Abstract
Purpose:
The Hip Worries Inventory (HWI) is an 11-item questionnaire developed to assess parental concerns related to developmental dysplasia of the hip (DDH) in early infancy. This study aimed to translate the HWI into Dutch and evaluate its psychometric properties.
Methods:
The HWI was translated using a standardized forward-backward translation process. Psychometric evaluation followed COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines and included assessments of readability, interpretability, responsiveness, reliability and content validity. Parents of infants aged 3–13 months diagnosed with DDH and managed with active surveillance or abduction bracing with a Pavlik harness were included.
Results:
The Dutch version of the HWI was completed by 59 parents prior to treatment and by 50 of them during treatment. In addition, 56 parents completed the questionnaire twice around their child’s first birthday. The Dutch HWI demonstrated good readability (Flesch Reading Ease Score = 84.1) and adequate interpretability (mean score 21.53, standard deviation 6.11). Responsiveness was supported by confirmation of 89% of the predefined hypotheses. Internal consistency was good (Cronbach’s α = 0.77–0.79) and test-retest reliability was excellent (Intraclass Correlation Coefficient = 0.95; 95% Confidence interval 0.91–0.97). Content validity assessment identified concerns regarding the relevance of items 3–6 for non-brace treatment, the sequencing of items, the use of negatively worded items and the absence of items addressing practical and informational needs.
Conclusions:
The Dutch HWI is a promising instrument for assessing parental concerns related to DDH in early infancy. However, limitations in content validity highlight the need for refinement, incorporating input from parents, caregivers and healthcare professionals to enhance content validity and clinical applicability.
Keywords
Introduction
Developmental dysplasia of the hip (DDH) is a common paediatric orthopaedic condition encompassing a spectrum of hip joint abnormalities. 1 Primary treatment in early infancy includes observation with serial ultrasound imaging (active surveillance) or abduction bracing, most commonly with a Pavlik harness.2–4 Timely diagnosis and appropriate treatment are essential to prevent hip pain, gait disturbances and premature hip osteoarthritis later in life. 4
Historically, research on DDH has focused primarily on clinical and radiological outcomes, with limited attention to the psychosocial impact. However, growing evidence indicates that the diagnosis and treatment of DDH can be stressful for parents and caregivers.5,6 Numerous studies have highlighted the multifaceted challenges parents face when caring for a child with hip dysplasia. These include insufficient medical and practical information,6–8 difficulties in parenting – particularly in bonding and the mother-child relationship during abduction brace treatment6,9–10 – as well as concerns about treatment acceptance, outcomes and future expectations.6,11 Additionally, parents frequently report emotional and psychological burdens, including feelings of being overwhelmed by the diagnosis, challenges in coping with their child’s condition and negative reactions from their surroundings regarding their child in an abduction brace.6,10,12
Despite growing recognition of these challenges, there remains a lack of validated instruments to systematically assess the impact of the diagnosis and treatment of DDH on parents. Reliable outcome measures are essential to support tailored care, improve psychosocial and practical guidance and evaluate interventions aimed at improving family-centred care.
The Hip Worries Inventory (HWI) is a questionnaire originally designed to assess parental concerns related to DDH in early infancy. 5 However, it has not yet undergone psychometric validation. This study aimed to translate the HWI into Dutch and to evaluate its psychometric properties in a cohort of Dutch parents of children with DDH during their first year of life. Based on prior qualitative research, 6 we hypothesized that the HWI would be an appropriate instrument to assess parental concerns, although it may not capture the full scope of the construct.
Methods
Study design
The study was conducted in two phases: (1) translation and cross-cultural adaptation of the HWI into Dutch and (2) validation of the psychometric properties of the Dutch HWI. The methodology adhered to the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) guidelines. 13 Approval for this study was obtained from the Institutional Review Boards (N23.050/2023-MMC-060 and MEC-2025-0122). Written informed consent was obtained from all parents.
Hip Worries Inventory
The HWI was originally developed by Gardner et al. 5 to assess maternal concerns in infants at risk for DDH. The questionnaire was evaluated among parents of both splinted and non-splinted infants at 8 weeks and 1 year of age. The original version comprised 12 items rated on a 5-point Likert scale (1 = strongly disagree to 5 = strongly agree) with higher scores indicating greater parental concern regarding the infant’s hips. One item was removed during the original development process due to poor reliability. This resulted in a final version of 11 items (Figure 1). Items 1–10 were administered when the infant was 8 weeks old, whereas items 1, 2, 8–11 were completed at 1 year of age, reflecting the varying relevance of specific concerns at different developmental stages.
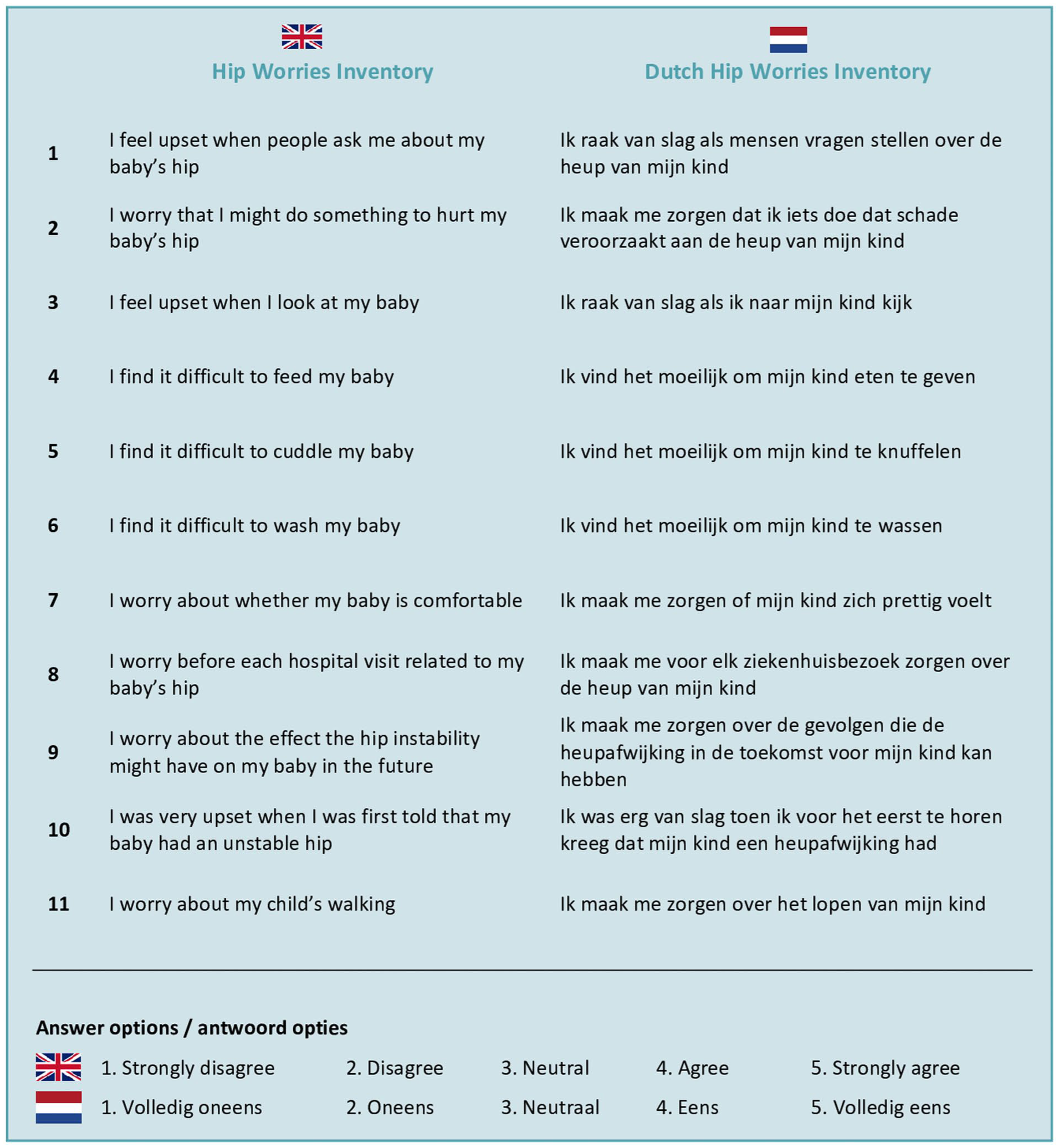
Overview of items of the HWI and Dutch HWI.
Participants
Parents were recruited from the paediatric orthopaedic outpatient clinics of the Máxima Medical Center and the Erasmus Medical Center between February 2024 and June 2025. Both mothers and fathers of children diagnosed with DDH were eligible for inclusion. Infants, aged 3–13 months, were treated with either active surveillance or a Pavlik harness. Parents with insufficient proficiency in the Dutch language were excluded.
To validate the Dutch version of the HWI, the questionnaire was administered at four timepoints during the treatment process aligned with the Dutch DDH care pathway:
A change in parental concerns was expected in the treatment phase (T1–T2), as this phase involves the diagnosis and the early experience of treatment. This period is known to evoke emotional responses, 6 which is the core construct that the Dutch HWI is intended to measure. In contrast, no major change in parental concerns was anticipated in the follow-up phase (T3–T4), as no active treatment was ongoing. An exception may be the small group of infants with residual acetabular dysplasia at 1 year of age, 14 for whom follow-up imaging and treatment may elicit renewed parental concerns. In addition, a subset of parents assessed at T1 participated in interviews to evaluate the content validity of the Dutch HWI.
Translation and cross-cultural adaptation
The translation and cross-cultural adaptation process followed the standardized multistep methodology described by Beaton et al. 15 This approach ensured both linguistic accuracy and cultural equivalence, preserving the content validity of the Dutch HWI across different populations. Two certified Dutch-native translators independently translated the original English HWI into Dutch. Two native English speakers then back-translated the Dutch version without prior knowledge of the original instrument. Discrepancies were resolved in a consensus meeting with the translators and one healthcare professional, resulting in a pre-final version. This pre-final Dutch HWI was pilot tested among five parents of children screened for DDH to assess general comprehension. In addition, the department of patient communication at the Máxima Medical Center reviewed the questionnaire and recommended linguistic adjustments. Based on the combined feedback from both parents and the communication department, the final version of the Dutch HWI was established (Figure 1 and supplemental material). This version consisted of 11 items and was scored in accordance with the original HWI on a 5-point Likert scale. Not all 11 items were administered at every time point due to developmental stage relevance. In the treatment phase (T1 and T2), items 1–10 were used (total score = 10–50 points). Item 11, assessing the child’s ability to walk, was excluded in this phase, as walking is not yet relevant in early infancy. In the follow-up phase (T3 and T4), when infants were 1 year old, items 1–3, 7–9 and 11 were included (total score = 7–35 points). This contrasts with the original HWI, in which items 3 and 7 were not administered at the 1-year follow-up, while item 10 was included.
Validation of psychometric properties
Readability
The readability of the Dutch HWI was assessed using the Flesch Reading Ease Score. 16 Scores range from 0 (academic language) to 100 (suitable for children aged 10–11 years). 16 To ensure the questions would be easily understood by most parents, a target readability score >80 points, corresponding to a sixth-grade reading level, was pursued. 17
Interpretability
Interpretability refers to the extent to which qualitative meaning can be ascribed to a quantitative score or a change in score. 18 To evaluate this, the distribution of the Dutch HWI scores prior to treatment (T1) was analyzed, focusing on the mean and standard deviation (SD). Floor and ceiling effects were considered present if >15% of the respondents scored within the lowest or highest 5% of the possible scores. 19 Interpretability was considered adequate if no floor or ceiling effects were observed.
Responsiveness
Responsiveness reflects the ability of a questionnaire to detect change over time. In the absence of a gold standard, responsiveness was assessed using hypothesis testing. 18 All hypotheses were defined a priori by an expert panel, consisting of a orthopaedic surgery registrar, paediatric orthopaedic surgeon and clinical researcher (WT, JT, MS) (Table 3). 20 Changes in Dutch HWI scores in the treatment phase (T1 and T2) were analyzed. Responsiveness was deemed adequate if at least 75% of the predefined hypotheses were confirmed. 18
Reliability
Reliability is the ability of a questionnaire to produce consistent results over time for participants who have not changed. 18 Parents who reported ‘no change in concerns’ on the anchor question at T4 were included. Reliability was assessed by evaluating internal consistency, test-retest reliability and measurement error. Internal consistency was measured using Cronbach’s α. Test-retest reliability was assessed using a two-way random effects model of the Intraclass Correlation Coefficient (ICC) for absolute agreement. Values ≥0.70 for both Cronbach’s α and ICC were considered indicative of good reliability. 21 The Standard Error of the Measurement (SEM) was calculated as: SEM = SD × √(1-reliability), where the ICC reflects the reliability. 19 The Smallest Detectable Change (SDC) was computed as: SDC = 1.96 × √2 × SEM. 22
Content validity
Content validity, defined as the extent to which an instrument adequately reflects the construct it is intended to measure, was assessed through semi-structured individual interviews with professionals and parents.18,23 Relevance, comprehensiveness and comprehensibility were evaluated according to the COSMIN guidelines. 13 Each of these domains was scored independently for both professionals and parents. A positive rating for relevance, comprehensiveness and comprehensibility was assigned if ≥85% of the items fulfilled the COSMIN criteria. Content validity was considered sufficient only if all three domains met this threshold independently. 13 Interviews were conducted by a single researcher (AI), audio-recorded, transcribed and independently analyzed by one orthopaedic surgery registrar (WT) and one researcher (AI).
Results
The Dutch version of the HWI (Figure 1) was completed by 59 parents prior to treatment (T1), of whom a representative group of 50 parents also responded during the treatment phase (T2). The follow-up group consisted of 56 parents who completed the questionnaire twice around the age of 1 year of their child. Characteristics of these groups are presented in Tables 1 and 2.
Characteristics of participants in the treatment phase (T1 and T2) for interpretability and responsiveness analysis.
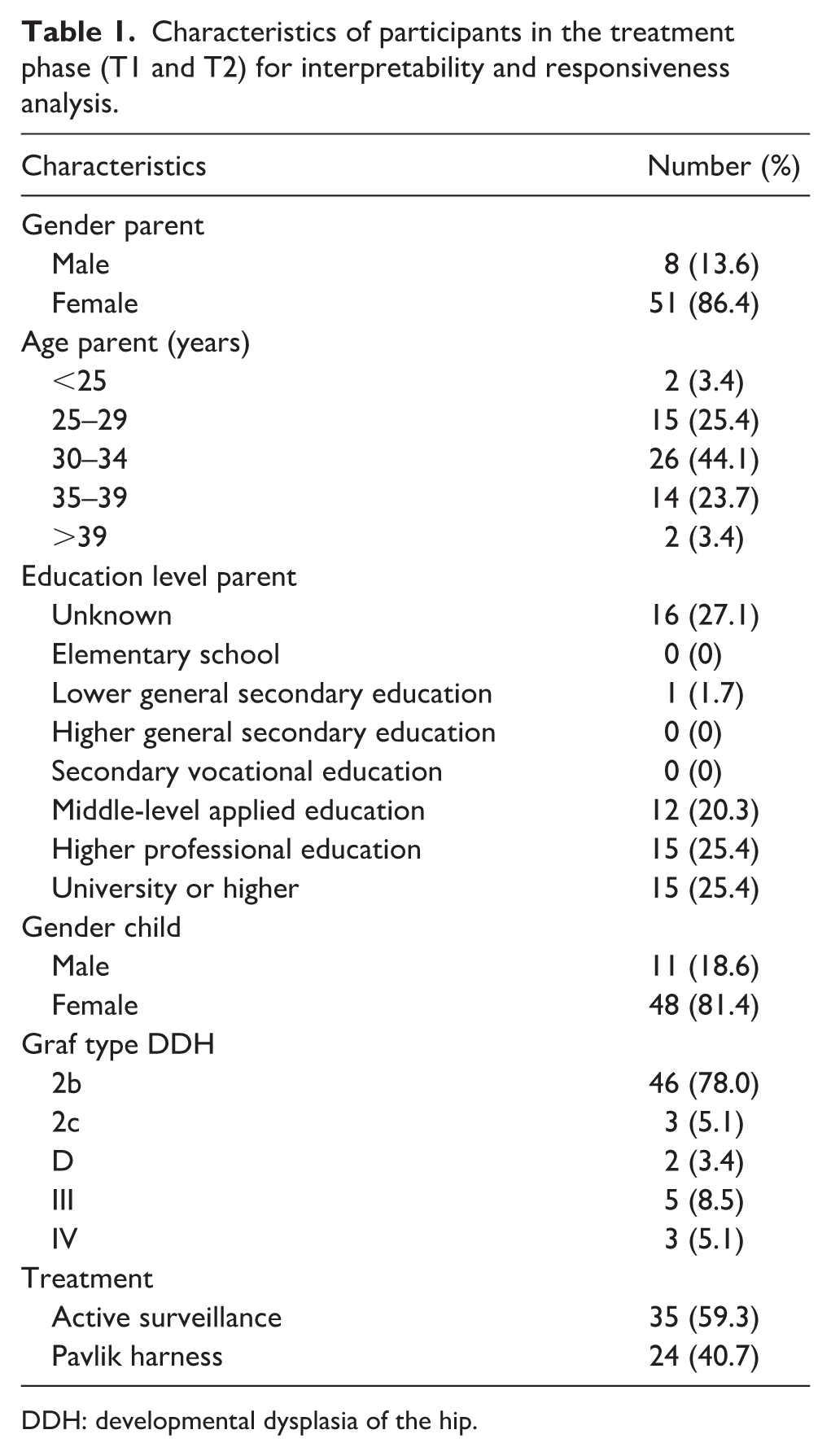
DDH: developmental dysplasia of the hip.
Characteristics of participants in the follow-up phase (T3 and T4) for reliability analysis.
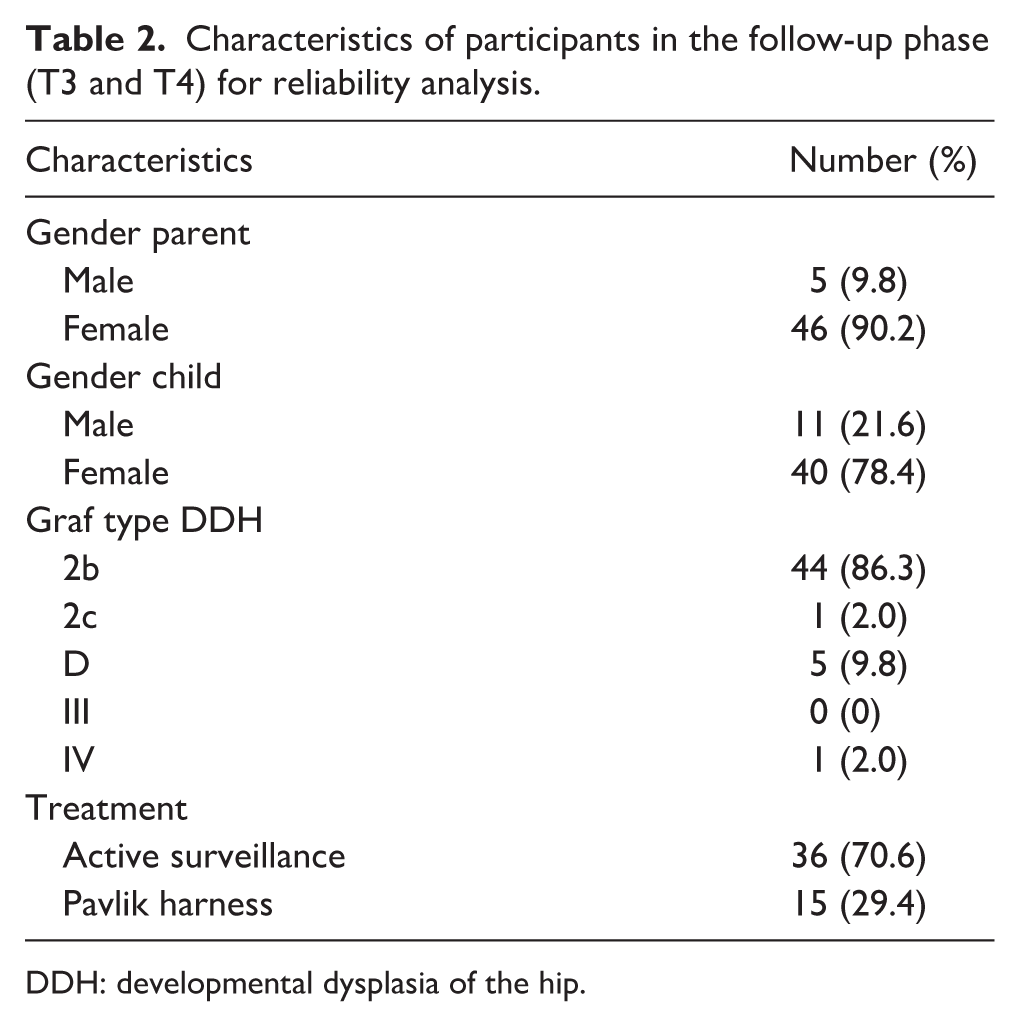
DDH: developmental dysplasia of the hip.
Readability
The Dutch HWI demonstrated good readability with a Flesch Reading Ease Score of 84.1. This corresponds to a reading level suitable for children aged 11–12 years (sixth grade) and meets recommended standards for patient-reported outcome measures (PROMs). 17
Interpretability
Interpretability was assessed in the 59 parents who completed the Dutch HWI prior to treatment (T1) (Table 1). The mean total score was 21.53 (SD 6.11), suggesting an adequate level of interpretability. No floor or ceiling effects were identified and the minimum and maximum scores were respectively 12 and 43.
Responsiveness
A total of 50 parents completed the Dutch HWI prior to treatment (T1) and during the treatment phase (T2). In response to the anchor question at T2, 21 parents reported fewer concerns, 24 parents reported no change and 5 parents reported increased concerns. Table 3 presents the results of the responsiveness analysis, including test statistics and outcomes for each predefined hypothesis. Eight of nine predefined hypotheses (89%) were confirmed, indicating that the Dutch HWI is responsive to changes in parental concerns over time.
Hypotheses for responsiveness analysis.
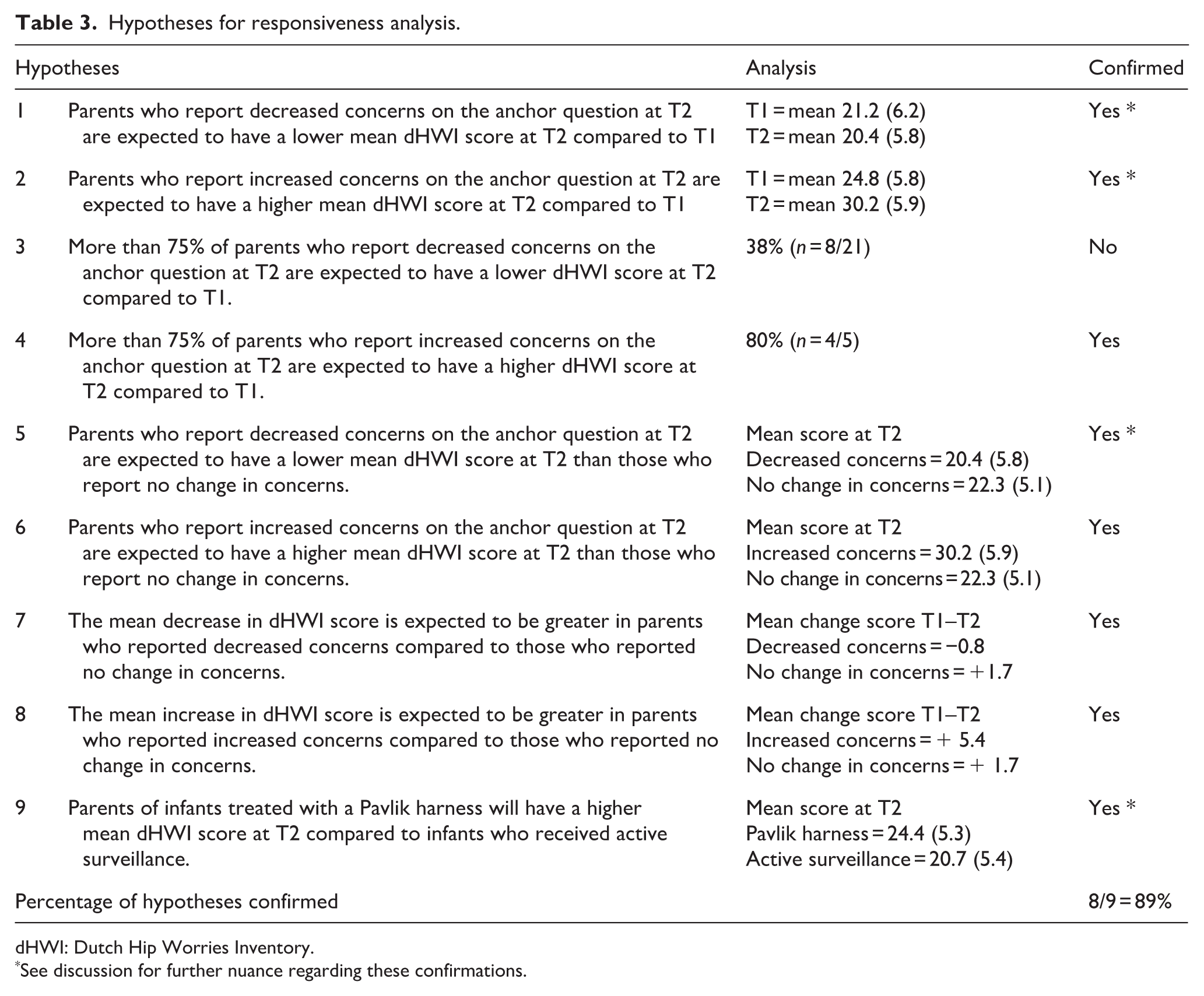
dHWI: Dutch Hip Worries Inventory.
See discussion for further nuance regarding these confirmations.
Reliability
Of the 56 parents assessed, 51 parents (91%) reported no change in concerns between the 2 time points and were included in the reliability analysis (Table 2). Internal consistency of the Dutch HWI was good with a Cronbach’s α of 0.79 at T3 and 0.77 at T4. Single item-deletion analyses showed Cronbach’s α ranging from 0.73 to 0.80 at T3 and from 0.72 to 0.78 at T4, indicating that no single item unduly influenced internal consistency. Test-retest reliability was excellent with an ICC of 0.95 (95% Confidence interval 0.91–0.97). The SEM was 1.40 and the SDC was 3.88 on a total score scale ranging from 7 to 35.
Content validity
To assess content validity, interviews were conducted with 7 healthcare professionals involved in the DDH healthcare trajectory (4 paediatric orthopaedic surgeons, 2 paediatric orthopaedic nurses and 1 youth healthcare doctor) as well as 10 parents of children with DDH. The parent group included six mothers of a child treated with a Pavlik harness, two mothers of a child managed with active surveillance and one mother and one father whose child underwent both active surveillance and Pavlik harness treatment.
Relevance
The relevance of the Dutch HWI was rated positive by 71% (5/7) of professionals and 60% (6/10) of parents. Several items were perceived as less relevant when active surveillance was used as the treatment strategy. Specifically, both professionals and parents considered item 3 (professionals 43% and parents 50%), item 4 (professionals 43% and parents 70%), item 5 (professionals 14% and parents 30%) and item 6 (professionals 43% and parents 50%) only relevant in the context of treatment with an abduction device. Consequently, both professionals and parents recommended restricting these items solely to situations involving treatment with a Pavlik harness or other abduction device. Furthermore, concerns were raised about the sequencing of items. Two professionals recommended repositioning item 10 at the beginning of the questionnaire; noting that item 1 of the Dutch HWI may be overly emotionally charged for an introductory item. In addition, several parents reported that item 10 is most relevant immediately after diagnosis and loses relevance in later stages of follow-up.
Comprehensiveness
A positive assessment of the comprehensiveness was reported by 43% (3/7) of professionals 50% (5/10) of parents. Professionals reported that the Dutch HWI lacked content in several key areas, including parental worries regarding: access to relevant information on DDH, practical problems for daily management, a potential need for surgical intervention and the effect on the development of the child in the long-term. On the other hand, parents identified missing content in the Dutch HWI on: access to relevant information on DDH, impact on child’s sleep and impact on child’s clothing.
Comprehensibility
The comprehensibility of the Dutch HWI was rated positively by 71% (5/7) of professionals and 70% (7/10) of parents. The main concern among both groups related to negatively worded items, particularly those beginning with phrases such as ‘I worry’ or ‘I find it difficult’.
Discussion
This study assessed the psychometric properties of the Dutch version of the HWI, an 11-item questionnaire designed to evaluate parental concerns in the context of DDH in early infancy. The instrument demonstrated good readability and acceptable levels of interpretability, responsiveness and reliability. Nonetheless, limitations in content validity were identified, indicating that further refinement is necessary to ensure the instrument captures the full spectrum of parental concerns.
Research in DDH has predominantly focused on clinical and radiological outcomes. While essential for guiding treatment decisions, such outcomes fail to reflect the broader psychosocial impact of DDH on parents and caregivers. Emerging evidence suggests that the diagnosis and treatment of DDH can cause substantial parental stress, including uncertainty about treatment outcomes, disruption of daily life and emotional burden.5–12 Treatment with abduction braces, such as the Pavlik harness, may introduce additional challenges, including limited infant mobility, interruption of parent-child bonding and concerns about comfort and care.6,8,9 Despite these challenges, there remains a scarcity of validated instruments to assess the psychosocial burden in DDH care experienced by parents. The most frequently used PROMs in paediatric orthopaedic practice are generic health-related quality of life instruments, such as the Infant and Toddler Quality of Life Questionnaire (ITQOL), 24 TNO-AZL (Nederlandse Organisatie voor toegepast-natuurwetenschappelijk onderzoek - Academisch Ziekenhuis Leiden) Preschool children Quality of Life questionnaire (TAPQOL) 25 and Paediatric Quality of Life Inventory (PedsQL). 26 These instruments are time-consuming (97, 43 and 23 items respectively) and lack specificity to the context of DDH. This highlights the growing need for a DDH-specific, psychometrically validated instruments that can accurately capture the parental concerns in DDH care. Such a tool could support paediatric orthopaedic surgeons in recognizing and addressing parental needs in clinical care and facilitate research into interventions aimed at reducing caregiver burden.
The original HWI underwent limited psychometric evaluation, demonstrating good internal consistency (Cronbach’s α = 0.83 and 0.85) and moderate stability over time (r = 0.58). Essential psychometric properties, including readability, interpretability, responsiveness, reliability and content validity, were not addressed. In the present study, the Dutch HWI was extensively tested across the full range of psychometric properties. Readability was high, supporting its use among parents with varying levels of health literacy. 17 Interpretability was adequate, with a mean total score of 21.53 (SD 6.11), indicating a wide range of responses. The absence of floor and ceiling effects suggests that the Dutch HWI captures a broad spectrum of parental concerns without clustering at either end of the scale. With regard to reliability, internal consistency of the Dutch HWI at 1 year of age (Cronbach’s α = 0.77) was marginally lower than that of the original version. This difference may be attributed to variations in item selection, as items were chosen based on their relevance to the specific developmental stage within the Dutch DDH care pathway. The Dutch HWI retained items 3 and 7, which were excluded in the original version, while omitting item 10 due to limited relevance at 1 year of age. Test-retest analysis supports the stability of the Dutch HWI among parents whose concerns remained unchanged over time. Although the original HWI has not been externally validated, it has been used in subsequent research. Notably, Hoffer et al. 27 reported significantly higher HWI scores among parents of infants with DDH treated with a Pavlik harness compared with those whose infants were only screened but not treated.
Responsiveness analysis demonstrated that 89% of the predefined hypotheses were confirmed, indicating good responsiveness of the Dutch HWI. The outcome of hypothesis 9 aligned with the findings from the original HWI, which also showed that parental concerns were greater in the abduction brace group compared to the active surveillance group. This likely reflects the practical challenges associated with brace treatment, which complicates daily care and serves as a constant visual reminder of the condition. Furthermore, being informed that their child requires active treatment may further elevate parental concerns. 5 However, the 89% hypothesis confirmation rate warrants cautious interpretation. Although hypotheses 1, 2, 5 and 9 (Table 3) were confirmed, the absolute differences in mean scores between groups were small and remained within one SD. This suggests that, despite confirmation of these hypotheses, these changes may not reflect clinically meaningful differences in parental concerns. As such, the responsiveness of the Dutch HWI should be interpreted with care, particularly in settings where minor score variations may not translate into relevant clinical changes in parental concerns. Nonetheless, the value of the Dutch HWI extends beyond dectecting changes in overall scores; it may also facilitate the identification of specific areas of parental concern that can be targeted during follow-up consultations.
Despite the overall strengths of the Dutch HWI, the content validity was rated as insufficient. Treatment strategies for DDH in general range from active surveillance and abduction bracing to more invasive interventions such as closed or open surgical reduction and pelvic osteotomies.2,3 The original HWI was developed and the Dutch version of the HWI was validated exclusively in infants managed with active surveillance or abduction bracing. 5 Items 3–6, which address practical challenges associated with abduction bracing, were frequently judged as irrelevant by parents whose children were not treated with a Pavlik harness. This raises concerns regarding the instrument’s ability to adequately capture parental concerns in cases managed by non-bracing strategies, such as active surveillance. Moreover, the applicability in surgically treated infants was not assessed, further limiting its generalizability across the full spectrum of DDH management.2,3 Concerns were also raised about item sequencing, with the first question perceived as confronting and potentially distressing for parents. This is known as the ‘order effect’, which refers to the phenomenon that the sequence of items may influence how respondents interpret and respond to subsequent items. 28 Both healthcare professionals and parents also identified several relevant domains not addressed by the Dutch HWI. These include concerns related to information provision, infant sleep and clothing, the possibility of future surgery and long-term developmental outcomes and milestones. Previous studies have highlighted the significance parents place on these topics.5–12 These omissions suggest that the Dutch HWI may not fully reflect the multidimensional impact of DDH and its treatment on parents and caregivers. Finally, concerns were also raised regarding the negatively worded items, which may place disproportionate emphasis on the adverse aspects of DDH treatment. This negative framing may introduce response bias, potentially influencing parental responses and distorting the overall interpretation of the results.29,30
This study has several limitations. First, content validity was assessed at the final stage of the validation process, which precluded revision of the Dutch HWI items in response to parental feedback. Second, most respondents were mothers, potentially limiting the generalizability of findings to the broader parental population. However, given that mothers are typically the primary respondents in clinical practice, 31 the current validation may still be appropriate for this group. Nonetheless, concerns particularly relevant to fathers may be underrepresented, a limitation commonly noted in questionnaire-based research. 32 Finally, the Dutch HWI focuses exclusively on parental concerns in the first year of life and does not capture worries related to long-term outcomes or concerns of parents whose children underwent surgical treatment for DDH.
The findings of this study indicate that the Dutch HWI may benefit from further refinement. The relevance of individual items should be reconsidered in relation to treatment modality, as parental concerns during active surveillance may differ substantially from those experienced during abduction brace treatment or surgical intervention. One potential approach is the development of a modular instrument, comprising a general core section and treatment-specific modules. This would enable parents to complete only those items relevant to their child’s clinical context, thereby reducing response burden and improving relevance.33,34 Recently, a novel PROM was developed for infants with DDH: Evaluation Measure for BRACe Experience (EMBRACE). 35 Specifically designed for parents of infants undergoing abduction brace treatment, EMBRACE facilitates the collection of more holistic outcomes by incorporating the caregiver perspective and aligning with a family-centred core outcome set. 36
The Dutch version of the HWI is a promising step toward a standardized instrument for assessing parental concerns in children with DDH in the Netherlands. It demonstrated favourable psychometric properties in terms of readability, reliability, interpretability and responsiveness. Limitations in content validity highlight the need for further refinement to ensure the instrument captures the full spectrum of parental concerns across the DDH treatment trajectory. Future iterations of the Dutch HWI should incorporate feedback from parental, caregivers and healthcare professionals to enhance its comprehensiveness and clinical applicability.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251411734 – Supplemental material for Assessing parental concerns in developmental dysplasia of the hip in early infancy: Validation of the Dutch Hip Worries Inventory
Supplemental material, sj-pdf-1-cho-10.1177_18632521251411734 for Assessing parental concerns in developmental dysplasia of the hip in early infancy: Validation of the Dutch Hip Worries Inventory by Wesley W.E.S. Theunissen, Jaap J. Tolk, Annika B. Ito, Britt J.M.H. Dörssers, Hesther A. Oldenkamp, Evi Van Heeswijk, Florens Q.M.P. Van Douveren and Maria C. Van der Steen in Journal of Children's Orthopaedics
Footnotes
Author contributions
WT: data analysis, data collection, funding acquisition, study design, writing original manuscript
JT: data collection, study design, writing review and editing
AI: data analysis, data collection, writing review and editing
BD: data collection, writing review and editing
HO: data collection, writing review and editing
EvH: data collection, writing review and editing
FvD: data collection, writing review and editing
MvdS: data analysis, funding acquisition, study design, writing review and editing
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Commission Research and Innovation of the Máxima Medical Center to cover the translation costs (grant code: COI 2023.coi.007).
Ethical statement
Ethical board: Institutional Review Boards from Máxima Medical Center (Veldhoven, the Netherlands) and Erasmus MC (Rotterdam, the Netherlands).
Approval numbers: N23.050/2023-MMC-060 (Máxima MC) and MEC-2025-0122 (Erasmus MC).
We confirm that parents gave written informed consent
Data availability statement
The dataset generated and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
