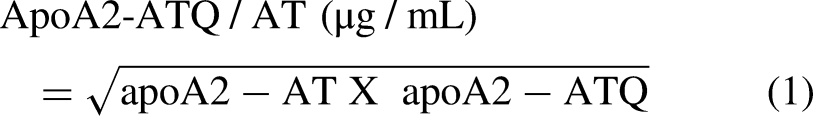Abstract
Pancreatic cancer is a rare and refractory cancer, and the development of blood biomarkers for the enrichment of high-risk individuals who have risk factors for pancreatic cancer from the asymptomatic population is an unmet medical need. We identified abnormalities in the C-terminal truncation of the apolipoprotein A2 dimer (apoA2-isoforms: apoA2-i) in the blood of pancreatic cancer patients through proteomic analysis, and we have reported the potential for diagnosing resectable pancreatic cancer by detecting these abnormalities. We successfully developed enzyme-linked immunosorbent assay (ELISA) reagents for measuring apoA2-i for research use only, and then the basic data for diagnosing pancreatic cancer were accumulated by several studies using these reagents. In 2023, ELISA for measuring apoA2-i was regenerated by the regulation under the Japanese Quality Management System (QMS), it received marketing approval in Japan as an in vitro diagnostic (IVD) kit to aid in the diagnosis of pancreatic cancer, and it is now used in clinical practice. This review chronicles the journey from the initial discovery through omics research, to demonstrating clinical utility via multicenter studies in Japan and international collaborative research using the research reagent and validating the clinical performance of the IVD ELISA kit through a regulatory, science-guided, clinical trial in Japan, and finally to recent activities in the USA.
Introduction
The American Cancer Society estimates that, by 2024, there will be more than 66,000 cases of pancreatic cancer and approximately 52,000 deaths from it (https://www.cancer.org/cancer/types/pancreatic-cancer/about/key-statistics.html). Given that the 5-year relative survival rate of patients with pancreatic cancer from 2013 to 2019 was 12.5%, which is extremely low compared to that of patients with other solid tumors, and that the most reliable treatment for pancreatic cancer is surgery, it is desirable to efficiently detect and initiate treatment for curatively resectable pancreatic cancer to reduce the mortality rate.
If pancreatic cancer is suspected by imaging tests such as magnetic resonance cholangiopancreatography (MRCP), endoscopic ultrasound (EUS) and endoscopic retrograde cholangiopancreatography (ERCP) are performed (https://www.cancer.org/cancer/types/pancreatic-cancer/about/key-statistics.html). However, examinations using these diagnostic modalities are typically performed only after the onset of clinical symptoms, when the window for resection surgery may have already passed.
Conversely, the United States Preventive Services Taskforce (USPSTF) does not recommend pancreatic cancer screening using imaging tests in asymptomatic populations due to the low age-adjusted incidence rate of pancreatic cancer (about 12.9 per 100,000 persons). The USPSTF found no evidence of benefit from pancreatic cancer screening in the asymptomatic population due to the low positive response rate and high false-positive rate. Furthermore, the study concluded that the potential benefits of pancreatic cancer screening for an asymptomatic population do not outweigh the harms, given the presence of more than moderate harms associated with pancreatic cancer treatment detected through screening. 1
However, the USPSTF removed the recomendation for high-risk groups from this guideline, because it is outside the scope of this recommendation with respect to familial pancreatic cancer and other cancers associated with a high risk of pancreatic cancer. Familial pancreatic cancer, defined as a family history of pancreatic cancer in first-degree relatives, is present in 5–10% of all pancreatic cancer patients. 2 The risk of developing pancreatic cancer increases with the number of first-degree relatives with pancreatic cancer, with a 6.4-fold increase for two first-degree relatives and a 32-fold increase for three or more relatives. This risk is significantly higher than the risk reported in the general population. Therefore, if the high-risk group can be concentrated to the extent of familial pancreatic cancer, there is a possibility of implementing pancreatic cancer screening using imaging tests and other methods.
Thus, a blood biomarker that can narrow down pancreatic cancer risk before imaging tests is highly promising as an unmet medical need given the above background.
Risk factors for pancreatic cancer include chronic pancreatitis and intraductal papillary mucinous neoplasm (IPMN), pancreatic cyst, and pancreatic duct dilation, and it is desirable to develop high-resolution blood biomarkers that will trigger imaging tests for the pancreatic cancer risk population with these risk factors. 3
However, there are not many blood markers for pancreatic disease in clinical use. Representative tumor markers for pancreatic cancer include carbohydrate antigen 19-9 (CA19-9), Duke pancreatic monoclonal antigen type 2 (Dupan-2), s-pancreas-1 antigen (Span-1), and carcinoembryonic antigen (CEA). CA19-9 is widely used but lacks specificity and is not expressed in approximately 5–10% of patients, limiting its effectiveness. Dupan-2 and Span-1 are also useful for monitoring disease progression but have lower sensitivity for early-stage pancreatic cancer.4,5 In addition, these biomarkers do not react IPMN, pancreatic cysts that are considered as high-risk individuals of pancreatic cancer. Clinical research has been actively conducted targeting proteins, DNA/RNA, exosomes, metabolites, and other factors, but there remains a critical need for the development and clinical application of biomarkers that exhibit high performance and are affordable.
Discovery of a characteristic cleavage pattern of the C-terminal amino acids of apolipoprotein-A2 homodimer (apoA2-isoforms) in blood found in pancreatic cancer patients and in a high-risk population for pancreatic cancer
We comprehensively profiled proteins in plasma samples from both pancreatic cancer patients and healthy controls using matrix-associated laser desorption ionization mass spectrometry (MALDI MS), and we found that proteins with a mass-to-charge ratio (m/z) of 17,252 were significantly lower in pancreatic cancer patients than in healthy controls. 6 The amino acid sequence of the protein with a mass of 17,252 m/z was determined by tandem mass spectrometry (MS/MS) analysis. It was identified as a dimer of apolipoprotein A2 (apoA2), a component of high-density lipoprotein (HDL) circulating in plasma. 7
In addition, MALDI MS profiles showed peaks at 17,380 m/z and 17,124 m/z near the 17,252 m/z peaks. These results indicated that these proteins are apoA2 dimeric isoforms (apoA2-isoforms; apoA2-i) with different C-terminal amino acid cleavage states. The C-terminal amino acids were designated apoA2-ATQ/ATQ (17,380 m/z, apoA2 heavy chain), apoA2-ATQ/AT (17,252 m/z, apoA2 intermediate chain), and apoA2-AT/AT (17,124 m/z, apoA2 light chain) according to their sequence, starting from the highest mass.7,8
From the mass spectrometry results, it was found that the level of apoA2-ATQ/AT, the apoA2 intermediate chain, is significantly lower in the plasma of pancreatic cancer patients than in healthy people. On reviewing the apoA2 profile in the plasma of patients with pancreatic cancer, the C-terminal amino acids of the apoA2 dimer appeared to be in a hyperprocessing pattern in which apoA2-i light chain (apoA2-AT/AT) was dominantly observed, or a hypoprocessing pattern in which apoA2-i heavy chain (apoA2-ATQ/ATQ) was dominantly observed. In the over-cleavage state, the apoA2 light chain, apoA2-AT/AT, increases, and the heavy chain, apoA2-ATQ/ATQ, decreases. It is reasonable to expect that apoA2-ATQ/AT, the intermediate chain, would decrease in patient plasma whether the apoA2 dimer is in the over-cleaved or under-cleaved state (Figure 1). 8
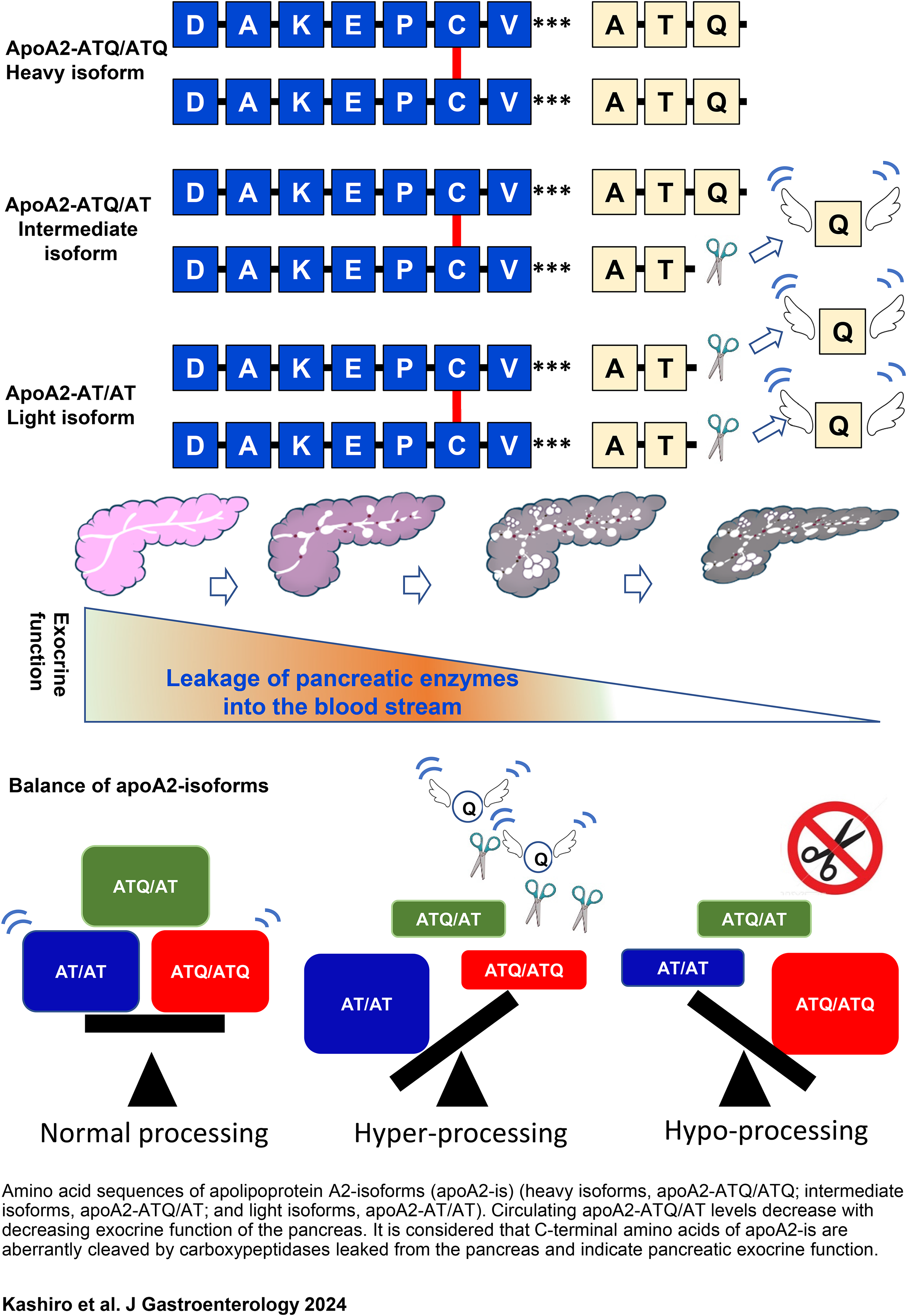
Scheme of apoA2-dimers and aberrant processing of C-terminal amino acids in circulating apoA2-dimers, as reflected in pancreatic exocrine function.8,17 Amino acid sequences of apolipoprotein A2-isoforms (apoA2-is) (heavy isoform, apoA2-ATQ/ATQ; intermediate isoform, apoA2-ATQ/AT; and light isoform, apoA2-AT/AT). Circulating apoA2-ATQ/AT levels decrease with decreasing exocrine function of the pancreas. It is considered that C-terminal amino acids of apoA2-is are aberrantly cleaved by carboxypeptidases leaked from the pancreas and indicate pancreatic exocrine function.
Proof-of-concept study of the potential of apoA2-i for detecting pancreatic cancer using a research-use ELISA
Clinical testing using mass spectrometry can be complex when measuring multiple specimens, particularly in terms of data standardization and precision control of testing procedures. To address this, specific antibodies against apoA2-ATQ and apoA2-AT were prepared in collaboration with Toray Industries Inc., and reagents for a sandwich ELISA were developed to measure apoA2-ATQ and apoA2-AT concentrations. Although the details of the test principle are not discussed here, the synergistic average of apoA2-ATQ and apoA2-AT concentrations measured by ELISA correlated well with the qualitative concentration of the apoA2 intermediate chain (apoA2-ATQ/AT) derived by mass spectrometry. This correlation led to the definition of a formula to estimate the apoA2-ATQ/AT concentration (Equation 1).
The point estimate of the area under the curve (AUC) for receiver-operating characteristic (ROC) curve analysis was 0.944, higher than that of CA19-9 (0.899).
Furthermore, the point estimates of AUC to discriminate benign pancreatic cancer-related diseases such as chronic pancreatitis, IPMN, endocrine tumors, mucinous cystic neoplasm (MCN), and serous cystic neoplasm (SCN) from healthy individuals were 0.992, 0.92, 0.84, 0.816, and 0.983, respectively. These results suggest that this biomarker has the potential to diagnose not only pancreatic cancer, but also diseases considered as a risk group for pancreatic cancer. 9
The potential of this biomarker to grade IPMN was further investigated using the developed ELISA reagents, since grading of IPMN is crucial for determining subsequent treatment strategies. IPMN high-grade dysplasia (HGD) is synonymous with the previously used term non-invasive IPMN, which is pathologically considered to be an intraepithelial carcinoma condition. In a collaborative study with the University of Heidelberg, a high-volume center for pancreatic surgery worldwide, the sensitivity of apoA2-ATQ/AT to detect IPMN HGD was 70.6%, compared to 14.5% for CA19-9. When discriminating IPMN HGD from healthy individuals, the AUC for apoA2-ATQ/AT was 0.910, significantly higher than the 0.686 for CA19-9 (p < 0.01, Bootstrap test). 10
Physiological significance of pancreatic exocrine function and apoA2-i
The pancreas is rich in carboxypeptidase, a digestive enzyme that cleaves the C-terminal amino acid of proteins. It is hypothesized that the onset of microcarcinoma increases intraductal pressure in the pancreatic duct, leading to leakage of C-terminal digestive enzymes into the peripheral circulating blood. This enhances the C-terminal cleavage of apoA2-i, resulting in excessive cleavage, with apoA2-AT becoming dominant over apoA2-ATQ. Conversely, when pancreatic exocrine dysfunction or devastation progresses, the C-terminal cleavage activity decreases, and apoA2-ATQ becomes predominant (Figure 1).
Several reports have suggested a relationship between apoA2-i and pancreatic exocrine function. For instance, Kobayashi et al. reported that the cleavage-suppressed form of apoA2-i is significant in patients with autoimmune pancreatitis (AIP). 11 In addition, Hayasaki et al. reported a shift from an apoA2-AT-dominant over-cleaved form to an apoA2-ATQ-dominant cleavage-suppressed form before and after preoperative radiation chemotherapy for pancreatic cancer. 12 These changes are thought to be induced by the loss of cleavage of the C-terminal amino acid of the apoA2 protein due to the decline or loss of pancreatic exocrine function. In fact, Futagami reported that blood levels of apoA2-i correlated with the results of the pancreatic function diagnostic testing (PFD test), which estimates pancreatic exocrine function. 13
Experimental pancreatic cancer screening study using apoA2-i ELISA research reagents
We conducted an experimental prospective screening study for pancreatic cancer or pancreatic risk diseases in an asymptomatic population aged 20 years or older. This study was conducted in collaboration with Kobe University, using the apoA2-i research-use ELISA. A total of 5221 individuals were enrolled in the study, and 84 (1.6%) tested positive. Of them, 54 subjects voluntarily underwent further imaging tests because this research was an observational study. Of these 54 subjects, 26 showed abnormal findings on imaging. These included 1 case of pancreatic cancer, 9 cases of IPMN, 5 cases of pancreatic cysts, and 3 cases of pancreatitis. The positive predictive value of abnormal pancreatic findings on imaging was 48.1% (26/54), suggesting that apoA2-i may be an efficient blood biomarker to increase the probability of pre-imaging diagnosis. However, there was a limitation in this study. The target asymptomatic population was 20 years of age or older, which is a young age group for pancreatic cancer screening, and the imaging rate was not high (64.3%), since only 54 of the 84 positive cases underwent precise imaging examinations.14,15 Therefore, in collaboration with the Kagoshima and Hokkaido chapters of the Japan Cancer Society, a prospective screening study has been initiated using the apoA2-i research reagent. This study targets an asymptomatic population aged 50 years and older.15,16 To date, more than 13,800 participants have been enrolled in this study. The cancer incidence status of nearly all participants is currently being monitored using information from regional cancer registries.
Though there are challenges in accuracy control due to the use of research reagents, it is anticipated that the detection rate of pancreatic cancer, approximate sensitivity and specificity of screening, and the risk ratio of pancreatic cancer incidence in the positive population compared to the biomarker-negative population can be calculated. 16
Development of APOA2-iTQ, an in vitro diagnostic kit to aid in the diagnosis of pancreatic cancer
Since the concept of the clinical performance of apoA2-i as a research reagent was proven, efforts were initiated to secure approval for its use as an in vitro diagnostic kit in Japan. To use apoA2-i in actual clinical settings, a clinical performance study was required, which was conducted after consulting with the Pharmaceuticals and Medical Devices Agency (PMDA) regarding the protocol. Subsequently, Toray Industries, Inc. redesigned “APOA2-iTQ”, an ELISA kit for measuring apoA2-i, to serve as an in vitro diagnostic reagent with a different configuration from that of the research reagent. A clinical performance study was conducted under QMS Ministerial Ordinance, and the kit was manufactured and tested for clinical performance in accordance with the QMS ordinance (Figure 2). 17
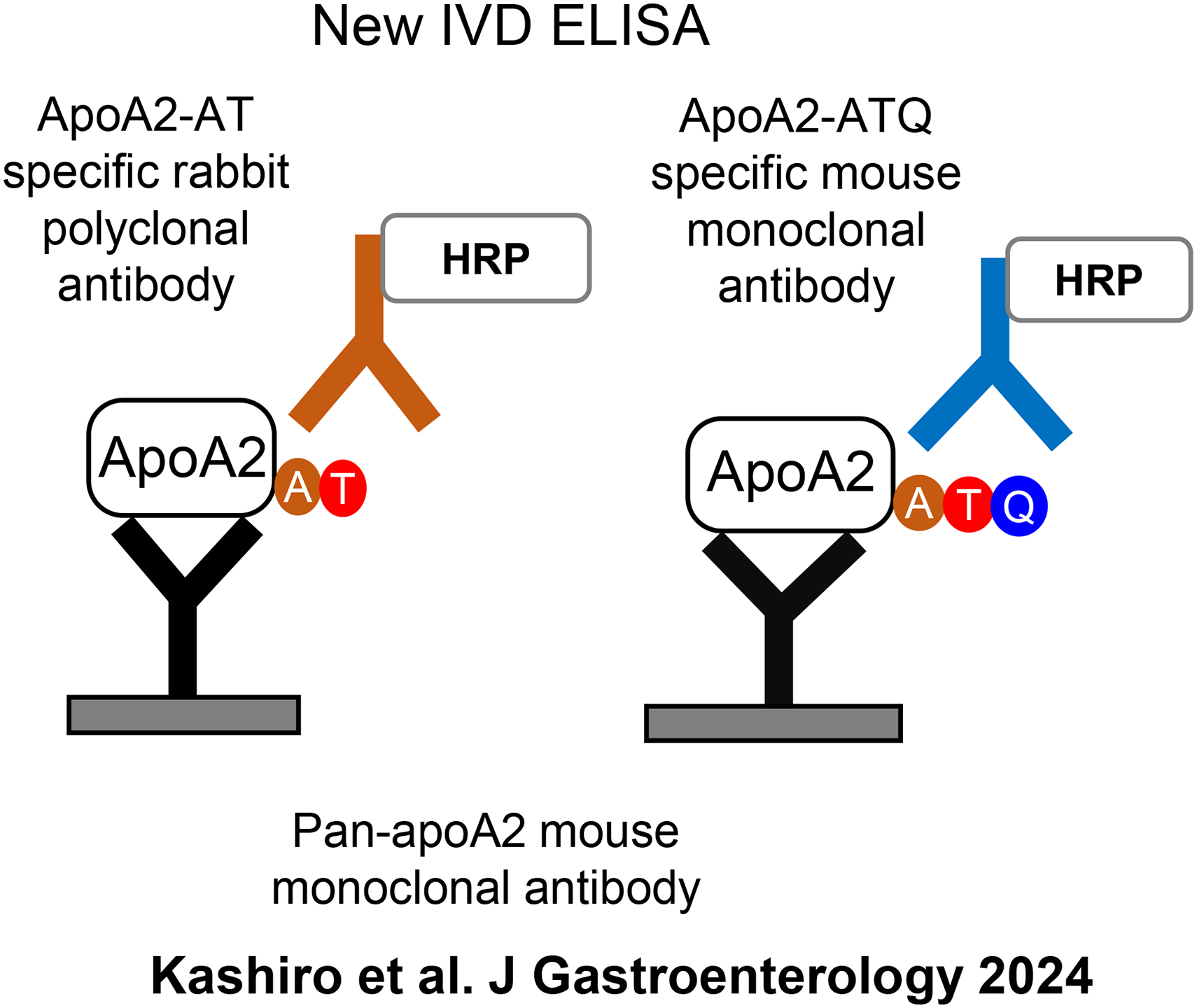
Configuration of the apoA2-i ELISA kit for in vitro diagnostics. 17
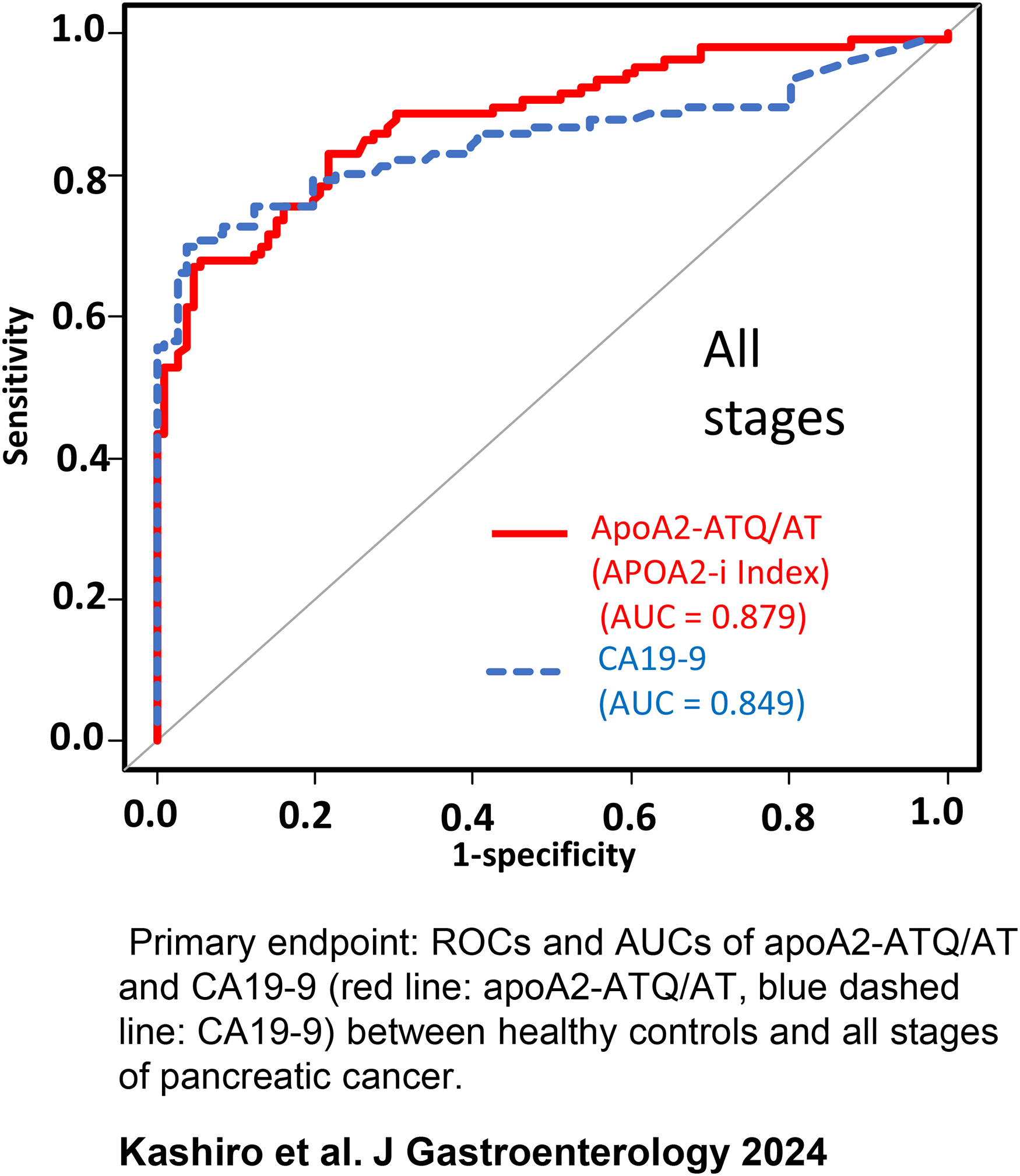
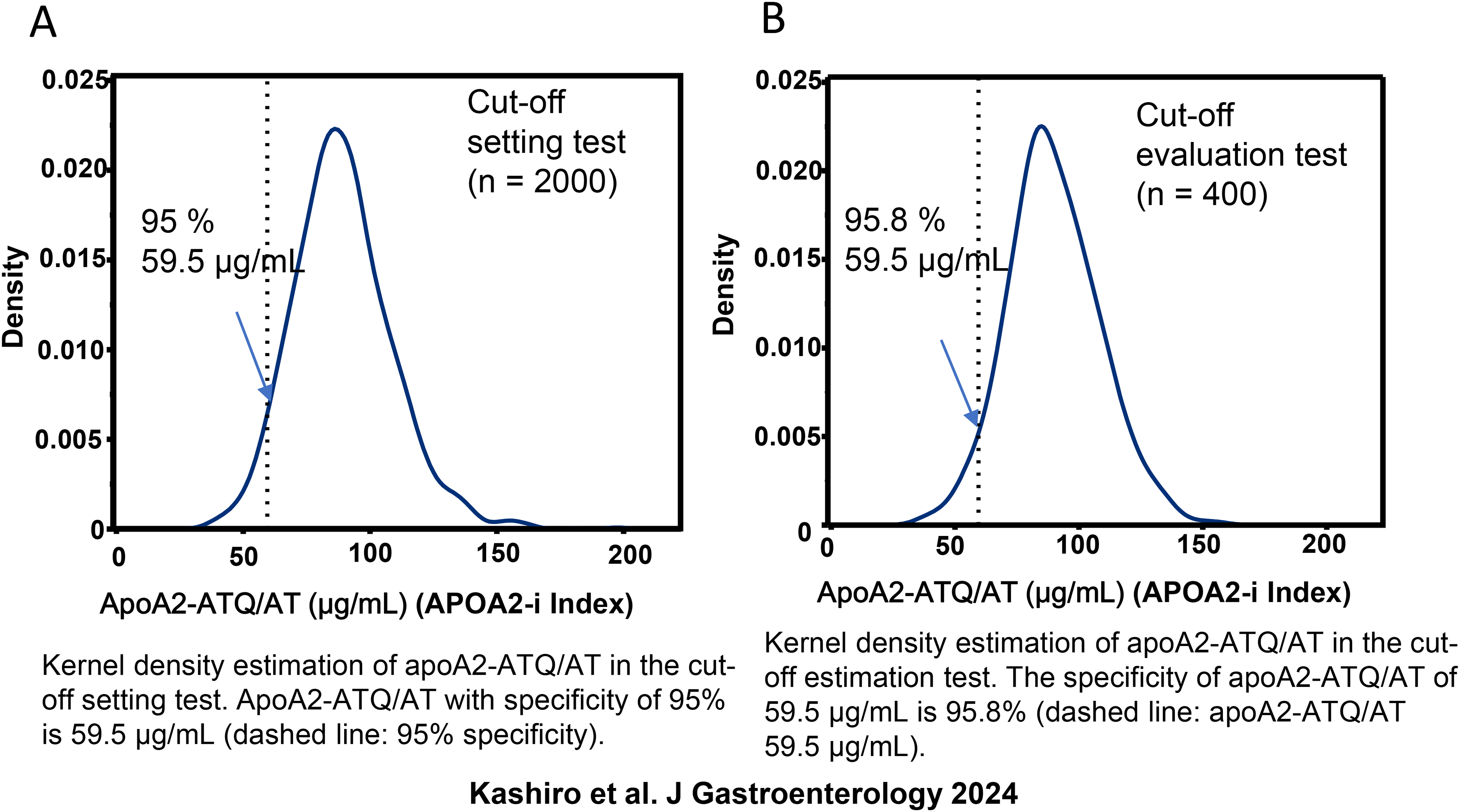
In APOA2-iTQ, the apoA2-ATQ/AT concentration was estimated as the synergistic average of apoA2-ATQ and apoA2-AT (Equation 1), as in the case of research reagents, and this result was called the “apoA2-i Index”. To establish the appropriate cut-off concentration, 2000 plasma samples from healthy individuals, collected using the same standard operating procedure (SOP), were measured with APOA2-iTQ. The cut-off value was defined as 59.5 μg/mL, which is the lower 95% point of the apoA2-i Index. The primary endpoint was the AUC point estimate to discriminate 106 pancreatic cancer patients from 106 healthy subjects. The target to be achieved was an AUC point estimate of apoA2-i Index not less than −0.05 of the AUC point estimate of CA19-9, considering that CA19-9 is used in actual clinical practice for diagnosis (Equation 2).
17
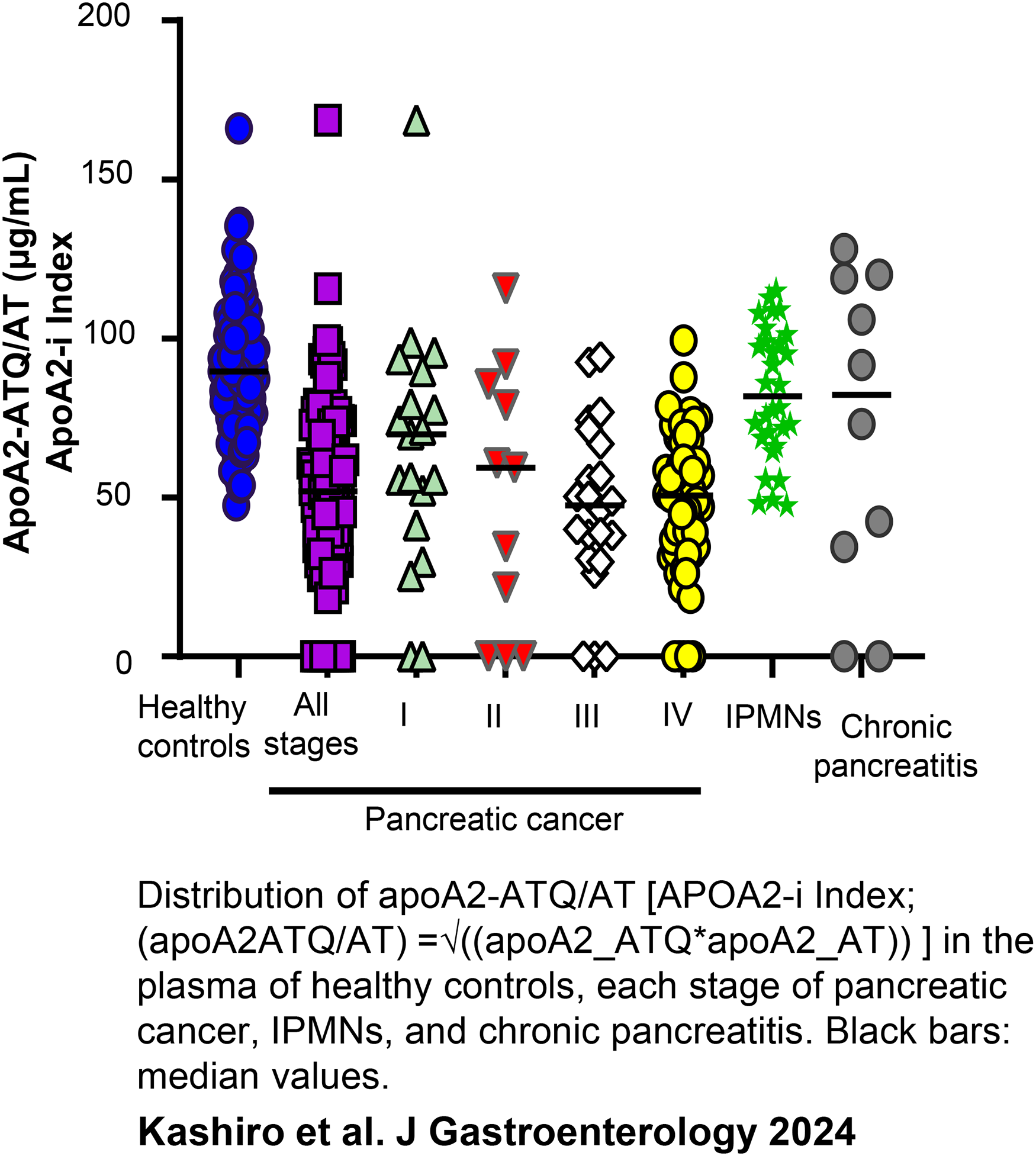
Distribution of apoA2-ATQ/AT (apoA2-i Index) in the plasma of healthy controls, each stage of pancreatic cancer, IPMNs, and chronic pancreatitis. Black bars: median values. 17
To determine the appropriateness of the previously defined cut-off values, the reproducibility of these values was assessed using 400 plasma samples from healthy controls that were not used to set the cut-off values. The points defined with a cut-off value of 59.5 μg/mL were found to be in the lower 95.8%, thus confirming reproducibility (Figure 5). 17
In the clinical performance study evaluating the primary endpoint, apoA2-i Index demonstrated a sensitivity of 63.2% in detecting pancreatic cancer in 106 healthy controls. The point estimates for sensitivity to detect Stage-I pancreatic cancer were 47.4% for APOA2-iTQ and 36.8% for CA19-9. The sensitivity to detect Stage-I/II pancreatic cancer increased to 70% when APOA2-iTQ was combined with CA19-9, compared with 46.7% for CA19-9 alone. 17
The detection performances of both the combination of apoA2-i Index and CA19-9 and the combination of CA19-9 and DUPAN2 are presented in Table 1. This table also includes the sensitivity and specificity of each biomarker for detecting pancreatic cancer in healthy individuals.
Positive rates of DUPAN2, CA19-9, apoA2-ATQ/AT, and those combination for healthy controls and pancreatic cancer.
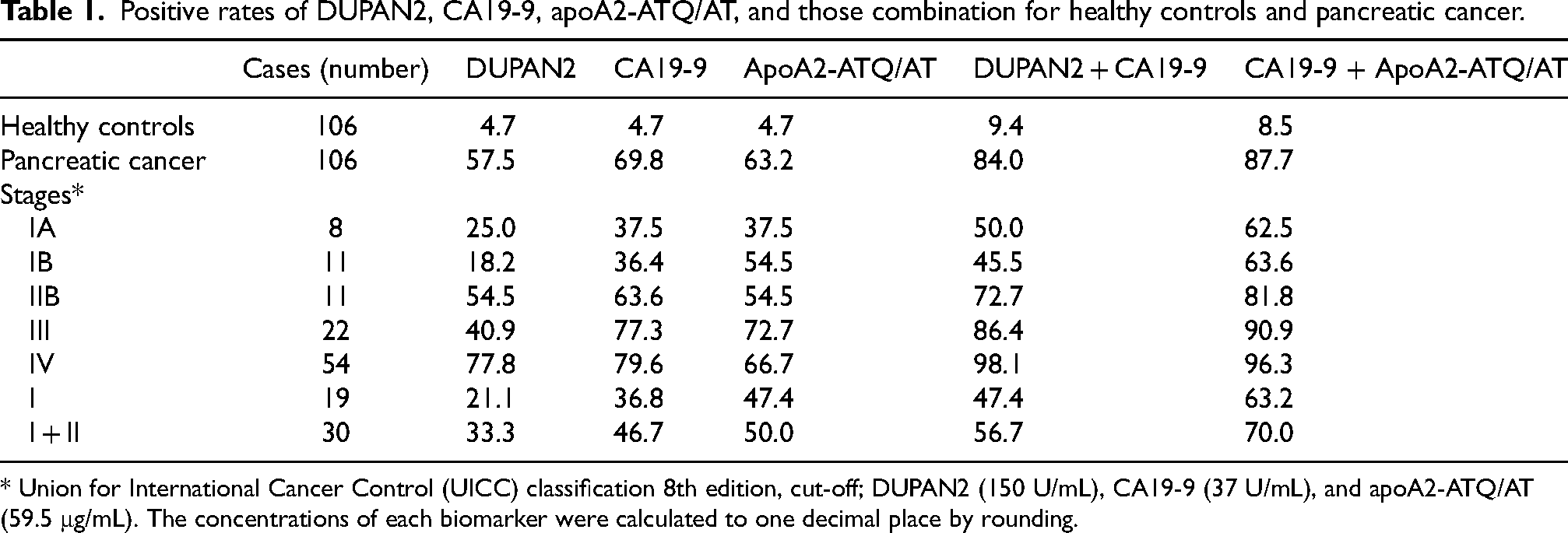
* Union for International Cancer Control (UICC) classification 8th edition, cut-off; DUPAN2 (150 U/mL), CA19-9 (37 U/mL), and apoA2-ATQ/AT (59.5 μg/mL). The concentrations of each biomarker were calculated to one decimal place by rounding.
As a result of the clinical performance trial, APOA2-iTQ received manufacturing and marketing approval in Japan in June 2023. It is now used as an in vitro diagnostic kit to aid in the diagnosis of pancreatic cancer in clinical practice.
Blind validation of APOA2-iTQ with the NCI EDRN's pancreatic cancer reference set
The clinical performance studies involving Stage-I/II pancreatic cancers were conducted with a relatively small sample size of 30 cases. However, the Early Detection Research Network (EDRN) of the National Cancer Institute (NCI) has developed reference sample sets for blinded measurement to quickly and fairly validate candidate biomarkers for early detection of cancer. 8
One of these sets, the Pancreatic Cancer Reference Set, contains 98 samples of Stage-I/II pancreatic cancer. These samples were blindly measured using APOA2-iTQ at Toray's U.S. Clinical Laboratories Improvement Amendments (CLIA)-certified clinical laboratory in Brisbane, CA. The point estimates of the AUC to discriminate Stage-I/II from healthy individuals were 0.836 (95% CI: 0.774–0.898) and 0.783 (95% CI: 0.710–0.855) for apoA2-i Index and CA19-9, respectively. The combination of apoA2-i Index and CA19-9 yielded a higher AUC of 0.897 (95% CI: 0.848–0.946). The reproducibility of the clinical performance test conducted in Japan was further confirmed by blinded validation conducted in the U.S. When the specificity for healthy individuals was set at 95.1%, the detection sensitivity for Stage-I/II (98 samples) was 60.2% for apoA2-i Index and 54.1% for CA19-9 (Table 2). 17
Blinded confirmation study for positive rates of apoA2-ATQ/AT and CA19-9 by NCI EDRN.
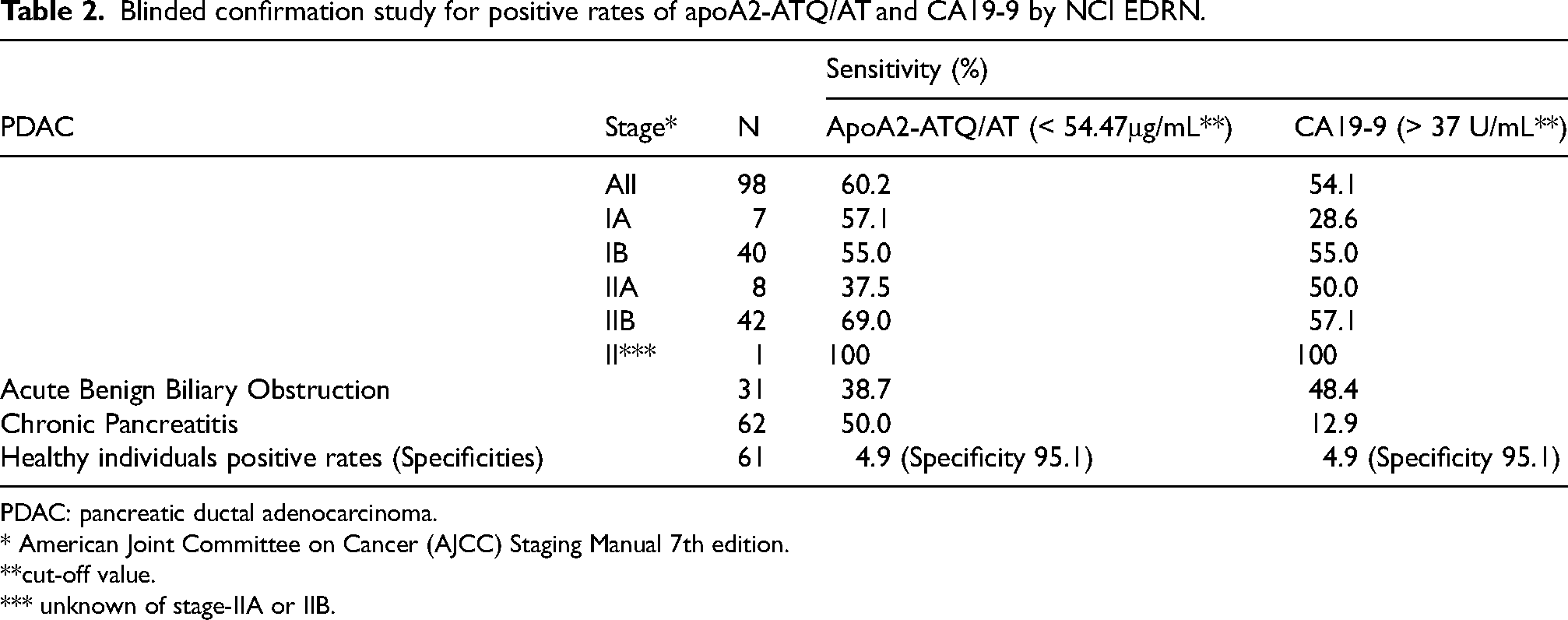
PDAC: pancreatic ductal adenocarcinoma.
* American Joint Committee on Cancer (AJCC) Staging Manual 7th edition.
**cut-off value.
*** unknown of stage-IIA or IIB.
Toward the next generation of liquid biopsy development
Recently, liquid biopsy has attracted attention. This technique involves conducting tests based on information derived from trace amounts of cells, nano-sized vesicles, nucleic acids, proteins, sugar chains, and metabolites circulating throughout the body. Notably, the development of Multi Cancer Early Detection (MCED) panels, which profile and diagnose multiple types of cancer by converting liquid biopsy information into panels, is primarily progressing in the USA. 18
However, the accumulation of genetic mutations in early-stage pancreatic cancer is low, and the contribution of MCED to early detection has not been fully explored. Unlike the conventional liquid biopsy concept, apoA2-i has a high potential to detect conditions that may lead to the development of pancreatic cancer by sensitively identifying changes in pancreatic exocrine function.
The clinical significance of apoA2-i may be similar to that of serum pepsinogen, which determines the degree of gastric mucosal atrophy, the motherland of gastric cancer, 19 and serum Mac2-binding protein glycosylated isomer (M2BPGi), a liver fibrosis serum marker. 20 The development of next-generation liquid biomarkers that are not limited to trace biomolecules leaked from cancer, but comprehensively evaluate the carcinogenic organ environment, is desirable.
Footnotes
Author contributions
Conception: K.H
Interpretation or analysis of data: A.K., J.G., and K.H.
Preparation of the manuscript: A.K., J.G., and K.H.
Revision for important intellectual content: J.G., and K.H.
Supervision: J.G., and K.H.
Funding
Some of the research described in this review was conducted with support provided by the Japan Agency for Medical Research and Development (AMED) Innovative Cancer Therapy Practical Application Research Project, AMED the Next Generation Cancer Therapy Creation Research Project, the Princess Takamatsu Cancer Research Grant, and joint research funds provided by Toray Industries, Inc.
Declaration of conflicting interests
KH and JG are the inventors of this biomarker, hold a patent for the invention, and KH serves as an advisor to Toray Industries, Inc.