Abstract
Background
Recent evidence suggests a link between glycoprotein non-metastatic melanoma protein B (GPNMB) and Parkinson's disease (PD) pathogenesis. Although elevated plasma GPNMB levels associated with disease severity have been reported in PD, cerebrospinal fluid (CSF) alterations remain elusive.
Objective
To explore CSF GPNMB alterations and its clinical significance in PD.
Methods
This study enrolled 118 sporadic PD patients and 40 controls. We examined the potential associations between CSF GPNMB levels and the clinical characteristics or biomarkers of neurodegenerative pathogenesis.
Results
PD patients had higher CSF GPNMB levels than controls (p = 0.0159). In the PD group, CSF GPNMB levels correlated with age (age at examination: rs = 0.2511, p = 0.0061; age at onset: rs = 0.2800, p = 0.0021) and the severity of motor and cognitive dysfunction (MDS-UPDRS III score: rs = 0.1998, p = 0.0347; Mini-Mental State Examination score: rs = −0.1922, p = 0.0370). After correcting for multiple comparisons, the correlation with age at onset remained significant. CSF GPNMB levels were also positively correlated with CSF soluble triggering receptor expressed on myeloid cells 2 (sTREM2) levels in both the PD (rs = 0.3582, p < 0.0001) and control (rs = 0.4743, p = 0.0023) groups. Furthermore, multiple regression analysis revealed CSF sTREM2 level as the strongest determinant of CSF GPNMB levels in the PD group (t-value = 3.49, p = 0.0007).
Conclusions
Elevated CSF GPNMB levels, linked with age and microglial activation, may be a valuable marker for understanding the interplay between aging, neuroinflammation, and PD pathology.
Keywords
Introduction
Parkinson's disease (PD), the second most prevalent neurodegenerative disease, affects approximately 6.1 million people worldwide. 1 Notably, both the incidence and prevalence of PD have increased in recent years. 2 In PD patients, a genome-wide association study uncovered several new genetic variants associated with autophagy–lysosomal pathway function, including polymorphisms in the gene encoding glycoprotein non-metastatic melanoma protein B (GPNMB). 3 GPNMB is an endogenous type 1 transmembrane glycoprotein that was identified in the context of tissue damage and inflammation, and reportedly affects mitochondrial function 4 and endo-lysosomal biology. 5 Several studies have indicated a relationship between single nucleotide polymorphisms in GPNMB and PD development.6–11 In addition, increased expression of GPNMB was proved in the substantia nigra of PD patients. 12 These above studies indicated a close association between elevated expression of GPNMB and PD pathology.
In addition to its role as a genetic risk factor, evidence proposes that GPNMB participates in the PD pathological process. Previous studies have shown elevated GPNMB protein levels and their association with disease severity and progression, primarily in lysosomal storage disorders.13–15 Recently, consistent with the autophagy–lysosomal pathway abnormalities demonstrated in PD, an autopsy study found increased GPNMB levels in the substantia nigra of PD patients, and the experimental modeling of lysosomal stress caused elevated GPNMB levels in several brain regions in mice. 5 Furthermore, GPNMB facilitates the neuronal uptake of alpha-synuclein, a protein that is associated with PD pathology, and elevated plasma GPNMB levels are correlated with more severe PD symptoms. 6 However, alterations in GPNMB in cerebrospinal fluid (CSF) and their clinical significance in PD have not yet been thoroughly investigated.
In neurodegenerative diseases, including PD, microglial activation plays a crucial role in the inflammatory response, which contributes to disease pathogenesis. 16 This microglial activation is characterized by the upregulation of a subset of genes, including GPNMB. 17 Similarly, the gene encoding the triggering receptor expressed on myeloid cells 2 (TREM2), an innate immune receptor found on microglia, 18 is also highly expressed during microglial activation. Soluble TREM2 (sTREM2) reflects this activation, and its levels in bodily fluids have been shown to correlate with disease severity in neurodegenerative conditions such as Alzheimer's disease (AD)19,20 and PD. 21 This suggests its potential as a marker of disease progression. Therefore, measuring sTREM2 levels alongside GPNMB may provide additional insights into the microglial contribution to PD pathology and further elucidate the relationship between neuroinflammation and disease progression, potentially offering a more comprehensive understanding of the mechanisms at play.
The objective of this study was to elucidate the link between GPNMB in the central nervous system and clinical presentations in PD. To do this, we compared CSF GPNMB levels between patients with PD and controls, and examined the relationships between CSF GPNMB levels and comprehensive clinical characteristics (such as age, sex, disease duration, and motor and non-motor manifestations). In addition, we assessed its associations with CSF or plasma biomarkers of neurodegenerative pathogenesis, including CSF sTREM2.21–24
Materials and methods
Participants
We enrolled 118 consecutive PD patients who were admitted to Fujita Health University Hospital during the period from May 2020 to June 2023. All PD cases met the Movement Disorder Society (MDS) clinical diagnostic criteria for PD. 25 Patients with PD were further subdivided into PD with normal cognition (PD-NC), PD with mild cognitive impairment (PD-MCI), and PD with dementia (PDD) groups. 26 PD-MCI and PDD were diagnosed according to the MDS Task Force Level I.27,28 Patients who did not meet the criteria for PD-MCI or PDD were defined as PD-NC patients. Additionally, we enrolled 40 age- and sex-matched participants. We are conducting an ongoing aging registry study that collects CSF from patients without neurodegenerative disease and undergoing epidural anesthesia for surgical treatment of urinary tract stones or benign prostatic hyperplasia in the urology department at Fujita Health University, Japan.
This research received approval from the ethics committee at Fujita Health University Hospital (approval numbers: HM23-205 and HG21-015), and all participants granted written informed consent for their participation, with the option to withdraw from the study.
Clinical evaluation
Motor and non-motor symptoms associated with PD were evaluated using the and the Japanese version of the Movement Disorder Society's Unified PD Rating Scale (MDS-UPDRS). Motor severity was calculated using Hoehn and Yahr and MDS-UPDRS III scores. Cognitive assessments were conducted using the Frontal Assessment Battery, Addenbrooke's Cognitive Examination-Revised (ACE-R), Mini-Mental State Examination (MMSE), and Japanese version of the Montreal Cognitive Assessment. We also assessed subjects using the Parkinson's Disease Questionnaire-39 Summary Index, Geriatric Depression Scale-15, Odor Stick Identification Test for Japanese, Japanese version of the REM Sleep Behavior Disorder Screening Questionnaire, Scales for Outcomes in Parkinson's Disease-Autonomic, Epworth Sleepiness Scale, and Japanese version of the Questionnaire for Impulsive-Compulsive Disorders in Parkinson's Disease. The levodopa equivalent daily dose (LEDD) was calculated using the latest conversion formula. 29 All neurological evaluations of PD patients were conducted during the ‘on’ condition. The interval period between clinical evaluation and CSF collection in the PD group was 7.6 ± 16.1 days (mean ± SD, range 0-137). In the control group, clinical evaluation and CSF collection were performed on the same day.
CSF sample preparation
CSF was obtained via standard lumbar puncture during fasting. Within 2 h of collection, CSF specimens were centrifuged at 1500 × g for 10 min at 4°C. They were then aliquoted into 500 µL portions in polypropylene tubes and stored at −80°C until their use in assays.
Measurements of CSF GPNMB and sTREM2 levels
The levels of CSF GPNMB and sTREM2 were measured using commercial enzyme-linked immunosorbent assay kits (DY2550; R&D Systems, Minneapolis, MN, USA, and ab224881; Abcam, Cambridge, UK). For both GPNMB and sTREM2 measurements, CSF samples were diluted by factors of 1 in 8 to obtain optical density measurements within the standard range. Absorbance at 450 nm was determined with the Bio-Rad Benchmark Microplate Reader and Microplate Manager version 5.2.1 (Bio-Rad Laboratories, Hercules, CA, USA). Samples and standards underwent duplicate measurements, and the means of these duplicates were used for statistical analysis. The average values of coefficient of variation across all samples analyzed for GPNMB and sTREM2 were 11.6% and 5.3%, respectively. Assays for CSF GPNMB were performed using the same reagents on the same day. Besides, replicate samples for sTREM2 on different days exhibited high reproducibility (Pearson r = 0.92).
Plasma sample collection and assays of ad-related biomarkers
Of the 118 patients with PD, we conducted measurements of AD-related biomarkers in the plasma of 65 consecutive PD patients admitted to Fujita Health University Hospital during the period from May 2020 to September 2021. To obtain plasma, blood samples were collected by venipuncture using EDTA blood collection tubes, following a fasting period of over 6 h. After collection, these samples were subjected to centrifugation at 1500 × g for 10 min, and 500 µL aliquots of plasma were transferred into polypropylene tubes, and promptly frozen and stored at −80°C until further analysis. Plasma levels of glial fibrillary acidic protein (GFAP), neurofilament light chain (NfL), amyloid-beta 40 (Aβ40), Aβ42, and tau phosphorylated at threonine 181 (pTau-181) were determined by single-molecule array using the Simoa Human Neurology 4-Plex E kit and Simoa pTau-181 V2 Advantage kit (Quanterix, Billerica, MA, USA). Each plasma sample was tested in duplicate.
We also compared the clinical characteristics between the 65 patients whose AD-related biomarkers were measured and the other 53 patients. Only the MDS-UPDRS IV score was notably higher in the measured group; the other clinical characteristics were not significantly different (Supplemental Table 1).
Statistical analysis
Statistical analyses were carried out using JMP software version 16 (SAS Institute, Cary, NC, USA). Statistical significance was defined as p < 0.05. To compare the distribution of sexes, Fisher's exact test was employed. The normality of variables and homoscedasticity were assessed with the Shapiro–Wilk and Levene's tests, respectively. Depending on the adherence to assumptions of normality or homogeneity of variance, comparisons of continuous variables between two groups were performed using Student's t-test or the Wilcoxon rank-sum test. As for the evaluation of CSF GPNMB, we added a generalized linear model for CSF GPNMB levels based on the presence of PD, age at examination, and sex. Spearman's rank correlation test was used to assess the correlations between continuous variables. To correct for multiple comparisons, we used a False Discovery Rate (FDR) correction using the Benjamini-Hochberg Procedure, 30 and the cutoff value for FDR was set at 0.05. In addition, we used a generalized linear model to evaluate the effects of the presence of PD, age at examination, and sex on the CSF GPNMB levels. Principal component analysis (PCA) was conducted on CSF GPNMB, CSF sTREM2, and AD-related plasma biomarkers. Principal component (PC) loading plots were used to identify the primary sources of variability among these biomarkers in PD patients. PC score plots were used to visualize individual people clustering based on biomarker profiles. To clarify the main factors related to CSF GPNMB levels, we conducted a multiple regression analysis with several explanatory variables based on simple regression analyses using Spearman's rank correlation test. Continuous variables are presented as the mean ± standard deviation.
Results
Participant characteristics
Table 1 presents the clinical features of participants in the control and PD groups. No notable differences were observed between the two groups in age at examination (p = 0.0845) or sex (p = 0.1661). In the PD group, 77 patients (65.3%) were identified as having PD-MCI, 26 (22.0%) as having PDD, and the remaining 15 (12.7%) as having PD-NC. The proportions of these three subgroups based on cognitive function were consistent with previous reports when disease duration was taken into account.31,32 No significant differences were found between the PD and control groups in CSF protein (PD 50.23 ± 23.43 mg/dL, control 57.83 ± 37.38 mg/dL; p = 0.1797) or albumin (PD 30.48 ± 16.38 mg/dL, control 33.74 ± 19.92 mg/dL; p = 0.1906) levels.
Clinical characteristics of participants in the PD and control groups.
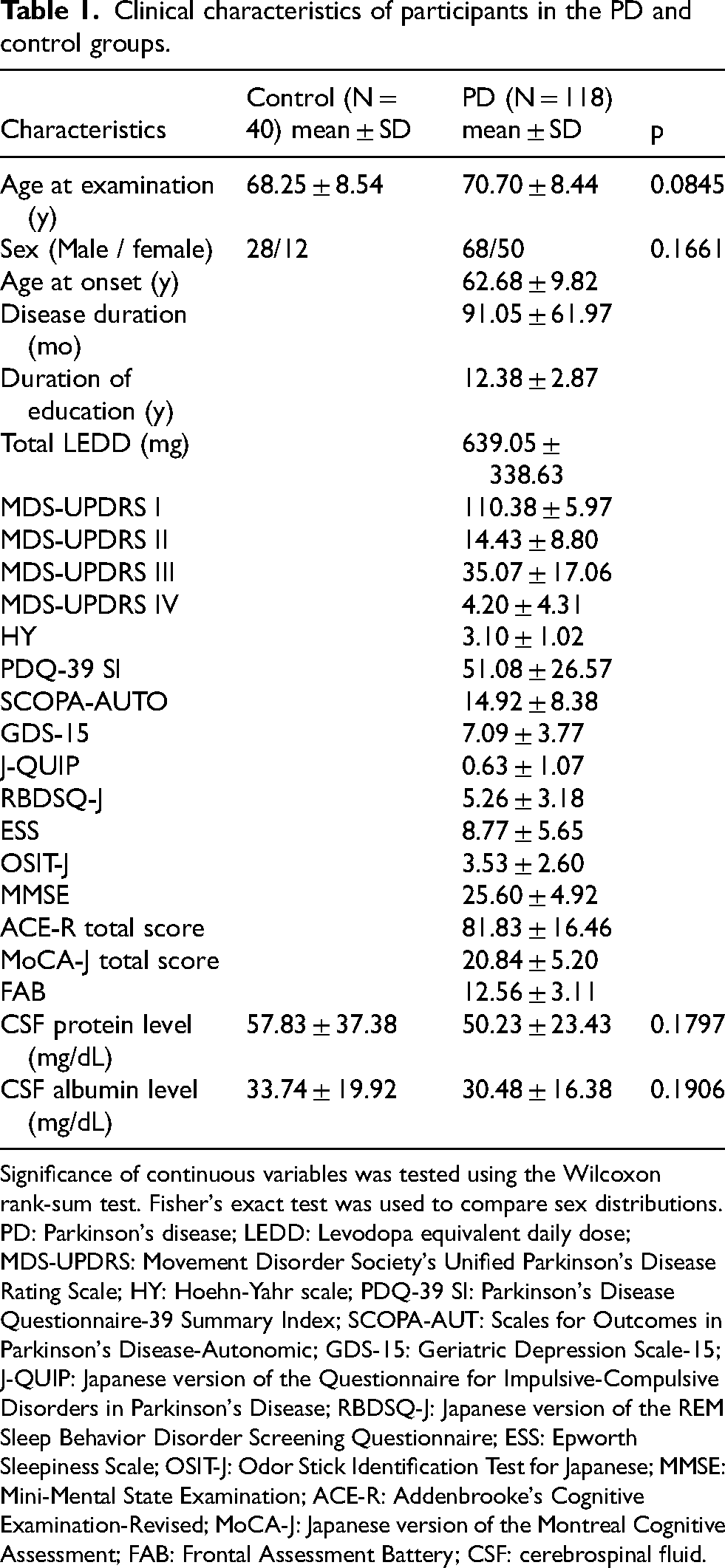
Significance of continuous variables was tested using the Wilcoxon rank-sum test. Fisher's exact test was used to compare sex distributions. PD: Parkinson's disease; LEDD: Levodopa equivalent daily dose; MDS-UPDRS: Movement Disorder Society's Unified Parkinson's Disease Rating Scale; HY: Hoehn-Yahr scale; PDQ-39 SI: Parkinson's Disease Questionnaire-39 Summary Index; SCOPA-AUT: Scales for Outcomes in Parkinson's Disease-Autonomic; GDS-15: Geriatric Depression Scale-15; J-QUIP: Japanese version of the Questionnaire for Impulsive-Compulsive Disorders in Parkinson's Disease; RBDSQ-J: Japanese version of the REM Sleep Behavior Disorder Screening Questionnaire; ESS: Epworth Sleepiness Scale; OSIT-J: Odor Stick Identification Test for Japanese; MMSE: Mini-Mental State Examination; ACE-R: Addenbrooke's Cognitive Examination-Revised; MoCA-J: Japanese version of the Montreal Cognitive Assessment; FAB: Frontal Assessment Battery; CSF: cerebrospinal fluid.
Comparison of CSF GPNMB concentrations between the PD and control groups
Student's t-test confirmed significantly elevated log-transformed CSF GPNMB levels in the PD group compared with the control group (p = 0.0159) (Figure 1). Additionally, a generalized linear model analysis, including age at examination and sex as covariates, also revealed a significant effect of the presence of PD on CSF GPNMB (p < 0.0001) irrespective of the covariates (Supplemental Table 2).
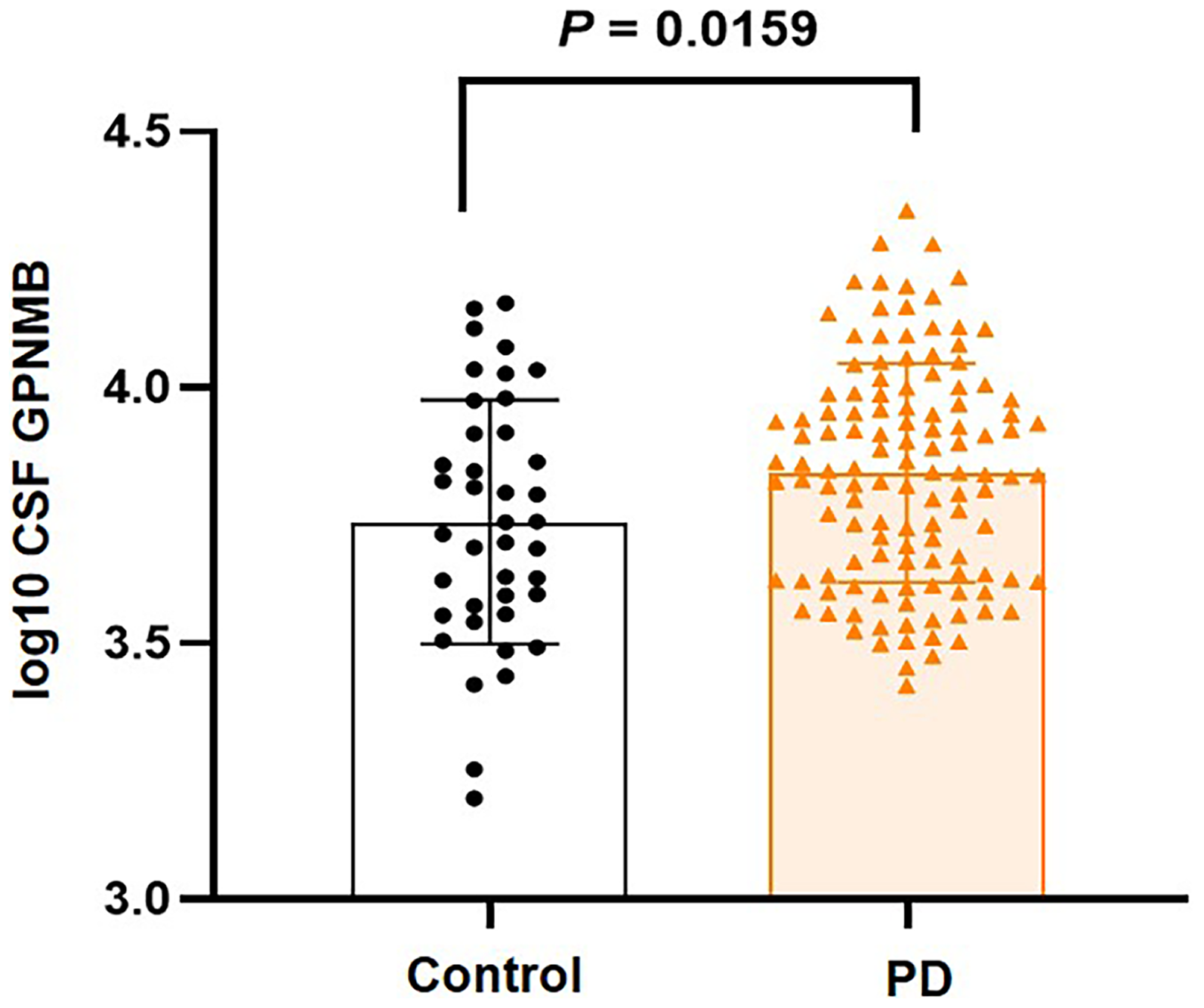
Comparison of CSF GPNMB levels. Comparison of log10 CSF GPNMB between the control and PD groups. Significance was tested using Student's t-test. CSF: cerebrospinal fluid; GPNMB: glycoprotein non-metastatic melanoma protein B; PD: Parkinson's disease.
Associations between CSF GPNMB and clinical metrics and CSF biomarkers in each group
Clinical metrics. In the PD group, CSF GPNMB levels exhibited a positive correlation with age at examination (rs = 0.2511, p = 0.0061), age at onset (rs = 0.2800, p = 0.0021), and MDS-UPDRS III score (rs = 0.1998, p = 0.0347) (Table 2). Moreover, in this group, CSF GPNMB levels demonstrated a negative correlation with the duration of education (rs = −0.2455, p = 0.0074), and MMSE score (rs = −0.1922, p = 0.0370). Furthermore, to investigate the effect of CSF GPNMB on motor and cognitive function in the PD group, we conducted multiple regression analysis on MDS-UPDRS III score and MMSE score, with CSF GPNMB included as a potential confounder. As shown in Supplemental Table 3, there was a trend towards a correlation between CSF GPNMB and the MDS-UPDRS III score, regardless of whether LEDD was included as a covariate, although this trend was not statistically significant (p = 0.0585, without LEDD; p = 0.0636, with LEDD). Meanwhile, no clear trend was found with the MMSE score.
Correlations between CSF GPNMB levels and clinical parameters or CSF biomarkers in the PD and control groups.
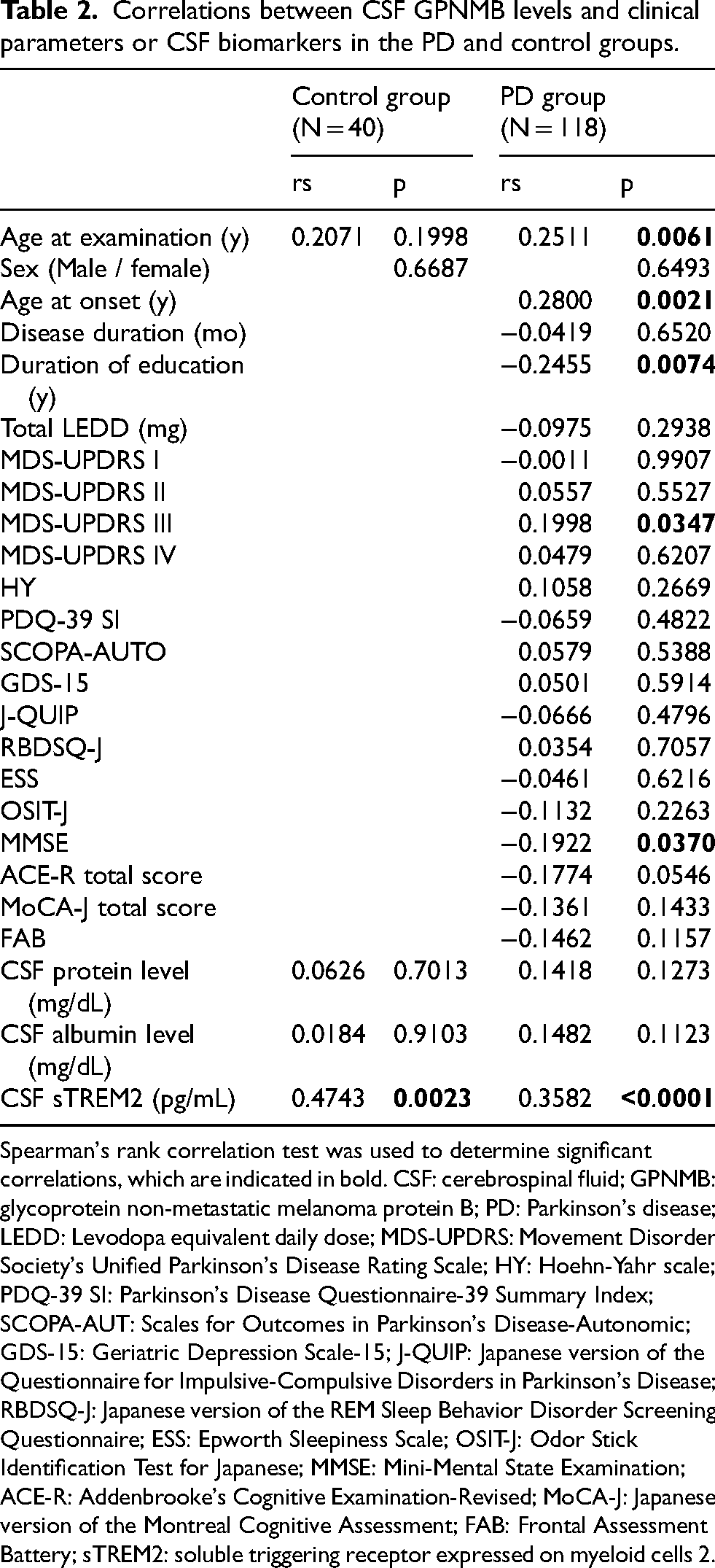
Spearman's rank correlation test was used to determine significant correlations, which are indicated in bold. CSF: cerebrospinal fluid; GPNMB: glycoprotein non-metastatic melanoma protein B; PD: Parkinson's disease; LEDD: Levodopa equivalent daily dose; MDS-UPDRS: Movement Disorder Society's Unified Parkinson's Disease Rating Scale; HY: Hoehn-Yahr scale; PDQ-39 SI: Parkinson's Disease Questionnaire-39 Summary Index; SCOPA-AUT: Scales for Outcomes in Parkinson's Disease-Autonomic; GDS-15: Geriatric Depression Scale-15; J-QUIP: Japanese version of the Questionnaire for Impulsive-Compulsive Disorders in Parkinson's Disease; RBDSQ-J: Japanese version of the REM Sleep Behavior Disorder Screening Questionnaire; ESS: Epworth Sleepiness Scale; OSIT-J: Odor Stick Identification Test for Japanese; MMSE: Mini-Mental State Examination; ACE-R: Addenbrooke's Cognitive Examination-Revised; MoCA-J: Japanese version of the Montreal Cognitive Assessment; FAB: Frontal Assessment Battery; sTREM2: soluble triggering receptor expressed on myeloid cells 2.
After correcting for multiple comparisons with FDR, the statistical significance between CSF GPNMB levels and age at onset remained significant (q-value = 0.0469) (Supplemental Table 4). In the control group, there were no significant associations between CSF GPNMB levels and age at examination (rs = 0.2071, p = 0.1998) or sex (p = 0.6687).
CSF biomarkers. Table 2 also shows the associations between CSF GPNMB and CSF biomarkers in the PD and control groups. There was a significant positive correlation between CSF GPNMB and CSF sTREM2 levels in both the PD (rs = 0.3582, p < 0.0001) and control (rs = 0.4743, p = 0.0023) groups (Figure 2A, B).
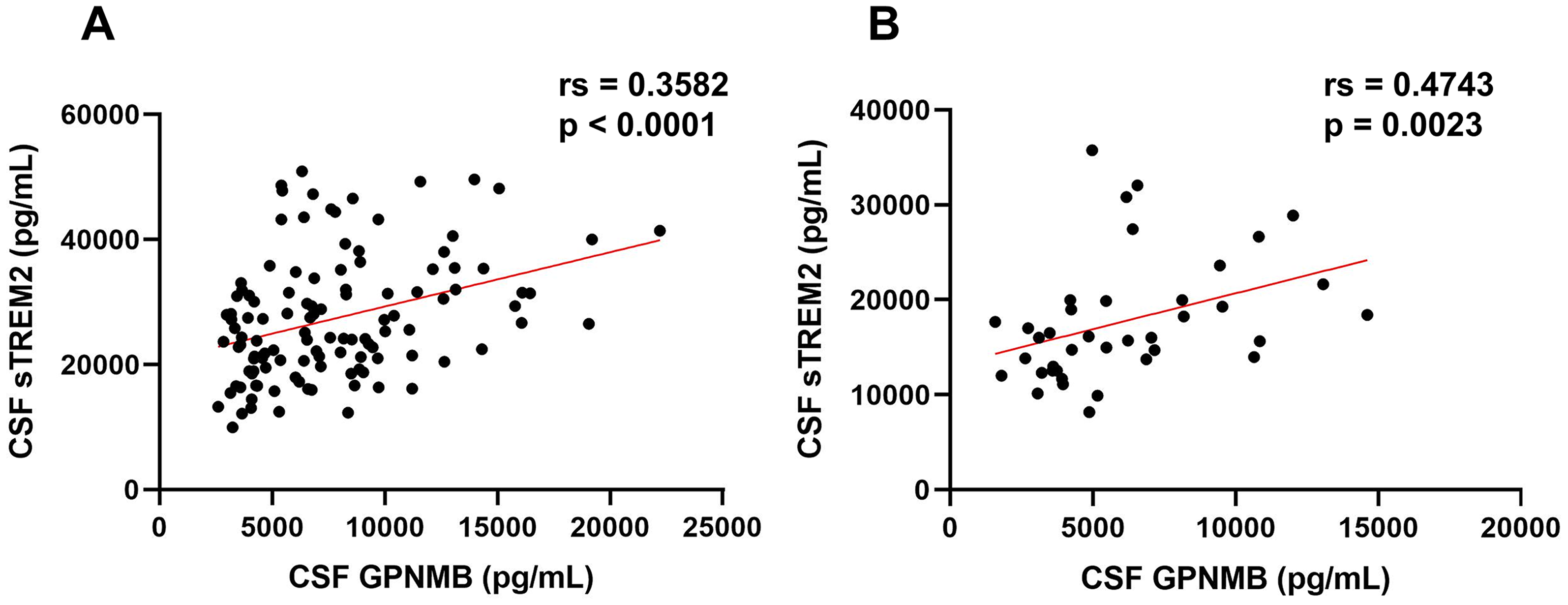
Correlation between CSF GPNMB and sTREM2 levels in each group. (A) Correlation between CSF GPNMB and sTREM2 levels in the PD group. (B) Correlation between CSF GPNMB and sTREM2 levels in the control group. Spearman's rank correlation test was used to determine significant correlations. CSF: cerebrospinal fluid; GPNMB: glycoprotein non-metastatic melanoma protein B; sTREM2: soluble triggering receptor expressed on myeloid cells 2; PD: Parkinson's disease.
Regression analysis for potential confounders of CSF GPNMB levels in the PD group
Considering the potential confounding variables from the aforementioned Spearman's rank correlation, we conducted multiple regression analysis to identify the determinants of CSF GPNMB levels in the PD group. As depicted in Table 3, the CSF sTREM2 level was the predominant influencer (t-value = 3.49, p = 0.0007), followed by the duration of education (t-value = −2.04, p = 0.0444).
Multiple regression analysis for CSF GPNMB and potential confounding factors in the PD group.
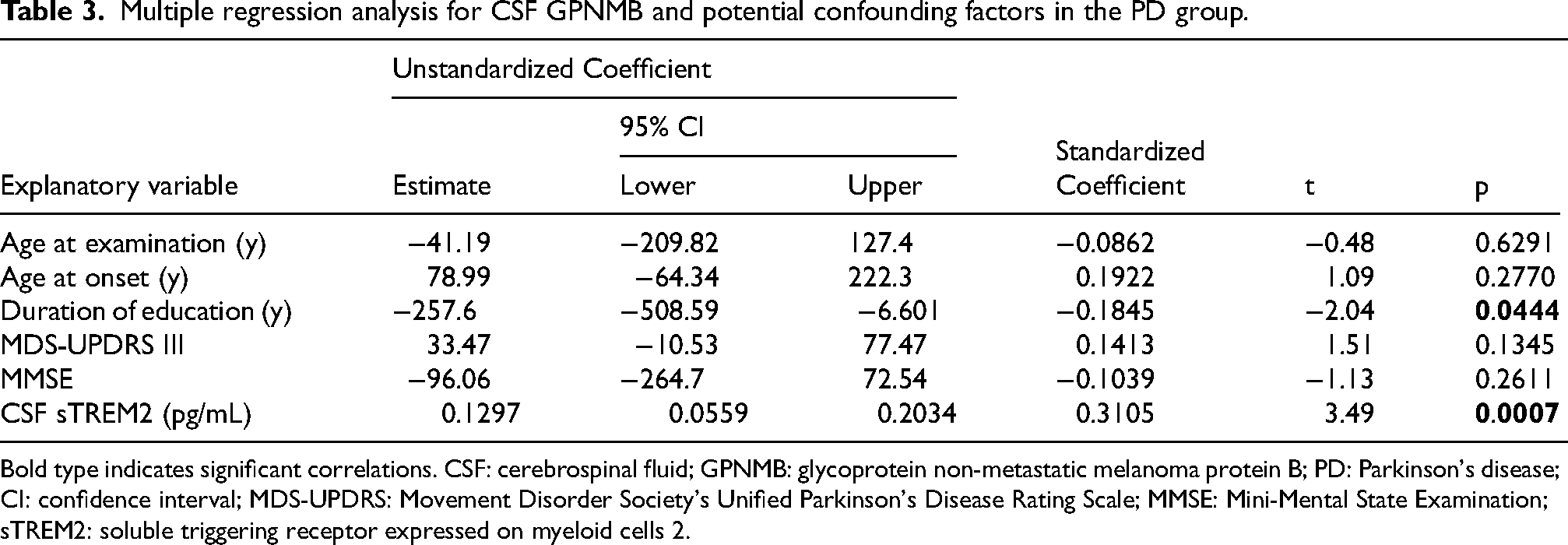
Bold type indicates significant correlations. CSF: cerebrospinal fluid; GPNMB: glycoprotein non-metastatic melanoma protein B; PD: Parkinson's disease; CI: confidence interval; MDS-UPDRS: Movement Disorder Society's Unified Parkinson's Disease Rating Scale; MMSE: Mini-Mental State Examination; sTREM2: soluble triggering receptor expressed on myeloid cells 2.
Associations between CSF GPNMB levels and AD-related plasma biomarkers in the PD group
Regarding the observed negative correlation between CSF GPNMB and MMSE, we investigated whether elevated CSF GPNMB level in PD reflects AD pathology, considering the previous reports suggesting the presence of AD pathology in cognitive decline in PD.33,34 We measured six AD-related plasma biomarkers (Aβ40, Aβ42, Aβ42/Aβ40, GFAP, NfL, and pTau-181), and examined their relationships with CSF GPNMB levels. Our results showed that those plasma biomarkers were not significantly associated with CSF GPNMB levels, except for plasma NfL (rs = 0.2681, p = 0.0308) (Supplemental Table 5). Figure 3 elucidates the interconnections between CSF and plasma biomarkers in the PD group. Spearman's rank correlation coefficients are systematically summarized in a correlation matrix, accompanied by their corresponding p-values (Figure 3A). A notable array of correlations was observed among the six AD-related plasma biomarkers. However, CSF GPNMB and sTREM2 demonstrated independence from those plasma biomarkers, with the exception of plasma NfL.
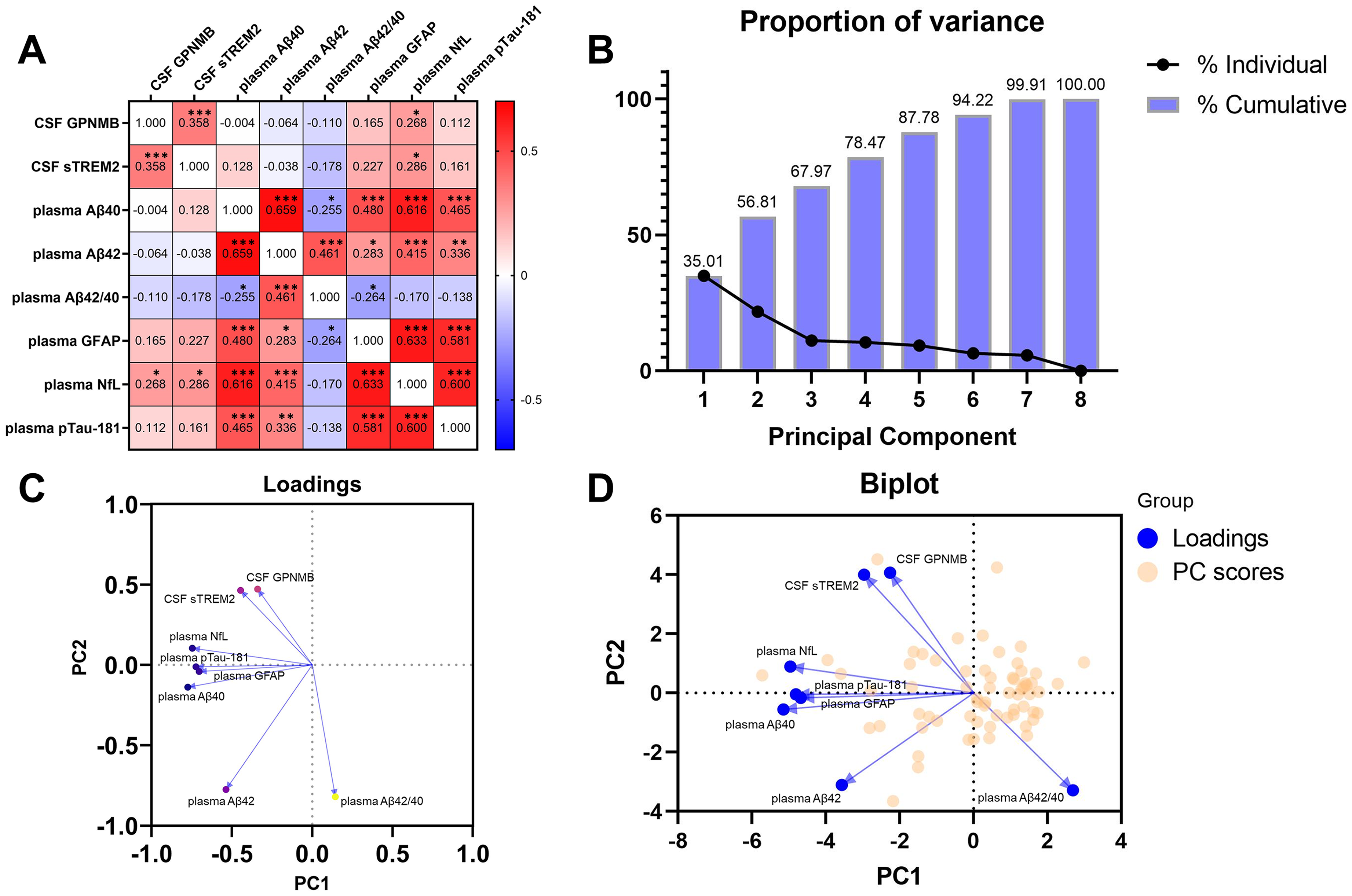
Interconnections between CSF and plasma biomarkers in the PD group. (A) Spearman's rank correlation between plasma biomarkers in the PD group. Spearman's rank correlation test was used to determine significant correlations. *p < 0.05; **p < 0.01; ***p < 0.001. (B-D) Results of principal component analysis on CSF GPNMB, CSF sTREM2, and AD-related plasma biomarkers. (B) Proportion of variance for each principal component. Individual proportion was depicted with dotted lines, and cumulative proportion was depicted with bars. (C) PC loadings plot, showing an involvement of each biomarker in PC1 variance and PC2 variance. (D) Superimposed biplot with the PC loadings plot and the PC scores plot, which shows the distribution of individual patients in relation to PC1 and PC2. CSF: cerebrospinal fluid; PD: Parkinson's disease; GPNMB: Glycoprotein non-metastatic melanoma protein B; sTREM2: soluble triggering receptor expressed on myeloid cells 2; PC: principal component; AD: Alzheimer's disease; Aβ: amyloid-beta; GFAP: glial fibrillary acidic protein; NfL: neurofilament light chain; p-Tau181: tau phosphorylated at threonine 181.
PCA revealed two principal components. The PC loading plot demonstrated that the primary contributors to PC1 variance were plasma NfL, p-tau 181, GFAP, and Aβ40. Conversely, CSF GPNMB and sTREM2 emerged as the primary determinants of PC2 variance, suggesting the independence of CSF GPNMB and sTREM2 from AD-related biomarkers in our PD patient group (Figure 3B-D).
Discussion
In the present study, CSF GPNMB levels were markedly elevated in the PD group compared with those in the control group. Further analysis in the PD group revealed that this elevation was significantly positively correlated with patient age and severity of motor and cognitive function. The age at onset remained significant after correction for multiple comparisons with FDR. A significant association was also observed between CSF GPNMB and sTREM2 levels in the PD group. This association was independent of AD-related plasma biomarkers except for NfL. Taken together, our study reveals that elevated CSF GPNMB levels in PD patients are associated with age at onset and show a unique correlation with sTREM2, while remaining independent of AD-related biomarkers, underscoring their potential relevance in the distinct pathology of PD.
Human GPNMB is expressed in the microglia of the central nervous system. The role of GPNMB in inflammation and its involvement in the pathology of neurodegenerative diseases have been underscored in several publications.17,35,36 Experiments involving GPNMB overexpression in transgenic mouse models have provided evidence of its neuroprotective and anti-inflammatory effects.35,37,38 Autopsy studies have shown that microglia in the midbrains of patients with PD have a pro-inflammatory trajectory, including elevated GPNMB levels.5,39 Consequently, the specific role of GPNMB in inflammation in PD remains unclear. Thus, further research is necessary to investigate the potential of GPNMB, especially in the CSF, as an indicator of inflammation in the central nervous system of PD patients. A recent study reported increased GPNMB levels in the plasma but not CSF of PD patients. 6 This discrepancy with our study's findings might be attributed to variations in sample size and patient backgrounds between the studies. To our knowledge, the present study is the most extensive examination conducted to date on patients with PD. Considering these factors, our extensive cohort not only amplifies the robustness of our findings but also provides a crucial contribution to the existing body of knowledge regarding the role of GPNMB in PD.
Diaz-Ortiz et al. observed a correlation between higher plasma GPNMB levels and more severe PD motor symptoms. 6 We also investigated those clinical associations of CSF GPNMB, and revealed that CSF GPNMB levels correlated with age at examination and onset, suggesting a possible relationship between GPNMB and aging. In particular, after adjustment for multiple comparisons by FDR, the positive correlation between GPNMB and the age of onset remained significant. Previous evidence has revealed a close relationship between onset at an older age and increased severity of the clinical phenotype of PD. 40 Moreover, the temporal progression of motor and non-motor milestones in PD is influenced by both disease duration and age at diagnosis.41–43 Given the well-documented relationships between aging, age at onset, and disease severity in PD, 44 our study aligns with previous findings that emphasize the role of GPNMB in aging and in the progression of patients with PD.4,45 Furthermore, the relationship between GPNMB and MDS-UPDRS III or MMSE scores observed in this study could be affected by age-related factors. Alternatively, higher GPNMB levels in older people with PD might be a potential risk factor for the decline in motor and cognitive functions, as reflected in the deterioration of MDS-UPDRS III or MMSE scores. This could be because GPNMB facilitates the faster spread of alpha-synuclein, a protein associated with PD pathology. 6 In line with this hypothesis, our multiple regression analyses on motor and cognitive functions revealed a trend indicating that CSF GPNMB levels might influence MDS-UPDRS III scores, although this trend was not statistically significant. Future research, particularly studies that evaluate alpha-synuclein, is needed to clarify the precise mechanisms underlying these associations.
Another finding from our study was the negative correlation between CSF GPNMB levels and MMSE score, indicating the potential influence of GPNMB on cognitive function in PD. Considering the complex pathology of cognitive decline in PD, we also investigated the relationship between CSF GPNMB levels and other biological markers that may reflect cognitive function in PD. 24 Notably, representative AD-related plasma biomarkers, such as Aβ 40, Aβ42, Aβ42/Aβ40, GFAP, and pTau-181, were not significantly associated with CSF GPNMB levels, except for NfL. As for the association with NfL, a marker of axonal damage which increases in various neurodegenerative diseases, 46 it might reflect the aspects of both CSF GPNMB and plasma NfL as disease progression biomarkers, involving both motor severity and cognitive decline in PD. 24
A noteworthy positive correlation was identified between CSF GPNMB and sTREM2 levels in PD patients. TREM2 serves as an innate immune receptor and is expressed by macrophages and microglia in the central nervous system.47,48 Additionally, TREM2 promotes microglial activation and is closely correlated with the pathogenesis of neurodegenerative diseases including AD. 49 Therefore, given its role in promoting microglial survival and inducing inflammatory responses, sTREM2 considered as a biomarker of microglial activation.47,50,51 Previous studies have shown that CSF sTREM2 levels are elevated in patients with PD and highlighting its potential as a predictor of cognitive decline in this disease.21,52,53 We found that CSF GPNMB and sTREM2 levels were independent of AD-related plasma biomarkers. This suggests that these microglial changes, as reflected by CSF GPNMB and sTREM2 levels, exhibit different dynamics from AD-related pathology associated with cognitive decline in PD. 24 At present, the mechanisms by which GPNMB and TREM2 function cooperatively in the context of PD are unclear. In a previous study on intracellular protein activity in a sarcoma model analyzed by single-cell RNA sequencing, the authors reported that a specific group of TREM2-expressing macrophages also expressed Gpnmb. 54 Other studies have shown that in PD, microglia are more prone to causing inflammation and have increased GPNMB levels. 39 These findings suggested that TREM2 and GPNMB may act together through microglial inflammatory signaling. However, further research is needed to explore the precise mechanisms and roles of GPNMB and TREM2 in PD pathogenesis.
Our study had several limitations. First, the control group lacked information regarding clinical features (except for age and sex) and AD-related plasma biomarkers. Second, we did not conduct a combined analysis of CSF and plasma GPNMB levels to clarify their association with central PD pathology. This decision was influenced by the fact that human GPNMB is expressed in diverse cell types across multiple organs, such as bone and skin. Given the distinct implications of GPNMB in plasma and CSF, our analysis focused solely on CSF GPNMB in this instance. Third, in this study, we did not investigate CSF GPNMB levels in atypical parkinsonian syndromes, such as multiple system atrophy (MSA) and progressive supranuclear palsy (PSP). Recent studies using plasma samples have included MSA and PSP along with PD, revealing that GPNMB levels are significantly lower in MSA compared to PD.55,56 Future investigations incorporating these conditions are necessary to evaluate the potential specificity of CSF GPNMB as a biomarker for PD. Fourth, we lack longitudinal clinical data and predictive models regarding CSF GPNMB. Fifth, this study did not include genetic testing for GPNMB, which could have contributed to a more comprehensive understanding of its involvement in the disease process. Lastly, the Simoa Advantage plasma Aβ42/Aβ40 assays are not sufficiently reliable to detect Aβ pathology, with low AUCs of ∼0.69 in detecting CSF and PET amyloid positivity. 57
Conclusions
In the present study, CSF GPNMB was significantly increased in PD, and its levels associated with patient age. Additionally, among known biomarkers related to neurodegenerative pathogenesis, CSF GPNMB levels were closely associated with CSF sTREM2 levels. Our study indicates that elevated CSF GPNMB levels in PD may reflect patient age and its related changes and might be associated with microglial activation.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241288712 - Supplemental material for CSF GPNMB in Parkinson's disease: A potential association with age and microglial activation
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241288712 for CSF GPNMB in Parkinson's disease: A potential association with age and microglial activation by Xi-Chen Zhu, Yasuaki Mizutani, Reiko Ohdake, Harutsugu Tatebe, Toshiki Maeda, Sayuri Shima, Akihiro Ueda, Mizuki Ito, Shinji Ito, Takahiko Tokuda and Hirohisa Watanabe in Journal of Parkinson's Disease
Supplemental Material
sj-xlsx-2-pkn-10.1177_1877718X241288712 - Supplemental material for CSF GPNMB in Parkinson's disease: A potential association with age and microglial activation
Supplemental material, sj-xlsx-2-pkn-10.1177_1877718X241288712 for CSF GPNMB in Parkinson's disease: A potential association with age and microglial activation by Xi-Chen Zhu, Yasuaki Mizutani, Reiko Ohdake, Harutsugu Tatebe, Toshiki Maeda, Sayuri Shima, Akihiro Ueda, Mizuki Ito, Shinji Ito, Takahiko Tokuda and Hirohisa Watanabe in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We are grateful to Dr Ryoichi Shiroki and his colleagues in our institute for their help with the collection of CSF samples from healthy controls.
Funding
The present work was supported by JSPS KAKENHI grant numbers JP21K07428 and JP22K07508 and AMED grant number 22dk0207055h0002.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
