Abstract
Purpose
The objective of this article was to explore the prevalence, pathophysiology, diagnosis, and management of sleep-related breathing disorders (SRBDs) in children with spina bifida and specifically myelomeningocele (MMC).
Summary
SRBDs, including obstructive and central sleep apnea, hypoventilation, and hypoxia, are prevalent in children with spina bifida, particularly those with MMC. This high prevalence, ranging from 40% to 80%, is often attributed to brainstem abnormalities, upper airway dysfunction, and restrictive lung disease. Despite the general efficacy of treatments like adenotonsillectomy in typical pediatric populations, these interventions show lower success rates in children with spina bifida. Comprehensive polysomnography is crucial for accurate diagnosis due to the atypical presentation of symptoms. Management strategies include surgical interventions, supplemental oxygen, noninvasive ventilation, and, in severe cases, tracheostomy. A multidisciplinary approach involving various specialties is essential for optimal care and improved outcomes.
Conclusion
Early diagnosis through polysomnography and a multidisciplinary management strategy are critical for effectively treating SRBDs in children with spina bifida, aiming to mitigate associated comorbidities and enhance overall quality of life.
Keywords
Introduction
Sleep-related breathing disorders (SRBDs) are characterized by abnormalities of respiration during sleep. The SRBD is grouped into obstructive sleep apnea (OSA) disorders, central sleep apnea disorders, sleep hypoventilation syndromes, and sleep-related hypoxia disorders. 1 OSA is one of the common SRBDs in children and is seen in 2–5% of general pediatric population. 2 Childhood obesity, craniofacial anomalies, some genetic syndromes such as trisomy 21 and achondroplasia, and neuromuscular disorders significantly increase the risks of OSA in children. SRBDs in children are linked to attentional and focusing deficits, learning difficulties, systemic and pulmonary hypertension, metabolic dysfunction, and overall decreased quality of life necessitating early diagnosis and interventions to prevent morbidities.3,4 Adenoid and tonsil hypertrophy is the most common reason for OSA in typically developing children; therefore, adenotonsillectomy is considered the first line treatment for pediatric OSA, with an overall success rates ranging from 60–80% in developmentally normal children. 5
Spina bifida (SB) is a childhood disability caused by the embryonic failure of fusion of the neural tube resulting in incomplete spinal development, ranging in severity from spina bifida occulta to the more severe myelomeningocele (MMC). MMC affects approximately four per 10,000 births in the United States. 6 MMC is almost always accompanied by Chiari malformations type 2 (CM-II), a condition of caudal herniation of the brain stem through foramen magnum, secondary to alterations in central nervous system development and consequent changes in cerebrospinal fluid flow. Hydrocephalus in present in most cases as well (80–90%), though it is seen is less commonly in children who have undergone prenatal repair of MMC. SRBDs are commonly reported in children with SB and MMC, with research studies showing a very high prevalence of 40–80%. The epidemiology, pathophysiology, and diagnosis and management of SRBDs in children with SB will be discussed here. For the purpose of this review, the term SB will be used to describe only children with associated MMC.
Materials and methods
This article is not a systematic review of the literature but includes the pertinent literature covering sleep breathing issues in children with SB.
Epidemiology of SRBD in SB
Respiratory disorders are well known in children with SB and MMC. Respiratory complications include but are not limited to abnormalities of control of breathing, upper airway dysfunction, restrictive lung disease, recurrent aspiration pneumonias, and pulmonary hypertension with resulting cor-pulmonale in severe cases. 7 In 1998, Waters et al. first reported a polysomnogram (PSG) confirmed a SRBD in about 63% (n = 52) of the children in Montreal Children's Hospital who screened positive for SRBDs. 8 Since then, various studies have reported a high prevalence of SRBDs in children with SB and MMC with a prevalence around 40% when a PSG was used as a screening tool and up to 80% when PSGs were performed in children with positive symptoms for SRBD.9–13 Consistent with the previous literature, a recent case control study and literature review by Lazzareschi et al. reported SRBD prevalence of 45% in children with CM-II/MMC compared to 5% of control subjects with 20% of their cases presenting with severe SRBDs. 14 The prevalence of SRBDs is reported to be much higher in neonates and infants, with studies showing a prevalence of 60% to almost 100% in neonates with MMC. In a more recent study by Stark et al. on comparison of SRBD outcomes between neonates who have undergone prenatal and post-natal repair of SB (MMC), 15 not only was a very high prevalence of SRBD reported in 97–100% of these neonates, but there was also no difference in SRBD outcomes after the prenatal repair. Some studies have identified risk factors for SRBDs in these children, with males being at higher risk than females, higher prevalence of SRBDs in more rostral regions than caudal lesions, and SRBDs being more common in children with impaired lung function.8,12 There is an association between SRBDs and sudden unexplained death in SB and MMC, with studies reporting 12–21% incidence of death among children with a SRBD followed over time.8,16
Pathophysiology of SRBDs in SB
SRBDs manifest as OSA, central sleep apnea, periodic breathing, sleep-related hypoxia, and sleep hypoventilation in children with SB. Brain stem abnormalities are thought to be the central cause of SRBDs in these children. MMC and hind brain herniation cause compression of the respiratory center nuclei as well as the lower cranial nerve nuclei that control the vocal cords and pharyngeal muscles maintaining airway patency. 17 In addition, neuropathological examination of children with MMC who died has shown brain stem dysplasia with hypoplastic or aplastic cranial nerve nuclei. 18 The brain stem changes, either developmental or acquired, result in impaired respiratory control, reduced airway patency, and poor arousal mechanisms resulting in a variety of sleep breathing disorders in these children.8,10 OSA and central sleep apnea are commonly reported SRBDs, with studies reporting presence of both central and obstructive events in most of these children.8,12,14,15 Upper airway soft tissue hypertrophy such as adenoid and tonsil enlargement, the most common etiology for obstructive SRBDs in children, should be considered and evaluated for all children presenting with a SRBD. Additionally, even as early as 11 weeks of gestation, changes in facial development are evident, potentially further narrowing the airway. 19 Children with SB have a higher incidence of obesity when compared with the general population, which in turn can impact risk for OSA. 20
These children are often insensitive to hypoxia and hypercarbia, secondary to reduced peripheral and central chemoreceptor sensitivity with reduced arousal mechanisms, resulting in sustained sleep-related hypoxia and sleep hypoventilation similar to children with congenital central hypoventilation syndromes.21,22 In addition, some children have kyphoscoliosis with restrictive lung disease secondary to lower motor neuron lesion. The reduced lung capacity from restrictive lung disease with impaired intercostal muscle movement in sleep can lead to severe gas exchange abnormalities particularly in rapid eye movement sleep.8,23 Some children have intrinsic lung disease with abnormal lung parenchyma from chronic airway aspiration. This contributes to sleep-related hypoxia and further decline in oxyhemoglobin saturation with respiratory events. Once again, obesity is a known risk factor for SRBDs, and truncal obesity can increase the severity of SRBDs in these children. 20
Diagnosis of SRBDs in SB
Children with SB may have no signs or symptoms of a SRBD, in stark contrast to typically developing children. Patient history of snoring or poor subjective sleep cannot be relied upon as an indication for a PSG for a child with SB. Because infants and children with SB generally do not present with the typical symptoms of SRBDs, formal polysomnography is necessary.12,14,24,25 Recent work by Eisenberg et al. demonstrated a lack of signs and symptoms that would predict an abnormal PSG other than previously witnessed apneas [10]. The sensitivity and specificity of sleep screening questionnaires such as the Pediatric Sleep Questionnaire for children with SB are reported to be much less compared to the general pediatric population, obligating the need for PSGs in most children. 26 A nocturnal oximeter can detect the presence and severity of nocturnal oxyhemoglobin desaturations. However, oximetry cannot differentiate the type of SRBD and can easily miss sleep hypoventilation. In-lab comprehensive PSG is considered the gold standard for diagnosis of SRBDs. An overnight PSG measures both respiratory and non-respiratory parameters at night including sleep stages, respiratory events, carbon dioxide, oxygen, heart rate, limb movements, and body position with audio and video recordings of the night. Respiratory events are calculated, and a severity score is assigned based on the number of apnea and hypopnea events per hour. The definition and scoring of SRBD events is detailed in the scoring manual by Berry et al. in 2007. 27 Of note, home PSGs in children are not typically effective at capturing needed data given current technology, though this may alter as technology improves. 28
The specific definitions of pediatric SRBDs are detailed in the scoring guidelines and international classification of sleep disorders set forth by the American Academy of Sleep Medicine.27,29 OSA, one of the common forms of SRBD, is characterized by episodes of partial or complete airway obstruction in children resulting in gas exchange abnormalities and sleep fragmentation. It is defined as apnea hypopnea index of more than two events per hour in children. Central sleep apnea, a less common SRBD in children, is complete cessation of breathing lasting more than 20 s or lasting more than two breaths with oxyhemoglobin desaturation of more than 3% of baseline or a brain arousal. Central apnea index of more than two events per hour is considered abnormal in children. Periodic breathing is characterized by episodes of recurrent central pauses in breathing with associated oxyhemoglobin desaturation events. Periodic breathing is common in the first few months of life due to immature respiratory centers, and most infants stabilize their breathing, and outgrow periodic breathing, by six months of age. Sleep hypoventilation is defined as carbon dioxide of more than 50 mm of Hg for more than 25% of the night on an overnight PSG. Sleep related hypoxia, usually seen in children with underlying lung disease, is defined as a total duration of more than three minutes of oxyhemoglobin saturation of less than 90% on an overnight PSG. This can be seen secondary to underlying lung disease, in children with restrictive lung disease and sleep hypoventilation, or in children with reduced chemoreceptor sensitivity. Studies report a higher incidence of OSA and central sleep apnea in children; the exact prevalence of sleep hypoventilation and sleep-related hypoxia is unclear in this population. Figure 1 shows an overnight hypnogram (a summary of sleep study findings) that demonstrates the combination of obstructive and central events with associated desaturations in a four-year-old child with MMC and SRBD. Sleep apnea screening should be offered at every clinic visit and there should be a lower threshold to refer these children for an overnight PSG for a thorough evaluation of SRBDs.
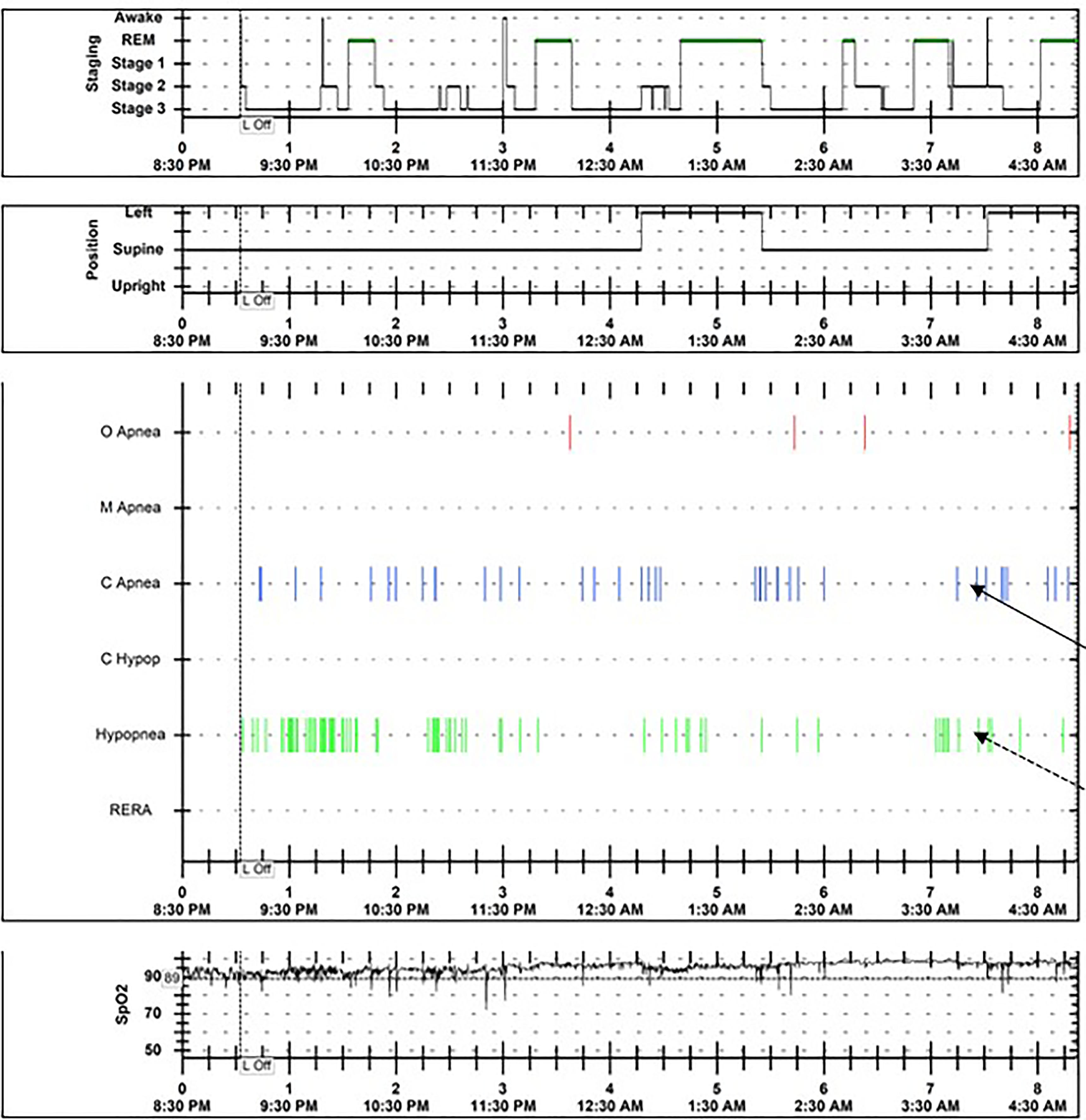
Hypnogram from an overnight polysomnogram showing recurrent central events (black arrow) and obstructive hypopneas (dotted arrow) for a four-year-old child with spina bifida and myelomeningocele.
Management of SRBDs in SB
There is limited data on management of SRBD in this population, and treatment mostly depends on the type of SRBD.11,12,30,31 Adenoidectomy and tonsillectomy are the first line treatment for OSA in otherwise typical children, with a success rate up to 80%. The success rate of adenotonsillectomy in the SB population is much less than the general population (less than 50%), but adenotonsillectomy should still be considered as a first line therapy, particularly in children with evidence of tonsil and adenoid enlargement.8,31 Previous literature has reported that neurosurgical interventions such as posterior fossa decompression improve the severity of SRBDs in some cases, but the neurosurgical community has shifted away from these interventions in recent years owing to lack of efficacy. 32 Antenatal repair of MMC is beneficial to prevent hydrocephalus and SRBDs, but a recent study reported no significant changes to SRBD outcomes with this antenatal intervention. 15 The primary/developmental changes in the brain stem likely explain the insignificant improvement in SRDBs post cranial decompression as well as prenatal repair of MMC in these children. 18 Supplemental oxygen has remained a mainstay of treatment for neonates and children with SRDBs and sleep related hypoxia.11,15 Caffeine/theophylline therapy is considered in some cases with predominantly central events and sleep-related hypoxia.13,31 Noninvasive ventilation support (continuous positive airway pressure or bilevel positive airway pressure) therapy is beneficial in children with residual OSA post adenotonsillectomy or neurosurgical interventions, and in children with sleep hypoventilation. It is often a challenge to find an appropriate noninvasive ventilation interface for children and tolerance and compliance are suboptimal in most children, but studies have shown improved compliance with parent education and close clinical follow ups. 33 Tracheostomy and invasive ventilation remain the mainstay of therapy for severe cases as well as for children who fail other therapies.31,34 Invasive ventilation adds more to the burden of care for patients and families, but this remains the most secure and safest way to maintain gas exchange in some of these children. 32
A multidisciplinary program including relevant specialists is the gold standard and essential to the effective care of children with MMC and SRBD. Ideally, such programs should facilitate collaboration between pulmonology, neurosurgery, urology, and physical medicine and rehabilitation at a minimum. Neuropsychology, orthopedics, social work, physical therapy, occupational therapy, clinical nutrition, and gastroenterology team members may also impact the successful management of SRBDs in MMC, given its multifaceted nature.35,36
Conclusion
SRBDs are common in children with SB, and a PSG should be strongly considered for early diagnosis and treatment of SRBD in these children. The treatment, though complex, can minimize the comorbidities and prevent complications. A multidisciplinary team approach with sleep physicians as team members is essential to improve the outcomes in these children.
Footnotes
Acknowledgements
The authors would like to acknowledge the patients and families caring for children and adults with SB as well as the multidisciplinary team members at their institutions for the knowledge gained in this field. This research did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical considerations
This study, considered a literature review, is exempt from the Institutional Review Board approval.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
