Abstract
Background
Timely recruitment and effective retention of participants are challenging for clinical Parkinson's disease (PD) research studies. Not achieving this can affect delivery costs and bias results.
Objective
To identify successful recruitment and retention methodologies for clinical research studies in PD.
Methods
We conducted a systematic scoping review using a Population, Intervention, Comparator, Outcome and Study design (PICOS) framework to search and screen literature within five databases for published quantitative and mixed methods studies evaluating recruitment and retention strategies of interventional and observational trials in PD.
Results
Searches identified 4608 records, of which six papers covering eight studies were included in the review following screening for eligibility. No papers on retention strategies were found that met eligibility criteria. Effective strategies for rapid recruitment included research registries and trial finders (n = 3), digital marketing campaigns (n = 3), site recruitment support (n = 1) and multiple referral sources (n = 1). Recruitment diversity was increased by online campaigns, mandatory 10% minority targets set by funders and supplemental site funding.
Conclusions
This scoping review has identified methods that can be used to increase the rate and diversity of recruitment to PD clinical studies. Retention methodologies are under-researched.
Plain language summary
Recruiting patients to clinical studies and ensuring participants complete them (known as “retention”) is challenging in Parkinson's disease (PD). If studies take longer this increases costs, and if the right people do not take part in studies this can mean that results may not be representative of the PD community. Our objective is to find out what effective strategies there are to support recruitment and retention to clinical research studies in PD. We reviewed literature within five study databases. We used the population, intervention, comparator, outcome and studies (PICOS) framework to create a search strategy based on our research question, and inclusion criteria for study selection. Important criteria were: (1) studies had to test a recruitment or retention strategy in a PD clinical research study, (2) studies had to provide evidence on how well the strategy worked. We found 4608 unique research papers, six of which described and evaluated recruitment strategies. No papers on retention strategies were found that met eligibility criteria. Successful recruitment strategies included digital marketing (n = 3), research registries (n = 3), supporting study sites (n = 1) and multiple sources to refer people with Parkinson's (PwP) to studies (n = 1). Recruitment of PwP from diverse backgrounds was enhanced by (1) online campaigns, (2) funders requesting that 10% of study participants come from diverse backgrounds 3) additional funding for research teams.
This scoping review identified methods that can be used to increase the rate and diversity of recruitment to PD clinical studies. Retention methodologies are under-researched.
Introduction
Parkinson's disease (PD) is a debilitating, slowly progressive neurodegenerative disease, and no therapies have been shown to halt or slow progression. PD prevalence in the United Kingdom (UK) is expected to increase to 168,000 by 2025. 1 Clinical trials are therefore required to improve health outcomes for people with PD (PwP).
Less than half of all clinical trials meet their recruitment targets, despite extensions to the recruitment window. 2 The reasons for this are multifaceted, including lack of research awareness, mobility and travel issues (particularly for elderly participants), as well as a mistrust in clinicians and fear of harm. 3 Furthermore, once recruitment has taken place, studies can struggle to retain participants, with around a quarter of trials experiencing attrition rates of more than 10%. 4 Problems with recruitment and retention not only affect the timely delivery of trials, but risk introducing selection bias leading to a study being underpowered, under-representative of the target population and with reduced accuracy and applicability of results. 5 Disease-modification trials in PD are particularly challenging, 6 as assessments in the OFF medication state and longer follow-up periods are often necessary. 7 Representative recruitment to PD trials is difficult with a reported mismatch of trial participants and the intended user group of certain PD therapies, where trial populations are on average younger and with less comorbidities compared to the real-world treatment population. 8
The aim of this scoping review is to understand strategies and methodologies that effectively improve recruitment and retention to clinical research studies in PD.
Methods
Study protocol
This scoping review protocol follows the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guide for scoping reviews 9 and is registered on the Open Science Framework (https://osf.io/er2jz).
Search strategy development
Following the protocol, a search strategy was developed based on the research question “What strategies/methodologies effectively improve recruitment and retention to clinical research studies in PD?” and mapped to the Population, Intervention, Comparator, Outcomes and Study design (PICOS) framework 10 (Supplemental Material 1).
There are no existing MeSH terms for ‘recruitment’ or ‘retention’, due to their broad meaning. To inform the development of a comprehensive search strategy that would allow the capture of relevant literature, a preliminary search was carried out to identify papers that meet eligibility criteria. Four papers were identified from preliminary searches. NVIVO software (Version 14; released 14 March 2023) word cloud generator was used to find common terms based on abstracts, titles and keywords. In collaboration with a University of Plymouth Information Specialist a final search strategy was developed and reviewed in accordance with peer review of electronic search strategies (PRESS) guidelines. 11 The final search strategy is shown in Supplemental Material 2.
Five databases were searched to find peer reviewed articles for consideration for inclusion: MEDLINE, CINAHL, Embase and Web of Science, as well the Study Within a Trial (SWAT) database. Additional articles were identified through the screening of citations and references of any articles that met eligibility criteria throughout the study selection process.
Study selection
Quantitative and mixed method studies in English, indexed from 01/01/2008 to 05/10/2023 were included in the review. This date was selected due to the timing of considerable changes in the use of digital interventions for research recruitment. Only randomized and non-randomized Clinical Trials of an Investigational Medicinal Product (CTIMPs) or observational studies were included in our review, and we excluded surgical and cell and gene therapy trials due to their differing nature in terms of invasiveness and motivation for participation. Search results were first screened for any duplicate entries. The title and abstract from the remaining search results were screened according to the PICOS framework. RP screened all records and a second independent review was provided by either MLZ, TB or MB who were masked to each other's decisions. The full text was also reviewed in cases where inclusion was not clear from the title and abstract. Reasons for exclusion were recorded along with screening decisions. Where there was disagreement on decisions, the full article was reviewed by both reviewers followed by a discussion to reach consensus. Where consensus could not be reached, a decision was made via consultation with a third expert reviewer (CBC).
Data extraction and synthesis
One reviewer (RP) extracted the data from the full articles (Supplemental Material 3). A second reviewer verified the extracted data (MLZ) and any differences which remained outstanding were resolved by a third expert reviewer (CBC).
Parent study characteristics
For all papers information was extracted regarding the overall aim. Study design, patient characteristics and recruitment target was then extracted for each parent study featured in a single paper.
Recruitment and retention strategies
Description of strategies used within each parent study were extracted, including modality of delivery and duration.
Comparator and outcome measures
Details of any comparators used to measure the effectiveness of strategies and their key findings were extracted, as well as a description and key findings of all other outcomes measured.
All extracted data was then narratively synthesized and presented as overall suggestions for future clinical research studies.
Results
Database search results
We identified 4608 studies through database searches. After duplicates were removed, 1659 studies remained, of which 629 were excluded by title and abstract screening. The remaining 30 studies were assessed for eligibility via full-text screening, six of which met inclusion criteria (Figure 1).
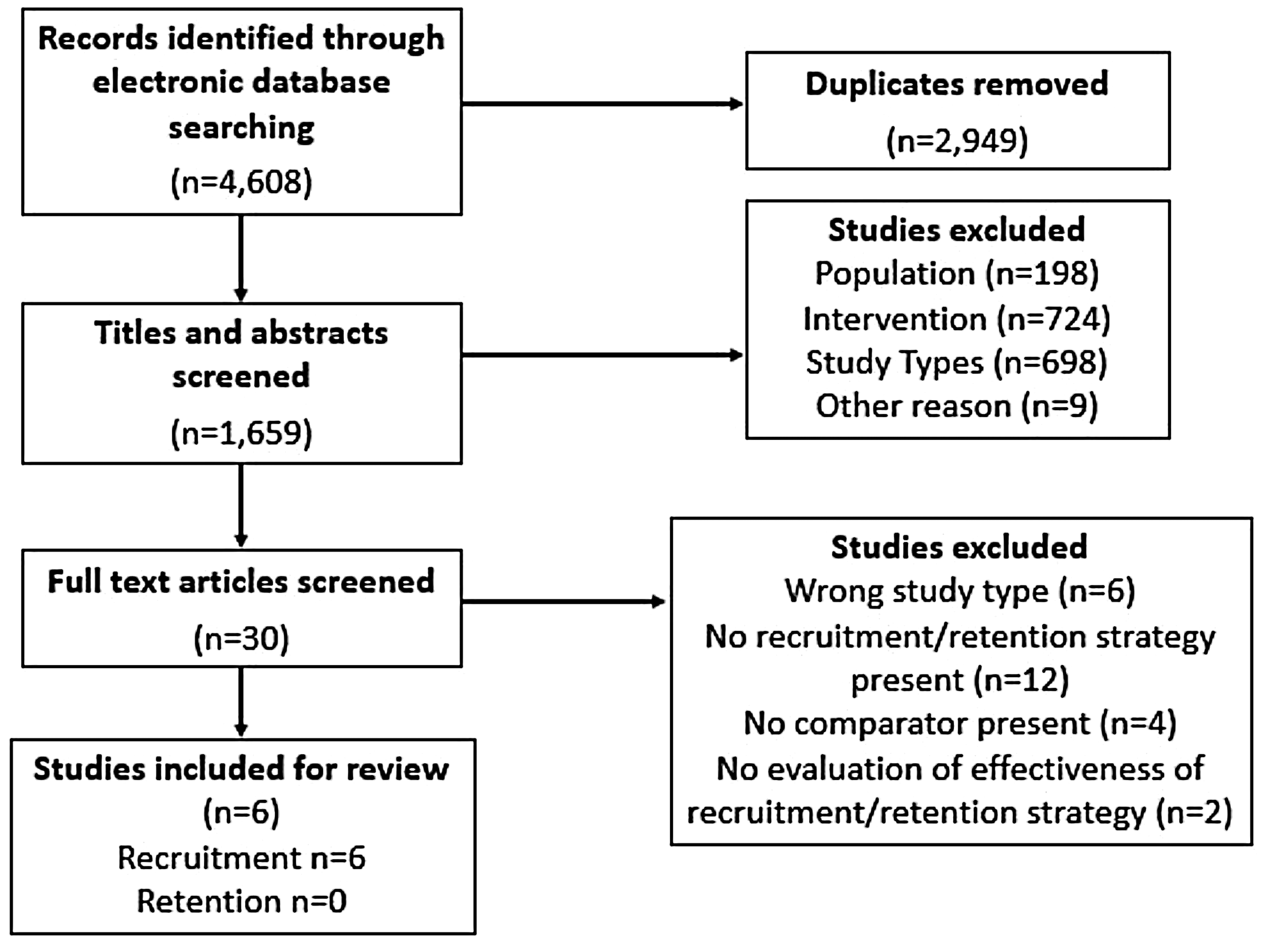
PRISMA diagram for included studies.
Characteristics of included papers
All six papers included for review evaluated recruitment strategies; none evaluated retention methods. Five of the six papers were purely quantitative, and two included qualitative data.12,13 Three papers evaluated recruitment to CTIMPs12–14 and three papers to observational studies.15–17
Four papers evaluated different recruitment activities within a single parent study12,13,15,17 and two compared recruitment activities of different parent studies.14,16 Three papers’ main focus was on the recruitment of racial and ethnic minorities13–15 (Table 1).
Characteristics of selected papers and their individual parent studies.
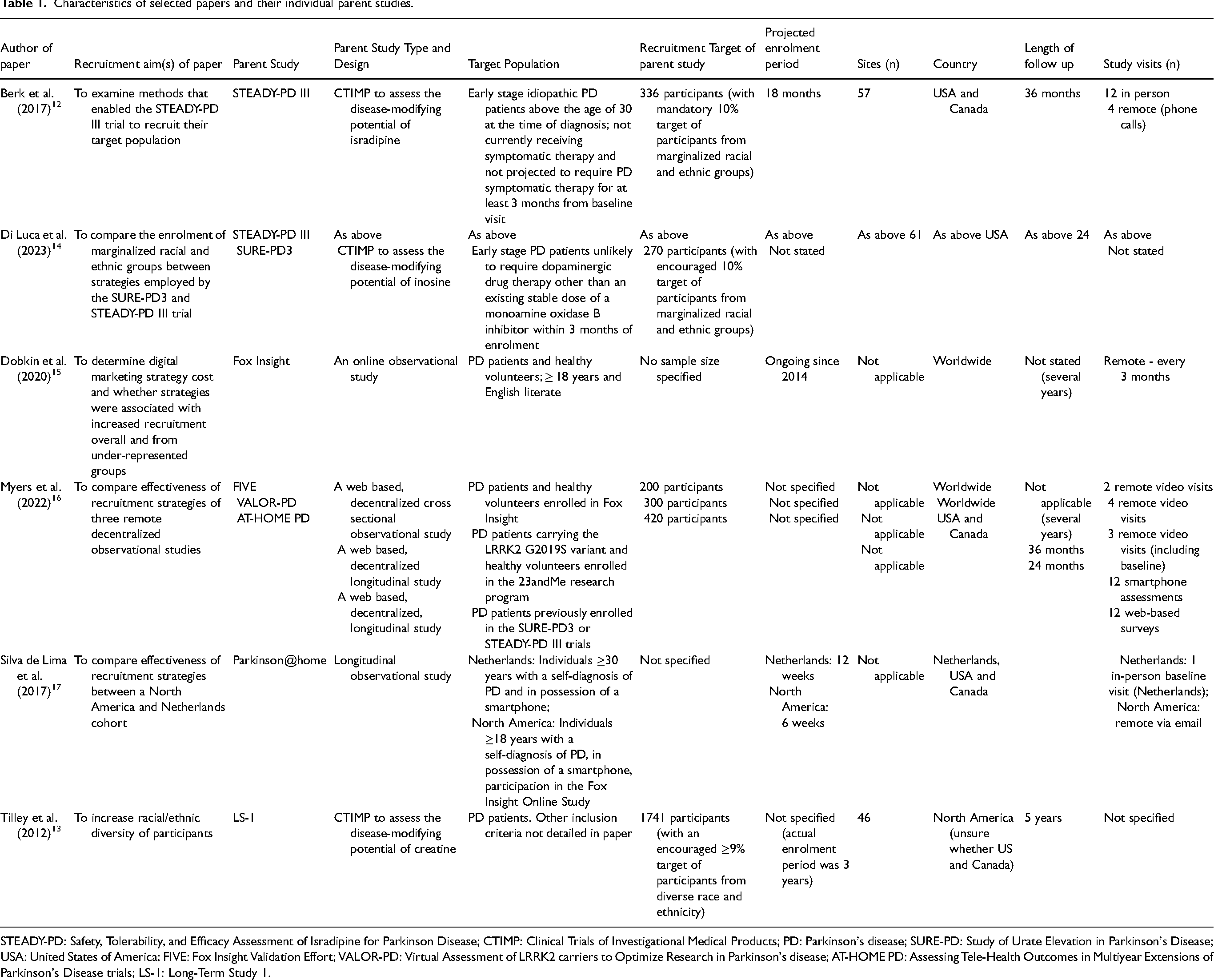
STEADY-PD: Safety, Tolerability, and Efficacy Assessment of Isradipine for Parkinson Disease; CTIMP: Clinical Trials of Investigational Medical Products; PD: Parkinson's disease; SURE-PD: Study of Urate Elevation in Parkinson's Disease; USA: United States of America; FIVE: Fox Insight Validation Effort; VALOR-PD: Virtual Assessment of LRRK2 carriers to Optimize Research in Parkinson's disease; AT-HOME PD: Assessing Tele-Health Outcomes in Multiyear Extensions of Parkinson's Disease trials; LS-1: Long-Term Study 1.
Details of recruitment strategies and their results can be found in Table 2 and are summarized below and in Table 3.
Recruitment strategies used per study and key conclusions.
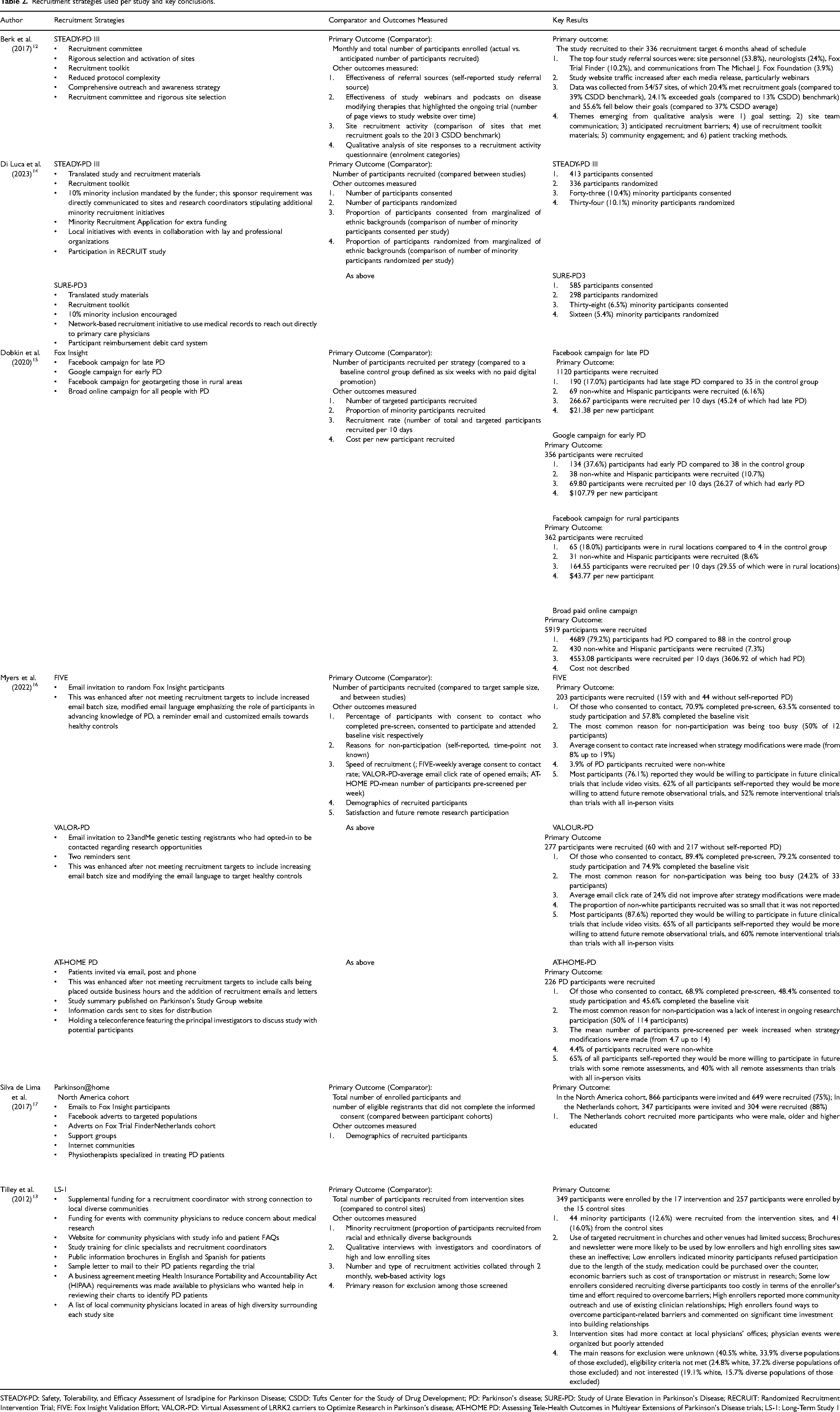
STEADY-PD: Safety, Tolerability, and Efficacy Assessment of Isradipine for Parkinson Disease; CSDD: Tufts Center for the Study of Drug Development; PD: Parkinson's disease; SURE-PD: Study of Urate Elevation in Parkinson's Disease; RECRUIT: Randomized Recruitment Intervention Trial; FIVE: Fox Insight Validation Effort; VALOR-PD: Virtual Assessment of LRRK2 carriers to Optimize Research in Parkinson's disease; AT-HOME PD: Assessing Tele-Health Outcomes in Multiyear Extensions of Parkinson's Disease trials; LS-1: Long-Term Study 1
Summary of recruitment strategies and their effectiveness.
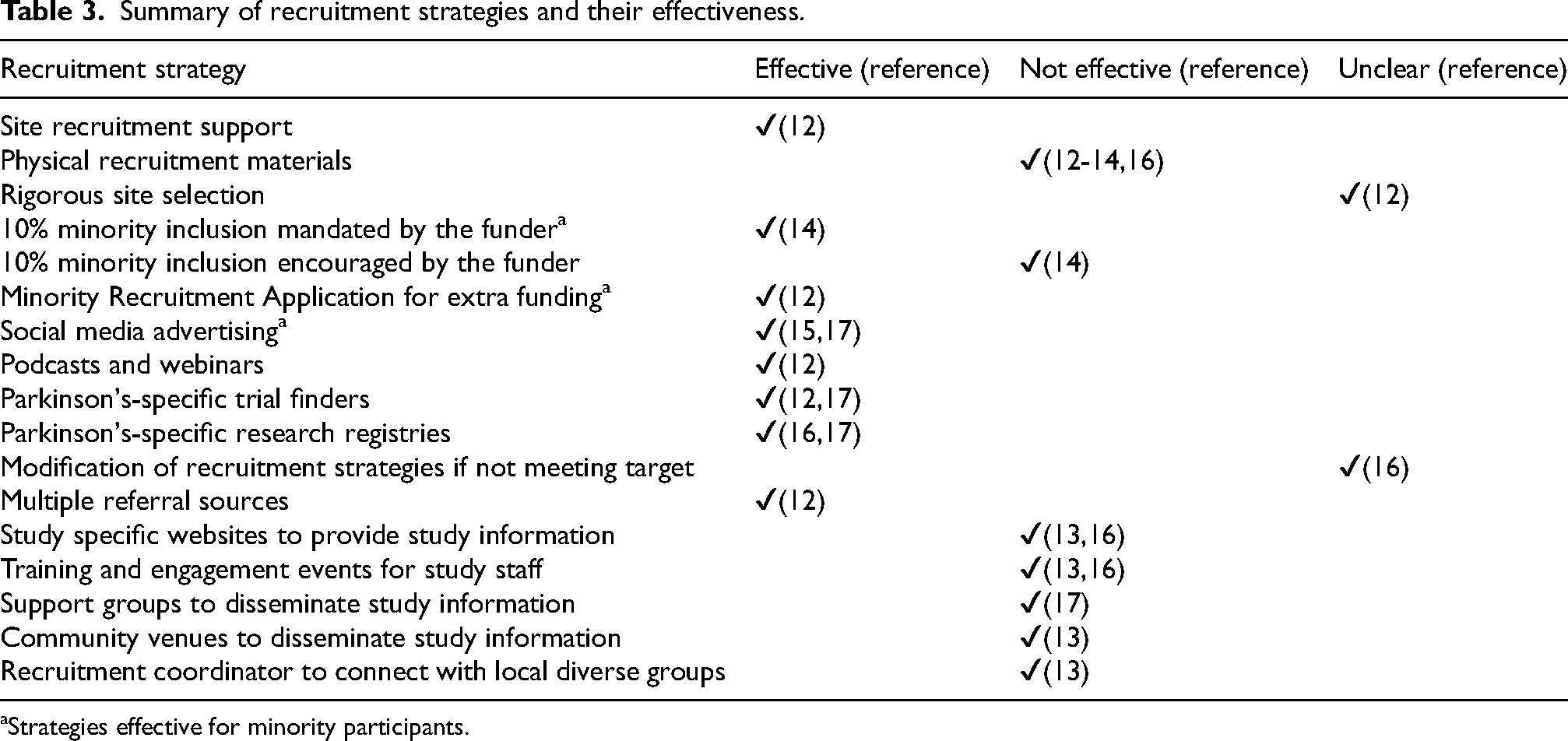
Strategies effective for minority participants.
Recruitment strategies and their effectiveness
We classified the recruitment methodologies reported in the included papers into 5 groups: (1) digital marketing campaigns which included outreach via social media, webinars, podcasts, study-specific and other website; (2) recruitment from an existing registry of participants who had previously consented to contact for future research opportunities or from trial finder platforms; (3) strategies specifically targeting minority participants; (4) physical recruitment materials including posters/flyers; (5) all other recruitment strategies. We will discuss the effectiveness of each. The strategies used varied considerably and in most papers (n = 5) several strategies were grouped together to form an overall recruitment package (Table 2).
Digital marketing campaigns
Three of the eight studies used a digital marketing strategy which included the use of social media, podcasts, press releases and television appearances,12,15,17 all of which met or exceeded their recruitment target, mainly attributed to the use of social media. Other online strategies included use of a study website, either to give PwP or study staff information about the study.13,16
Research registries and trial finders
Three out of four studies that used a trial finder 12 or registry database15,16 as part of their recruitment package met or exceeded their recruitment target. One study 17 found that a North American cohort who utilized both a trial finder and research registry as part of their recruitment package recruited more effectively than a participant cohort based in the Netherlands who did not use these strategies. However, because all parent studies evaluated the use of a registry or trial finder as part of a whole recruitment package, the individual effectiveness is not clear.
Minority recruitment
The aim of three of the papers, which covered four parent studies, was to recruit a representative sample of minority participants.13–15 Of these, two parent studies recruited successfully. Berk et al. 12 recruited a representative sample of 10.1% racial and ethnic minorities. Recruitment strategies employed in this study included translated study and recruitment materials, local initiatives in collaboration with minority PD organizations such as the Muhammed Ali Foundation, a mandatory 10% minority inclusion target set by the study funder and a minority recruitment funding source which study sites could apply for to gain additional recruitment funding. Dobkin et al. 15 used targeted social media advertising to improve recruitment of rural participants. This strategy resulted in the recruitment of 65 (18%) rural participants, compared to four (3%) in the control group who did not utilize any digital marketing strategies. The study also found that their Google advertising campaign recruited 10.7% non-white and Hispanic participants; however, as participants comprised both healthy controls and PwP, it is not known how many participants in these groups had PD.
Further insight into minority recruitment was achieved during qualitative interviews with site staff in one other paper. 13 Here it was found that minority participants were less likely to want to be involved in the trial due to its length and economic barriers including the cost of transportation. Low enrolling sites in particular commented on the time and effort it takes to mitigate these barriers.
Physical recruitment materials
Physical study materials such as cards, brochures and thank you cards were utilized by four of the studies,12–14,16 but in these cases they were evaluated as part of a whole recruitment package and so their individual effectiveness is unknown. However, one study 13 used qualitative interviews to discuss how low enrolling sites were more likely to use brochures and newsletters than high enrolling sites. High enrolling sites also perceived mailing study brochures to local physicians to be ineffective.
Other recruitment strategies
Additional recruitment strategies included the use of multiple referral sources, forming a recruitment committee for rigorous site selection and comprehensive recruitment support for sites, as well as reduced protocol complexity. 12 Although this study managed to recruit to target six months ahead of schedule and reach 10% minority inclusion, these strategies were not individually assessed and so their effectiveness is unknown.
Three studies that were compared in one paper 16 enhanced their recruitment strategies to include more comprehensive email or phone contact methods. However, the outcomes for each study were measured differently, with one of these studies seeing no improvement on email click rate, while another saw an increase in the consent to contact rate for database participants. The other study in this paper utilized the addition of calls made outside business hours which the authors concluded resulted in improvements in the weekly pre-screening rate.
Unsuccessful recruitment strategies included the use of community settings such as PD support groups, 17 as measured by comparative recruitment with a study cohort who used different recruitment methods. The use of churches and other venues 13 to disseminate study information was also deemed unsuccessful, as described in qualitative interviews.
Also suggested as being unsuccessful were attempts to directly engage clinicians and site staff by teleconference and training events,13,16 both of which were reported as being poorly attended. However, their effect on recruitment is not directly assessed and so their individual effectiveness is unknown.
Discussion
This review aimed to identify strategies that have been shown to be effective for supporting recruitment and retention of people with PD to clinical research studies. Evidence from six papers reporting on interventions to support recruitment in three CTIMPs and five observational studies was included. There was considerable heterogeneity in aim, recruitment strategies, and measurement of effectiveness, with many reporting the success of a package of recruitment methods rather than individual strategies.
Retention
The lack of studies that measured or investigated the effectiveness of retention strategies was a key finding of this review, highlighting an important knowledge gap. There is qualitative data exploring participant trial experience and the barriers and facilitators to retention;7,18–20 however, this knowledge has not been translated into evaluable retention strategies. Insights from similar studies may be used to devise strategies that could be formally tested within trials.
The effect of the recruitment strategy on retention was not evaluated in any of the papers. Such evaluation has been carried out in a range of other disease areas providing valuable insights. 21 The failure of the papers included in this scoping review to assess the impact of their recruitment strategies on study retention could be considered a lost opportunity.
Recruitment
Several strategies were found to enhance recruitment; these included the use of research registries or trial finders, digital marketing campaigns, site recruitment support and multiple referral sources. Some recruitment strategies were suggested to have little or no effectiveness. These included the use of physical recruitment materials, the use of support groups and community settings to disseminate study information, and training and engagement events for site staff.
All but one study that employed PD specific research registries or trial finders recruited more participants compared to the study's recruitment target or study cohort that did not use this strategy. The one study that found this method ineffective 16 utilized a genetics research registry and was limited to LRRK2 G2019S carriers which may explain why their recruitment target was not met. Research registries and trial finders have previously been assessed as a successful means of trial recruitment in brain diseases 22 including dementia. 23 However, both trial finders and registries require significant infrastructure and support for information governance, data privacy and data storage requirements which are significant to support the large user base required to cover a breadth of representation that caters for specific recruitment needs meeting a particular study's eligibility criteria. 24
The use of digital marketing had a positive effect on recruitment for the two observational studies that employed this strategy. Dobkin et al. 15 aimed to recruit a proportion of PD patients to their online observational study, using various online campaigns aimed at the general public. Between 17 and 37.6% of study participants recruited via targeted social media campaigns had PD, while a broad paid online campaign recruited a proportion of 79.2% PD patients. The latter recruitment strategy also included the use of emails to the Michael J Fox Foundation community, which has over 380,000 members, and a television appearance by Michael J Fox, the publicity value of which may have influenced the response rate of participants interested in the study. Berk et al. 12 demonstrated increased website traffic resulting from a heightened online presence in the form of webinars and podcasts of disease-modifying therapies that featured the parent study, which the authors consider contributed to their successful study recruitment.
Social media has been demonstrated to be an effective tool in recruiting to CTIMPs in other disease areas25,26 including in older adults, 27 although almost always deployed in conjunction with other, more traditional recruitment methods. The targeting of participants based on predefined demographic criteria is a particular advantage of social media approaches. 24 However, its use could introduce bias and limit representativeness of a study population: social media users tend to be younger and more computer literate; additionally some ethnic and racial groups tend to be more active on social media than others. 28 Nevertheless, Dobkin et al. 15 found participants recruited via social media to be older, with more complicated medical histories and lower educational attainment compared with the baseline group recruited with no paid digital promotion.
Although paid social media campaigns are costly, a recent systematic review found that social media (mainly the use of Facebook) was almost twice as cost-effective as traditional recruitment methods, and more so than other forms of paid online advertising, although interpretability of these findings is limited by variability in patient population, target sample size, study type, location and recruitment methods leading to large variability in effect sizes. 29
Ongoing, systematic recruitment monitoring is essential to mitigate challenges and barriers to enrolment. 30 Berk et al. 12 used a recruitment committee to contact each study site monthly and amended their individual recruitment strategies, including new outreach opportunities, ways to leverage technology and the revision of recruitment materials. Although additional site support had a very positive impact on a quarter of the 57 sites in the study, deploying a such a strategy involving modifications and monthly meetings would require significant resource, particularly for longer-term trials with higher numbers of trial sites. This could be mitigated with the use of comprehensive central monitoring and adopting a risk-based approach to contacting sites who are struggling to attain their recruitment goals.
The proportion of under-served groups, including those from racial and ethnic minority backgrounds, recruited to studies remains low. Ongoing initiatives are exploring these communities’ knowledge and attitudes towards PD research. 31 Of the three papers that addressed this in their aims,13–15 two identified strategies which may be successful in increasing recruitment diversity. A Google advertising campaign resulted in the recruitment of 10.7% non-white and Hispanic participants to an online observational study, 15 however due to the recruitment of healthy controls it is not known how many participants in this group had PD. When comparing the three CTIMPs which aimed to recruit a target proportion of racial and ethnic minorities, the study that utilized the provision of funding specific to minority recruitment, and setting of mandatory minority recruitment goals recruited a higher proportion of marginalized racial and ethnic participants 12 than those that had encouraged goals, 14 or had a singular strategy of a recruitment coordinator to connect with local diverse groups. 13 Due to the multifaceted nature of research participation barriers, recruitment strategies for minority PD patients should be both multi-layered and tailored to the individual needs of specific minority populations at the time of study design.32,33
Several strategies, including the use of physical recruitment materials and interventions aimed at study recruiters were suggested to not improve recruitment. Two recent systematic reviews found recruitment interventions targeting site staff were of insufficient quality to allow conclusions to be drawn.2,34 Although once a core route of targeted recruitment, the ability for site staff to access potential study participants’ medical histories and discuss eligibility recruitments with clinicians has been impacted following the adoption of more innovative remote trial designs following the COVID19 pandemic. 35 Additionally, the former systematic review found the use of churches and other community venues to disseminate study information may enhance recruitment. The broader entry criteria compared to those of PD studies, and the fact there may have been a low prevalence of PD patients attending church settings may reflect why this conclusion is not supported by the findings of this scoping review.
Another aspect to consider is the types of study identified in this review. Recruitment to an observational study is a very different proposition to recruitment to a more complex and intensive CTIMP. Only two of the three CTIMPs in this review recruited to target, with one achieving this 6 months ahead of schedule. 12 The simplified protocol, including less stringent eligibility criteria and using change in total UPDRS score in the medications’ ON state as the primary outcome measure may have contributed to this more rapid recruitment, particularly as PD patients may have difficulty with OFF assessments in longer-term trials. 7 Additionally, the country in which a study was based may have influenced the results as each has their own healthcare and research infrastructure.
Study limitations
The findings in this review are limited by the exclusion of grey literature and studies published in non-English languages. A lack of formal critical appraisal also means the quality of the papers included was not assessed. A narrow definition of the research question may have limited eligibility of studies. Finally, the presence of possible publication bias, particularly considering the limited number of studies, may have resulted in the overrepresentation of the efficacy of identified strategies.
Implications for future Parkinson's disease trials
It is not possible to draw firm conclusions from this review regarding recruitment strategies for PD trials, due to the limited scope of evidence and heterogeneity of reported strategy content and evaluation. Costs of strategies were only reported for digital marketing campaigns in a single paper and there were no data on further resources such as personnel time. These factors may affect applicability of particular strategies for individual trials. It is also not known whether any of the recruitment activities were co-created with Parkinson's patients. Involvement of the target population in all aspects of trial design is known to reduce barriers to participation, particularly for older adults, 36 and so their input into the development of recruitment strategies is vital. Nevertheless, several suggestions can be made in terms of recruitment interventions for PD trials and how these could be applied (Table 4).
Recruitment suggestions and their potential application.

Conclusion
This review has highlighted different strategies that may increase not only the rate, but the diversity of recruitment to PD clinical research studies. Further studies evaluating retention strategies in PD studies are needed.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241291986 - Supplemental material for Improving recruitment and retention of people with Parkinson's disease to clinical studies: A scoping review
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241291986 for Improving recruitment and retention of people with Parkinson's disease to clinical studies: A scoping review by Rebecca Petty, Veena Agarwal, Jennifer Allison, Sandra Bartolomeu-Pires, Michèle Bartlett, Timothy Boey, Rebecca Croucher, Helen Collins, Sally Collins, Emma Davies, Joy Duffen, Romy Ellis-Doyle, Cristina Gonzalez-Robles, Jemma Inches, Georgia Mills, Sheila Wonnacott, Thomas Foltynie, Victoria Allgar, Tom Thompson, Camille B. Carroll, Stephen Mullin, Marie Louise-Zeissler and in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We would like to thank Chris Johns, Information Specialist from the University of Plymouth who assisted with search strategy development, and all additional PPIE working group members for their input.
ORCID iDs
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was conducted as part of the Edmond J Safra Accelerating Clinical Trials in Parkinson's Disease (EJS ACT-PD) Initiative, which is funded by the Edmond J Safra Foundation. This work was also supported by funding from the MRC-NIHR Trials Methodology Research Partnership (TMRP) Doctoral Training Partnership (DTP).”
Declaration of conflicting interests
CBC and TF are Editorial Board members of this journal, but were not involved in the peer-review process nor had any access to information regarding its peer review.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during this study
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
