Abstract
Background
Research over the past twenty years has shown that gait outcomes have a high sensitivity for diagnosing Parkinson's disease (PD), for detecting the effects of interventions, and for monitoring disease progression, even in early disease. However, the lack of standardization in protocols and reported gait measures is impeding data aggregation across study sites and contributes to heterogeneity in the results, thus limiting the adoption of gait outcomes in clinical trials.
Objective
To provide recommendations for a minimum set of gait measures to be adopted in projects evaluating people with PD to enhance standardization across the field.
Methods
The Gait Advisors Leading Outcomes for Parkinson's (GALOP) committee is an advisory committee for the MJFF. Based on a five-step approach, GALOP generated recommendations for standardizing protocols that assess quantitative gait measures, following expert consensus on best practices.
Results
Built on the literature and consensus amongst experts, we recommend a minimum set of meta-data to accompany gait protocols and a minimum gait assessment protocol to be performed at a comfortable speed. Suggestions on challenging testing are provided.
Conclusions
To support and empower the scientific community, we have generated recommendations to collect and share gait data gathered from people with PD using an open data repository. Standardizing gait protocols and outcomes in PD has the potential of accelerating research and clinical trials, harmonizing protocols across study sites, fostering collaborations, and in the long run, improving patient care and quality of life.
Introduction
Specific gait impairments are one of the earliest diagnostic signs of Parkinson's disease (PD) and also reflect severity and progression of the disease.1,2 Yet, in the gold standard clinical motor assessment, the Movement Disorders Revised Unified Parkinson's disease Rating Scale (MDS-UPDRS) Motor Score (Part III), 3 has only one item evaluating “Gait” and one item evaluating “Freezing of Gait (FoG)”, both on a Likert scale of 0 to 4. “Problems with walking” are reported as increasingly important in people with PD as the disease progresses;4,5 however, this is not yet reflected in a widely used, quantitative, and sensitive assessment.
Unfortunately, clinical trials that aim to prevent or slow the progression of immobility in PD may face limitations given the low sensitivity of gold-standard outcome measures, which depend on subjectively rated scales that are based on expert opinions.2,6,7 Sensitive and objective markers of disease progression are also needed for earlier, more precise detection and characterization of subtle motor deficits in early phenotypic PD as well as in high-risk, pre-symptomatic populations.8–10
Moreover, certain gait issues are often levodopa-resistant. 11 Objective, quantitative measures can address gaps in the advancement of disease modifying therapies by evaluating meaningful effects that are not masked by currently approved symptomatic dopaminergic treatments.
Quantitative gait characteristics, as measured in gait laboratories, are abnormal at the time of diagnosis and inevitably decline with PD progression.7,12,13 Apart from gait impairments that are visible during routine clinical observation, such as the average reduction in gait speed, stride length and arm swing, there are a number of aspects of gait that are not easily observed visually but have shown to be highly sensitive and specific to PD 14 when quantitatively measured using reliable and valid methods. For example, increased gait asymmetry and stride-to-stride variability (i.e. unsteadiness or inconsistency and arrhythmicity of stepping) can be measured throughout the disease. 14 These metrics have been identified even in individuals who are considered at risk of developing PD,10,15,16 as well as in those recently diagnosed and who are still medication free. 17 The magnitude of variability also tends to increase with disease severity.2,14,18 Gait variability is closely associated with falls in older adults19–24 and represents a measure sensitive to a number of other neurological diseases.22,25–31 However, despite its potential value, gait variability is often not reliably obtained or reported in PD studies.32–34 In fact, due to poor standardization of gait protocols, there are reports calculating gait variability from intermittent, short walks, e.g. only 5 meters, and others from long walks, over minutes. This broad range in the testing condition can have a large impact on the results32,33,35 and in discriminating people with PD from healthy controls. 36 Similarly, some studies include the entire walking trial including steps of acceleration and deceleration, for example before and after a turn to change direction,36,37 while other studies only consider steady-state gait.38,39
Inconsistency in protocol treatment of the confounding variable of turning is problematic for interpreting data. Turning (i.e. duration, velocity, and number of steps to make a turn) is often impaired in people with PD compared to healthy individuals of similar age even when gait speed is similar.40,41 Moreover, turning is a key contributor to FoG in people with PD42,43 and may present distinct features, when compared to basic straight-line walking.
Some gait protocols include straight line comfortable speed walking trials while others also include challenging dual tasks44–46 to increase neural overload, also known as ‘cognitive-motor’ interference. 47 Variations also exist in the types of walking evaluated, with some studies using short walks while others evaluating endurance walking, and in the environments where gait is analyzed, such as clinical settings versus real world conditions. Additionally, the methodologies of data acquisition and processing differ dramatically making it challenging to draw consistent conclusions.
This wide variety means that even if the actual data can be harmonized, the differences in the protocols are a barrier to interpretation. Consistency is critical to demonstrate the efficacy of gait variability, the role of turns, and the impact of steady-state walking as measurable clinical outcomes. In the existing literature, there are no guidelines on a minimum protocol to obtain valid gait measures in people with PD, nor which protocol and gait measures would be more sensitive to use in the early versus late stage of the disease. Protocols and algorithms to assess gait quantitatively are extremely varied, received little to no attention on standardization, and their designs depend on the available instrumentation (shorter walks with motion analysis and pressure mat versus wearable technology), the environmental constraints (longer spaces in laboratory versus clinic, and shorter distances in daily life), and the original hypotheses. Although several papers point to the need for standardizing gait testing protocol and sensing systems in PD,33,34,36,48 no guidelines have been published. To the best of our knowledge, the only document providing guidelines on a common data set for gait metrics in PD was recently put together by the National Institute of Neurological Disorders and Stroke (NINDS). However, these guidelines targeted solely the use of wearable sensors for research and clinical trials in PD, were not specific to gait, and did not include recommendations on a specific gait protocol, which is crucial for validity of gait measures. There is, therefore, a great need for standardization of the field, to enable harmonization between studies, allow for aggregation of information, and inform a better understanding of the value of gait measures for diagnosis, monitoring, prediction and responsiveness to interventions.
Here, our objective was to recommend a common protocol for multicenter, natural history, and intervention studies to support the inclusion of specific gait measures for clinical trials in PD. Recommendations are provided based on aggregated information from the literature and expert panel consensus.
Methods
The guidelines for a unifying protocol and a set of gait measures have been developed in a step-wise, tiered manner by first reviewing the existing literature and then having discussions (first subgroup and then plenary) to reach a consensus among an international and diverse group of experts on gait and PD.
The group of experts is part of an advisory committee for the Michael J. Fox Foundation for Parkinson's Research (year 2023). The group includes senior and junior experts in the field of gait, from academia to clinical care, who aim to progress research and treatment of gait impairments in PD. Specifically, the group was formed of ten members (MM, JH, PB, EP, RC, TE, JK, LG, AF, AN) and a chair (AM). Out of the 11 members, 4 are PT PhDs, 3 are MD movement disorders specialists, 3 are bioengineers, and one is an individual with PD living with gait issues.
The flow describing how the guidelines were developed is represented in Figure 1.

Overview of the flow used to develop guidelines and minimum protocol for gait standardization in people with PD.
The data collection process was partly in line with the ACcurate COnsensus Reporting Document (ACCORD) procedures to achieve consensus. 49 Briefly, it included 5 stages. First, in Stage 1, a sub-committee (MM, JH, PB, EP, and AM) was formed to carry out the proposed work, in which the scope and timelines were defined during a virtual meeting. In Stage 2, the sub-committee discussed a narrative review of the recent literature, including reviews on gait in PD and the recently developed NIH guidelines on the reporting of digital outcomes for PD (part of the NINDS Common Data Elements initiative for PD, https://www.commondataelements.ninds.nih.gov/Parkinson%27s%20Disease). Then, a first outline was drafted (MM) and reviewed by the sub-committee (AM, JH, PB, EP). In Stage 3, the first draft of the guidelines was presented to the full committee during a virtual meeting and feedback was obtained. In Stage 4, gait standardization items were finalized, a guide, a data dictionary, and examples on how to report data were created (MM and AM), circulated to the sub-committee and full committee and agreed upon by all members. Lastly, Stage 5 consists of: a) consultation with patients and patient representatives to receive feedback, see Supplemental Material, and b) dissemination through article publication and presentations.
During the virtual meeting, 100% agreement was reached on the minimum set of items to report in the gait standardization document and final approval from the full committee was obtained at each stage.
Results
Figure 2 summarizes the recommended demographic and clinical measures, as well as the protocol guidelines and gait measures to report while investigating gait in people with PD. The following demographics should be reported, in reference to the time of testing: age, sexual orientation and gender identify (SOGI), height, weight, years of education, disease duration, race and ethnicity.
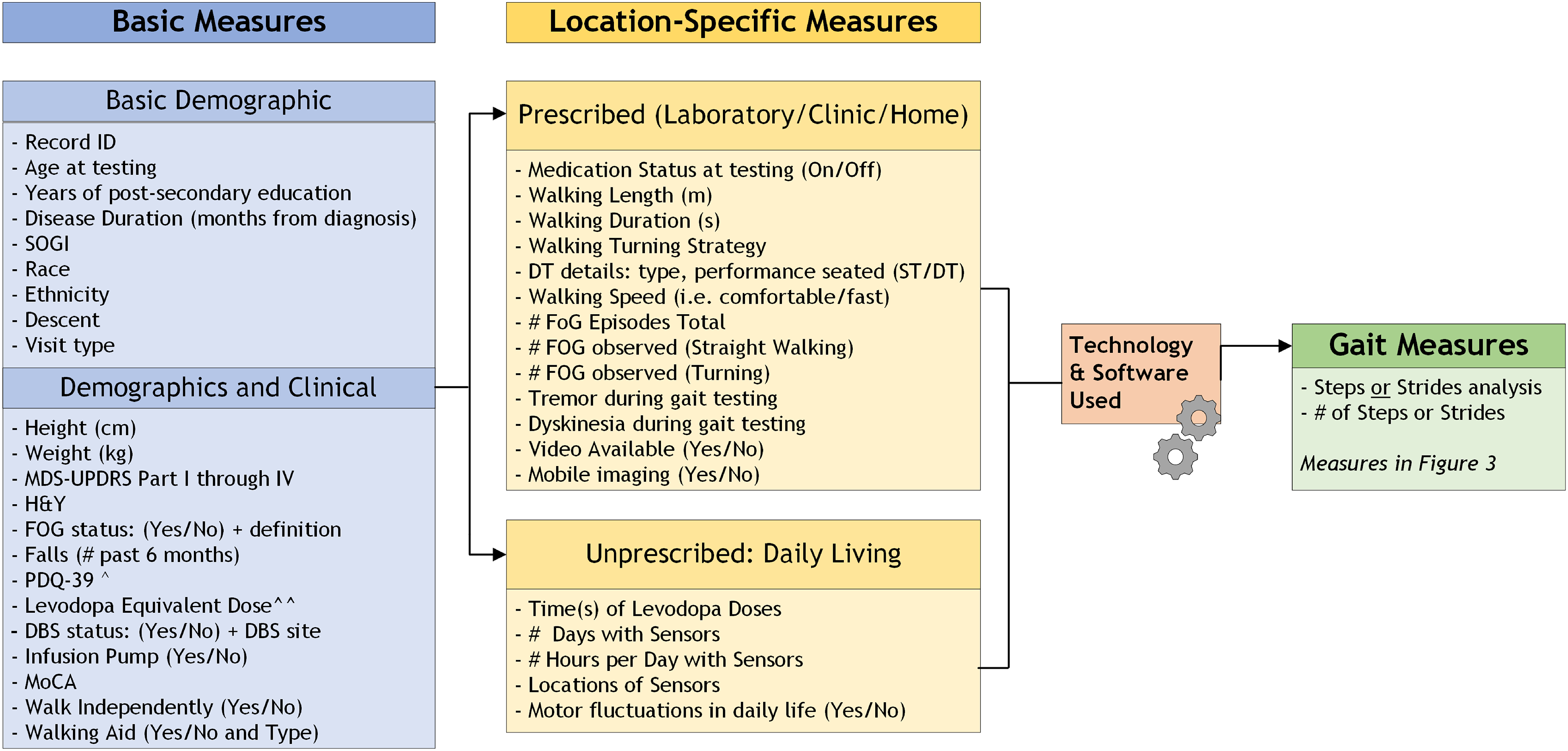
Diagram to visualize summary data to report. Visit Type cross-sectional for a one-time visit. For a study with two or more visits (within a few weeks to one month), enter visit1, visit2 and so forth. For longitudinal studies over years, enter baseline, 6months, 12months, 18months, and so forth. ^PDQ-39 summary score. ^^To report the levodopa equivalent dose (mg), please refer to Jost et al. 50
The committee deemed the reporting of the demographics and clinical measures of importance, in that: 1) they characterize the participant and allow integration data from similar projects, and 2) some may influence expected or actual gait performance. For some of those demographics and clinical measures, the effects on gait have been previously investigated (age, disease duration,2,18,51 DBS, 52 cognition,53,54), while for others, their influence on gait is not yet clear but warrants further consideration and may be important for future trials (SOGI55,56). Regardless, all the SOGI questions have an option of “prefer not to answer”.
For the clinical measures, the MDS-UPDRS and Hoen & Yahr stage should each be reported; we recommend reporting all items as well as the total MDS-UPDRS score, as well as the commonly calculated sub-scores (i.e. PIGD, Tremor scores). 3 Freezing of Gait (FOG) status and past falls (recollection of 6 months prior) should be collected. Information on whether participants have a deep brain stimulation (DBS) implanted, the anatomical target of DBS (including unilateral vs. bilateral implantation), or whether they have an infusion pump should be collected. We recommend the Montreal Cognitive Assessment (MoCA) 57 and the Parkinson Disease Questionnaire (PDQ-39) 58 to characterize overall cognition and patient-reported assessment of quality of life, respectively.
Levodopa equivalent dose should be calculated following recent recommendations. 50 As the guidelines are aimed at addressing a wide population of people with PD, walking aids are permitted if strictly necessary. However, information on whether the individual is using a walking aid (and the type) should be reported. It is important for the validity and interpretation of the gait measures that the individual can walk unassisted (although an examiner guarding gait to ensure safety is allowed, as long as there is no interference to the walking and turning of the individual).
To help standardize the heterogeneity across gait protocols (i.e. short versus long walks, technology, simple versus challenging tasks), the committee recommended a minimum protocol, see Figure 3, summarized in the Location-Specific Measures of Figure 2. Specifically, if the protocol includes collecting gait in prescribed conditions (with an examiner present explaining the protocol) in a laboratory, clinic or home, the committee recommends, at minimum, that the protocol include: 1) one continuous gait task of at least 1 min in duration, 2) over a path of 10-meters in length (allowing 1 m at each end for a safe turn), and 3) with participants performing 180 degrees turns at each end (ideally passing a line and turning back to the opposite end). The rationale for the choice of a continuous walk of at least 1 min over 10 meters was agreed upon to ensure the validity and reliability of the gait measures, in fact, variability measures need at least 30 to 50 steps.32,33,36 The duration of 1-min was thought to be a good compromise in case multiple gait testing conditions can be assessed without making the testing excessively long and while still ensuring that an appropriate number of steps and strides are recorded. The length of 10-meters was proposed to ensure reaching steady-state gait before decelerating prior to a turn and accelerating after a turn to change direction. 59 If a 10-m path is not available, investigators should report the specific distance used and they should be aware that the reliability and validity of certain gait measures may be compromised. We acknowledge that many clinics may carry out the gait testing directly in the clinic space or hospital corridors and that this situation will expose the tested participant to interference of other individuals possibly passing by. For this reason, we recommend trying to minimize the interferences with possibly a wait sign in the area and instructing participants to carry out their test without acknowledging people passing by.
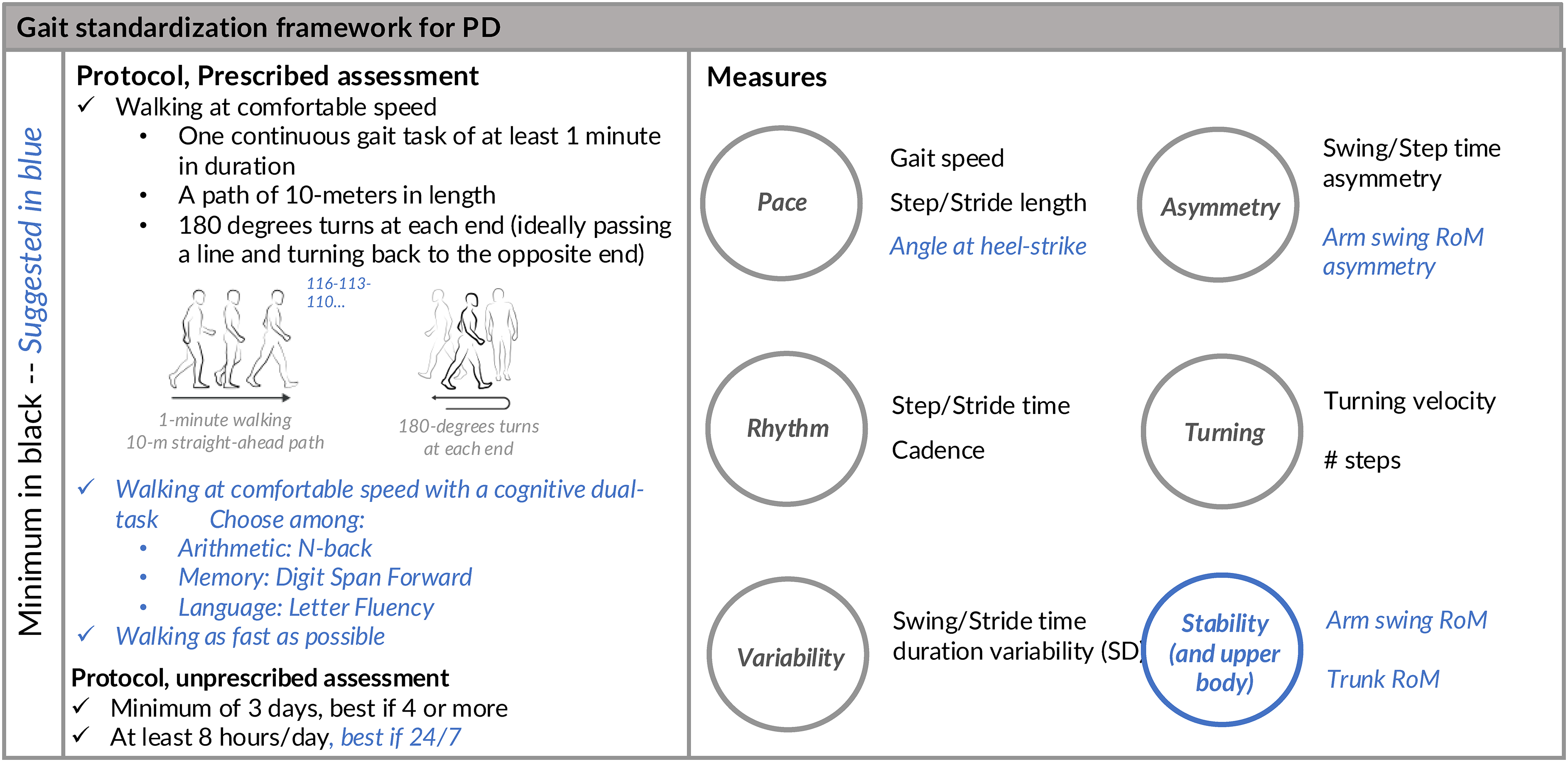
Proposed basic protocol and set of measures for gait assessment in ambulatory people with Parkinson's disease. Tasks and measures in darker color indicate a minim set, tasks and measures in lighter color indicate suggested items. Participants should not use any walking aid or assistive device if possible.
Gait should be collected at least during comfortable walking speed and the medication status at the time of testing should be reported (e.g. ON, OFF, time since the last intake of PD medication). Specific measures are recommended to collect data on walking duration, length, turning strategy, walking speed, number of strides or steps used for the analysis, and the number of FOG episodes, if observed. The committee agrees on the importance of including turning metrics when reporting gait. Difficulties turning in PD occur early in the disease 41 and are associated with clinical outcomes, such as falls, fear of falling, disease severity, and FoG.60–63
Additional suggested protocol elements and measures are also provided, beyond the minimum recommendations of the consensus committee, as shown under “suggested” in Figure 3. Specifically, more challenging gait protocols are advised, if possible, such as a dual-task walking, using one of the 3 cognitive challenges, and/or a fast walk. If using a dual-task walking condition, we also suggest collecting seated, baseline cognitive task performance. The committee agrees on the importance of challenging gait. It is known that people with PD have difficulties performing a second task while walking, such as walking and talking. 46 As a consequence of performing two tasks at the same time, gait and/or their performance on the secondary task at hand deteriorates.46,64,65 These difficulties in dual task walking often lead to increased disability, increased FoG and fall risk, and reduced quality of life.46,64,66 The commission is aware of the possibility of emerging new biomarkers that arise to detect and predict freezing of gait in early disease.12,67,68 Therefore, updating the current guidelines is indicated in the near future.
The technology and software used should be reported and provide validation details for use in PD.33,38,69–71 Specifically, as detailed in the Supplemental Material, the following should be stated regarding the used technology: 1) type of technology (e.g. gait mat, motion analysis, IMUs); 2) manufacturer, make and model; 3) sampling frequency; and 4) references on the validity of the specific technology in measuring gait. For the used software, the following should be specified: 1) ad-hoc or commercial algorithms; 2) definition used to identify bouts of gait; 3) references on the validity of the algorithm in people with PD, and 4) explanation on how the gait measures are calculated.
The specific selection of gait measures was partly in agreement with the recent recommendation of the NIH common data elements and demonstrated criterion validity and reliability, according to the COSMIN framework. 72 As gait is multifactorial, several studies in the past have used conceptual gait models to identify independent gait domains.73–75 Based on this information, we recommend as shown in Figure 3 to report, at a minimum, measures related to domains of: 1) pace, with gait speed, step or stride length; 2) rhythm, step or stride time, cadence; 3) asymmetry, swing or step time asymmetry using a normalized/logarithmic formula described in Supplemental Material; 4) variability, step or stride standard deviation; and 5) turning, turning velocity and number of steps/turn. These domains were chosen based on their consistent use in literature, although other domains or slightly different subdivisions have been proposed.74,75 Additionally, if the technology allows and measures of upper-body/stability are available, then the angle of the foot at heel-strike,39,76,77 arm swing range of motion and its asymmetry41,78 should be reported, as well as the trunk coronal range of motion while walking. 41
All the variables presented in Figure 2 should be made available in summary/derived form in a .csv file or a similar and de-identified version of data, meeting ethical and regulatory requirements, and should be deposited in an open access platform of choice. The Supplemental Material provides an example given to investigators on MJFF's supported studies on how to report results and definitions consistent with a data dictionary.
Discussion
In this paper, the Gait Advisors Leading Outcomes for Parkinson’s (GALOP) committee presents guidelines designed to provide recommendations for a minimum protocol to quantitatively assess gait in PD for non-expert and expert researchers, clinicians and health specialists interested in measuring gait. We intended to facilitate standardization of gait to enable harmonization of gait data from different studies in the PD research community in order to advance quantified gait as a clinical outcome measure and to move from small studies based on data collected locally or in a specific project to analyses based on large datasets, like those that have been done with respect to ageing.79,80
A minimum set of data to accompany gait protocols (including basic demographics and clinical data), and a minimum gait protocol of at least 1-min of walking in a straight-line path of 10 m, including 180 degrees turns to be performed at a comfortable speed, have been agreed upon. This recommendation is similar to recent guidelines from a consensus on digital measures of gait in people with spinocerebellar ataxia. 81 In addition, this committee also recommends adding additional challenging conditions such as fast-paced walking or dual-task to enable a comprehensive assessment of mobility. To note, prescribed, gait assessment in the laboratory has been extensively studied and is reliable for persons with PD.37,38,48,82 The drawback is that such assessments may not reflect mobility during daily life.83,84 The gait of people with PD is often paradoxically better when they know that they are being observed. 85 Unobtrusive community monitoring has the untapped potential to provide quantitative mobility measures that may be even more sensitive markers of subtle changes, compared to measures in the laboratory in people with PD. 84 Gait measured in the home and community during normal daily activities reflects real-world mobility affected by complex environments, multitasking, and daily motor fluctuations.83–88 Nowadays the technology allows for measuring gait in these patient-relevant, ecological conditions; however, there are several challenges regarding the best protocol to use, data processing and data storage. With the current state of the literature,83,84,86–90 at least 3 days, best if 4 or more, of continuous monitoring of gait in daily life are recommended, for at least 8 h a day, best if 24/7. 91
In the existing literature, there are also no guidelines on which protocol and gait measures would be more sensitive to use in the early versus late stage of the disease. Clearly, different metrics should be assessed at different stages of the disease2,18 and the spectrum of gait conditions should be chosen accordingly. With this in mind, the proposed minimum set is seen as a foundation on which additional gait tasks (i.e. turning in place or other tasks to elicit FoG), additional gait measures (i.e. EMG, mobile imaging, etc.), additional measures of balance capacity (i.e. MiniBEST 92 ), as well as additional demographics (i.e. genetic status, co-existing medical issues) can be laid. We acknowledge the potential limitations of research and clinical settings regarding time, effort, cohort and research questions, thus opted for a minimum set of data to allow inclusivity towards standardization and harmonization of data.
With the current review of the literature, we do strongly suggest incorporating challenging walking conditions, such as dual-tasking and/or fast walking, which may be useful in the prodromal state, for example. 10 Further, we intentionally did not specify outcomes to be gathered but only the common minimal set. However, we encourage a broad approach with transparency in reporting, to encourage the harmonization between studies. For example, studies show that gait measures of asymmetry (either lower or upper body) and variability, as well as turning, only when assessed during challenging conditions (fast or dual-task), are altered in individuals considered at risk of developing PD 16 (either for genetic mutations10,93 or RBD 94 ) compared to healthy controls. Instead, when gait is used to monitor disease progression, few studies show a decline of stride length,76,95,96 gait speed,,95–97 angle of the foot at heel-strike and gait variability 76 when measured during single-task gait at comfortable speed.76,95,96,98 Investigators should specify the approach(es) / technology proposed to collect quantitative gait outcomes and provide a detailed explanation for all data collected and the technical and clinical validation of proposed assessments. 99 Of course, the specific hypothesis and context of use should also be delineated.
In addition to sharing all summary measures, we want to stress the importance of making raw data available to the extent possible, as well as computer code/algorithms used for pre-processing. There are many open-source data and software platforms, such as Zenodo, Physionet, and Github, that can be used for these purposes. The authors do not yet recommend a specific platform and file structure to be used, although some examples are starting to come out in the literature, including the example Supplemental Material presented here.70,99,100
Strengths of the proposed recommendations include the large, international, multidisciplinary group of experts in PD and gait, the use of a collaborative process with open discussions, and active engagement of one individual with first-hand personal experience as a member of the committee. Our overall approach was based on a collaborative process with group participation and feedback. As such, the group members’ identities were known to each other, and the recommendations were written with group contributions and iterative feedback. Moreover, given the vast background and experience of the committee with multidisciplinary collaboration and teamwork, this type of group and teamwork flowed naturally to allow all members to participate openly. We acknowledge that the lack of a formal Delphi process may be a methodological limitation, though we also recognize that the literature supports various guidelines and statements without a formal Delphi method81,101 (examples are NINDS guidelines and MDS Task Force102,103).
The proposed set of guidelines is a first step in the direction of proposing a standardized gait assessment for a wide population of people with PD with the vision of being able to integrate datasets in the future and reach appropriate sample sizes for clinical trials. In the future, we envision that this protocol will be updated based on new knowledge and recommendations. As gait is crucial for moving independently in daily life, is one of the symptoms most important to people with PD, 4 and is impacted in other movement disorders, this framework could later be implemented for other neurological diseases to advance the understanding of those diseases and enable the identification of common and distinct gait alterations across different diseases.
Supplemental Material
sj-csv-1-pkn-10.1177_1877718X241305626 - Supplemental material for A framework to standardize gait study protocols in Parkinson's disease
Supplemental material, sj-csv-1-pkn-10.1177_1877718X241305626 for A framework to standardize gait study protocols in Parkinson's disease by Martina Mancini, Jeffrey M Hausdorff, Elisa Pelosin, Paolo Bonato, Richard Camicioli, Terry D Ellis, Jochen Klucken, Larry Gifford, Alfonso Fasano, Alice Nieuwboer, Catherine Kopil, Katharina Klapper, Leslie Kirsch, David T Dexter, Rosie Fuest, Vicki Miller, Angelica Asis, Martijn LTM Müller, Diane Stephenson and Anat Mirelman in Journal of Parkinson's Disease
Supplemental Material
sj-csv-2-pkn-10.1177_1877718X241305626 - Supplemental material for A framework to standardize gait study protocols in Parkinson's disease
Supplemental material, sj-csv-2-pkn-10.1177_1877718X241305626 for A framework to standardize gait study protocols in Parkinson's disease by Martina Mancini, Jeffrey M Hausdorff, Elisa Pelosin, Paolo Bonato, Richard Camicioli, Terry D Ellis, Jochen Klucken, Larry Gifford, Alfonso Fasano, Alice Nieuwboer, Catherine Kopil, Katharina Klapper, Leslie Kirsch, David T Dexter, Rosie Fuest, Vicki Miller, Angelica Asis, Martijn LTM Müller, Diane Stephenson and Anat Mirelman in Journal of Parkinson's Disease
Supplemental Material
sj-csv-3-pkn-10.1177_1877718X241305626 - Supplemental material for A framework to standardize gait study protocols in Parkinson's disease
Supplemental material, sj-csv-3-pkn-10.1177_1877718X241305626 for A framework to standardize gait study protocols in Parkinson's disease by Martina Mancini, Jeffrey M Hausdorff, Elisa Pelosin, Paolo Bonato, Richard Camicioli, Terry D Ellis, Jochen Klucken, Larry Gifford, Alfonso Fasano, Alice Nieuwboer, Catherine Kopil, Katharina Klapper, Leslie Kirsch, David T Dexter, Rosie Fuest, Vicki Miller, Angelica Asis, Martijn LTM Müller, Diane Stephenson and Anat Mirelman in Journal of Parkinson's Disease
Supplemental Material
sj-docx-5-pkn-10.1177_1877718X241305626 - Supplemental material for A framework to standardize gait study protocols in Parkinson's disease
Supplemental material, sj-docx-5-pkn-10.1177_1877718X241305626 for A framework to standardize gait study protocols in Parkinson's disease by Martina Mancini, Jeffrey M Hausdorff, Elisa Pelosin, Paolo Bonato, Richard Camicioli, Terry D Ellis, Jochen Klucken, Larry Gifford, Alfonso Fasano, Alice Nieuwboer, Catherine Kopil, Katharina Klapper, Leslie Kirsch, David T Dexter, Rosie Fuest, Vicki Miller, Angelica Asis, Martijn LTM Müller, Diane Stephenson and Anat Mirelman in Journal of Parkinson's Disease
Footnotes
Acknowledgments
ORCID iDs
Funding
MJFF-024749 and The Michael J. Fox Foundation for Parkinson's Research.
Declaration of conflicting interests
Drs. Alice Nieuwboer and Anat Mirelman are Editorial Board Members of this journal but were not involved in the peer-review process of this article nor had access to any information regarding its peer-review. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
