Abstract
Introduction
Depression often co-occurs with Parkinson's disease (PD), with up to 50% of patients with PD suffering from it. 1 Selective serotonin reuptake inhibitors (SSRI) and serotonin and noradrenaline reuptake inhibitors (SNRI) are the most widely prescribed antidepressants in North America and Europe2–4 and have been shown to be effective treatment options for depression in PD. 5 The mechanism of action of SSRIs and SNRIs is based on preventing the reuptake of the neurotransmitter serotonin, leading to an increase in its activity. 6 In addition to it, SNRIs also block the reuptake of noradrenaline, thereby increasing its activity, 7 but the effect on depression symptoms is similar to SSRIs. Emerging research suggests that neurotransmitters, including serotonin and noradrenaline, play a role in the pathophysiology of PD.8,9 The serotonergic system appears to influence motor and non-motor symptoms, 10 and serotonergic pathology may anticipate the development of motor symptoms and dopaminergic pathology, indicating a higher disease burden. 11 In addition, noradrenergic pathophysiology has also been reported as a common hallmark of PD and can precede dopaminergic pathology and corresponding motor symptoms. 12
Several studies have reported that SSRIs/SNRIs may be associated with worsening of motor symptoms, but some studies show no effect. 13 Some studies even show improvement in motor symptoms.13–22 For example, the SSRIs paroxetine and fluoxetine showed no effect on motor symptoms in PD, while they improved depressive symptoms,14–17 although one of the studies found an improvement in tremor. 17 On the other hand, worsening of PD after taking fluoxetine has also been reported. 18 There are also reports of cases of irreversible parkinsonism induced by fluvoxamine/sertraline 19 and worsening of parkinsonism induced by fluvoxamine. 20 However, some studies have also found a potential benefit of SSRIs for certain motor symptoms such as bradykinesia or freezing of gait 22 in PD patients treated with levodopa and citalopram. 21 In addition, motor side effects are occasionally observed in patients taking SNRIs, namely venlafaxine and duloxetine, such as worsening of tremor13,22 and dyskinesias. 22
In summary, the effects of SSRIs/SNRIs on motor symptoms in PD remain unclear. Some studies suggest a potential benefit on motor symptoms, while others suggest potential risks. Furthermore, the effect of SSRIs/SNRIs on motor symptoms in PD has so far been reported only in several studies, mostly case reports or in studies with a small number of patients. Therefore, we set out to assess the effects of SSRI/SNRI on motor symptoms in PD using data openly available from the Parkinson's Progression Markers Initiative (PPMI) database. 23
Methods
Study design
We created a matched-subject design with a target group that had taken SSRI/SNRI medication and a control group that had not. Patient data came from the PPMI database, a longitudinal, multicenter observational study that assesses progression of clinical features and other markers and outcomes from PD. 23 The Movement Disorders Society Unified Parkinson's Disease Rating Scale part III (MDS-UPDRS-III) total score and the axial (sum of items 3.9 to 3.13), bradyhypokinesia (sum of items 3.4 to 3.8 and 3.14), rigidity (item 3.3), and tremor (sum of items 3.15 to 3.18) subscores were assessed on two occasions. For the target group, these occasions were before and after taking the SSRI/SNRI medication and for the control group, these occasions were two equally spaced examinations. To analyze the data, a model was constructed with group (target/control), examination (first/second), levodopa equivalent daily dose (LEDD) at the time of the first examination (initial LEDD), depressed mood (MDS-UPDRS-I item 1.3), anxious mood (MDS-UPDRS-I item 1.4), and apathy (MDS-UPDRS-I item 1.5) as predictors for the MDS-UPDRS-III total score and subscores, with a one-to three-way interaction (between the first three predictors) possible. The main outcome measure was the difference between the target and control group in the change in the MDS-UPDRS-III total score between the two examinations. The secondary outcome measures were the difference between the target and the control group in the four motor subscores (axial, bradyhypokinesia, rigidity, and tremor) between the two examinations. We were primarily interested in examining the interaction between the group and the examination, i.e., whether the subjects who belonged to the target group had a different average rate of change in their MDS-UPDRS-III score and axial, bradyhypokinesia, rigidity, and tremor subscores compared to the subjects in the control group.
Subjects
Two groups of PD patients were selected from the PPMI database: (1) A target group of PD patients [N = 47, 14 females, median age 61.4 (55.3–69.9) years] who had, at some point, been taking an SSRI or SNRI, and (2) A control group of PD patients [N = 90, 33 females, median age 63.4 (58.7–69.3) years] who have never taken an SSRI or SNRI. The control group of patients was matched, patient per patient, to the target patients on their initial motor status, reflected in the overall MDS-UPDRS III score, the relevant MDS-UPDRS III subscores, and the initial LEDD (Table 1).
Demographic and clinical characteristics of the patients in the target and control group, and p-values indicating the significance of the differences between the first and the second examination. The results are presented as median (25th-75th percentiles) or proportion (percentage, %) where appropriate.

MDS-UPDRS I, II, and III: Movement Disorders Society Unified Parkinson's Disease Rating Scale part I, II, and III; LEDD: Levodopa Equivalent Daily Dose. The Axial subscore was caluclated as a sum of the MDS-UPDRS-III items 3.9 through 3.13, the Bradyhypokinesia subscore as a sum of the items 3.4a through MDS-UPDRS-III 3.8b and 3.14, the Rigidity subscore as a sum of the MDS-UPDRS-III items 3.3a through 3.3e and the Tremor subscore sum of the items MDS-UPDRS-III. 3.15a through 3.18.
Patient selection
The PD Features (PDFEAT) table as of 5 July 2021 from the PPMI registry, together with Participant Status table as of 9 July 2024, were used to select the patients from the target and the control group. Tables MDS-UPDRS Part III Treatment Determination and Part III: Motor Examination (NUPDRS3) and Concomitant Medication Log (CONMED) as of 5 July 2021 were also used in the selection process. The exact target and control patients’ identification number (PATNO) for the final target and control group patients included in the study and other relevant information is given in the Supplemental Table 1. Importantly, all the patients in the final target and control group were from the PPMI clinical cohort.
The first step in patient selection was to select the target group from the PPMI database. Each patient had to undergo two assessments: one before starting SSRI/SNRI treatment and one 2 to 7 months after starting treatment, and only if the treatment was still ongoing in that moment. Only the assessments performed OFF dopaminergic medication were considered, as the effect of dopaminergic medication might have influenced the results of the study. In the second step, we selected two control patients for each target patient. The control patients were selected so that their status at the first examination matched the first examination of the respective target patient according to several criteria (see below) and so that the time interval between their first and second examination was close to the time interval between the examinations of the respective target patient. For a graphical representation of the patient selection process, please see the flowcharts in the Supplemental Flowchart 1.
Only patients who had taken an SSRI or SNRI medication were selected for the target group. The presence of SSRI or SNRI medication was manually coded. The SSRI and SNRI medications from the database and the number of patients taking each of the drugs were as follows: citalopram (N = 11), escitalopram (N = 10), sertraline (N = 8), trazodone (N = 7), duloxetine (N = 3), venlafaxine (N = 3), paroxetine (N = 3), fluoxetine (N = 2). Patients who had only one examination were excluded. For the first target examination, the examination closest to or on the day of the start of SSRI/SNRI treatment was taken. Only examinations that took place within the 6 months before the start of the treatment were considered. If a patient had taken several SSRI/SNRI medications, the first medication was considered. It should be noted that only month and year were available for all events. For the second target examination, we looked for the examinations that occurred between 2 and 7 months after the start of SSRI/SNRI medication. This is because it takes some time for the SSRI/SNRI to take effect. In cases where there was a date for stopping the SSRI/SNRI medication, we made sure to exclude the examinations that took place after this date. If several examinations were found to meet the criteria, the last one was kept as the second target examination.
The first step in selecting the control group was to select only patients who were not taking SSRI/SNRI medication. For each potential control examination and each selected first target examination, the difference between the total MDS-UPDRS-III score and the LEDD was calculated. Only those pairs in which the difference in the MDS-UPDRS-III total score was less than or equal to 2 points and the difference in LEDD was less than 100 (approximately 0.5 standard deviations of the initial LEDD, SD = 215.1) were kept. The 2-point difference in the MDS-UPDRS-III was considered small enough to be clinically significant. 24 The Euclidean distance on the subscales of MDS-UPDRS-III between each first target examination and the potential control examination was calculated, and for each first target examination a match was found with the control examination with the smallest difference in the total MDS-UPDRS-III score and, in the case of several matches, the smallest Euclidean distance on the subscales of MDS-UPDRS-III. This was taken as the first control examination. Each control patient could only serve as a match for one target patient. The time difference between each pair of target examinations was calculated, as well as between all possible control patients’ examination pairs. The second control examination was found by selecting the first examination of the selected control patient that fell within the acceptable time frame (±31 days difference from the time difference between the examinations of the corresponding target patient). For the second round of control patients (as the design was two controls for each target patient), the matching procedure was repeated with the remaining potential control patients.
To summarize, all the patients in the target group were matched as closely as possible with two control patients on their initial MDS-UPDRS III status (including subscales) and their initial LEDD intake, as well as matching the duration of the observation period for each such target-control pair. The target and control groups were selected using scripts written in the Python programming language.
Statistical analysis
The total MDS-UPDRS-III score off medication, the axial, bradyhypokinesia, rigidity, and tremor subscores were used to assess the effect of SSRI/SNRI on the motor symptoms. The data were analyzed using hierarchical linear regression, also known as multilevel modeling. 25 We specified and estimated a linear growth model for the MDS-UPDRS-III score that allowed each patient to have their own initial MDS-UPDRS-III score and rate of change. We used group, examination, initial LEDD, depressed mood (item 1.3), anxious mood (item 1.4) and apathy (item 1.5) as fixed factors. Depressed mood, anxious mood and apathy were included as factors since they all can be manifestations of depression. The patients were treated as a random factor. We were mainly interested in two- (group × examination) and three-way interactions (group × examination × initial LEDD) between the factors. Moreover, Mann-Whitney U-test and Chi-square (χ2) tests were used to assess the differences between the groups, as well as between the examinations within each group. The data were presented as median (Mdn) and 25th-75th percentiles of the values. The analysis was performed using the nlme package in R statistical software. 26 The p-values below 0.05 were considered significant. All the tests were two-tailed.
Results
The final sample consisted of 47 target group patients, and 90 control group patients (Table 1). As already mentioned, every target patient had two control patients matched with it, except for four target patients, for which only one good match could be found. There was no difference in age (p = 0.41) or disease duration (p = 0.68), as well as in gender (p = 0.42), between the target and control group at first examination. We found significant differences between the target and the control group at first examination in the MDS-UPDRS-I total score (p < 0.001; Mdncontrol =5 (3–7), Mdntarget = 8 (6–12), depressed mood item 1.3 score (p = 0.006; Mdncontrol = 0 (0–0), Mdntarget = 0 (0–1), anxious mood item 1.4 score (p < 0.001; Mdncontrol = 0 (0–1), Mdntarget = 1 (0–2), cognitive impairment item 1.1 score (p = 0.02; Mdncontrol = 0 (0–0), Mdntarget = 0 (0–1), and apathy item 1.5 score (p = 0.02; Mdncontrol = 0 (0–0), Mdntarget = 0 (0–1), while the differences between groups on the other measures were not significant (all p > 0.05). Importantly, there were no significant differences between the groups in the measures on which they were matched: MDS-UPDRS-III total score (p = 0.92) and its subscale scores: axial (p = 0.68), tremor (p = 0.92), rigidity (p = 0.72), and bradyhypokinesia (p = 0.91), initial LEDD (p = 0.75), and time difference between the first and the second examination (p = 0.87).
Total motor (MDS-UPDRS-III) score change related to SSRI/SNRI intake
The main effect of group (Figure 1 and Supplemental Table 2a, b) was not significant (p = 0.43), suggesting that there were no differences in motor condition between groups at the time of initial testing, confirming that the matching on the MDS-UPDRS-III score was successful. There was a significant three-way interaction group × examination × initial LEDD (p = 0.03), suggesting that the lower initial LEDD in the target group was associated with greater worsening of motor scores, with an absolute estimate of 1, 95% CI [0.01, 2.20] to 2.5, 95% CI [0.26, 5.64] (calculated for the median LEDD values of 110 and 285 for the control and patient group respectively, Figure 1 and Supplemental Table 2a). The significant two-way interaction group × examination (p = 0.01) suggested steeper worsening of motor symptoms in the target group, with an absolute estimate of 4.24, 95% CI [0.98, 7.51] total MDS-UPDRS-III units (Figure 1 and Supplemental Table 2a). MDS-UPDRS I Apathy score (item 1.5) was a significant predictor of the total MDS-UPDRS-III score (p = 0.03) with an absolute estimate of 1.84, 95% CI [0.23, 3.44]. In other words, one apathy point predicted 1.84 of the MDS-UPDRS-III total score, suggesting worsening of the motor symptoms with increasing apathy.
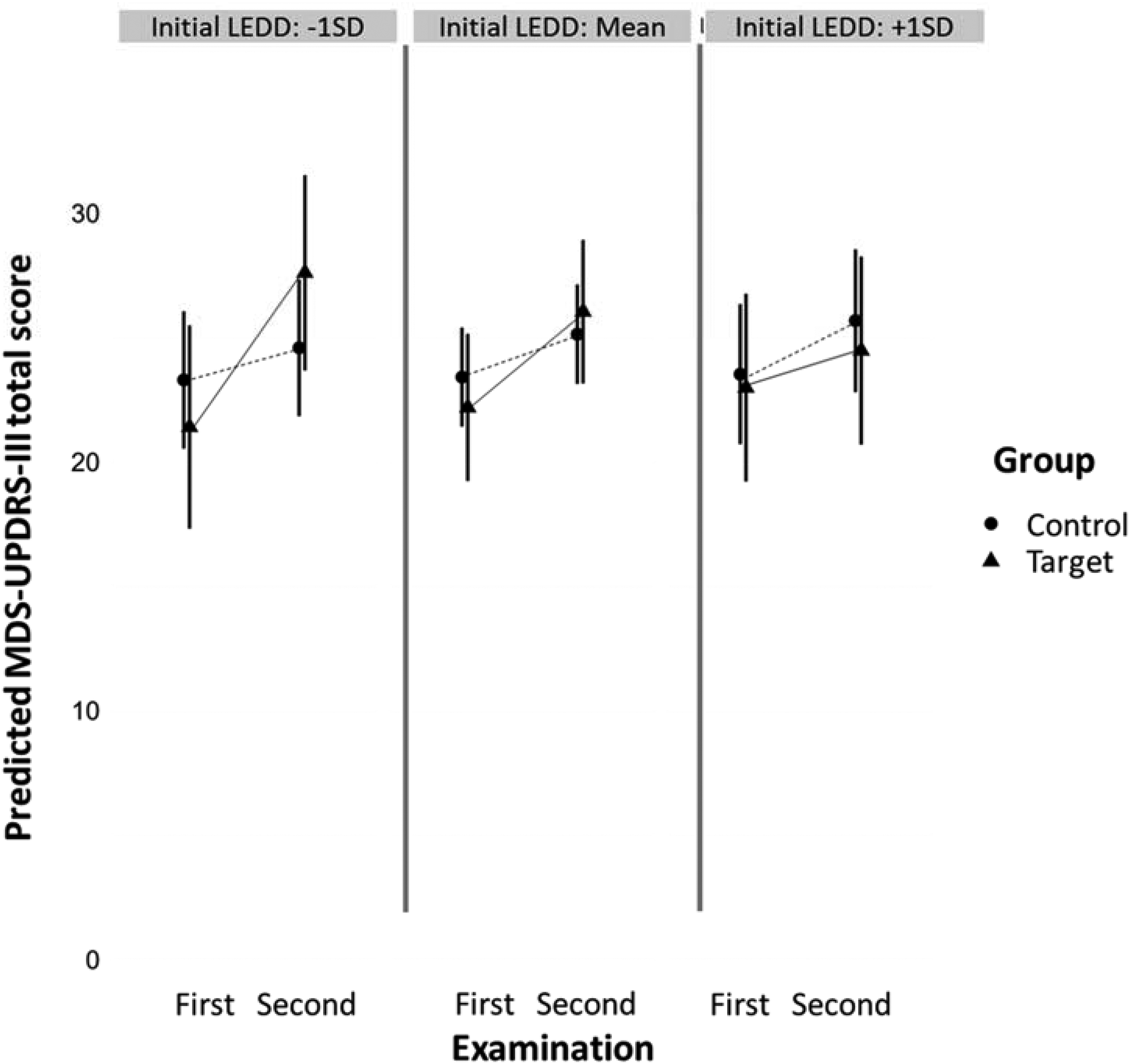
The figure shows the predicted values of the MDS-UPDRS-III total score on the y-axis, and examinations (first and second) on the x-axis, by group (target and control), represented by lines with different mark types. Three panes show this interaction between group and examination on three different levels of initial LEDD: −1 standard deviation, mean and +1 standard deviation.
Subscore (MDS-UPDRS-III) motor changes related to SSRI/SNRI intake
When predicting the axial subscore (Figure 2(a), Supplemental Tables 3a, b), there was a significant three-way interaction group × examination × initial LEDD (p = 0.01) suggesting that the lower initial LEDD in the target group was associated with greater worsening of the axial symptoms, with an absolute estimate of 0.2, 95% CI [0.05, 0.33] to 0.6, 95% CI [0.14, 0.85] (calculated for the median LEDD values of 110 and 285 for the control and target group respectively, Figure 2 and Supplemental Table 3a). In addition, the two-way interaction examination × group was also significant (p = 0.03), with an absolute estimate of 0.69 95% CI [0.09, 1.28], suggesting different rates of change in the MDS-UPDRS-III axial subscore between the groups with a steeper progression of axial symptoms in the target group. Similarly to the total MDS-UPDRS-III score, the MDS-UPDRS-I apathy score (item 1.5) significantly predicted the axial subscore (p = 0.03) with an absolute estimate of 0.29, 95% CI [0.03, 0.56], i.e., one apathy point predicted 0.29 of the MDS-UPDRS-III axial subscore, suggesting worsening of the axial symptoms with increasing apathy.
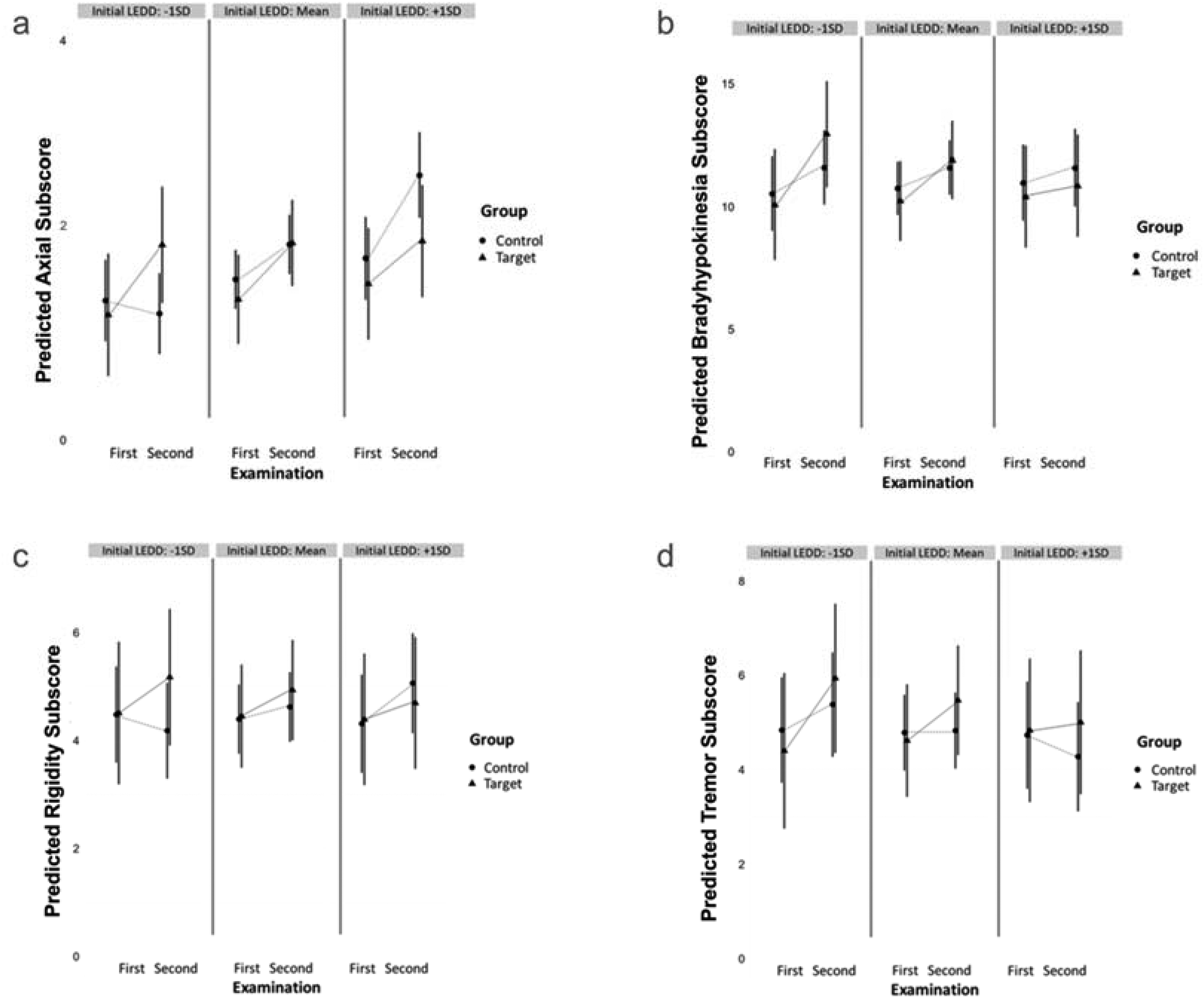
Figures show predicted values of MDS-UPDRS-III subscores on y-axes: axial (a), bradyhypokinesia (b), rigidity (c), and tremor (d). Each figure shows examinations (first and second) on the x-axis, by group (target and control), represented by lines with different mark types. Three panes within each figure show this interaction between group and examination on three different levels of initial LEDD: −1 standard deviation, mean and +1 standard deviation. The Axial subscore was caluclated as a sum of the MDS-UPDRS-III items 3.9 through 3.13, the Bradyhypokinesia subscore as a sum of the items 3.4a through MDS-UPDRS-III 3.8b and 3.14, the Rigidity subscore as a sum of the MDS-UPDRS-III items 3.3a through 3.3e and the Tremor subscore sum of the items MDS-UPDRS-III. 3.15a through 3.18.
For the bradyhypokinesia (Figure 2(b), Supplemental Tables 4a, b), the interactions were not significant (all p > 0.10), although the effect of the MDS-UPDRS-I apathy score (item 1.5) was significant (p = 0.02) with an absolute estimate of 1.06, 95% CI [0.15, 1.96], i.e., one point of apathy change predicted 1.06 of the bradyhypokinesia subscore, suggesting worsening of bradyhypokinesia with worsening apathy. For the rigidity (Figure 2(c), Supplemental Tables 5a, b), the two-way interaction examination × initial LEDD was significant (p = 0.04) suggesting that the association of the initial LEDD and rigidity subscore differs at the time of the first and the time of the second examination, with an absolute estimate of 0.2, 95% CI [0.11, 0.33] for the control and an absolute estimate of 0.6, 95% CI [0.03, 0.85] for the target group for the median LEDD of 110 and 285 respectively. For the tremor (Figure 2(d), Supplemental Tables 6a, b), the interactions were not significant (all p > 0.10), even though the target group seemed to show a similar trend of greater worsening of these symptoms with lower initial LEDD.
Discussion
The main finding of this study is that PD patients who had been taking SSRI/SNRI had a steeper worsening of motor symptoms. In addition, lower values of initial LEDD were associated with a greater motor worsening. Axial, bradyhypokinesia, rigidity and tremor symptoms all appeared to show a similar pattern of greater deterioration in the target group associated with lower initial LEDD, but this was only significant for axial symptoms. In addition, apathy was an independent predictor of general motor worsening, particularly worsening of axial symptoms and bradyhypokinesia.
The finding that patients on SSRI/SNRI show greater motor decline could have several explanations. Both SSRIs and SNRIs share serotonergic modulation as their main function 27 so possible mechanisms that explain our findings could apply to both classes of medication. One explanation would be that SSRI/SNRI themselves influence motor progression, causing steeper motor deterioration. According to some studies, SSRIs may cause motor deterioration, 28 and the start of SSRI use may quickly lead to need for a higher LEDD, 29 suggesting greater motor deterioration. The effect of SSRIs, and possibly SNRIs, in altering motor progression may lie in their mechanisms of action. Both SSRIs and SNRIs inhibit the reuptake of the neurotransmitter serotonin, resulting in an increase in serotonin activity. 6 SNRIs also increase noradrenaline activity. 7 This increase in serotonin or noradrenaline activity or the possible imbalance between these activities and other neurotransmitter activities (such as dopamine) could possibly lead to a different motor course of the disease. 8 Another explanation could be the influence of SSRIs and SNRIs on the metabolism of levodopa. 30 SSRI/SNRI treatment could lead to an altered effect of levodopa and thus less effective treatment, which could cause faster motor progression in the long term. However, clear evidence for this interference with levodopa metabolism has only been provided by behavioral studies.31,32 In addition, considering that it is common to receive dopamine agonists as the first dopaminergic treatment, 33 and also keeping in mind their antidepressant effects, 5 the combination of dopamine agonists and SSRIs or SNRIs could also explain our findings. For example, a double-blind clinical trial in patients with major depressive disorder it was reported that the combination of SSRIs with dopamine agonists may lead to a higher dropout rate, which is explained by intolerable effects in patients, such as severe worsening of mood or headache. 34 So, combining both medications in PD patients could lead to more adverse effects and possibly underdosing with dopaminergic medication, which could, in turn, cause worse motor symptoms. The underlying depression that these patients have could also offer an explanation. Starting antidepressants, especially SSRIs, in patients with a short history of depression has been associated with a higher risk of PD. 35 It is also known that depression is associated with motor disorders36,37 and may have a similar motor impairment as PD, 38 potentially altering the presentation of the motor symptoms over time. On the other hand, depression could be an indicator of worse disease progression, which would also be characterized by greater motor deterioration.39,40 Importantly, the difference of 3.42 total MDS-UPDRS-III points nevertheless higher than the suggested clinically insignificant threshold difference of 2. This should possibly be taken into account when considering treatment options of whether to prescribe SSRI/SNRI to PD patients depending on the level of depression.
The observation that poorer motor progression, albeit with small estimates, is predicted by lower initial LEDD in patients taking SSRIs/SNRIs may be these patients being neurobiologically at an earlier stage of deterioration and having more room for symptom worsening. In general, lower LEDD can indicate a good response to dopaminergic treatment and slower disease progression, 41 which seems to be the trend we see in our control group. However, in a recent study exploring the association between levodopa responsiveness and disease progression in PD in a large group of patients, LEDD was significantly higher at the time of the levodopa challenge test in definite responders, compared to the limited responders 42 ; limited levodopa responsiveness was associated with faster disease progression. 42 In addition, in a double blind, placebo controlled study, the severity of parkinsonism increased more in the placebo than in the groups taking levodopa, which is consistent with the notion that levodopa slows disease progression, 43 although the results of the neuroimaging ([123I] β-CIT SPECT of the dopamine transporter) from the same study data have suggested either that levodopa accelerates the loss of nigrostriatal dopamine nerve terminals or that its pharmacologic effects modify the dopamine transporter. Moreover, the data from one of the best naturalistic studies of PD progression published to date 44 showed that although levodopa is associated with motor fluctuations, nocturnal OFF disability was consistently less severe in patients taking levodopa than in patients not taking levodopa, regardless of disease duration. 44 The response to levodopa included axial signs that are generally considered non-levodopa-responsive, including postural instability and falls. 44 In our study the different subscales seem to follow a similar pattern as the total motor scores, where patients taking SSRI/SNRI show steeper deterioration, especially in those who had a lower baseline LEDD, although this was statistically significant only for the axial symptoms.
In our study, apathy was an independent predictor of the progression of motor symptoms in PD, especially axial symptoms and bradyhypokinesia. These results are consistent with previous work 45 in drug naïve PD patients in which an association between apathy and motor symptoms, as well as an association between apathy and male gender and apathy and depression was found. This suggests a common underlying pathophysiological mechanism between apathy, motor severity and motor progression in PD, which includes neurodegeneration, as apathy is more common in PD than in equally disabled patients with other chronic conditions. 46 Other studies found no association between apathy and motor progression in PD, 46 despite the association of apathy with cognitive deterioration. In our study we found no association between anxiety and depression and motor progression in PD, which is partly consistent with the results of other studies, that found an association between depression, but not anxiety, and disease progression. 46 These different findings are consistent with the idea that, although it is sometimes clinically difficult to distinguish between apathy, depression and anxiety, they probably have different pathophysiological mechanisms and represent different pathological entities. 47
A limitation in this study is a relatively small sample size, which could also account for the fact that not all the subscales show the same significant effects. However, this limitation is also partly addressed by the matched-subject study design, although this subject-by-subject matching imposed restrictions upon the choice of subjects, restricting the sample size, which could have resulted in reduced statistical power. On the other hand, by using the described matching process, we were able to minimize the impact of the between-subject differences which could have influenced our results. In addition, we did not control for the effect of doses of the drugs. However, patients taking different drugs with the same (SSRI) or similar (SNRI) modes of action were included in the study. Also, to the best of our knowledge, there is no good way of calculating SSRI/SNRI equivalent daily dose, hence we relied on how long (2 to 7 months) the patients have been taking the drug, rather than the dose of certain drugs they were on. We also did not consider the possible modifying effect of other non-motor symptoms that may have influenced motor progression. However, the predictors (anxiety, depression, apathy) most relevant to the aim of the study—to investigate the possible effect of SSRI/SNRI on the motor symptoms of PD—were included in the model.
In conclusion, SSRI/SNRI-use seems to be characterized by a steeper worsening of motor symptoms in PD, which can be predicted by a lower initial LEDD. Our study therefore contributes to understanding of the possible influence of SSRI/SNRIs on disease progression in PD. Future studies should focus on the long-term effects of SSRI/SNRI use in PD patients in a prospective setting, preferably with a larger sample size.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241296016 - Supplemental material for The effect of serotonin reuptake and serotonin-noradrenaline reuptake inhibitors on motor symptoms in Parkinson's disease: A PPMI-based matched-subject study
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241296016 for The effect of serotonin reuptake and serotonin-noradrenaline reuptake inhibitors on motor symptoms in Parkinson's disease: A PPMI-based matched-subject study by Teodora Matić, Martijn Hendriks, R Saman Vinke, Aleksander Sadikov and Dejan Georgiev in Journal of Parkinson's Disease
Footnotes
Acknowledgements
Data used in the preparation of this article were obtained on July 8, 2021 from the Parkinson's Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/access-dataspecimens/download-data), RRID:SCR 006431. For up-to-date information on the study, visit ![]() . PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Janssen Neuroscience, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics. This study has received support from the Slovenian Research and Innovation Agency (ARIS), within the Artificial Intelligence and Intelligent Systems research program (Grant No. P2-0209).
. PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Janssen Neuroscience, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics. This study has received support from the Slovenian Research and Innovation Agency (ARIS), within the Artificial Intelligence and Intelligent Systems research program (Grant No. P2-0209).
Funding
This study was supported by the Slovenian Agency for Research and Innovations (ARIS) grant number P2-0209.
Declaration of conflicting interests
RSV acts as an independent consultant for Boston Scientific.
Data availability
The data used in this study are available at http://www.ppmi-info.org/access-dataspecimens/download-data (Parkinson's Progression Markers Initiative (PPMI) database RRID:SCR 006431). For up-to-date information on the study, visit ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
