezing triggers have been identified, thus, typically, FoG rarely occurs during straight-line walking, but rather during gait initiation or gait modulation, such as turning or crossing narrow passages. Interference between a cognitive task and walking has also been reported to provoke FoG. 3 Environmental triggers can either provoke or alleviate FoG, 4 depending on their influence on the locomotor network. For example, external cueing (either visual or auditory) can alleviate FoG by altering balance and gait patterns to favor non-automated motor programs. 5
FoG is closely linked to postural instability, 6 but studying the balance and upright posture control system during walking and FoG is challenging using functional MRI (fMRI) due to the subject's static stance in the supine position. Paradigms such as gait imagination (mental motor imagery) or alternative flexion/extension movements of the feet or hands have been proposed.7–11 Virtual reality studies have reported functional decoupling of cortico-striatal circuits in the FoG phenomenon, and motor imagery studies have revealed increased recruitment of the parieto-occipital regions. 12
Deciphering the underlying mechanisms of FoG should also consider a network approach. Three main locomotor networks, known from animal and human studies, are used during walking. For standard walking in a straight line, without any obstacles and without the need to adapt the speed or direction according to cues, the network is quite direct: primary motor area, brainstem, central locomotion pattern generators (CPGs). This circuit is used exceptionally, because it does not correspond to the real conditions of walking, except in a laboratory (or alone on a wide dyke without intercurrent thought). Generally, a network involving the cerebellum is used preferentially for processing rhythmic signals, which modulates additional motor areas connected with the basal ganglia.13,14
Unfortunately, this network is impaired in PD due to damage to the basal ganglia, forcing patients to utilize other circuits, particularly the parieto-frontal attentional networks. This includes parietal input to analyze cues and premotor output capable of modulating the primary motor area, trunk nuclei, and CPGs. Deregulation of this circuit, marked by disconnection between parietal input (increased reliance on cues) and premotor output (difficulty aligning motor programs with cues), has been demonstrated in patients with FoG using MRI virtual paradigms. 12 However, the cerebellum would also modulate both parietal input and premotor output and cerebellar networks have been implicated in FoG physiopathology. 15 Understanding the role of the cerebellum, proprioceptive inputs, and the organization of metabolic networks can be better achieved in real gait and actual FoG.
The analysis of cues may also be impaired in PD patients with FoG. Visual dependency, referring to an over-reliance on visual input to maintain postural stability and navigate the environment, is often disrupted. As a result, these patients tend to depend heavily on visual cues to compensate for deficits in motor control. Several studies have explored the relationship between visual processing and FoG; PD patients with FoG symptoms exhibited slower visual processing speeds4,16 and were more prone to freezing episodes, especially in visually complex environments. 17 As a result, they tend to depend heavily on visual cues to compensate for deficits in motor control.
Common triggers for FoG include sudden visual changes, such as approaching doorways, or navigating tight spaces. 18 In these situations, the patient fails to process visual stimuli effectively, leading to a breakdown in motor planning and, ultimately FoG. However, the success of visual strategies to improve gait 19 highlights the paradoxical nature of visual dependency in PD. While an over-reliance on vision can exacerbate FOG in certain contexts, carefully controlled visual input can also serve as a powerful tool for overcoming it. This suggests that FOG in PD is not merely a result of motor dysfunction but is deeply intertwined with sensory processing abnormalities.
In a previous study, 14 we have proposed an original paradigm to investigate the neural basis of FoG using brain positron emission tomography (PET) with fluorodeoxyglucose ([18F]-FDG). This imaging modality reflects brain cell metabolism over the 20–30 min following the injection of the radiotracer. Consequently, if the subject engages in actual gait within the 30 min post-injection, the uptake would reflect brain metabolism during this motor activity, making it a valid measure of activation. 13 We focused on cognitively-preserved patients in an off-drug state to explore the motor aspects of this complex phenomenon. The study included two patients’ groups: freezers and non-freezers. We have observed deregulation of the frontoparietal attentional network (hypermetabolism in the sensory pole—paracentral lobule—contrasting with hypometabolism in the output area of the premotor cortex). Both groups activated their cerebellum and visual areas during gait, contrasting with the “rest” condition. 14
These considerations suggest that studying real walking, with the integration of complex sensorimotor, visual, vestibular signals, as well as postural, attentional, and visuospatial constraints, is feasible with metabolic imaging. PET connectivity allows the examination of interactions among different networks, similar to MRI connectivity, but focuses on metabolically active regions during real ecological walking.
In the current study, we propose to reanalyze these data by investigating the network aspects of FoG, studying brain metabolic connectivity in freezer and non-freezer patients with PD during rest and its modulation following real gait and actual FoG. This study is expected to highlight the compensatory networks activated in PD patients without FoG compared to those with FoG in a cognitively-matched population by observing the relationships between gait networks (direct locomotor network as well as compensatory networks, attentional, visuo-guided, and goal-controlled) during rest and gait with or without FoG.
Methods
Study population
Twenty-six patients with PD were recruited from the active case file of the Movement Disorders Department at Lille University Hospital (Lille, France). Among these patients, 12 had off-drug FoG, 20 with a positive answer to item 3 of the FoG questionnaire 21 and displayed FoG during a FoG trajectory 18 while the remaining patients were classed in the non-FoG group. For the inclusion of patients in the FoG group, patients were required to present FOG episodes during specific FoG trajectories in the off-drug state but not in the ‘‘supra-on-drug” state (i.e., after an acute intake of at least one and a half times the usual dose of levodopa). The two groups were matched in terms of age, sex and cognitive status.
The study was approved by the local institutional review board and each participant gave his/her prior, written, informed consent to study participation (CPP Nord-Ouest IV, 08/03/2011, n°2010-A01329-30, 14/03/2012, n°2012-27) according declaration of Helsinki. Lille Regional Hospital promoted this study and subscripted a policy (SHAM – n°127 795).
Experimental design
The patients underwent the full procedure, composed of two PET imaging session, while they were in OFF-drug condition. The two sessions correspond to the two conditions: rest and gait. A time interval of one week to four weeks separated the two acquisitions.
First, the patients were examined in rest condition. They lay still in the supine position throughout the 30-min tracer uptake and stabilization period. A 15-min PET imaging was then performed.
Second, regarding the gait condition, the patients performed FoG trajectories for the 30 min immediately following the injection of the tracer. The subsequent 15-min emission scan reflected brain metabolism during the FoG trajectories. All patients were then asked to walk and to pass through a 80 cm wide passage a few meters ahead, then they performed a full 360-degree turn to the right, a full 360-degree turn to the left, a 540-degree turn (one and a half turns) to the right, a 540-degree turn to the left, a full turn to the right as quickly as possible, a full turn to the left as quickly as possible, a 540-degree turn to the right as quickly as possible, a 540-degree turn to the left as quickly as possible, and finally, a “go” while counting down by threes (starting from a number between 100 and 200). The trajectories were carried out continuously during the 30 min between the radiotracer injection and the PET scan. The duration of the episodes was recorded in real-time by the neurologist who was present during the evaluation. The FoG trajectories were videotaped, and a second neurologist also recorded the duration of the episodes. Agreement between the two evaluations was excellent, and the average of the two evaluations was considered as the measure of total freezing time.
A FoG episode started when one of the following situations is observed: very small shuffling steps with minimal forward movement, leg trembling in the absence of effective forward motion, complete akinesia. The episode ended when the patient took an effective step with a relatively normal step length and swing phase.
PET imaging
All patients underwent a [18F]-FDG brain PET scan after 30 min of rest (“rest condition”) and another one after 30 min of real gait (“gait condition”), consisting of a succession of FoG trajectories, triggering episodes of freezing in all patients in the FoG group for around 8 min (cumulative time of FoG) out of the 30 min of gait. The protocol was described in full in. 14
Data were acquired on a GE Advance SL PET/CT device (GE Medical Systems). Patients were scanned after fasting. Blood glucose level was checked before intravenous injection of [18F]-FDG (185–198 MBq). Thirty min later, a low-dose CT scan of the brain was acquired for attenuation correction of the PET data, and emission images were subsequently acquired in 3D mode. Images were reconstructed iteratively using an ordered-subset expectation-maximization algorithm (with two iterations and 21 subsets) in a 256 × 256 resolution and a definition of 1.17 × 1.17 × 3.27 mm3.
PET images analysis
After individual intensity normalization, the images were normalized spatially according to the Montreal Neurological Institute (MNI) template (McGill University, Montreal, Canada) using an affine transformation in SPM 12 (Wellcome Department of Cognitive Neurology, London, UK). The registered images were labelled using the automated anatomical labelling (AAL) atlas composed of 116 regions of interest (ROI). Starting from the labelled images, a 116 × 116 connectivity matrix was built for each group by pairwise measurement of the correlations, using Pearson's coefficient, between the ROI mean uptake across all patients in the group (Figure 1).
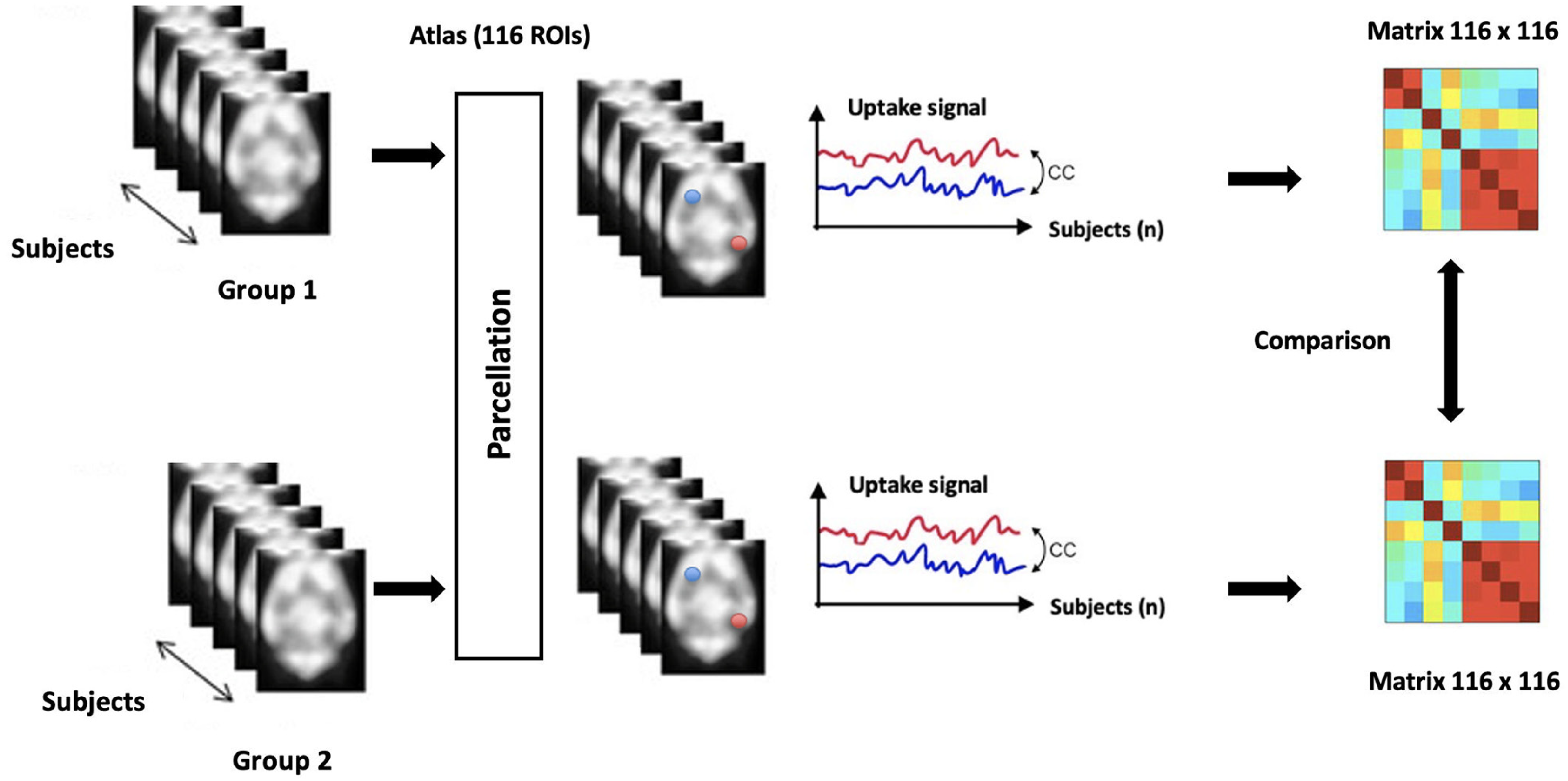
Metabolic connectivity analysis scheme: matrix building and comparison between groups and/or conditions.
Consequently, four connectivity matrices were built representing the metabolic connectivity in the two groups in the two conditions. Connectivity matrices were computed using a home-made script in Matlab (The MathWorks, Inc., Natick, MA, USA). Visualization was performed using the BrainNet toolbox. 22
Statistical analysis
For the clinical data, intergroup differences were evaluated using an unpaired Student's t test for continuous variables with a normal distribution (according to the Shapiro–Wilk test) or, in the event of a non-normal distribution, with a Wilcoxon test. A Chi-squared test was used for categorical variables.
To characterize the changes in metabolic connectivity patterns between the groups and the conditions, the corresponding matrices were compared. After Fisher's z-transformation of the correlation coefficients, analyze of variance (ANOVA) tests using permutation tests were run.
For all the analyses, statistically significance was set to p < 0.05.
Results
Intergroup comparisons (FoG vs. non-FoG) did not reveal any differences in demographic or cognitive data. There was a non-significant trend towards higher off-drug UPDRS part III scores in the FoG group. The FoG group had more axial symptoms, with higher scores for the gait item of the UPDRS part III, for the axial subscore and for the FoG-questionnaire, when compared with the non-FoG group (Table 1).
Main demographic and clinical data for the study population.
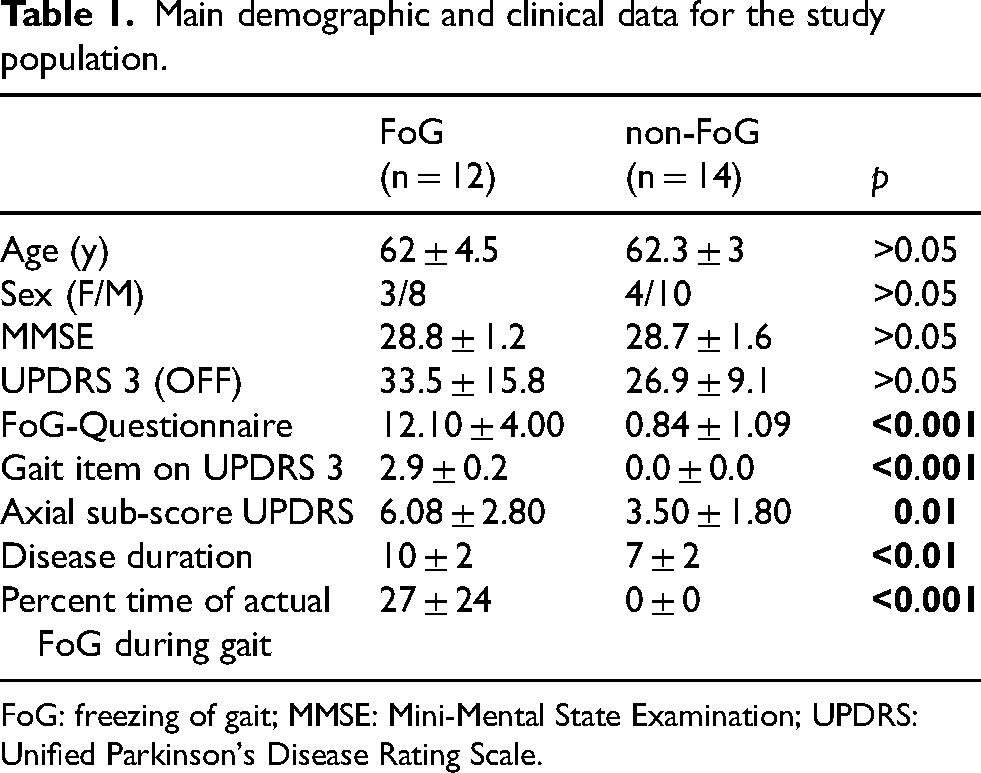
FoG: freezing of gait; MMSE: Mini-Mental State Examination; UPDRS: Unified Parkinson's Disease Rating Scale.
Figure 2 shows the mean connectivity matrices for the two groups in the two conditions. In the rest condition, the FoG group showed reduced connectivity between the brain regions compared to the non-FoG group. Statistical analysis confirmed this observation. Figure 3A shows the connections with higher correlations in the non-FoG group when compared to the FoG group in the rest condition: at rest the metabolic connectivity was disrupted in the thalamo-cortical and cerebello-cortical loops in the FoG group, as well as in the temporo-parietal networks and the frontal regions (Supplemental Table 1).
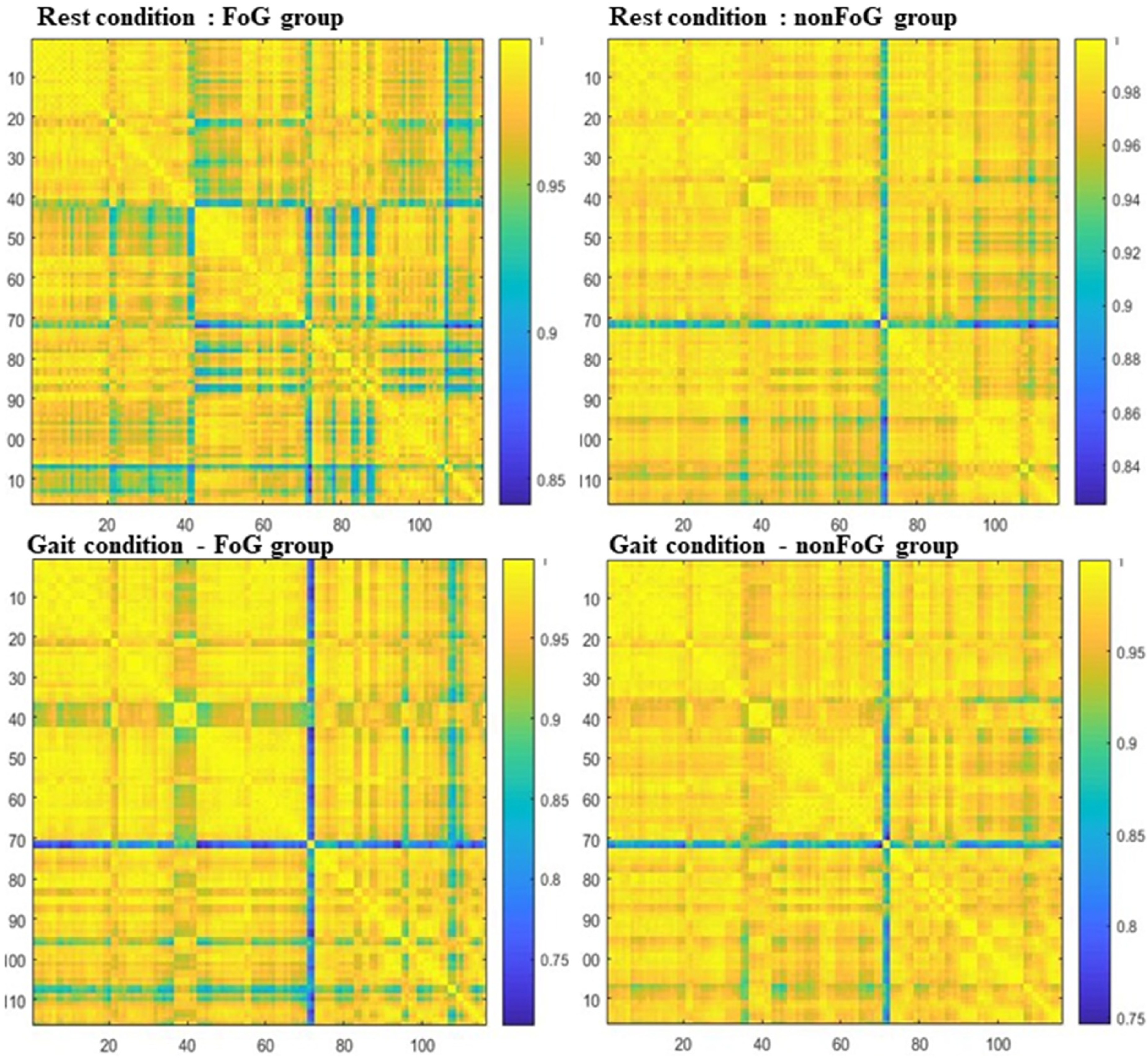
Mean connectivity matrices of two groups in the two conditions.
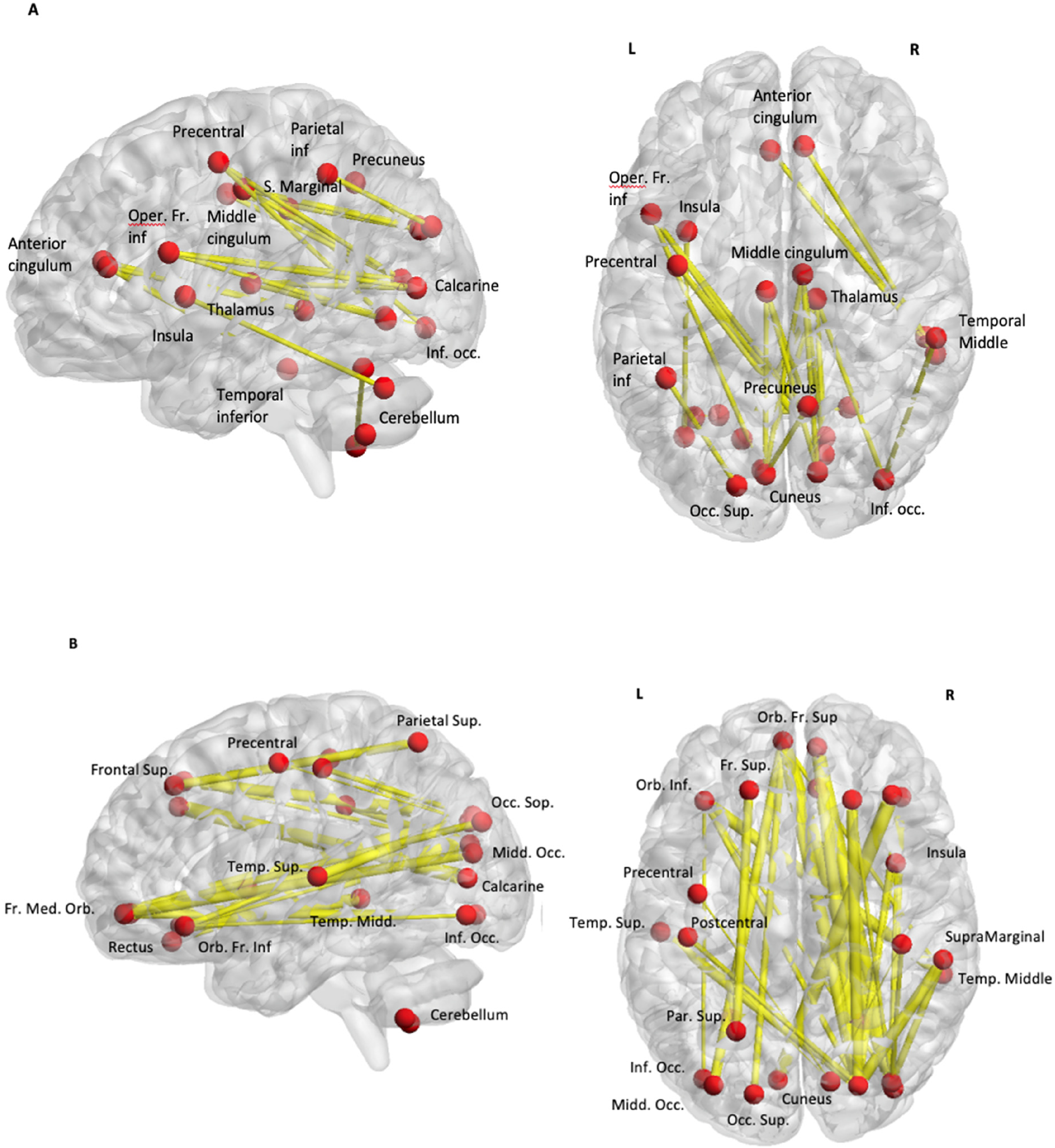
(A) Rest conditions. non-FoG > FoG. (B) FoG group: gait > rest. An exhaustive list of connections is presented in Supplemental Tables 1 and 2. Only significant covariations by contrasting two groups in rest conditions are represented (A) or conditions in the same group (B).
Between the gait and rest conditions, no significant differences were observed in the non-FoG group. Conversely, after the gait session, the FoG group partially restored these altered connections (increased functional connectivity shown in Figure 3B). During gait with FoG (in comparison to rest in FoG), a metabolic network activates, mainly involving the cerebellum, cerebello-cortical loops and parieto-frontal regions, with a high recruitment of the visual cortex (Supplemental Table 2).
However, after the gait session, the restoration of connectivity observed in the FoG group did not bring this group to the same level as the non-FoG group and differences still existed between them (Figure 4 and Supplemental Tables 3 and 4). The non-FoG group exhibited higher connectivity from the cerebellar regions and the FoG group displayed increased connectivity from the occipital and parietal regions towards the internal frontal regions (more limbic than motor ones).
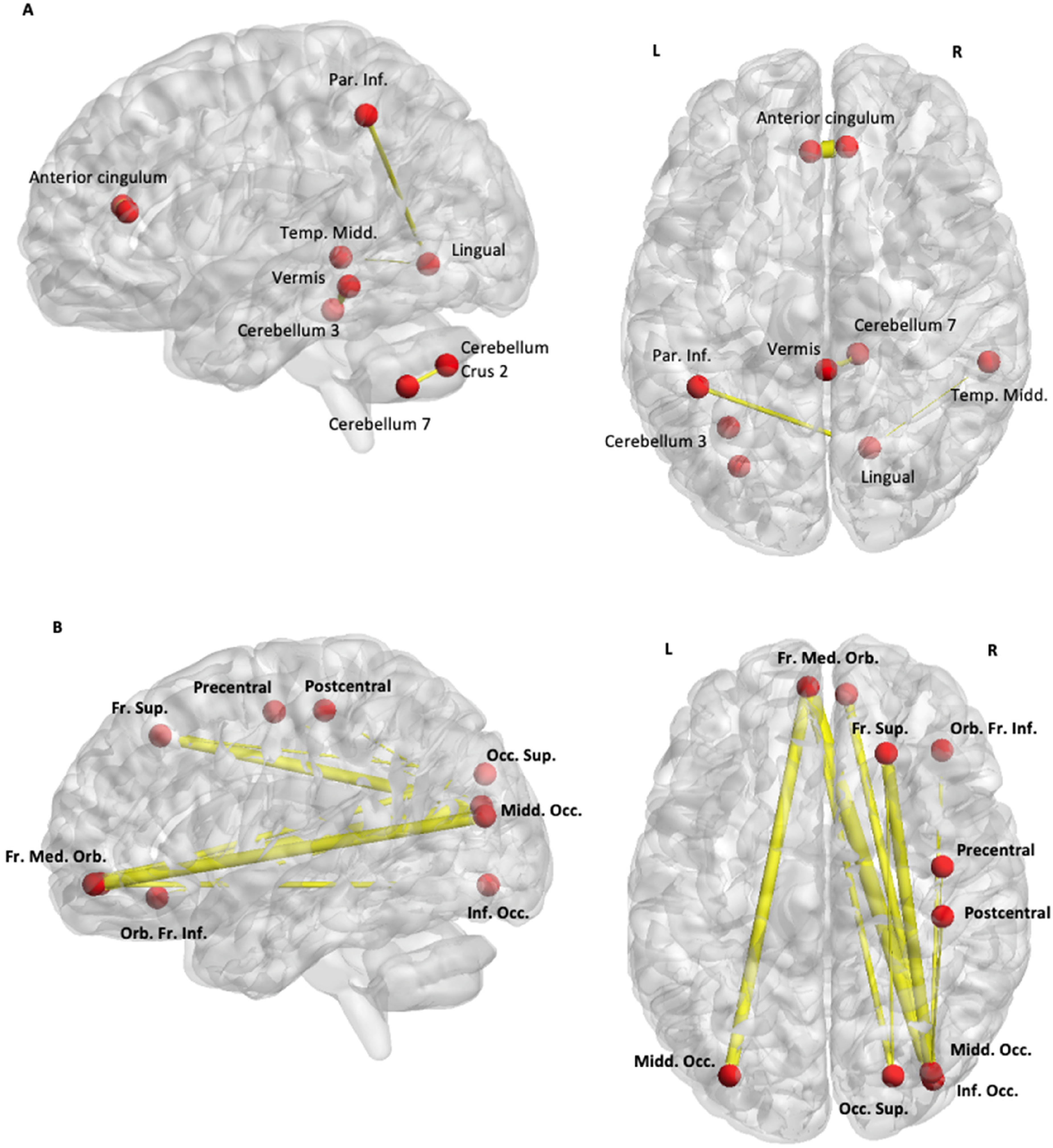
Gait conditions. (A) non-FoG > FoG. (B) FoG > non-FoG. Only the significant covariations by contrasting two groups in the gait condition are represented.
Discussion
This study demonstrates that patients with FoG at rest exhibit decreased metabolic connectivity across all networks, particularly between the cerebellum, midbrain regions, and cortical areas (sensorimotor as well as parieto-occipital). Non-FoG patients showed better metabolic connectivity, even at rest, with only weak modulations of these interconnected regions during actual gait. When performing a standardized gait trajectory that involves various triggers responsible for actual FoG (39 ± 23%) and FoG during gait, patients with FoG were able to activate partial cerebello-cortical networks, but still below the levels observed in non-FoG patients. They also failed to recruit other long-distance networks from the cerebellum to the sensorimotor areas that are normally activated during gait. In contrast, they inappropriately recruited both visual areas (which explains the visual dependence associated with the FoG phenomenon) and the orientation network (reflecting reliance on this compensatory network). The use of this network, also described in virtual MRI studies, was maladaptive here, as it was disconnected from the motor and cerebellar loops. The activation of limbic areas in FoG patients may reflect fear of falling and could contribute to this phenomenon.
This dramatic resting metabolic disorganization has also been reported in another neurodegenerative disease characterized by nigrostriatal degeneration, namely dementia with Lewy bodies. 23 The FoG group in our study was cognitively preserved, but exhibited significant disorganization and dedifferentiation in all metabolic networks, suggesting extensive degeneration of dopaminergic, cholinergic and noradrenergic systems. 24 Neurodegeneration affects molecular pathways and local circuits in specific brain regions, leading to reconfigurations, loss of function in higher-order neural networks, and alterations in large-scale resting states.25–28 Our results argue for greater neurodegeneration in patients with FoG, even before cognitive decline, evidenced by anatomical connectivity studies between the cerebellum and subcortical structures, including the mesencephalic locomotor regions and basal ganglia.29–31
Regarding the recruitment of the parieto-frontal network during gait, which is known to serve as a compensatory strategy for the lack of automaticity in PD patients, our results showed that this network is already activated during rest in non-FoG patients, with no specific recruitment during gait. In contrast, connectivity partially increased during FoG though not reaching levels observed in non-FoG patients). This may explain why, in virtual gait paradigms, increased activation in the frontoparietal regions and reduced activity in the basal ganglia, thalamus, and pre-supplemental motor area were observed during equivalents of FoG on upper limbs. 11
The most surprising result of our study was the absence of modulation of the metabolic networks in the non-FoG group, despite the additional integration of postural and locomotor programs supporting gait initiation and modulation, as well as cerebellar mechanisms triggering time-locked body movements. The metabolic networks reflect neurons that consume glucose together at rest; it is well known that at rest, neurons continuously depolarize and repolarize. In non-FoG patients, all these networks are already activated at rest and are not modified by actual gait. Conversely, in FoG patients during walking, anxiety and environmental cues (turns, narrow passages) lead to partial activation of networks that do not function automatically, particularly the visual areas and orientation networks, without restoring normal operation—likely because these networks are too impaired by neuronal loss and are no longer connected to the locomotor networks. In our study, the connection between the occipital and orbitofrontal regions was stronger in the FoG group during gait than in the non-FoG group, illustrating the involvement of both anxiety and visual triggers (and the reciprocal potentiation of these two modulators) in the FoG phenomenon. Attempting to modulate these factors in light of the severe network alterations at rest seems illusory.
The supraspinal locomotor network is recruited when the subject needs to integrate external stimuli to precisely adjust gait parameters (direction, speed, amplitude), involving the cerebellum, basal ganglia, visual cortex, prefrontal cortex (premotor and dorsolateral prefrontal cortices), and temporo-parietal junction. Rhythm generators are also engaged, involving the cerebellar vermis and paravermal cortex. 32 In this study, large disruptions in the cerebello-cortical networks were observed in the FoG group, which were partially functional when the necessary elements for walking were restored (e.g., standing posture, cues) but in an unstable manner. Similar to neuromuscular blocks at an unstable junction, these temporary interruptions in the motor program could indicate very poor functioning of these complex gait compensatory networks in FoG patients.
One limitation of this study in cognitively unaffected patients is that it did not explore further aggravation of the FoG phenomenon due to dysexecutive impairment. We observed profound disruption of the metabolic networks prior to cognitive failure in patients with FoG. In patients with severe dysexecutive syndrome or cognitive impairment, more severe alterations in connectivity would be expected but with large heterogeneity. 33 On the other hand, the study confirms the cortical pathophysiological hypotheses of FoG, namely perceptual and executive dysfunction, with abnormal connectivity within the parieto-frontal network in PD patients activated during walking, but not sufficiently during FoG. This interference problem is exacerbated by poor bilateral rhythmicity and coordination, as well as incorrect integration of visual information: weak activation of the cerebellar-cortical loops, heightened activation of the visual cortices, but disconnected from the motor networks. The study also highlights the severe disruption of most metabolic networks, leading to extensive denervation, resulting in untimely breakdowns during motor adaptation and generating an abnormal gait pattern due to the decoupling of cortical and subcortical regions.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241305713 - Supplemental material for Metabolic connectivity of freezing in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241305713 for Metabolic connectivity of freezing in Parkinson's disease by Céline Tard, Arnaud Delval, Luc Defebvre, Pierre Lenfant, David Devos, Caroline Moreau and Nacim Betrouni in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors acknowledge the assistance of Florence Lejeune, Franck Semah and Damien Huglo.
Funding
The study was supported by Lille University Hospital.
Declaration of conflicting interests
Professor Caroline Moreau is an Editorial Board member of this journal, but was not involved in the peer-review process of this article nor had access to any information regarding its peer review. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data and the code used in this study can be made available by request sent to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
