Abstract
Purpose:
To assess ultrasonographic features of hip morphology in mucopolysaccharidosis type I Hurler.
Methods:
Acetabular bony rim, acetabular cartilaginous roof, alpha and beta angles, echogenicity, and hip coverage were analyzed in eight mucopolysaccharidosis type I Hurler syndrome children before and after hematopoietic stem cell gene therapy.
Results:
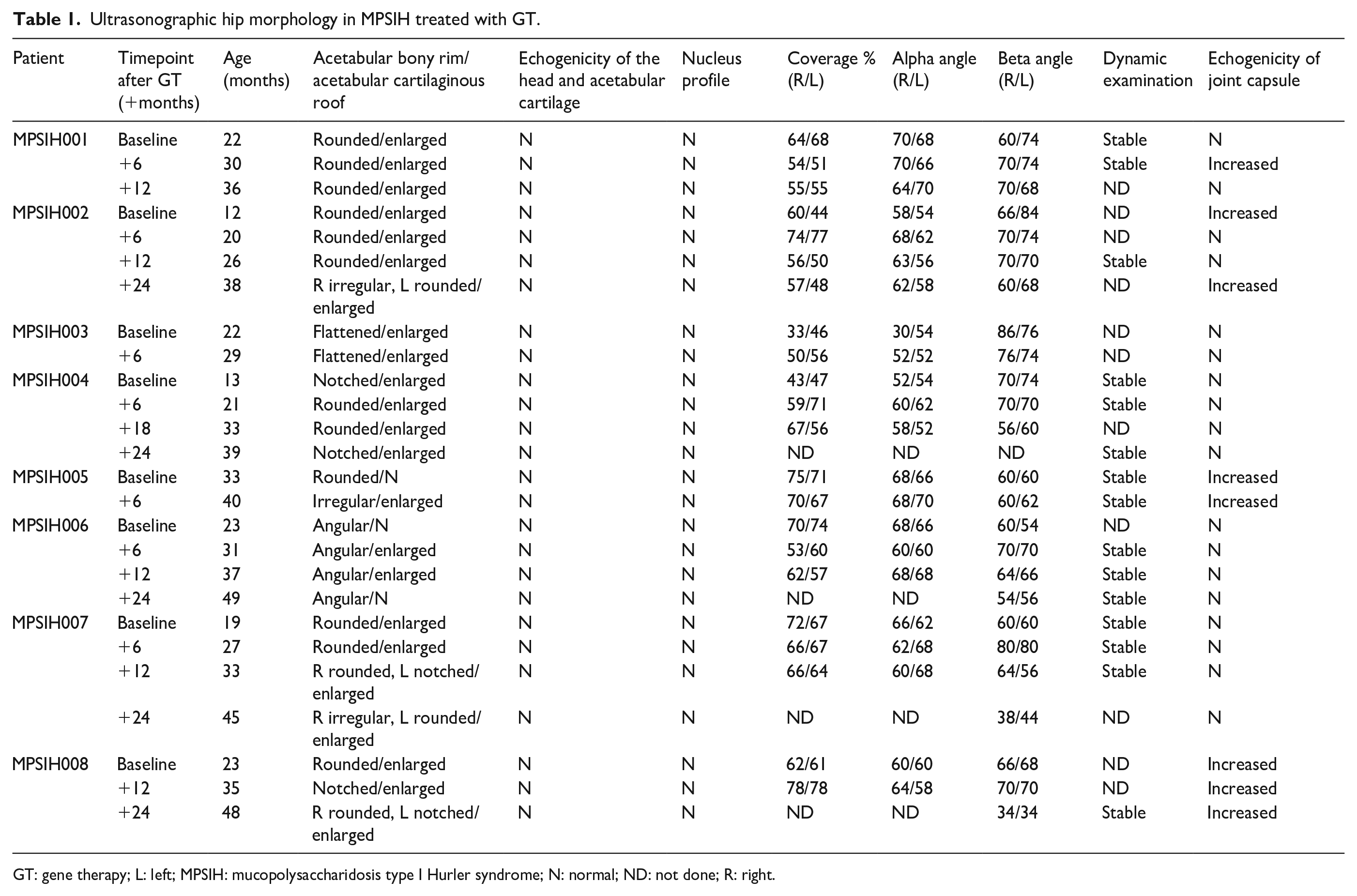
Sixteen hips at baseline, 10 at +12 months, and 10 at + 24 months after hematopoietic stem cell gene therapy were evaluated. The median age was 22, 35, and 45 months at baseline evaluation, +12, and +24 months, respectively. Acetabular bony rim at baseline was angular in 2/16, rounded in 10/16, notched in 2/16, and flattened in 2/16; at +12 months, angular in 2/10, rounded in 5/10, notched in 3/10; at +24 months, angular in 2/10, rounded in 3/10, irregular in 1/10, and notched in 4/10. Acetabular cartilaginous roof at baseline was normal in 4/16, enlarged in 12/16; at +12 months, enlarged in 10/10 and at +24 months, enlarged in 8/10 and normal in 2/10. Echogenicity of the joint capsule at baseline was normal in 10/16, increased in 6/16; at +12 months, normal in 8/10, increased in 2/10; at +24 months, normal in 6/10, increased in 4/10. The mean femoral head coverage was 60% at baseline (16/16), 62% at +12 months (10/10), and 52% at +24 months (2/10). The mean alpha angle was 60° at baseline (16/16), 64° at +12 months (10/10), and 60° (2/10) at +24 months. The mean beta angle was 67° at baseline (16/16), 65° at +12 months (10/10), and 49° at +24 months (8/10).
Conclusions:
Hip morphology of children with mucopolysaccharidosis type I Hurler syndrome before and after hematopoietic stem cell gene therapy can be evaluated by available ultrasound techniques until a median age of 45 months.
Introduction
Mucopolysaccharidosis type I Hurler syndrome (MPSIH) is a rare autosomal recessive lysosomal storage disorder characterized by alpha-L-iduronidase (IDUA) deficiency with subsequent accumulation of glycosaminoglycans (GAG) in cells and tissues1 –3 and multiorgan clinical manifestations. GAG storage in the skeleton is associated with a severe and progressive skeletal dysplasia, which is characterized by dorso-lumbar kyphosis, odontoid hypoplasia, genu valgum, and hip dysplasia (HD). HD represents one of the most prevalent skeletal features in MPSIH patients.4 –7
Morphological changes such as bony roof deficiency (acetabular dysplasia), hip dislocation, and femoral head deformity are reported with increasing age in MPSIH.6 –8 The region of interest is the area of the bony and cartilaginous roof, which represents the bone-cartilage interface in physiological hip development and pathological hips affected by HD. In MPSIH, GAG accumulation in the growth plate is responsible for abnormal growth of the acetabulum, both osseous and cartilaginous.9,10 Allogeneic hematopoietic stem cell transplantation (HSCT) and enzyme replacement therapy (ERT) represent the standard of care for MPSIH, although ineffective on the progression of skeletal manifestations, including HD.4,6 –8,11
A novel strategy based on genetic engineering of autologous HSC to drive supranormal IDUA activity that is distributed to affected tissues through the turnover of resident hematopoietic cells was designed to treat eight MPSIH children in a phase I/II clinical trial. Autologous ex vivo HSC gene therapy (HSC-GT, also known as OTL-203; NCT03488394) showed preliminary evidence of metabolic detoxification (i.e. clearance of GAGs) 12 and early beneficial effect on typical skeletal features in terms of clinical, functional, and radiological parameters. 13
While hip morphology has been previously investigated in HSCT and ERT-treated MPSIH patients by X-ray and magnetic resonance imaging (MRI),4,7,14 only one article reported data of ultrasound (US) evaluation. 15
This prospective study aimed to evaluate hip morphology in MPSIH children before and after HSC-GT using available US techniques.
Materials and methods
Eight MPSIH children (six males and two females; mean age ± SD at treatment: 1.9 ± 0.5 years) were enrolled in a phase I/II, non-randomized, single-center study (NCT03488394, EudraCT 2017-002430-23) 12 and treated with HSC-GT between July 2018 and December 2019.
Autologous HSCs were collected by leukapheresis on two consecutive days after mobilization from the bone marrow niche with granulocyte colony-stimulating factor and plerixafor. CD34+-enriched cells were transduced with a lentiviral vector encoding human IDUA complementary DNA under the control of the human phosphoglycerate kinase 1 gene promoter, and the cells were cryopreserved. The drug product was infused intravenously after conditioning regimen based on rituximab (375 mg/m2 of body-surface area on day −15), busulfan (from days −5 to −2, single daily intravenous dose starting with 80 mg/m2, then adjusted to a myeloablative total area-under-the-curve target of 85,000 μg × h/L based on daily therapeutic drug monitoring), and fludarabine (40 mg/m2 intravenously from days −5 to −2).
After HSC-GT, patients were followed up at specific time points: +30, +60, +90, +180 days, +9 months, +1 year, and yearly. Safety and efficacy assessments as per protocol were performed.
No conservative orthopedic treatment for HD was performed on the patients.
Hip morphology was studied with US at baseline (first evaluation after enrollment of the patient), 6, 12, and 24 months after HSC-GT by the same pediatric orthopedic surgeon (MDP) experienced in US technique and expertise in osteochondrodysplasias, HD, and rare skeletal diseases. Hip US was performed according to the Graf technique 16 with patients positioned in an examination cradle using Siemens Acuson X300 with a multifrequency VF8-3 linear Transducer (manufactured by Siemens Healthcare GmbH, Henkestr. 127, 91052 Erlangen, Germany).
Ultrasonographic features of hip joints in MPSIH children before and after HSC-GT were studied after identification of the anatomical landmarks (Figure 1). The following aspects were qualitatively assessed: acetabular bony rim and acetabular cartilaginous roof according to Graf terminology, 16 echogenicity of the femoral head and the acetabular cartilage, and femoral head nucleus presence. Hip coverage percentage was assessed according to Morin et al. (Figure 2(a)), knowing that in a normal hip, the acetabulum should cover at least 50% of the femoral head. 17 Alpha and beta angles were measured according to Graf’s method (Figure 2(b)). The alpha angle quantifies the bony roof and is considered normal if >60°.16,18 Beta angle represents a parameter for the cartilaginous roof, as it quantifies the cartilaginous component of the acetabulum roof: the smaller the angle, the smaller the cartilaginous components of the acetabulum. Considering physiological variability of hip morphology, the beta angle is considered normal if below 70°.16,18
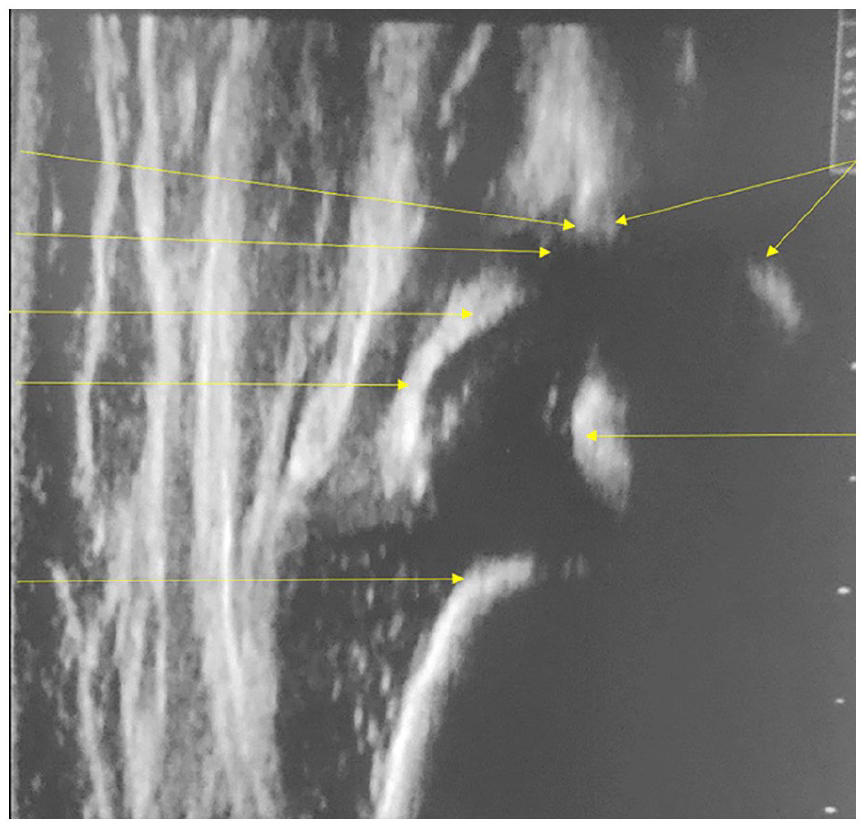
Ultrasonographic hip image in a 6-month old healthy child. Anatomical landmarks according to the Graf technique. 16
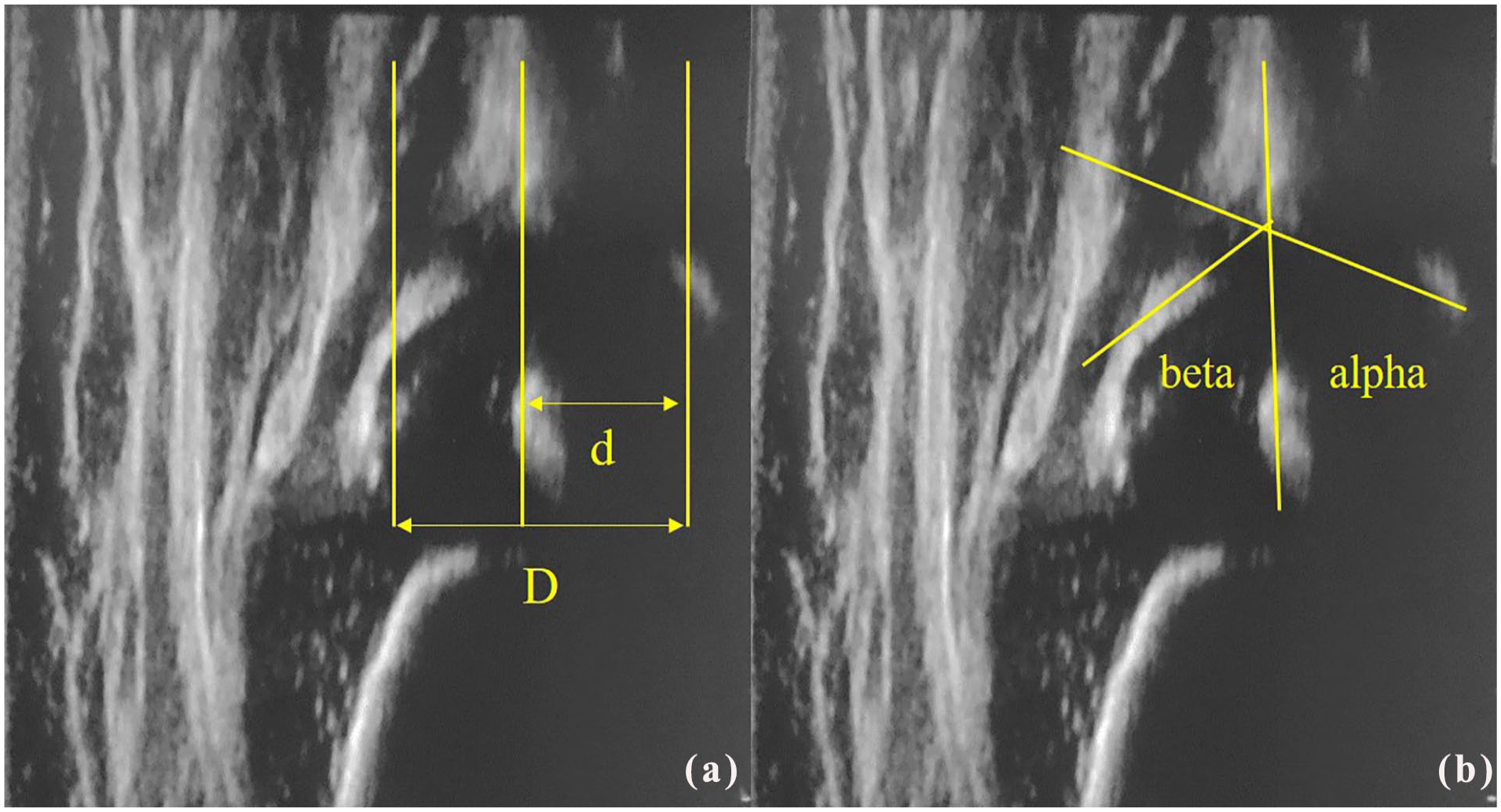
Echogenicity of the joint capsule, with respect to the gain, which maintained the same calibration in all US examinations, and hip stability by dynamic examination were evaluated.
In those patients at +24 months where the large dimension of the nucleus prevented the visualization of the inferior margin of the ilium in the fossa acetabuli, the US slice with the larger femoral head dimension in the standard plane, with the labrum clearly visible, was chosen. In these cases, only the bony rim and the cartilaginous roof were evaluated, and only the beta angle was measured.
Results
We evaluated 16 hips at baseline, 10 hips at +12 months, and 10 hips at + 24 months after HSC-GT. One patient (MPSIH003) showed hip dislocation before treatment and was evaluated only up to 6 months after HSC-GT, confirming hip dislocation. Thereafter, this patient underwent surgical correction in her country of origin and was excluded from this analysis. Two patients (MPSIH004 and MPSIH005) did not perform +12 months after HSC-GT follow-up evaluation due to the SARS-CoV2 pandemic lockdown; one of them (MPSIH004) was evaluated at +18 and +24 months after HSC-GT, while the other (MPSIH005) missed all three examinations due to inability to travel to the study site. One patient (MPSIH001) did not evaluate at +24 months after HSC-GT due to poor compliance.
Patient’s median age at baseline was 22 months (range 13–33 months), at +12 months after HSC-GT 35 months (range 26–37 months), at +24 months was 45 months (range 38–49 months).
At baseline, the acetabular bony rim showed angular in 2/16, rounded aspect in 10/16 hips, notched in 2/16, and flattened in 2/16, while at +12 months after HSC-GT was angular in 2/10 hips, rounded in 4/10, notched in 2/10, rounded on one side and notched on the other in one patient, at +24 months angular in 2/10, rounded on one side and irregular on the other side in two patients, notched on one side and rounded on the other in one patient, notched in 2/10. We used the term “irregular” to describe the profile of an acetabular bony rim showing an irregularity and interruption of the echogenic bony surface (Figure 3). The term “notched” was used to describe the acetabular bony rim with lateral bone deficiency visualized as a “step.” 19
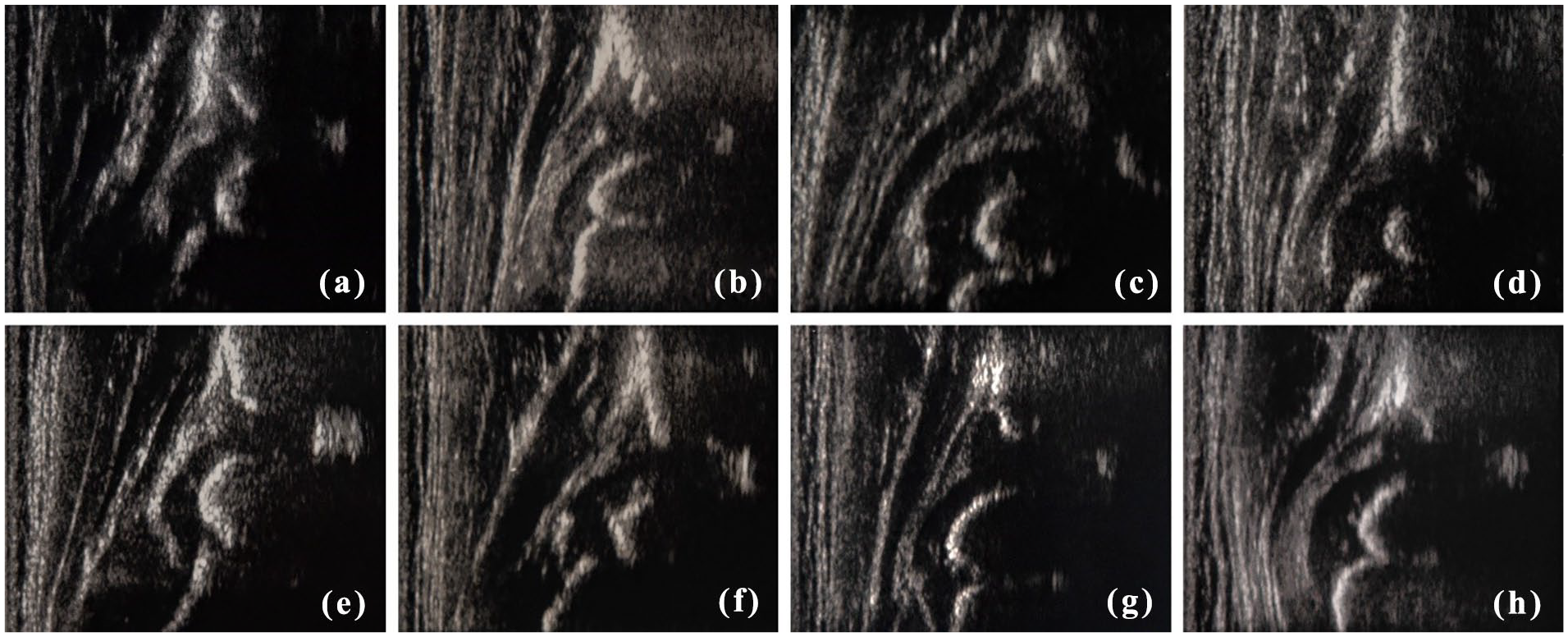
Ultrasonographic hip images of all patients at different timepoints and ages with morphological features according to Table 1 terminology. (a) MPSIH001: left hip, baseline, 22 months old, rounded acetabular bony rim and enlarged roof cartilage. (b) MPSIH002: right hip, +24 months, 38 months old, irregular acetabular bony rim, enlarged roof cartilage, and increased capsule echogenicity. (c) MPSIH003: right hip, baseline, 22 months old, flattened acetabular bony rim and enlarged roof cartilage. Dislocated femoral head. (d) MPSIH004: left hip, baseline, 13 months old, notched bony rim and enlarged roof cartilage. (e) MPSIH005: left hip, baseline, 33 months old, rounded acetabular bony rim and increased capsule echogenicity. (f) MPSIH006: right hip, +6 months, 31 months old, angular acetabular bony rim and enlarged roof cartilage. (g) MPSIH007: left hip, +12 months, 33 months old, rounded acetabular bony rim and enlarged roof cartilage. (h) MPSIH008: left hip, +12 months, 35 months old, notched acetabular bony rim, enlarged roof cartilage, and increased capsule echogenicity.
As described above, hip symmetry was present in four children, while in the remaining four patients, asymmetrical features of the hips were present (MPSIH007 at +12 months after HSC-GT and MPSIH002, MPSIH007, and MPSIH008 at +24 months) without clear pathological features.
Acetabular cartilaginous roof showed normal aspect in 4/16 hips and enlarged in 12/16 at baseline, while it was enlarged in 10/10 at +12 months and normal in 2/10 and enlarged in 8/10 at + 24 months after HSC-GT.
Echogenicity of the femoral head and the acetabular cartilage was normal in all patients at baseline and at the subsequent timepoints up to +24 months after HSC-GT.
The femoral head nucleus was present in all 16/16 hips at baseline and also at all assessments post-HSC-GT. Moreover, it showed a normal, regular lateral bone profile.
The femoral head coverage showed an average value of 60% at baseline (SD ± 13%) and 62% (SD ± 9%) at +12 months after HSC-GT. It was measurable only in one patient (MPSIH002) at +24 months after HSC-GT and was 52% (SD ± 4%; Table 1).
Ultrasonographic hip morphology in MPSIH treated with GT.
GT: gene therapy; L: left; MPSIH: mucopolysaccharidosis type I Hurler syndrome; N: normal; ND: not done; R: right.
Alpha angle showed an average value of 60° (SD ± 10°) at baseline and of 64° (SD ± 4°) at +12 months after HSC-GT. It was measurable only in one patient at +24 months after HSC-GT and was 60° (SD ± 2°; Table 1).
Beta angle showed an average value of 67° (SD ± 9°) at baseline, 67° (SD ± 4°) at +12 months, 49° (SD ± 12°) at +24 months after HSC-GT (Table 1).
At dynamic examination, only patient MPSIH003, with a flattened bony rim, showed instability with hips dislocation, which was already present before HSC-GT and maintained at the following evaluations.
Echogenicity of the joint capsule at baseline was normal in 10/16 hips and increased in 6/16, while it was normal in 8/10 and increased in 2/10 hips at +12 months, and normal in 6/10 and increased in 4/10 hips at +24 months after HSC-GT.
Table 1 summarizes ultrasonographic hip morphology features of all patients at baseline and at follow-up examinations after HSC-GT.
Figure 3 shows hip morphology in all patients at different timepoints and ages.
Figure 4 reports the alpha angle, beta angle, and femoral head coverage trend in all patients before and after HSC-GT. The cartilaginous roof was clearly enlarged at the beginning and became smaller over time, with the beta angle decreasing. The bony roof maintained an almost normal shape with alpha angle measuring a normal value (>60°) over time. Moreover, US evaluations showed alpha angle stabilization, beta angle decrease, and femoral head coverage increase, confirming a beneficial effect of HSC-GT on these specific pathological features of HD.
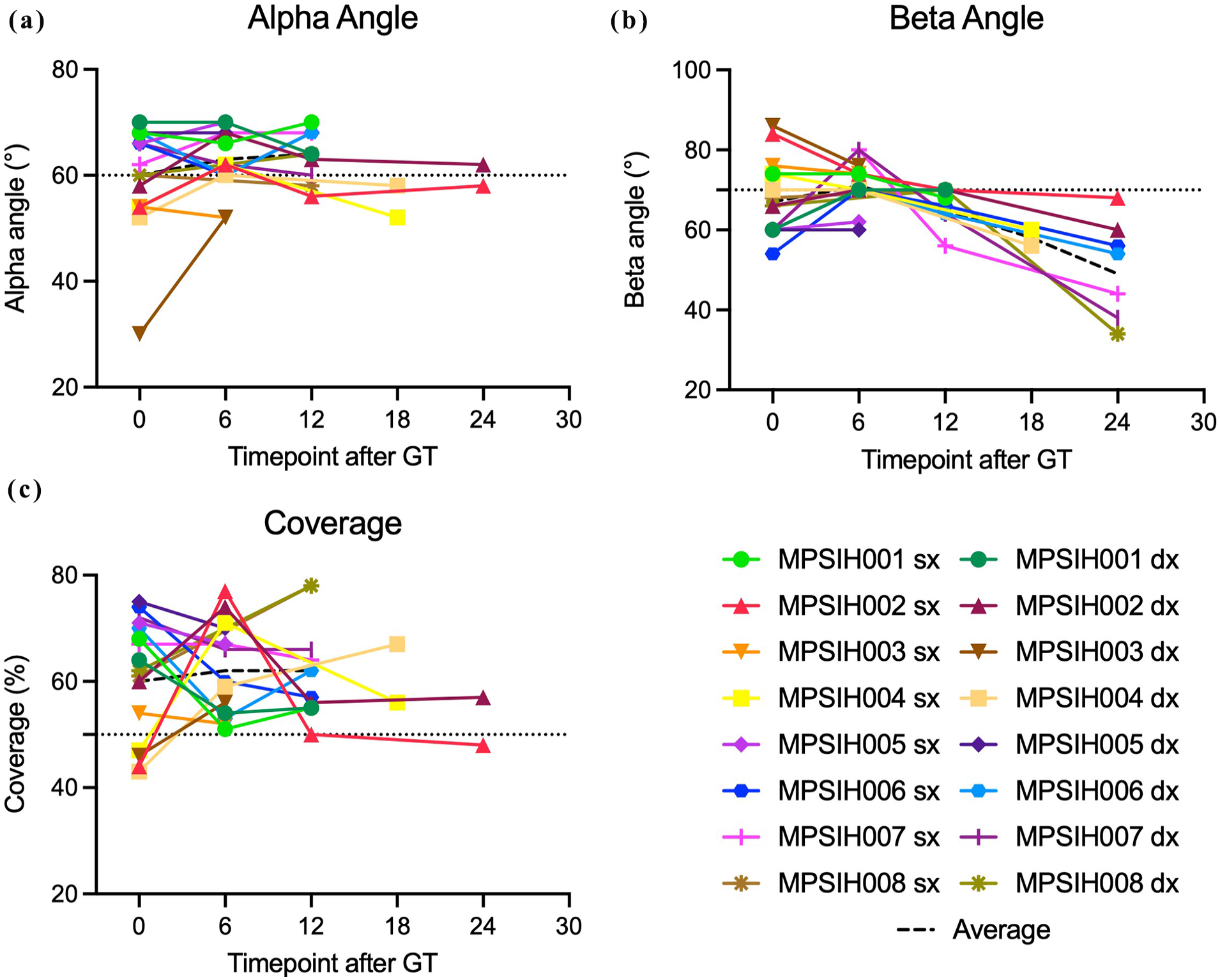
(a) Alpha angle trend before and after HSC-GT. (b) Beta angle trend before and after HSC-GT. (c) Head coverage trend before and after HSC-GT. Dotted lines indicate the threshold of normal hips for each parameter.
Discussion
Among mucopolysaccharidoses, MPSIH presents with some of the most severe skeletal complications, with HD being prevalent and progressive. Growth plate dysfunction due to accumulation of GAG in cartilage and bone causes progressive HD.9,10 The pathological changes are at the bone-cartilage interface of the lateral acetabular roof. This region, which is the area of major interest for developmental dysplasia, was evaluated in previous radiological studies6,7,14 and with US in our study. Langereis et al. described progressive acetabular dysplasia by analyzing 52 patients and 192 radiographs. 6
We recently reported for the first time that stabilization of HD parameters after HSC-GT in the same cohort of MPSIH patients such as acetabular bony roof (Acetabular Index, Head Coverage) using X-Ray and MRI.12,13 US evaluations showed similar results and allowed us to monitor hip morphology before and after HSC-GT. The bony rim provided pathological features with a rounded or flattened or notched rim at baseline and no angular shape, while the cartilaginous roof was enlarged in most cases.
Irregular and notched bony rim could be described as peculiar aspects of MPSIH and has also been described in other osteochondrodysplasias (spondylometaphyseal dysplasia, spondiloepiphyseal dysplasia, metatropic dysplasia, and Kniest dysplasia). 19 Like in osteochondrodysplasias, the failed endochondral ossification in Mucopolysaccharidosis (MPSs) results in a delay of the ossification.9,20
In our study, we evaluated patients by US up to a median age of 45 months (range 38–49 months). At baseline, the median age was 22 months (range 13–33 months), and hips were evaluated according to Graf methods in the presence of all the classical landmarks, including the inferior margin of the ilium in fossa acetabuli. At +12 months of follow-up and at a median age of 35 months (range 26–37 months), all patients were evaluated for all parameters. At +24 months, at a median age of 45 months (range 38–49 months), 4/8 patients could not be evaluated because of the large dimension of the nucleus and the absence of the inferior margin of the ilium. In these latter cases, it was not possible to determine the coverage of the femoral head expressed by the alpha angle and by Morin’s method. However, the beta angle and the cartilaginous roof dimension were evaluated, showing a decreased trend confirming the progressive cartilage-to-bone conversion. The nucleus itself showed normal and regular surface lateral profile in all cases. The shape of the nucleus was studied by X-ray and MRI in the same cohort of patients at the same timepoints, with no evidence of typical femoral head deformity, 13 while in HSCT and ERT-treated MPSIH patients, femoral head deformity was already present in early childhood.6,7
The bony roof lateral border for femoral head coverage, a typical anatomical landmark for radiological HD evaluation, was measurable in all US, allowing comparison with older ages. According to the X-ray evaluation of the pelvis, a line similar to the Perkin’s line could be drawn starting from the bony roof lateral border and coverage of the bony nucleus (Figure 5). Therefore, we compared the US image with the X-ray of the image to quantify the bony coverage of the nucleus (Figure 5(c) and (d)). Similarly, we compared MRI and US images of the pelvis of our patients at the same ages to measure the cartilaginous roof coverage (Figure 5(c)–(e)). MRI measurements were taken on the slice where the femoral head appears greater in its diameter, which corresponds to the biomechanically most important plane for the femoral head-acetabulum relationship, according to Huber et al. 21 As in the MRI evaluation, 21 we chose and measured the US slice with the larger nucleus dimension. Moreover, US allowed us to distinguish the anechogenic cartilaginous roof limit from the echogenic labrum and therefore to determine the dimension of the cartilage component of the roof.
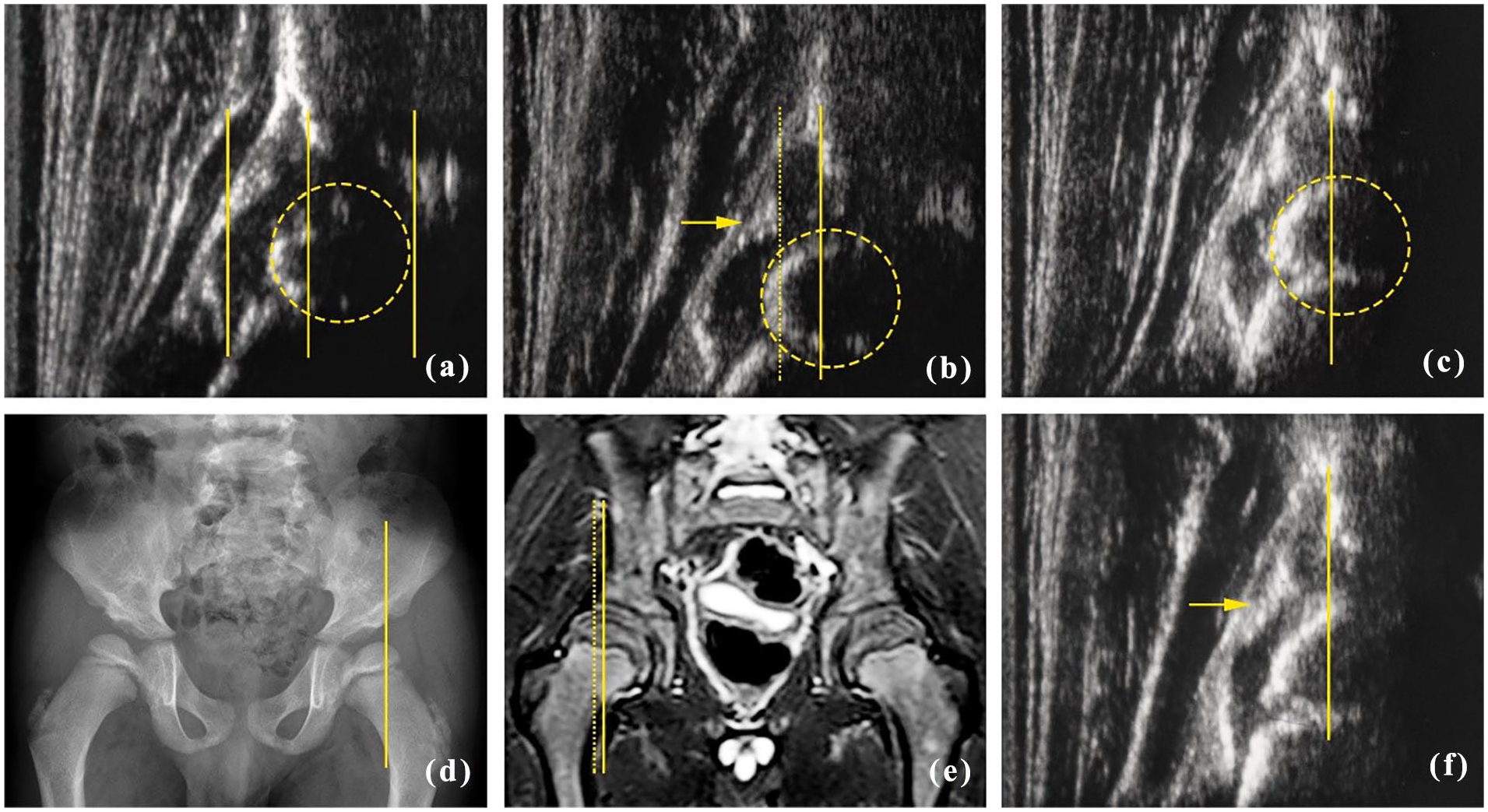
Ultrasonographic hip morphology of patient MPSIH006 at different follow-ups after HSC-GT in comparison with X-ray and MRI for bony and cartilaginous head coverage. (a) Right hip, head coverage at baseline and 23 months of life, 65% according to Morin et al. 17 (bony roof limit = central vertical line, cartilaginous medial and lateral femoral head limits = medial and lateral vertical lines, respectively). Ossific nucleus borders with the femoral head (dashed circle). (b) Right hip, head coverage of 62% according to Morin et al. at 12 months post-HSC-GT and at 37 months of age. 17 Enlarged cartilaginous roof (bony roof limit = vertical line, cartilaginous roof limit = vertical dotted line, labrum acetabulare = arrow). Ossific nucleus borders with the femoral head (dashed circle). (c) Right hip, head coverage at 24 months post-HSC-GT and at 49 months of age, with stable bony coverage (bony roof limit = vertical line, ossific nucleus = dashed circle). (d) X-ray of the pelvis at the same timepoint as (c) for comparison (bony roof limit = vertical line). (e) MRI image at the same timepoint of (c) (bony roof limit = vertical line, cartilaginous roof limit = vertical dotted line). (f) Left hip at the same timepoint of (c) (bony roof limit = vertical line, labrum acetabulare = arrow). In (c) and (f), the inferior margin of the ilium in the fossa acetabuli is covered by the large ossific nucleus and thus not visible.
Despite the fact that the children were over the usual age for US evaluation according to the Graf and Morin et al. technique,16,17 hips could be evaluated in half of the patients at follow-up +24 months after HSC-GT and at a median age of 45 months (range 38–49 months). In the remaining four patients, with a mean age of 45 months (ages 38, 45, 48, 49 months, respectively), US could not be performed due to the limitations cited above.
Only one study in the literature reported data from Mucopolysaccharidosis patients examined by US. 15 This study is not comparable with ours because it employs different techniques and because of the different patients’ ages. The three patients analyzed in Żuber et al., 15 aged 11, 12, and 32 years, respectively, were studied through a ventral longitudinal approach and not with the standard methods according to Graf and Morin et al.16,17 No data are available about coverage, acetabular bony, and cartilaginous roof, which are the most studied parameters in hip evaluation. Moreover, the echogenicity of the joint capsule was described as significantly increased in all patients. 15 In our study, echogenicity was increased in 50% of the patients at baseline with a median age of 22 months (range 13–23 months), decreasing to 40% at +24 months after HSC-GT at a median age of 45 months (range 38–49 months), possibly because of an early beneficial effect of HSC-GT on HD. Whether echogenicity will increase again in HSC-GT patients in the long term needs to be carefully monitored. Evaluation of the echogenicity is always a challenge due to the lack of a standard method to measure it. Only the comparison with the normal hips of healthy children with the same age and sex, together with the echogenicity of the nearby structures, may support this statement. 22 Moreover, echogenicity can be influenced by the gain and the position of the transducer. We always considered these points, maintaining the same gain value in all US examinations and paying attention to avoid tilting errors of the transducer.
In all US images of this article, the labrum is well represented with its echogenicity and triangular shape; it is only possible by keeping the transducer perpendicular to the hip structures, as known according to the Graf technique.
Despite the absence of the landmark at +24 months of follow-up in the patients with a median age of 45 months (range 38–49 months), the bony rim shape, the cartilaginous roof, and the coverage could be evaluated. The regular nucleus lateral profile distinguishing the anterior, central, and posterior parts in the US scanning allowed us to select the slice in the standard plane with the largest nucleus, thus the most central part of the nucleus.
Since there is no literature regarding US examination in MPSIH patients, to corroborate our findings, we compared US images with the corresponding X-rays and MRI images at the same timepoint after HSC-GT (Figure 5). This comparison of methods showed similar results and therefore indicates the reliability of our US technique and evaluations for bony and cartilaginous head coverage.
The main limitation of this technique is that hip US can be used according to Graf 16 only until the ossific nucleus of the femoral head allows the US waves to reach the inferior margin of the os ilium in the fossa acetabuli, which is a fundamental landmark, making sure to evaluate the central part of the fossa acetabuli. Considering the abnormalities of hip development in MPSIH patients, 9 US examination could be extended over the first years of life in line with previous data analyzing morphology of the bony and cartilaginous roof in osteochondrodyplasias, despite the absence of the inferior margin of the ilium in the acetabular fossa.20,21
This article reports the first ultrasonographic evaluation experience in a rare genetic disorder characterized by severe skeletal involvement. The anatomy of a MPSIH pelvis is not normal, as well demonstrated in literature5,6,7,13,14 (by X-ray and MRI) and also as shown in this article in Figure 5(d). Particularly, the shape of the acetabulum and the lateral acetabulum region, where the failures of endochondral ossification in the cartilage-to-bone conversion process are visible, is altered. For this reason, the contour and the silhouette of the iliac bone over the lateral acetabulum border cannot be represented as in a normal hip anatomy, but are notched and irregular. However, the lateral acetabulum shape could be well described in this study. Moreover, in all US images of this study, the landmarks for a correct evaluation of the hip morphometry are present.
Conclusion
Our study shows that US allows not only a qualitative evaluation of hip features such the bony roof, the cartilaginous roof, and the femoral head nucleus, but also the quantitative measurement of hip morphology with parameters such as alpha angle, beta angle, and femoral head coverage, and it represents a valid tool for monitoring hip joint development in MPSIH patients. This study may suggest a beneficial effect of HSC-GT on the ultrasonographic features of hips in MPSIH patients.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251349438 – Supplemental material for Ultrasonographic hip morphology in mucopolysaccharidosis type I Hurler after hematopoietic stem cell gene therapy
Supplemental material, sj-pdf-1-cho-10.1177_18632521251349438 for Ultrasonographic hip morphology in mucopolysaccharidosis type I Hurler after hematopoietic stem cell gene therapy by Maurizio De Pellegrin, Chiara Filisetti, Matilde Cossutta, Michele Colombo, Giulia Consiglieri, Francesca Tucci, Alessandro Aiuti and Maria Ester Bernardo in Journal of Children’s Orthopaedics
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AA was the study PI between May 2018 and July 2020, and MEB has been the PI since July 2020. GC, MC, FT, CF, AA, and MEB are investigators of the trial. MEB has acted as ad hoc consultants for an Orchard Therapeutics advisory board in 2020. MEB has also received occasional reimbursement of travel costs and registration fees for scientific congress presentations from Orchard Therapeutics. MDP has a service contract with Telethon Foundation. The other authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Clinical trial funding
Fondazione Telethon was the initial financial sponsor; in May 2019, when lentiviral vector-hematopoietic stem cell-based gene therapy for patients with MPSIH was licensed to Orchard Therapeutics Ltd (OTL), and OTL became the financial sponsor.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
