Abstract
Objective:
The aim of this study was to investigate the effects of manual diaphragmatic relaxation combined with diaphragmatic breathing on respiratory muscle strength, pulmonary function, chest expansion, and sitting ability in children with diplegic cerebral palsy (CP).
Materials and Methods:
This randomized controlled study was conducted with 15 children with diplegic CP aged 5–15 years. Participants were randomly divided into two groups (control and intervention); both groups received conventional physiotherapy two days a week (45 minutes) for eight weeks. The intervention group additionally received manual diaphragm relaxation and diaphragmatic breathing exercises. The primary outcome was respiratory muscle strength (maximum inspiratory pressure and maximum respiratory pressure. Secondary outcomes were pulmonary function test (PFT), chest expansion and sitting dimension of the Gross Motor Functional Measurement (GMFM-B). (NCT05559346)
Results:
After eight weeks of intervention, the intervention group showed improvement in respiratory muscle strength, chest mobility, and GMFM-B score (p < 0.05). Respiratory muscle strength was significantly higher in the intervention group than in the control group (p < 0.05), but PFT parameters and GMFM-B scores were not significantly different between the two groups (p > 0.05)
Conclusion:
Adding manual diaphragmatic relaxation and diaphragmatic breathing exercises to the physiotherapy program in children with CP may contribute to the rehabilitation program.
Keywords
Introduction
Cerebral palsy (CP) is a developmental disorder that affects movement and posture due to a permanent but non-progressive lesion in the brain. It is often accompanied by other impairments in cognition, perception, communication and musculoskeletal system and co-morbidities such as epilepsy. Although CP is non-progressive, symptoms can change over time, making it important to recognize and manage accompanying disorders such as neurological, cognitive, sleep, gastrointestinal, nutritional, urinary, and respiratory problems.1,2
Respiratory system problems are frequently observed in children with CP due to the nature of neurological and anatomical dysfunction present at different levels. Recurrent pneumonia, atelectasis, bronchiectasis, and restrictive lung disease are among the most common respiratory disorders in CP. Adults with CP have a 14 times higher risk of death from respiratory diseases compared to the general population; pneumonia has been reported as the leading cause of death and hospitalization in children and young adults with CP.3,4
The diaphragm is the primary respiratory muscle, responsible for approximately 80% of all respiratory work during normal tidal breathing. 5 It is a core muscle that works in synergy with the abdominal muscles, which control both respiration and posture during rest, movement and exercise. 6 The diaphragm is directly related to postural stability and exerts its influence through modulation of intra-abdominal pressure. 5 In the initial stages of motor development, the diaphragm primarily functions as a respiratory muscle. However, at approximately six months, coinciding with the assumption of an upright position, it begins to serve a dual role as both a respiratory and postural muscle. 7 It has previously been demonstrated in research studies that an increase in respiratory demand has the effect of reducing the role of the diaphragm in maintaining trunk stability.8,9 The term “postural stability” is defined as the result of postural control in the context of activities of daily living. 10 Children with CP, which is characterized by a deficit in postural control, experience difficulty in maintaining postural stability in an upright position. 11 The attainment of sitting, the first upright posture, occurs at an early stage of development and plays a pivotal role in facilitating a range of functional, perceptual, cognitive and social opportunities for the child. 12
The vulnerability of individuals with CP to respiratory diseases is well known, but the mechanisms and risk factors involved are still under investigation. Potential risk factors include dysphagia, motor dysfunction, impaired protective swallowing reflexes, and decreased muscle strength and coordination, particularly in respiratory muscles. The coordination of diaphragmatic, abdominal, chest, neck, and pharyngeal muscle movements, as well as trunk and head stability, are all essential for normal breathing. Motor impairments in individuals with CP are a risk factor for respiratory problems.13,14 Children with CP are more susceptible to respiratory diseases, and this risk escalates as their Gross Motor Functional Classification System (GMFCS) level rises. Treatment should aim to maximize musculoskeletal function and minimize progressive complications. Children with CP have worse lung function and respiratory muscle strength compared to typically developing peers, with function worsening as the child's GMFCS level increases. There is a positive relationship between respiratory muscle strength, self-care and social function competence levels in activities of daily living.13–16
In 2020, the first consensus statement on respiratory diseases in CP noted limited evidence for effective respiratory interventions and recommended further research, including into conventional physiotherapy approaches and the effectiveness of exercise. 17 Interventional studies on respiratory system disorders in CP have increased in recent years and are limited. A 2021 systematic review and meta-analysis found that various forms of physiotherapy have the potential to improve pulmonary function in children with CP, and different approaches should be investigated. 18 Another 2022 systematic review and meta-analysis study found significant improvements in inspiratory muscle strength and pulmonary function after respiratory training added to conventional rehabilitation, but there is no consensus on the frequency, type, or intensity of respiratory exercises for children with CP, highlighting the need for further research. 19 Diaphragmatic breathing exercises help the diaphragm in inspiration and expiration, decreasing work of breathing and increasing diaphragm excursion. 20 The stiffness of the rib cage and thoracic wall in CP appears to disrupt the normal resting position of the diaphragm, resulting in the shortening of muscle fibers and a consequent reduction in the efficiency of inspiration. 21 A reduction in diaphragmatic movement and a decrease in the expansion of the thoracic wall have been observed in children with CP in comparison to healthy controls.22,23 Manual diaphragm relaxation could enhance diaphragmatic contraction by activating the muscle spindle and Golgi tendon organ, while also aiding the partial recruitment of motor units in diaphragmatic muscle fibers affected by brain damage in CP. 24 Furthermore, in the context of muscle stiffness, which can be defined as the loss of muscle fascia elasticity, the muscle is restricted, resulting in weakened biomechanical properties. This can lead to a reduction in muscle strength, endurance and motor coordination. 25 For these reasons, manual diaphragmatic exercises may be helpful in children with CP.
However, data on specific interventions such as diaphragmatic relaxation is almost non-existent and has never been compared to conventional physiotherapy alone. It is a comfortable exercise that can be easily administered by a trained physiotherapist, and it can be helpful in improving respiratory functions in patients with pulmonary diseases. 19 The ease of application and its potential benefits make diaphragmatic relaxation a potentially useful technique in children with CP.
The aim of this study was to determine the effects of manual diaphragmatic relaxation and diaphragmatic breathing exercises added to conventional physiotherapy on respiratory muscle strength, thoracic cage mobility, respiratory functions and sitting ability in children with diplegic CP. It was hypothesized that the addition of diaphragmatic breathing and manual diaphragm relaxation exercises to conventional physiotherapy would primarily improve respiratory muscle strength. As a secondary outcome measure, improvements in pulmonary function, chest expansion, and sitting ability in children with diplegic CP were also expected.
Materials and methods
Trial design and ethical considerations
This study was conducted in the Pediatric Rehabilitation Unit of Marmara University Pendik Training and Research Hospital. The study was approved by Marmara University Faculty of Medicine Clinical Research Ethics Committee with decision number 09.2022.76 dated 07 January 2022. Parents of participating children were given detailed information about the study. It was stated that participation was voluntary, and an informed consent form was signed by the parents of all children participating. In addition to obtaining written informed consent from the parents, verbal assent was obtained from all participating children prior to enrolment. The study was prospectively registered in ClinicalTrials.gov (NCT05559346).
Subjects and settings
Participants were recruited consecutively from the outpatient pediatric rehabilitation clinic of a university-affiliated training and research hospital during the study period. Power analysis was performed with the G*Power package program (version 3.1.9.4) to determine the minimum number of participants to be included in the study. The maximum inspiratory pressure (MIP) parameter of a similar study was used in power analysis. With type I error α=0.05 and type II error β=0.15 (power=85%), the minimum sample size was calculated as 14, with seven participants in each group. Considering the rate of loss, 16 participants were included in the study. This study was planned as a prospective, randomized, controlled and single-blind study. Participants were randomly assigned to intervention or control groups using computer-generated random numbers, with allocation concealed in sealed, opaque envelopes prepared by an independent researcher. The study was completed with 15 participants, eight in the control group and seven in the intervention group. The flow chart is shown in Figure 1.
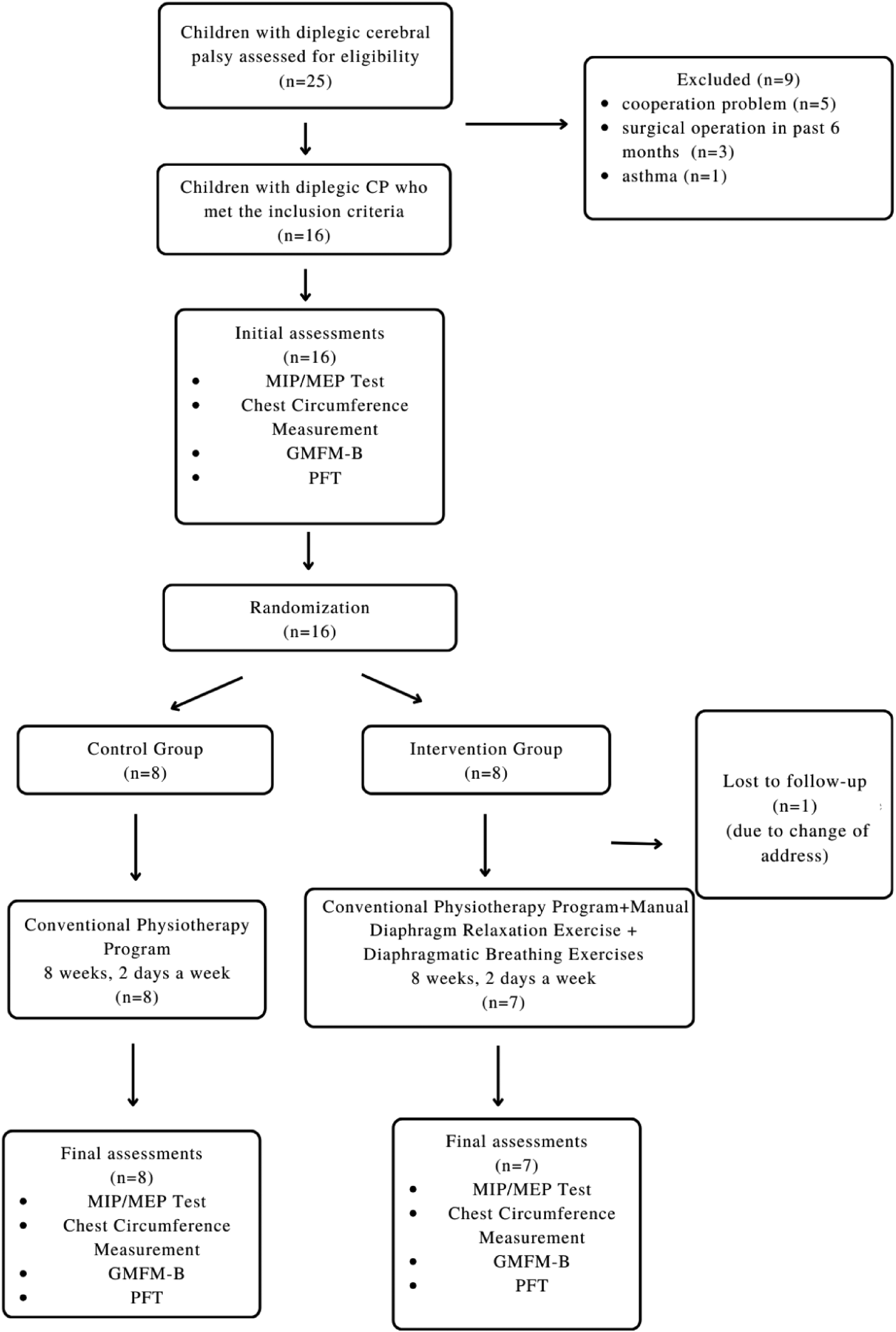
Flow chart.
Inclusion criteria were 1) diagnosis of diplegic CP by a pediatric neurologist (mild upper extremity involvement was included with a Manual Ability Classification System [MACS] level of I and II); 2) age 5-15 years; 3) GMFCS level I, II or III; 4) a suitable cognitive level to understand and apply the instructions in the tests; and 5) sufficient oral motor function, as observed during pre-assessment, to seal the mouthpiece and follow instructions for respiratory measurements. Exclusion criteria were 1) a surgical operation in the last six months; 2) scoliosis detected over 30 degrees; 3) diagnosed chronic respiratory disease; 4) a botulinum toxin injection for spasticity in the last six months; or 5) current baclofen use.
Assessment and outcome measures
All clinical measurements were performed with the child in a sitting position on a stable surface with hips and knees flexed at approximately 90°, feet supported, and hands free for movement. Similarly, respiratory pressure (MIP/maximal expiratory pressure [MEP]) tests and pulmonary function tests (PFTs) were conducted while sitting, while chest expansion was measured at three thoracic levels (axillary 4th intercostal space, epigastric xiphoid process, and subcostal 9th intercostal space). The Gross Motor Function Measure (GMFM-B) was administered on a mat in accordance with the standardized testing manual, using the appropriate starting positions for each item (i.e., supine, sitting, or standing), as specified in the original GMFM-88 procedure.
Demographic and clinical information form
Demographic and clinical information of each participant was recorded before the clinical evaluations. In the demographic and clinical information form, participant’s age, sex, height, weight, body mass index (BMI), history, level according to the GMFCS, level according to the MACS, and classification according to the Communication Function Classification System (CFCS) were recorded.
Respiratory muscle strength
MIP and MEP were recorded in the evaluation of respiratory muscle strength. The respiratory muscle strength measurements were taken using the Micro RPM portable manometer device. Assessments were conducted in accordance with ATS/ERS criteria. 26 In the MIP test, the child was asked to exhale and then perform an effortful inhalation against the device for as long as possible (1-3 seconds) with as much effort as possible. In the MEP test, the child was instructed to inhale to their full lung capacity and then exhale forcefully into the device for up to three seconds without puffing out their cheeks. The maneuver was explained in detail to each child, and they were shown how to do the test which was practiced through multiple trials. A child was considered to have learned the maneuver when they were able to form a proper lip seal, perform the test without air leakage, and follow the timing instructions correctly. Once the maneuver was successfully performed in at least two consecutive attempts, three formal measurement trials were conducted. The best value was recorded, provided that the results of the three trials did not differ by more than 20%. 27 Rest intervals were provided between the assessments. In the interpretation of the measurements, normal values based on sex and age were determined according to the reference equations. 28
Chest expansion
To assess chest expansion, measurements were taken at three sites: axillary (4th intercostal space), epigastric (xiphoid process) and subcostal (9th intercostal space), using a tape measure marked at 0.1 cm intervals while the child was in a sitting position. Circumference measurements of all three sites during maximal voluntary inspiration and expiration were performed by instructing the children to inhale and exhale as much as possible. For each measurement, the highest value at maximum voluntary inspiration and the lowest value at maximum voluntary expiration were recorded in three trials. The difference between the circumference measurement values at maximal voluntary inspiration and maximal voluntary expiration was recorded as chest expansion.29,30
PFT
Pulmonary function was tested using a spirometer device (Minispir™, USA) with forced vital capacity (FVC) maneuver in accordance with ATS/ERS guidelines. 31 The FVC maneuver was explained to the children in detail and demonstrated. FVC, forced expiratory volume in first second (FEV1), FEV1/FVC and peak expiratory flow (PEF) values and their percentages according to expected values were recorded.
Sitting function
In the evaluation of the sitting function of the children, the B section of the GMFM-88, the sitting section, was used. The GMFM is a standardized criterion-referenced measurement tool designed to measure gross motor function over time in children with disabilities aged five months to 16 years. The GMFM is used for clinical and research purposes to measure change in motor function over time and the effectiveness of interventions that affect change. The sitting dimension of the GMFM consists of 20 items and each item is scored between 0 and 3 (0 points = does not initiate; 1 = initiates; 2 = partially completes the item; 3 = completes the item independently). 32 The Turkish version of the GMFM, which has been shown to have excellent internal consistency and intra/inter-rater reliability in children with CP, was used in this study. 33
All clinical assessments were carried out by the same assessor at the outset and culmination of the intervention for both groups.
Intervention programs
Children in both groups received conventional physiotherapy sessions for 45 min two days a week for eight weeks. All physiotherapy sessions were conducted by the same physiotherapist. The content of the conventional physiotherapy program applied to both groups was planned according to individual needs after evaluations and generally consisted of passive stretching exercises for spastic muscles, strengthening exercises for antagonists of spastic muscles, exercises to improve weight transfer, balance and correction reactions, and postural control.
In addition to the conventional physiotherapy program, manual diaphragm relaxation and diaphragmatic breathing exercises were added to the program of the intervention group at the end of each session. The physiotherapist applied the manual diaphragmatic relaxation technique, which is proposed to enhance subsequent inspiratory contractility by potentially lengthening the diaphragm muscle fibers and/or modulating fascial and tissue properties. To perform manual diaphragm relaxation, the child lay comfortably supine while the physiotherapist positioned themselves at the head of the child. Using the pisiform and hypothenar regions of both hands, along with the last three fingers, the physiotherapist palpated the seventh to tenth ribs to facilitate rib elevation during inhalation. During exhalation, the physiotherapist gently pulled the contact points towards the child's head and slightly laterally, applying consistent resistance and gradually increasing pressure towards the inner margins of the ribs. In subsequent breathing cycles, the depth of contact within the costal margin was progressively enhanced. 34 The maneuver was repeated in two sets of 10 breaths at one minute intervals. (Figure 2). The duration of the pulmonary intervention was approximately 25 min.
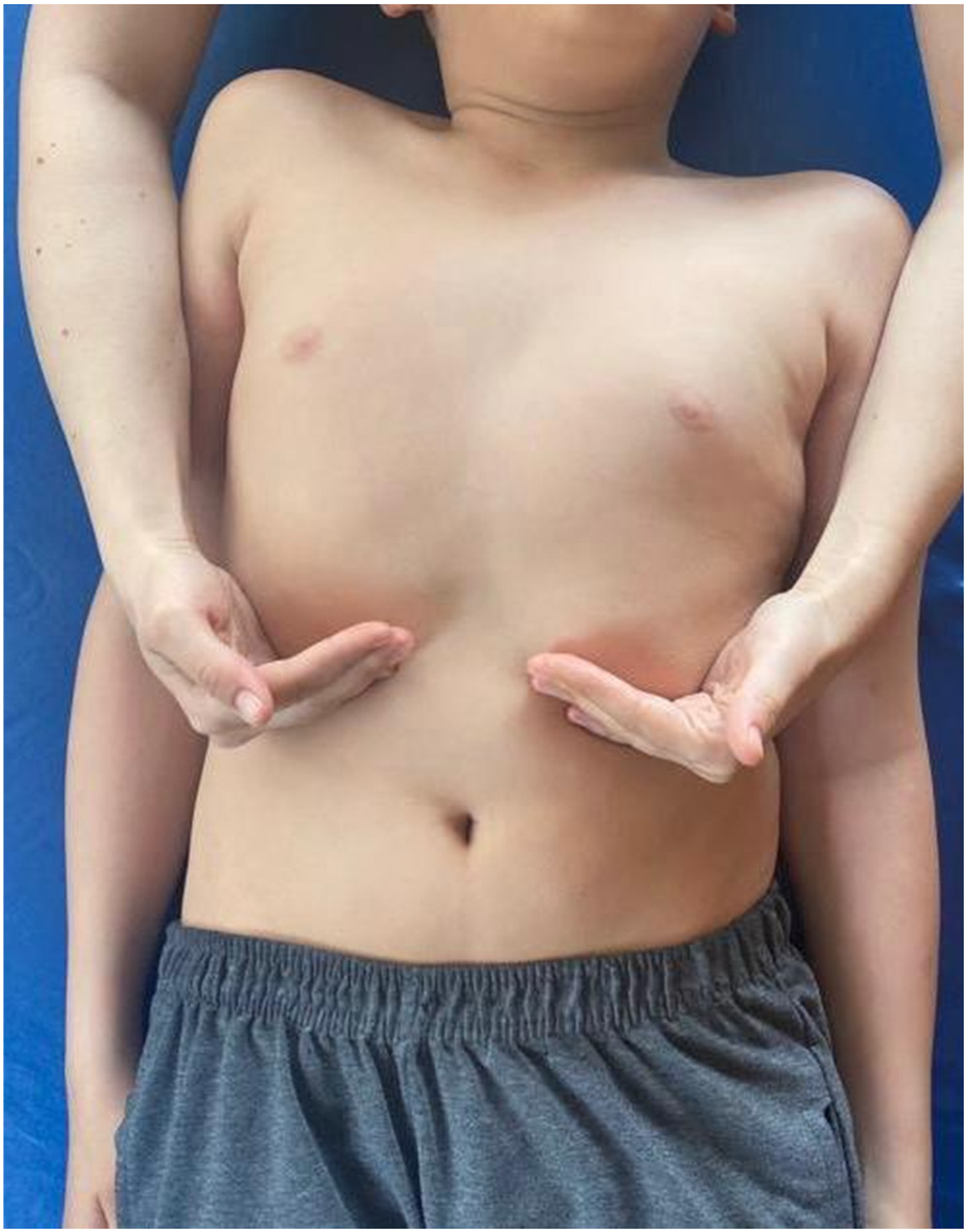
Manual diaphragmatic relaxation technique
The session continued with diaphragmatic breathing exercises with an emphasis on activating and strengthening a diaphragm motor plan and learning to produce greater amplitude of excursion as skill level improved over the eight weeks. The exercises were performed with the child in a supine position, lying comfortably on a mat with relaxed shoulders and upper chest. In the diaphragmatic breathing exercise, the child was asked to place their hand on the rectus abdominus muscle at the end of the ribs and take a deep and slow breath under their hand while their shoulders and chest remained relaxed. The patient was asked to inhale through the nose by inflating the abdomen, holding the breath, and exhaling with pursed lip breathing with the mouth half open. They were instructed to take a breath consisting of three seconds of inspiration, three seconds of breath holding and six seconds of expiration 35 and to resist movement of the rib cage and shoulders during this breathing exercise. This exercise was performed in three sets of 10 repetitions. Patients were corrected if they had difficulty in performing the exercise properly and only proper repetitions were counted.
Statistical analysis was conducted using IBM SPSS 20.0 package program. In descriptive statistics, categorical data were expressed as frequency (%), numerical data as mean ± standard deviation and median, minimum, maximum values. Chi-squared and Fisher’s exact chi-squared tests were used for qualitative data. In intragroup and intergroup comparisons, non-parametric tests were used because the sample size was less than 30. Mann Whitney U test was used for intergroup comparisons and Wilcoxon signed rank test was used for intragroup comparisons. Cohen effect size (d) measurement was used to determine the magnitude of change in the evaluated parameters. Effect sizes (Cohen’s d) were interpreted as small (0.2), moderate (0.5), and large (0.8) based on conventional thresholds. Statistical significance was accepted as p < 0.05.
Results
In total, 16 children met the inclusion criteria. They were randomly assigned to two groups: control and intervention. Fifteen completed the study. One child from the intervention group was excluded from the study due to poor attendance in the intervention program. In the intervention group, two (28.6%) were female and six (71.4%) were male. The control group consisted of five girls (62.5%) and three boys (37.5%).
The mean, standard deviation, median, minimum, and maximum values of age, height, weight, and BMI of the patients in the intervention and control groups are shown in Table 1. The mean age was calculated as 9.57 ± 3.64 years for the intervention group and 9.13 ± 3.31 for the control group. The comparison of the age, height, weight and BMI values of the children in both groups revealed that the two groups were similar (p > 0.05).
Demographic characteristics of the participants.
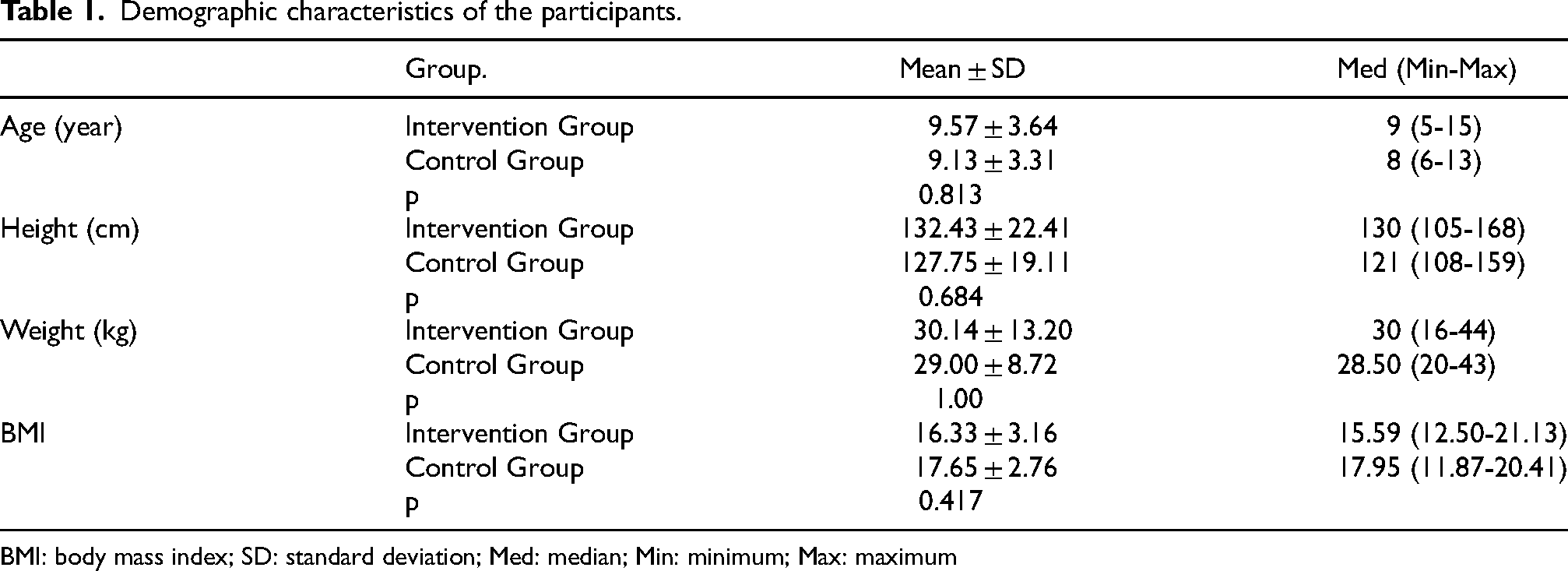
BMI: body mass index; SD: standard deviation; Med: median; Min: minimum; Max: maximum
The distribution of the intervention and control groups according to the GMFCS, MACS and CFCS is shown in Table 2.
Functional status of the participants.
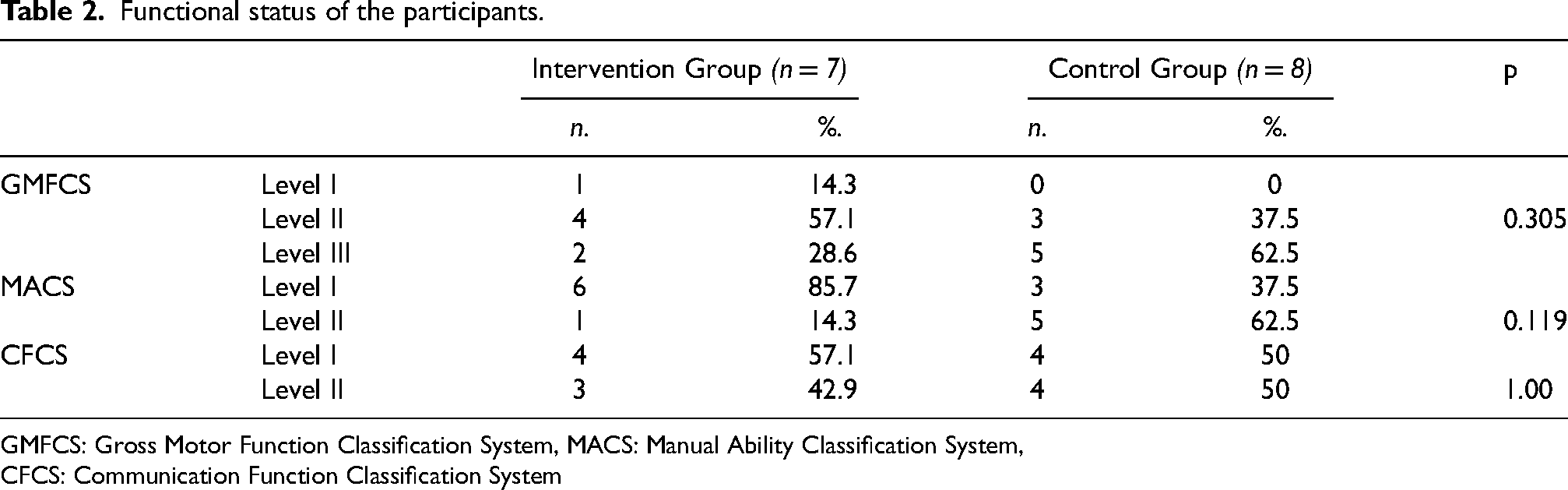
GMFCS: Gross Motor Function Classification System, MACS: Manual Ability Classification System,
CFCS: Communication Function Classification System
Respiratory muscle strength values before and after the ntervention are shown in Table 3. In intragroup comparison, a statistically significant increase was found in the MIP, MIP%, MEP and MEP% values of the intervention group and only in the MEP% value of the control group (p < 0.05). After the intervention, MIP values were significantly higher in the intervention group compared to the control group (p = 0.027), with a large between-group effect size (d = 1.06). MEP values also showed a significant between-group difference (p = 0.032), with a large effect size (d = 1.28).
Comparison of respiratory muscle strength pre- and post-intervention.
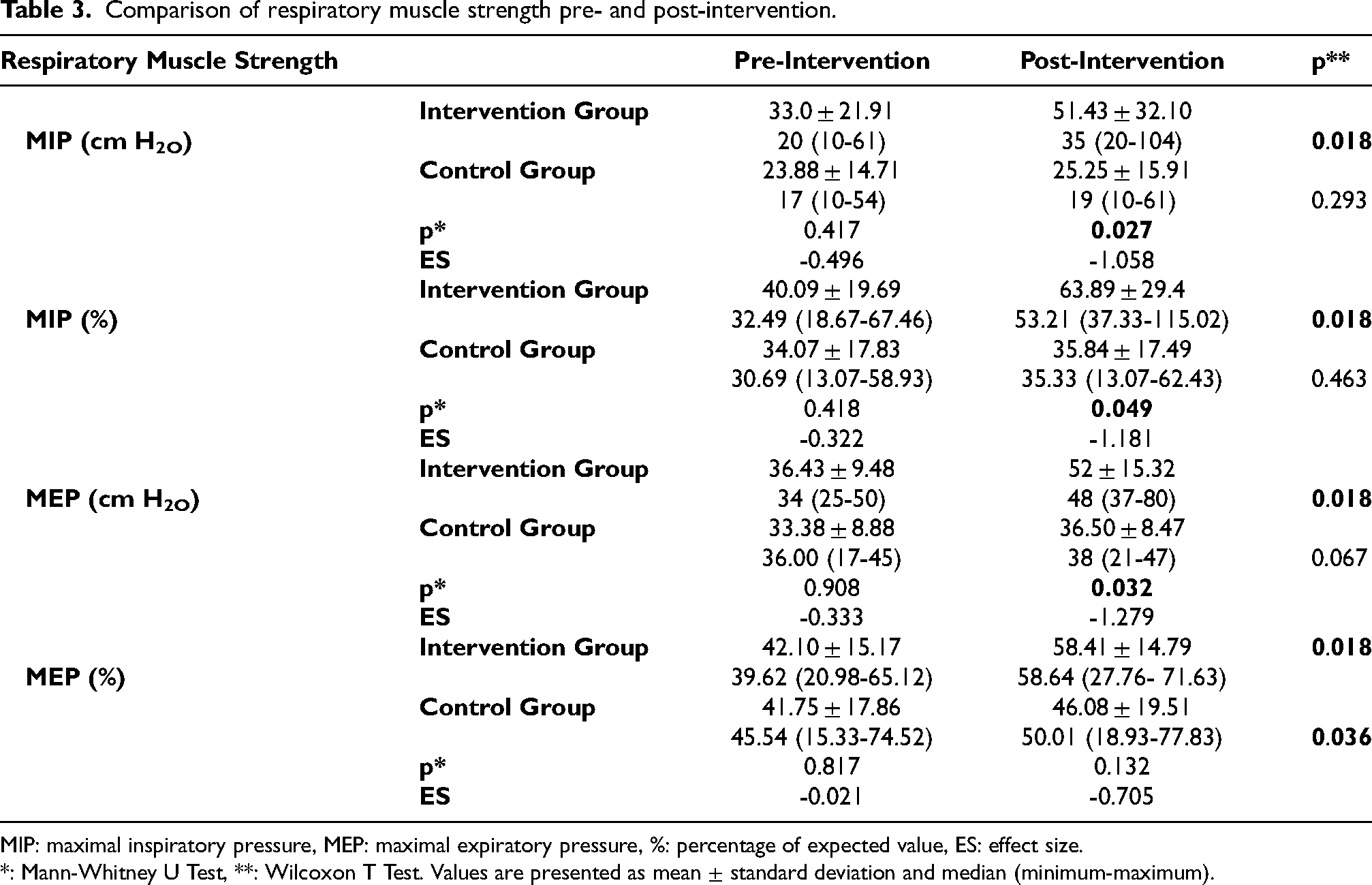
MIP: maximal inspiratory pressure, MEP: maximal expiratory pressure, %: percentage of expected value, ES: effect size.
*: Mann-Whitney U Test, **: Wilcoxon T Test. Values are presented as mean ± standard deviation and median (minimum-maximum).
Intragroup and intergroup comparisons of chest expansion at axillary, epigastric and subcostal levels of the intervention and control groups before and after intervention are shown in Table 4. Significant improvements in chest wall expansion were observed at multiple sites. In the intervention group, chest expansion showed significant improvement at the epigastric (p = 0.017) and subcostal (p = 0.017) levels. The control group also showed a significant within-group improvement at the subcostal level (p = 0.041), but not at the epigastric level.
Comparison of chest expansion pre- and post-intervention.
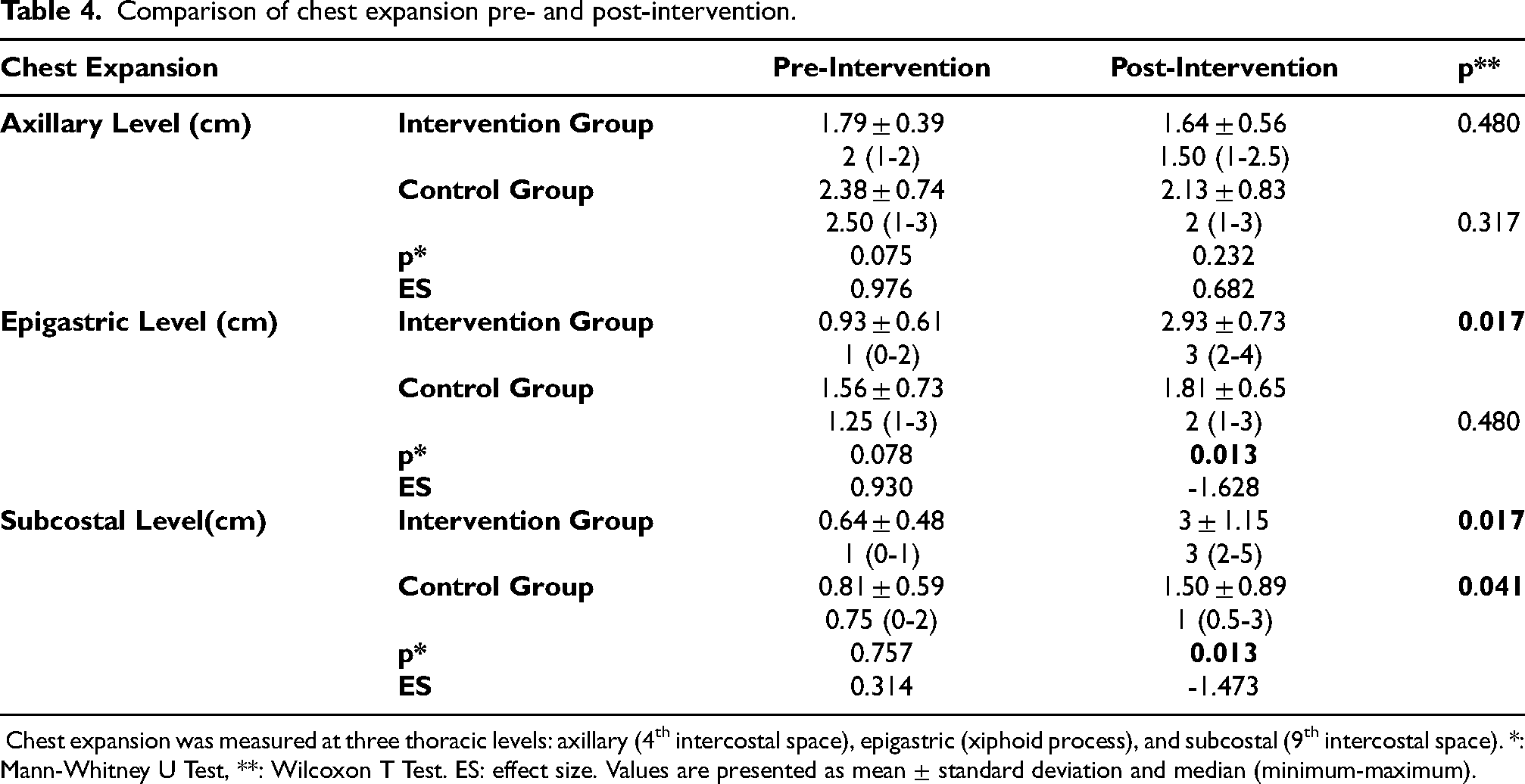
Chest expansion was measured at three thoracic levels: axillary (4th intercostal space), epigastric (xiphoid process), and subcostal (9th intercostal space). *: Mann-Whitney U Test, **: Wilcoxon T Test. ES: effect size. Values are presented as mean ± standard deviation and median (minimum-maximum).
In the between-group analysis, the intervention group showed significantly greater chest expansion than the control group at both the epigastric and subcostal levels (p = 0.013 for both), with large effect sizes observed at each site (d = 1.63 and d = 1.47, respectively).
Intragroup and intergroup comparisons of the GMFM-B scores are shown in Table 5. Both groups showed a statistically significant increase within the group (p < 0.05). In the intergroup comparison, no statistically significant difference was found between the GMFM-B scores (p > 0.05).
Comparison of sitting function pre- and post-intervention.
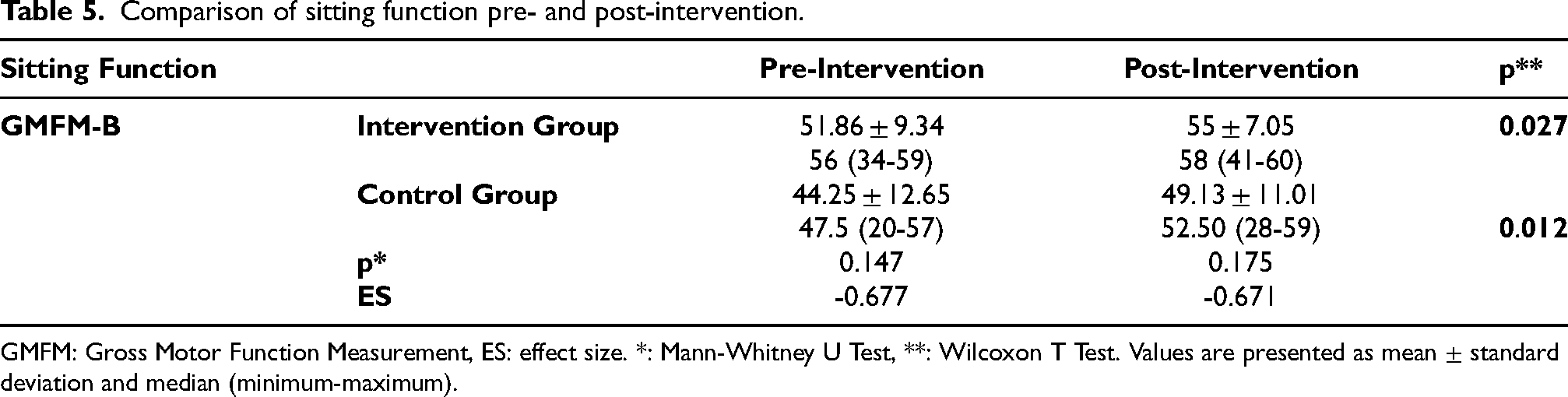
GMFM: Gross Motor Function Measurement, ES: effect size. *: Mann-Whitney U Test, **: Wilcoxon T Test. Values are presented as mean ± standard deviation and median (minimum-maximum).
Intragroup and intergroup comparisons of PFT parameters are shown in Table 6. In intragroup comparisons, a statistically significant difference was found in FVC, FVC%, PEF and PEF% parameters in the intervention group (p < 0.05). In the control group, a statistically significant difference was found in FEV1, FEV1%, PEF and PEF% parameters (p < 0.05). In the comparison of PFT parameters between the groups after the intervention, no statistically significant difference was found in any parameter (p > 0.05).
Comparison of pulmonary function pre- and post-intervention.
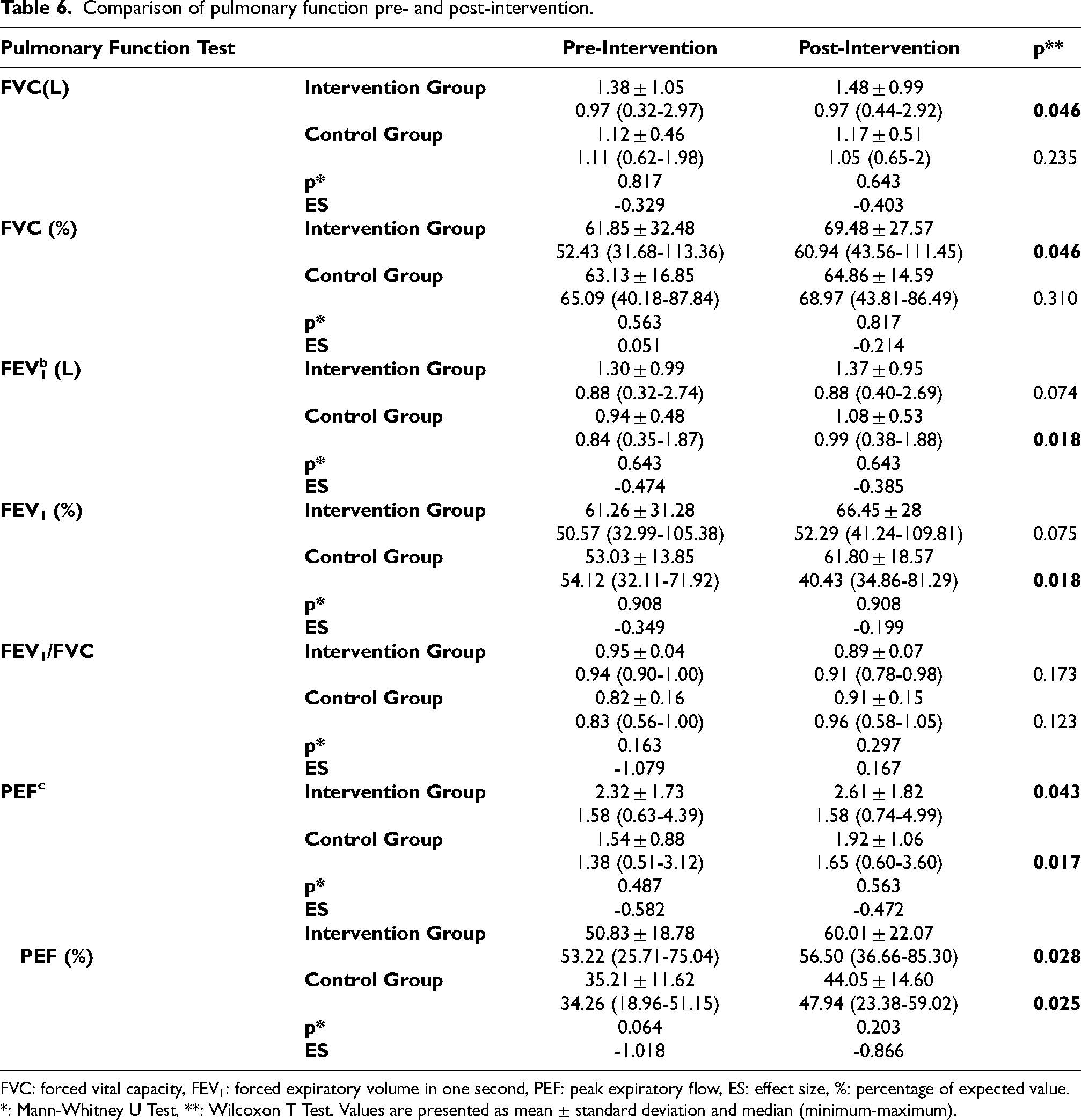
FVC: forced vital capacity, FEV1: forced expiratory volume in one second, PEF: peak expiratory flow, ES: effect size, %: percentage of expected value.
*: Mann-Whitney U Test, **: Wilcoxon T Test. Values are presented as mean ± standard deviation and median (minimum-maximum).
The MIP, MEP, FVC and FEV1 values for each participant before and after the intervention are shown in Figure 3, separately for the intervention and control groups. (Figure 3)
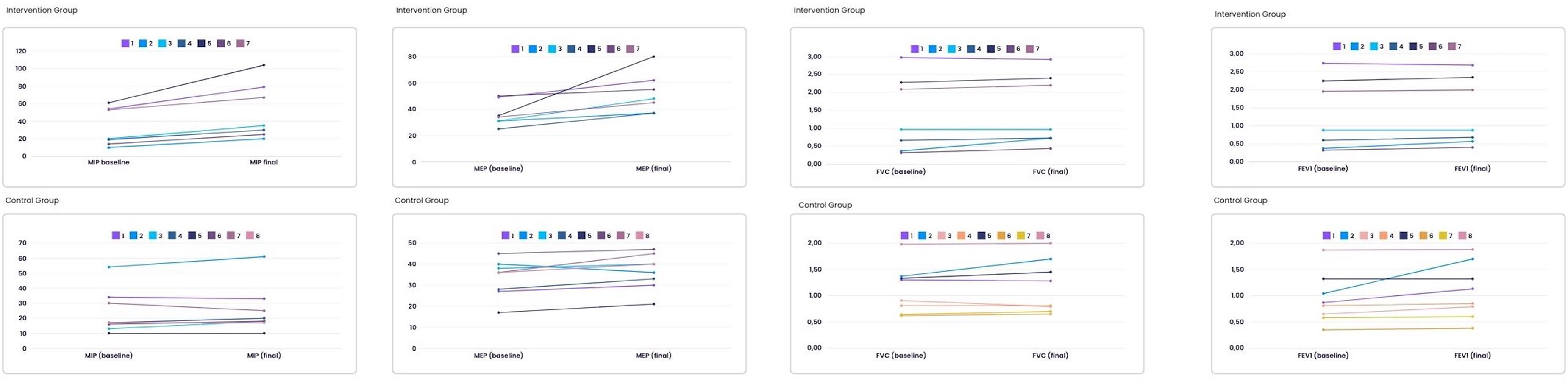
Subject comparison pre-and post-intervention control and intervention groups.
Discussion
This study investigated the effects of manual diaphragmatic relaxation and diaphragmatic breathing exercises added to conventional physiotherapy in children with diplegic CP. At the end of the eight week intervention, both the intervention and control groups showed within-group improvements in several respiratory and functional outcomes. However, when comparing the two groups, significant between-group differences were found only in respiratory muscle strength (MIP, MIP%, and MEP) and chest expansion at the epigastric and subcostal levels, in favor of the intervention group.
These results may reflect the influence of conventional physiotherapy on both groups. Physiotherapy interventions, in any form, may have increased the patients’ ability to breathe more effectively by facilitating the use of respiratory muscles during general movement-based exercises. It must also be kept in mind that during initial recruitment, both groups exhibited restrictive spirometry patterns and impaired respiratory muscle function. The observed within-group improvements—even over a short intervention period—may therefore be more pronounced in this population due to their baseline functional limitations.
The respiratory muscle strength outcomes of this study are consistent with those previously reported in children with CP. In particular, the baseline MIP and MEP values align with those reported by Kwon and Lee in diplegic CP, while slightly lower than values reported in studies including children with hemiplegic CP, such as in Wang et al., where respiratory capacity tends to be better preserved.15–36 Unlike many previous studies that utilized device-based inspiratory muscle training or incentive spirometry, this intervention relied solely on manual diaphragm relaxation and diaphragmatic breathing exercises without the use of specialized equipment. Despite this, the intervention group demonstrated large between-group improvements in respiratory muscle strength (MIP: d = 1.06; MEP: d = 1.28). These findings are in line with previous studies that used device-based interventions and still reported significant improvements in MIP and MEP values. For example, El-Refaey et al. (2017) observed significant increases in both MIP and MEP following a four-week feedback respiratory training program. 27 Similarly, Keleş et al. (2018) and Kepenek-Varol et al. (2022) reported improved respiratory pressures after inspiratory muscle training conducted over six to eight weeks.37,38 Although the methods differ, the magnitude of respiratory strength gains observed in the current study suggests that manual techniques may offer a viable alternative, especially in low-resource settings.
The observed improvements in respiratory muscle strength in the present study may be explained by neuromuscular adaptations associated with skill-based breathing practice. It has been documented that repeated motor tasks enhance muscle activation through increased recruitment of motor units and improved coordination via central nervous system pathways. 39 Strengthened neuromuscular communication and progressive engagement of respiratory muscles, particularly the diaphragm, may have contributed to the observed increases in maximal inspiratory and expiratory pressures. 40 The significant between-group differences in MIP and MEP values observed in this study may be attributed to the specific characteristics of the breathing exercises employed. Manual diaphragmatic relaxation was intended to increase the mobility of the diaphragm and rib cage, while diaphragmatic breathing exercises aimed to improve the synergy and coordination between respiratory muscles. This improved coordination during inhalation and exhalation is thought to enhance respiratory efficiency, enabling the generation of greater inspiratory and expiratory pressures. Therefore, the marked improvement in respiratory muscle strength in the intervention group may be explained by these neuromuscular and biomechanical effects. It should also be noted that participants in the present study successfully adapted to the requirements of lip sealing and instruction following during MIP/MEP assessments. This observation, while ancillary to the main outcomes, highlights the practicality of applying volitional respiratory pressure tests within this study group and may inform future research on the reliability and clinical utility in this group.
Most previous studies assessing chest expansion in children with CP have limited measurements to the axillary level, corresponding to the fourth intercostal space.29–41 In contrast, the present study included chest expansion measurements at the axillary, epigastric, and subcostal levels. The inclusion of the lower thoracic regions may provide a more direct reflection of diaphragmatic movement and the mobility of the lower thoracic cage, which are commonly affected in children with CP. These lower measurement sites align with the anatomical location of diaphragmatic excursion and may better capture the effects of interventions targeting diaphragmatic activation. The findings of the present study are supported by Bennett et al. (2021), who also observed increased chest expansion measured at the xiphoid process following manual diaphragm relaxation. 21 Although that study did not report significant between-group differences in pulmonary function parameters, it did demonstrate a significant intergroup difference in chest expansion, consistent with the results of the present study.
In this study, statistically significant between-group differences were observed at both the epigastric and subcostal levels (p = 0.013 for both), with large effect sizes (d = 1.63 and d = 1.47, respectively). These findings may indicate that the intervention contributed to improvements in lower thoracic mobility. Furthermore, incorporating measurements at these levels could potentially offer additional clinical insight beyond standard axillary assessments, particularly in interventions targeting diaphragmatic function. PFTs are commonly used outcome measures in studies evaluating respiratory interventions in children with CP. Among these, incentive spirometry is the most frequently applied technique, usually in combination with conventional physiotherapy. Previous studies using incentive spirometry in CP have shown mixed results. Some reported significant within-group improvements in parameters such as FVC, FEV1, and PEF 42,43, while others found improvements only in PEF. 44 However, most of these studies did not report significant between-group differences, suggesting that improvements may be related to general activity rather than specific device effects. One study using a feedback-based respiratory endurance device (Spiro Tiger) showed significant gains in FVC and FEV1 (Lee et al., 2014), though this approach differs mechanistically from volume-oriented incentive spirometry. 45 Similarly, a study investigating manual diaphragm relaxation without devices found a significant increase in FVC% in the intervention group but no between-group difference, with changes in chest expansion more pronounced than changes in spirometry values. 21 In the present study, both groups showed within-group improvements in several PFT parameters. The intervention group demonstrated increases in FVC, FVC%, PEF, and PEF%, while the control group improved in FEV1, FEV1%, PEF, and PEF%. However, consistent with most previous research, no significant between-group differences were observed in any of the PFT parameters. This may be due to the inclusion of conventional physiotherapy in both groups, which is known to positively influence pulmonary function. Additionally, the limited sample size and relatively short intervention duration may have reduced the power to detect subtle differences in spirometric values between groups.
To the best of the authors’ knowledge, there are no published studies evaluating sitting function in children with CP following respiratory interventions using the GMFM-B section specifically. In the present study, both groups showed statistically significant within-group improvements in GMFM-B scores, but no significant between-group difference was observed. This may be partially explained by a ceiling effect, as baseline scores were already close to the upper limit, reducing the potential for further measurable improvement. Another factor may be the specificity of training. While the breathing intervention was performed in a supine position, the GMFM-B assesses trunk control in sitting. The mismatch between training and testing positions may have limited the observable effects of the intervention. The diaphragm and other respiratory muscles are known to contribute to postural stability by modulating intra-abdominal pressure. 6 Therefore, respiratory training could theoretically improve trunk control, as also suggested by a randomized controlled study by Keleş et al., which showed significant improvements in trunk control following inspiratory muscle training. 37 However, it is important to note that GMFM-B was not a primary outcome in this study, and the sample size was not powered to detect group differences in postural function. The inclusion of conventional physiotherapy in both groups may have contributed to improvements in postural control across participants. Moreover, variability in the intensity, duration, and frequency of respiratory interventions in the literature makes it difficult to determine the optimal protocol. While improvements in respiratory muscle strength may lead to better functional outcomes over time, this relationship may require longer intervention periods and follow-up assessments to be fully captured. Additionally, age-related differences in muscle development and respiratory plasticity may influence the effectiveness of such interventions. Future studies examining age-specific responses to respiratory exercise programs in children with CP would help clarify whether younger or older age groups benefit more, particularly in relation to muscle fiber maturation and motor control development.
This study has several limitations. First, the sample size may be insufficient to generalize the results to the broader CP population. Also, initial discrepancies in gender and functional level between groups may have influenced the outcomes. However, the specificity of the sample also represents a strength, as it allowed for focused evaluation within a well-defined subgroup. Second, the relatively short follow-up period limited the ability to assess whether the observed effects were sustained over time or translated into broader health outcomes such as reduced pulmonary infections or hospitalizations. Third, participants were recruited from a single outpatient unit within a clinical training and research hospital, which may limit the generalizability of the findings across different clinical contexts. Fourth, one participant in the intervention group showed unusually high post-intervention values, which may have slightly skewed group means due to the small sample size. Fifth, the study was not designed to evaluate structural changes in the pulmonary system, and future studies incorporating imaging techniques such as diaphragmatic ultrasound would be valuable. Lastly, the wide age range of participants may have influenced outcomes due to developmental differences in muscle structure and neuromotor control. Future studies stratified by age group may help clarify differential responses to respiratory interventions.
Conclusion
This study suggests that adding manual diaphragmatic relaxation and diaphragmatic breathing exercises to conventional physiotherapy may enhance respiratory muscle strength and chest expansion in children with CP at GMFCS levels I–III. Importantly, this intervention does not require specialized equipment, making it a cost-effective and accessible option, particularly in low-resource settings. While the results are promising, further research with larger, age-stratified samples and longer follow-up periods is needed to confirm the long-term structural and functional impact of these interventions.
