Abstract
Purpose
This study aimed to test a robotic supine gait training (RSGT) device's safety when treating children and adolescents with a variety of diagnoses, to ensure their safety and the standardization of clinical practices.
Methods
This retrospective observational study included 280 patients who underwent one or more treatment sessions with a RSGT device (DPA Med®) at the Nantes Regional Children's and Adolescent Health Care Center. These patients’ medical files, indexed in the digital medical file manager program, were examined in search of evidence of adverse events presumably associated with the treatment. The search also included when the adverse events took place, their severity, the required treatment, and the clinical outcomes.
Results
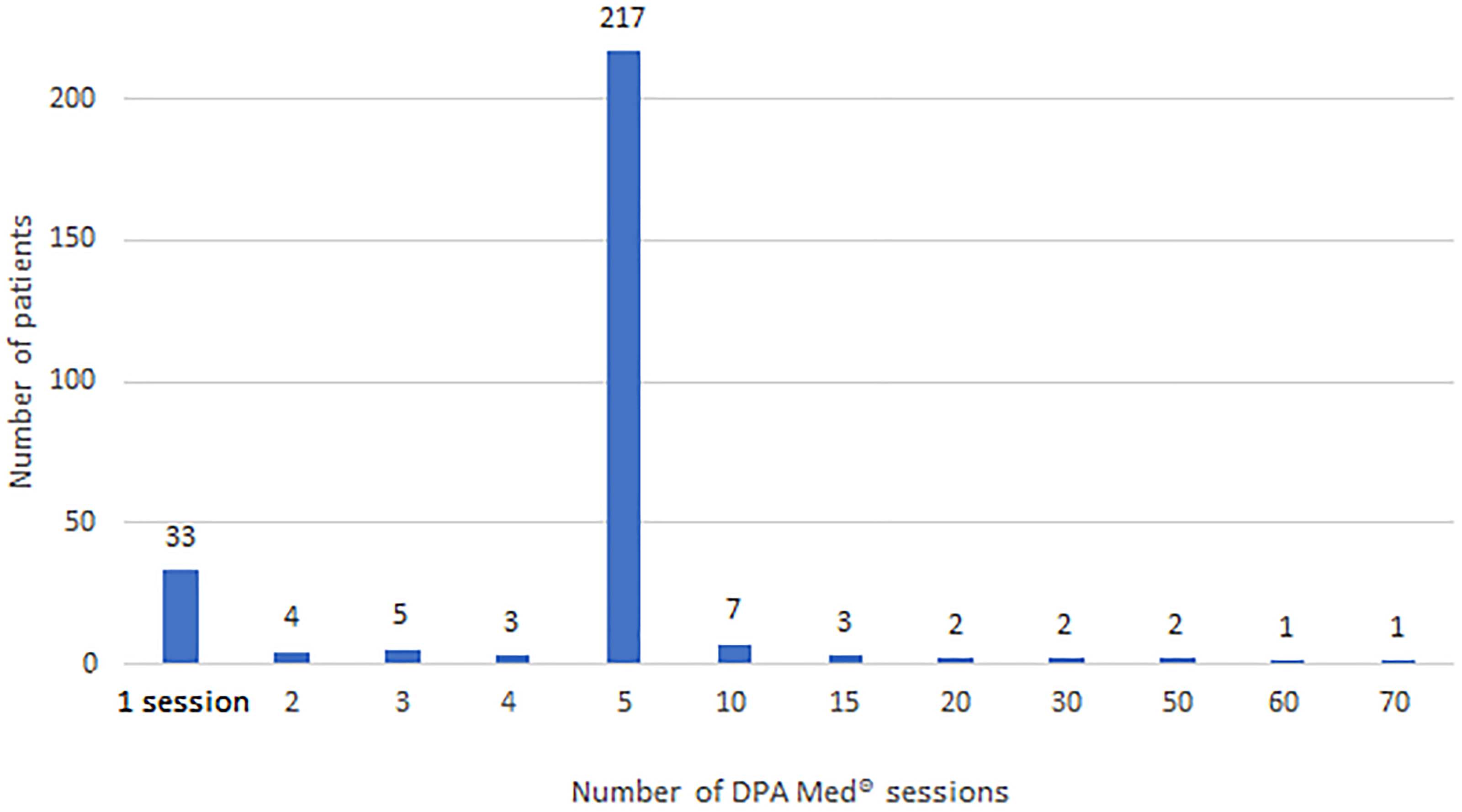
From January 2, 2019, to June 1, 2021 (a 2.5-year period), no adverse events were logged, presumed, or definitively linked to the treatment, for any of the 280 patients included in this study (100% of the sample). Patients underwent an average of 5.71 (standard deviation = 7.06) treatment sessions, for a total of 1598 sessions during that period.
Conclusion
No adverse events linked to RSGT were registered for the children and adolescents treated with the device in this preliminary study.
Introduction
Robotic supine gait training (RSGT) can be performed using a medical device (Figure 1) that induces axial traction and three-dimensional mobilization (lemniscate-shaped movements) of the lumbar-pelvic-femoral and thoracic-lumbar complexes in the supine position, with the legs at an angle of approximately 20°. It also generates pelvic and shoulder girdle dissociation, and pelvic mobility similar to that of typical gait. 1
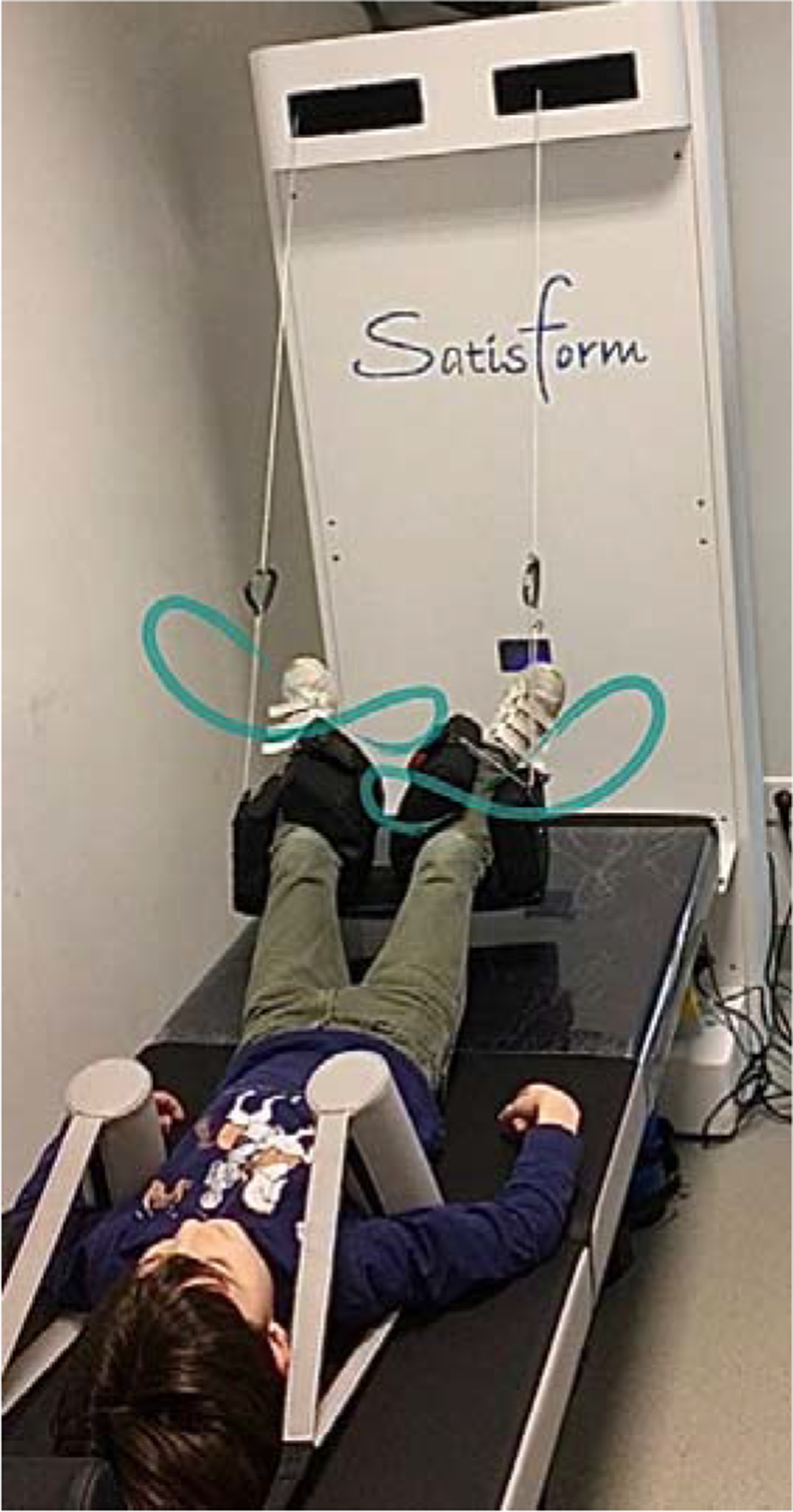
Robotic supine gait training device (DPA Med®).
The RSGT device is used in the treatment of lower back and radicular pain, gait disorders, spinal posture, and respiratory disorders. The mobilization of the lumbar-pelvic-femoral and thoracic-lumbar complexes, induced by the lemniscate-shaped movements of the RSGT, increases joint mobility and flexibility, and stimulates the patient's digestive tract. This is important because these patients are often non-ambulatory. It has been demonstrated that mobilization and spinal traction may reduce pain and enhance function in patients with chronic lower back pain.2-4 Some studies have suggested that mobilization and spinal traction have the additional benefits of increasing lumbar range of motion, granting mobility to patients with vertebral stiffness.5,6 This device would be a valuable tool to treat children and adolescents with similar afflictions, so ensuring its safety is paramount. This will reassure therapists and expand the RSGT device's range of applications for a growing population, without short-, medium- or long-term adverse effects. The purpose of this study was to test the safety of treating a cohort of young patients (children and adolescents) with a variety of diagnoses with the RSGT device.
Methods
The digital medical file manager program registry was used to identify all patients who underwent at least one RGST session using a medical dynamic traction table (DPA Med®, Satisform, Saint-Saturnin, France) between January 2, 2019, and June 1, 2021, at the Nantes Regional Children's and Adolescent Health Care (ESEAN) Center. The treatment sessions were prescribed by the medical doctors of the ESEAN Center.
All patients who met these criteria were included in this study, regardless of their diagnosis or the goals of the treatment session(s). Their medical files were then examined, searching for proof of adverse events such as pain, as evidenced by patient-reported pain Visual Analogue Scale scores or the Face, Legs, Activity, Cry, and Consolability scale, which is an observational pain scale. Reports of skin ulceration, muscle spasms, etc., with a presumed link to the treatment session were also reviewed. If an adverse event was found, information such as when it occurred, the location, and the severity, were extracted from the patient's file. A “Stop” button linked to the device, and if pressed by the user or physiotherapist, induced an instant security stop in case of acute adverse event.
In case of doubt or missing information, patients were contacted by telephone (when possible) to verify the accuracy of the information extracted from their medical records.
This study was approved by the Nantes Group for Ethics in the Medical Field registered under the ruling number 23-27-02-160. It was also compliant with the World Medical Association's Declaration of Helsinki and with European and French biomedical research regulations.
Total research time: five months Data recovery period: three months Data treatment period: two months
The patient's medical file included data focused on several key endpoints.
The primary endpoint involved information that may indicate any adverse effects associated with the use of the RSGT device (DPA Med) during rehabilitation sessions. Secondary endpoints encompassed a variety of details, including the patient's anthropomorphic data such as age, sex, body mass, and height.
Additionally, the file covered main diagnoses according to the International Classification of Diseases 11th Revision and disability classifications based on the International Classification of Functioning, Disability, and Health. 7
It also outlined the objectives of the RSGT rehabilitation sessions, the number of sessions per patient, the frequency and duration of these sessions, and the classification or type of each programmed session.
The inclusion criteria specified that patients must be between five and 19 years old and have undergone at least one RSGT session during their routine care at the ESEAN Center.
Statistical analysis
Microsoft® Excel software was used to perform the statistical analysis. Continuous variables (sex, age, height, body mass, RSGT sessions, adverse effect) were summarized using classic descriptive analysis parameters (mean, standard deviation, extreme values). Qualitative variables (sex, diagnoses, objectives for the RSGT session) were presented as frequencies and absolute percentages.
Results
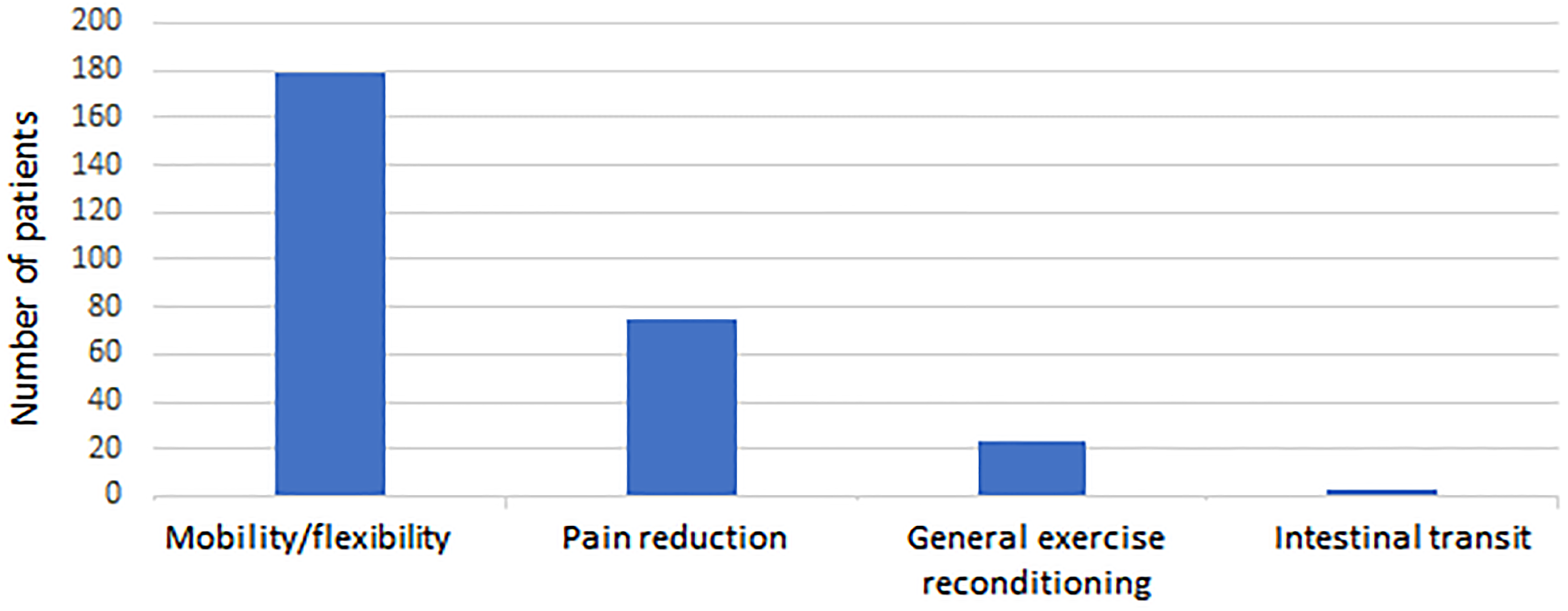
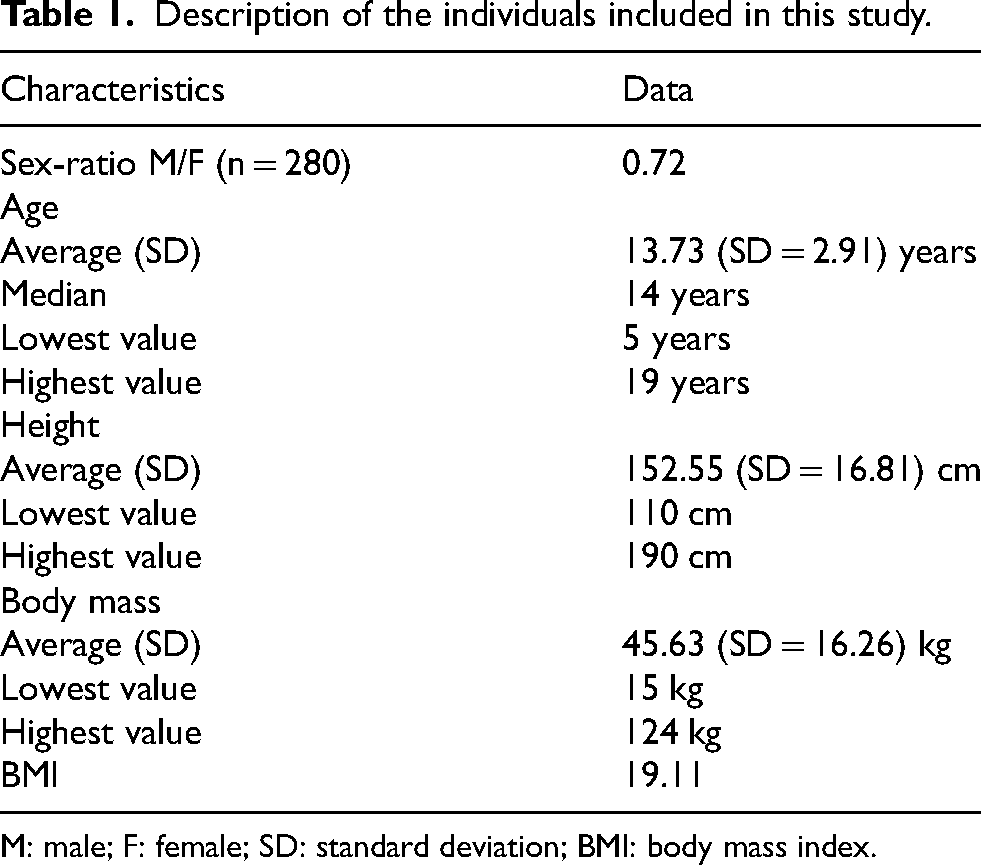
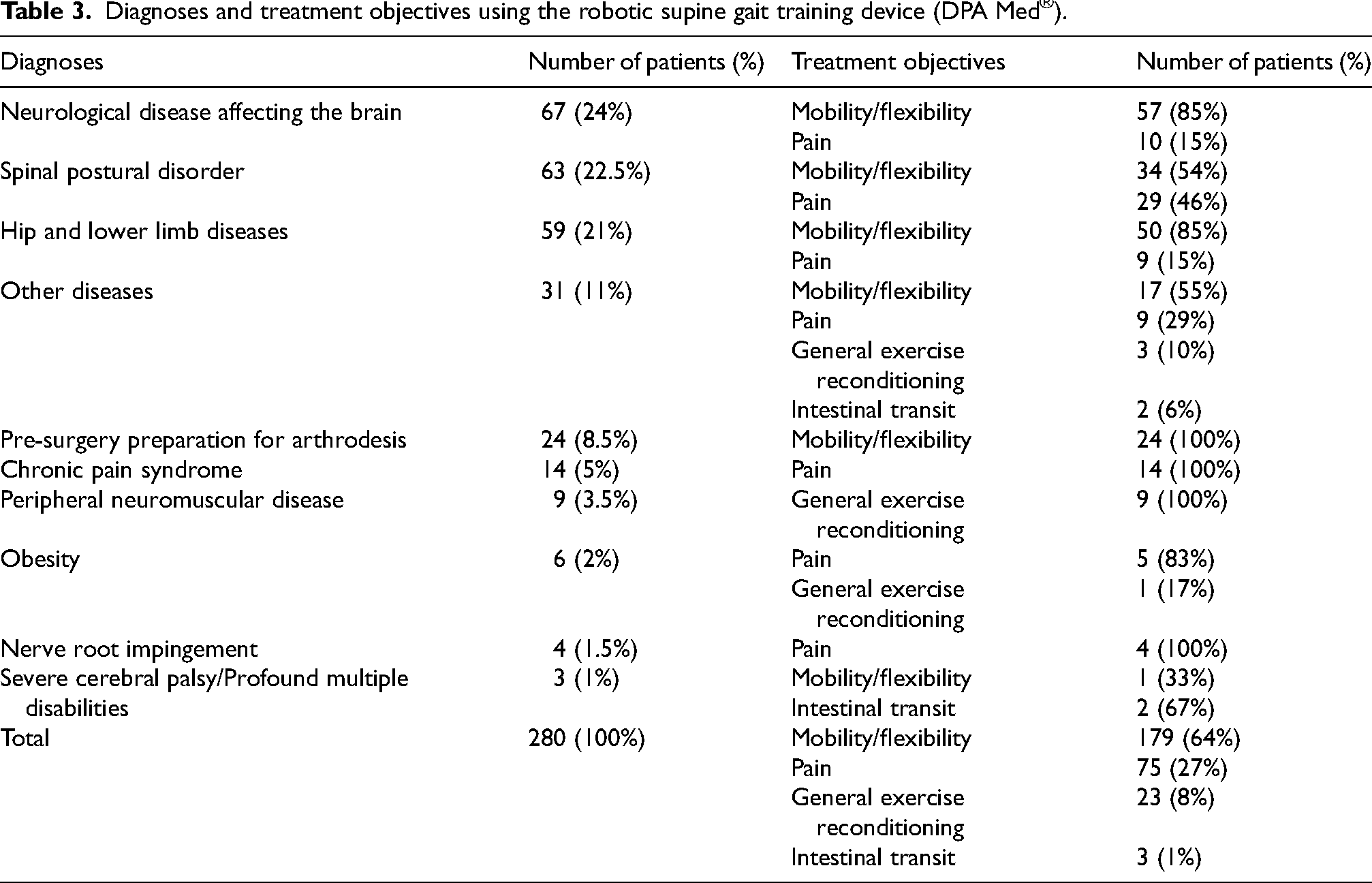
Between January 2, 2019, and June 1, 2021, 280 children and adolescents with an average age of 13.73 years (Table 1) underwent at least one RSGT session (Table 2 and Figure 2). The most common diagnoses were spinal postural disorders (24%) and hip and/or lower limb disorders (22.5%) (Table 3). The treatment objectives for these patients included increasing mobility and flexibility (64%), decreasing pain (27%), general exercise reconditioning (8%), or enhancing intestinal transit (1%) (Figure 3). No direct or indirect adverse effects associated with a RSGT session were found (Table 4). On the contrary, comments such as “pain relief” and “device well tolerated” were mentioned by providers at multiple points in the medical files. The emergency stop button was never pressed.
Distribution of the number of robotic supine gait training device (DPA Med®) sessions applied, per patient.
Distribution of the treatment objectives with the robotic supine gait training device (DPA Med®).
Description of the individuals included in this study.
M: male; F: female; SD: standard deviation; BMI: body mass index.
Number of robotic supine gait training (RSGT) device (DPA Med®) treatment sessions, per patient.

SD: standard deviation.
Diagnoses and treatment objectives using the robotic supine gait training device (DPA Med®).
Findings of the medical file analysis.

Discussion
This study confirmed the safety of using a robotic medical device (DPA Med®) for pediatric RSGT rehabilitation, as no adverse events were recorded among the 280 participants. Children and adolescents with neurological diseases, spinal postural disorders, and hip and/or lower limb disorders were included in this retrospective analysis. These findings are particularly significant as they provide evidence of the device's safety in routine clinical practice, which is critical for expanding its application in pediatric settings. The data gathered during this study was used to establish a minimum age limit of five years, minimum height limit of 110 cm and a minimum body mass limit of 15 kg when using the standardized optimal RSGT installation in safe conditions.
The results align with findings from studies on robotic rehabilitation devices, such as the Lokomat, widely used in pediatric populations. For instance, Cherni et al. demonstrated that robotic gait training significantly improved motor function and walking capacity in children and adolescents with cerebral palsy. 8 Similarly, a study by Gonzalez et al. reviewed various robotic devices and highlighted their potential in enhancing rehabilitation outcomes in children. 9 These studies provide a relevant context to support the safety and potential benefits observed in this research study.
Robotic devices have been increasingly integrated into rehabilitation to address neurological and musculoskeletal disorders, with promising outcomes. This study further supports the notion that such devices can be safely employed with children and adolescents, provided they are used under medical supervision and within the parameters defined by clinical guidelines. Importantly, this study establishes baseline criteria for safe usage, such as minimum age, height, and weight, which contribute to standardizing clinical practice.
The retrospective design may introduce inherent limitations, such as reliance on existing medical records and the potential for missing data. However, the rigorous review of patient files and follow-up procedures mitigates these concerns. The study was conducted in a single center, which might limit the generalizability of the findings to other settings. Future multicenter studies are needed to validate these results across diverse clinical environments.
While this study provides robust evidence of safety, future research should aim to assess the efficacy of the device in achieving specific rehabilitation goals, such as reducing pain or improving mobility. Prospective, multicenter trials could offer more comprehensive insights into the long-term benefits and safety profiles of robotic rehabilitation devices in pediatric populations. Additionally, integrating patient-reported outcomes and advanced monitoring tools could enhance the evaluation of both efficacy and safety.
Conclusion
This study of 280 children and adolescents with a variety of conditions and having undergone one or more RSGT sessions did not present evidence of adverse events following the use of the device. Even though the retrospective nature of this study makes it impossible to rule out the loss of information, the data is in favor of recognizing the safety of the RSGT device when used to treat children and adolescents. If the device is properly applied, it can be safely used for traction and three-dimensional mobilization of the lumbar-pelvic-femoral and thoracic-lumbar complexes. Nevertheless, after this preliminary study, it is necessary to run prospective, multicenter efficacy and safety studies specific to pediatric rehabilitation. This would substantiate these encouraging initial safety conclusions and demonstrate the contribution of the RSGT device to rehabilitation in this population.
Footnotes
Acknowledgments
The authors would like to thank César Cardoso, a licensed English language translator.
All co-authors have reviewed and validated the contents of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Christophe Bensoussan is the CEO of Satisform and Lise Maillard has been awarded a scholarship for her Master's internship by Satisform. The three other authors have no conflict of interest to report.
