Abstract
Excitotoxicity is a primary pathological process that occurs during stroke, traumatic brain injury (TBI), and global brain ischemia such as perinatal asphyxia. Excitotoxicity is triggered by an overabundance of excitatory neurotransmitters within the synapse, causing a detrimental cascade of excessive sodium and calcium influx, generation of reactive oxygen species, mitochondrial damage, and ultimately cell death. There are multiple potential points of intervention to combat excitotoxicity and downstream oxidative stress, yet there are currently no therapeutics clinically approved for this specific purpose. For a therapeutic to be effective against excitotoxicity, the therapeutic must accumulate at the disease site at the appropriate concentration at the right time. Nanotechnology can provide benefits for therapeutic delivery, including overcoming physiological obstacles such as the blood–brain barrier, protect cargo from degradation, and provide controlled release of a drug. This review evaluates the use of nano-based therapeutics to combat excitotoxicity in stroke, TBI, and hypoxia–ischemia with an emphasis on mitigating oxidative stress, and consideration of the path forward toward clinical translation.
The impacts and challenges of neurological disease
Acute brain injury is a common phenomenon associated with significant morbidity and mortality. Each year in the United States, around 800 thousand people suffer from a hemorrhagic or ischemic stroke, 60 thousand newborns experience perinatal asphyxia and subsequent hypoxia–ischemia (HI) encephalopathy, and three million people suffer from a traumatic brain injury (TBI). 1 –3 The costs associated with these diseases total over $113 billion per year and are not limited to the medical treatments, subsequent pathological complications, and loss of earned wages throughout life; additional unquantifiable costs also include the physical and emotional toll on the individual, along with their caregivers, family, and friends. 1,4,5
Acute brain injuries exhibit a set of characteristic mechanisms that contribute to neurological damage. The pathological sequelae include immediate cell death and tissue loss followed by ongoing excitotoxicity, inflammation, and oxidative stress. 6,7 Excitotoxicity is the pathological process of neuronal death due to dysregulated neuronal overstimulation by excitatory amino acids (EAAs) such as glutamate. Excitotoxicity leads to oxidative stress, where an excess of reactive oxygen species (ROS) exhausts native antioxidant systems. Understanding the etiology of neurological disease progression, especially the role of excitotoxicity and oxidative stress, is essential for determining ideal points of intervention for the development and implementation of effective therapeutics.
There have been extensive efforts in the fundamental sciences of pathology, immunology, and neuroscience to expand knowledge of excitotoxic neuronal death to elucidate key stages of intervention. Promising candidates include natural antioxidant, inorganic antioxidant-mimetic, and growth factor therapeutics, among others. However, developing a successful therapeutic requires a drug that not only has biochemical efficacy, but also effectively accumulates at the target site at therapeutic concentrations. In neurological disease, free drugs often fail to reach the target site due to in vivo degradation, systemic clearance mechanisms, and the barrier properties of the neurovascular unit. 8 Consequently, despite the research progress achieved, there are currently no approved therapeutics for targeting excitotoxicity or its direct sequelae after acute neurological injury. Nanotechnology can be leveraged to help overcome each of the aforementioned delivery obstacles to the brain. By conjugating drugs to nanoparticle surfaces, encapsulating drugs within nanoparticles, or utilizing materials with intrinsic therapeutic effects in nanoparticle form, nanoparticle therapeutics can stabilize labile free drugs and traverse steric obstacles to reach diseased sites. 8 Here, we provide an overview of the relevant disease mechanisms and points of intervention in excitotoxicity and subsequent oxidative stress including the advantages of nanotechnology, a summary of nanotherapeutic progress achieved thus far with a focus on in vivo work, and research strategies to implement for effective clinical translation.
Mechanisms of excitotoxicity in acute brain injury
Glutamate is the major excitatory neurotransmitter in the central nervous system (CNS), responsible for the sense of sight, smell, taste (umami), nociception, and hearing, as well as the more complex processes of memory formation and learning. 9 During neuronal activity, vesicles containing neurotransmitters in the presynaptic neuron merge with the membrane, expelling their contents, namely glutamate, into the synaptic cleft. 10 Glutamate then rapidly diffuses across the synapse to bind to ionotropic glutamate receptors (iGluRs) on the postsynaptic neuron, eliciting an influx of sodium, which triggers an action potential that propagates down the neuron, forming the basis of neuronal communication. 9 Astrocytic end feet enveloping the synapse rapidly uptake glutamate to recycle back to the presynaptic neuron to replenish vesicular stores. 11 Upon neuronal excitation, calcium also influxes and binds to post synaptic density protein 95 (PSD-95) to initiate the recruitment of neuronal nitric oxide synthase (nNOS) and production of nitric oxide (NO). 12 NO then activates the nicotinamide adenine dinucleotide phosphate oxidase (NOX) complex that generates superoxide radical anion (SOX, O2 −). 13
In acute brain injury, a number of pathological processes result in excitotoxicity depending on the nature of the insult. During stroke and HI, asphyxiation reduces ATP production leading to sodium/potassium ATPase pump failure and subsequent anoxic depolarization-induced glutamate release. 14 Under hypoxia, as cells switch to lactic acid production and undergo acidosis, ATP generation further decreases, inhibiting ATP-dependent astrocytic glutamate uptake. 15 In TBI, direct trauma induces immediate necrotic death of neurons and glia resulting in neuronal release of their glutamate stores, and subsequent dysregulation of cerebral blood flow leading to similar energy deficits as occurs in stroke and HI. 16,17 Once blood flow returns after a hypoxic event, reperfusion injury may exacerbate neuronal damage. 18 Regardless of the injury, excessive synaptic glutamate accumulation causes excitotoxicity. 12,19 Substantially elevated intracellular glutamate, ROS, and calcium levels lead to cell death, increased glutamate release from dying neurons, and propagation of excitotoxicity. 12,20
Mitochondria play a prominent role in excitotoxicity, oxidative stress, and cell death processes due to energy, calcium, and ROS dynamics. 21 During excitotoxicity, mitochondria uptake excess cytosolic calcium and produce high concentrations of SOX during mitochondrial respiration in response to excitatory postsynaptic currents. 22 NOX-derived SOX triggers even greater SOX production from mitochondria. 23 –26 SOX mediates mitochondrial damage, resulting in electron transport chain failure as well as reverse electron transport leading to greater SOX production. 23 Other sources of SOX include production by xanthine oxidase or invading neutrophils during reperfusion, monoamine oxidase, and uncoupled nNOS by SOX oxidation of the nNOS cofactor tetrahydrobiopterin. 18,27 –30 Abundant SOX reacts with NO to form peroxynitrite, which can cause lipid peroxidation, protein deactivation, DNA mutation, and poly(ADP ribose) polymerase (PARP) activation. 31,32 High intracellular calcium and SOX levels lead to extensive mitochondrial fission and mitochondrial permeability transition pore (mPTP) formation. 22,33 The exact mechanism of mPTP formation remains to be elucidated, but ROS contributes several roles. 34 –36 mPTP formation results in burst release of SOX, calcium, and other necrosis and apoptosis effectors into the cytosol. 37 Mitochondrial burst release of SOX further induces NOX SOX production and neighboring mitochondrial stress, reducing mitochondrial antioxidant functions and creating a reinforcing cycle. 23,38 As excitotoxicity, ROS generation, and mPTP formation are intimately linked to death processes, ROS scavenging is a promising therapeutic strategy after acute brain injury.
After acute neurological injury, a number of neuroinflammatory processes occur both alongside and due to excitotoxicity. Oxidative stress plays a prominent role in neuroinflammation as well. SOX generated from NOX and subsequent oxidation to hydrogen peroxide (H2O2) elicit microglial proliferation and other downstream inflammatory signaling pathways. 39,40 For instance, in perinatal asphyxia models, HI induces both excitotoxic neuronal death as well as neuroinflammatory microglial proliferation. 41 Inflammation can in turn cause further delayed excitotoxicity, as occurs in depression and Parkinson’s disease. 42,43 Therefore, therapies that manage oxidative stress seen after acute neurological injury have the combined promise of alleviating excitotoxicity as well as neuroinflammation. An in-depth discussion of neuroinflammation is beyond the scope of this review, and we refer readers to other reviews. 44 –46 Figure 1 illustrates the major processes of excitotoxicity along with therapeutic points of intervention, while Table 1 introduces promising therapeutic candidates at these points of intervention and the potential benefits that nanotechnology can provide.
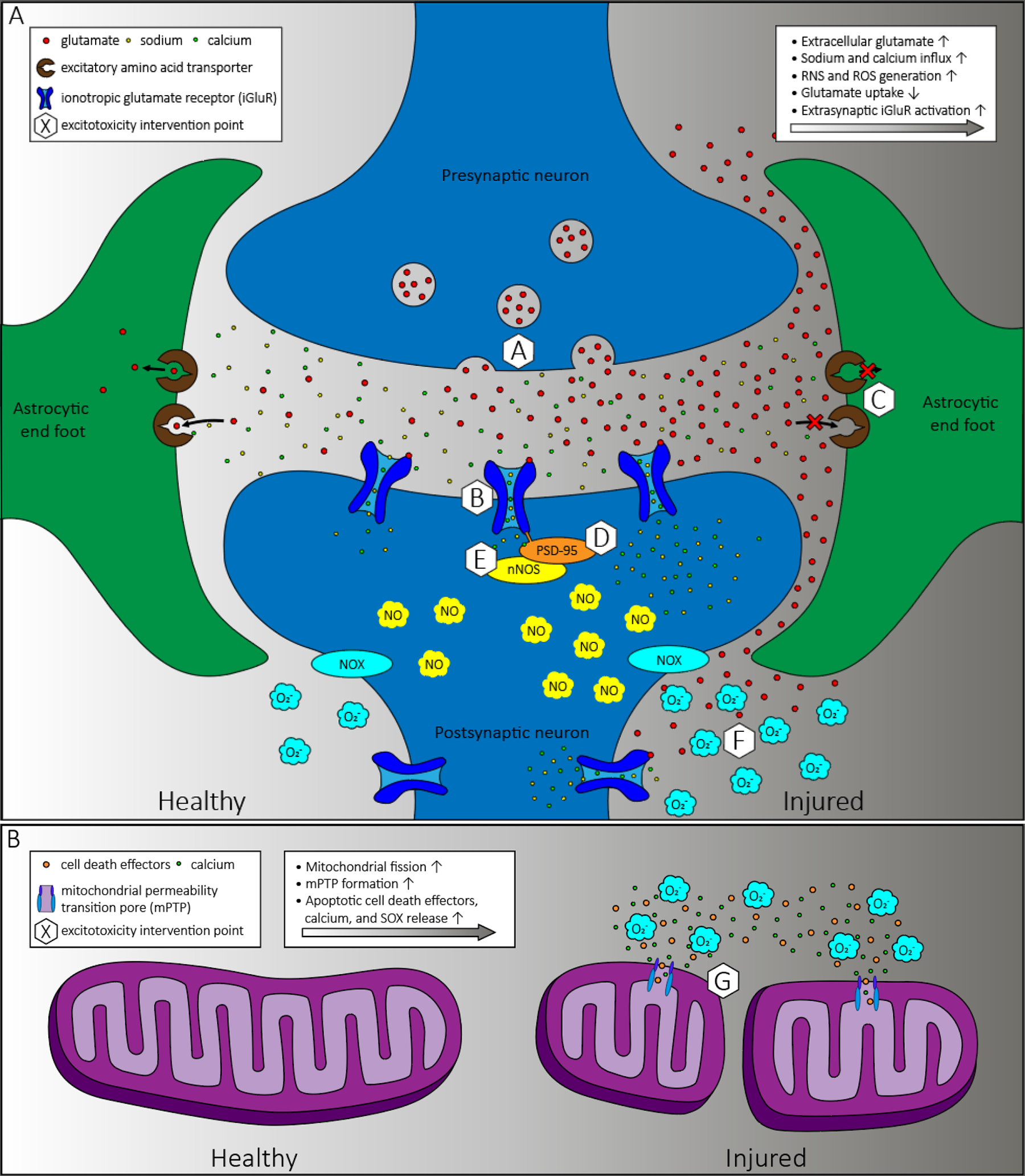
Schematic overview of excitotoxic and oxidative stress processes in acute brain injury with points of intervention (white hexagons labeled A–G) at the A) synapse and B) mitochondria, where the left side is healthy, and the right side is injured. Excessive glutamate concentrations in the synapse can lead to excitotoxicity through enhanced ionotropic glutamate receptor activation, sodium and calcium influx, and generation of NO and SOX. These processes amplify mitochondrial ROS stress, resulting in mitochondrial fragmentation and mPTP formation, and release of calcium, SOX, and apoptotic cell death effectors.
Promising treatments for excitotoxicity in acute brain injury corresponding to Figure 1 points of intervention, labeled in white hexagons as A–G. These therapeutics could potentially benefit from nanoparticle strategies to surpass biological barriers and increase site specific localization.
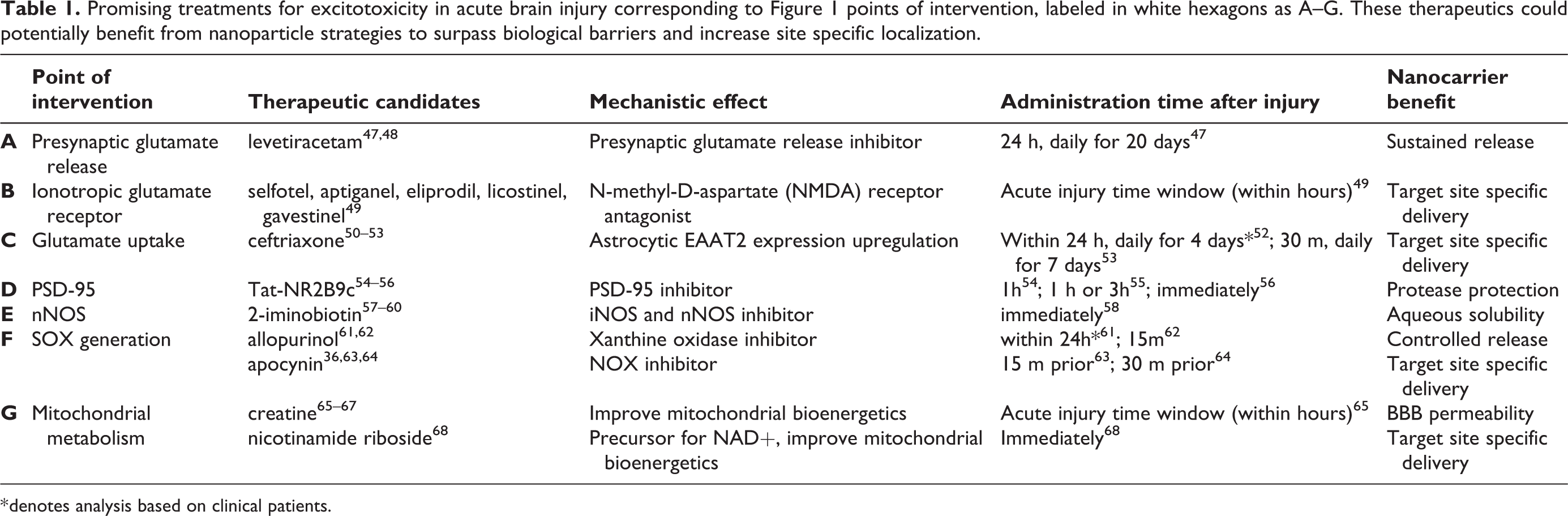
* denotes analysis based on clinical patients.
Therapeutic requirements for overcoming CNS barriers
To combat oxidative stress in excitotoxicity, a therapeutic must perform its biochemical function of scavenging ROS or inhibiting ROS generation but also be capable of reaching the diseased area. The mammalian body is highly efficient in the clearance of foreign substances. The kidneys readily filter out molecules smaller than 5–10 nm, while the liver metabolizes any molecule above 200 nm before reaching the brain. 69 Therapeutics also require an inert near-neutral surface charge to avoid non-specific adsorption to circulating serum proteins, extracellular matrix components, or cell membranes. 70 Without an inert surface, therapeutics can readily be opsonized for digestion by resident macrophages within many organs. 71 Furthermore, the in vivo microenvironment may affect drug stability with potential deactivation due to proteases, oxidation/reduction, hydrolysis, pH, and unfavorable binding. Therapeutic proteins are especially susceptible to rapid degradation from in vivo proteases. 72 –74 Even after overcoming systemic clearance, a therapeutic must be capable of crossing the highly restrictive blood–brain barrier (BBB). 75 The BBB inhibits passage of all macromolecules such as proteins, and 98% of small molecule drugs. 76 However, in acute brain injuries such as stroke and TBI, there is some BBB breakdown, resulting in “leakiness” of the BBB to a wider range of molecules. 77 –79 We refer readers to other reviews that cover how acute brain injury mediates BBB breakdown. 78,80 After admittance to the brain, a therapeutic must avoid expulsion by efflux transporters such as the P-glycoprotein, ATP-binding cassette, and solute carrier transporters, and then subsequently navigate the brain parenchyma to reach diseased areas. 81,82 Depending on the ultimate target, a therapeutic may also require a specific surface chemistry to undergo cell-type specific uptake.
Application of nanoparticles and their therapeutic benefits
Nanotechnology holds promise to both maintain therapeutic stability and overcome the barriers to brain delivery. There are a wide variety of nanoparticle platforms used for therapeutic delivery to the brain, including polymeric nanoparticles, liposomes, hydrogels, and dendrimers. Polymeric nanoparticles can provide controlled drug release, targeting capabilities, and prolonged drug action by protection from proteases, and have been shown to cross an intact or impaired BBB. 72,83 Nanoparticle size, shape, flexibility, and surface charge can be tailored to overcome steric clearance and non-specific binding to alter pharmacokinetics and improve brain accumulation. 84 –86 With a dense poly(ethylene glycol) (PEG) coating, nanoparticles exhibit increased systemic circulation time by reducing interactions that lead to clearance and opsonization. 75 Densely-PEG coated nanoparticles up to 114 nm are also capable of diffusive and convective transport through the brain parenchyma. 87,88 Drug-incorporation strategies include loading within nanoparticle matrices, cores, or lipophilic bilayers, or covalent conjugation to surface end groups. 8 To improve pharmacokinetics or cellular uptake, nanoparticles can also be further decorated with surface ligands or surfactants, while incorporation of biology-responsive materials can further specialize therapeutic delivery. 83,89 Incorporation of pH-sensitive groups can imbue nanoparticles with triggered release capabilities, only releasing drug when reaching the acidotic ischemic area, or when internalized within an acidic lysosome. 90,91 Similar strategies apply for attachment of protease-cleavable linkers and oxidation/reduction-sensitive bonds. 92,93 Superparamagnetic nanoparticles can also be guided to diseased tissue regions using magnetic resonance (MR). 94 Nanoparticles may also serve as biomarkers of disease, however, biomarker applications are outside the scope of this review and have been covered in other reviews. 95,96 By leveraging the benefits of nanoparticle drug delivery and understanding neurological disease processes at the biomolecular level, therapeutic agents can be better designed to combat excitotoxicity.
ROS scavenging antioxidant enzyme-loaded nanoparticles
Nanoparticles have been investigated for delivering therapeutics in in vivo models of stroke, TBI, and HI. One of the most promising enzyme therapeutic candidates is superoxide dismutase (SOD), a native antioxidant enzyme that converts SOX into H2O2 and water. SOD-loaded polymeric poly(lactic-co-glycolic acid) (PLGA) nanoparticles reduced infarct volume by 65% in a middle cerebral artery occlusion (MCAO) rat model, compared to only a 25% reduction from free SOD alone. 97 While 0% of rats survived without treatment after 4 weeks, SOD-nanoparticle application resulted in 75% survival. 98 Similarly, Yun et al. evaluated SOD delivery in liposomes, polybutylcyanoacrylate (PBCA) nanoparticles, and PLGA nanoparticles in a mouse model of multi-vessel ischemia (MVI). All three platforms resulted in 35–45% infarct volume reduction, with 50–60% reduction when conjugated with N-methyl-D-aspartic acid receptor 1 (NMDA-R1) antibodies. 99 The efficacy of SOD-loaded liposomes against bilateral common carotid artery occlusion (BCCAO) was also evaluated in gerbils, where treatment prevented a decrease in endogenous SOD levels and mitigated mitochondrial membrane lipid peroxidation but failed to reduce brain swelling. 100 From other studies, SOD-loaded liposomes reduced infarct size by 18–33% after focal cerebral ischemia in rats, and reduced BBB permeability and brain edema in cold-induced TBI rats. 101,102 In a MCAO mouse model, SOD-encapsulating polyion condensation complexes of PEG-b-poly(L-lysine) (PEG-PLL) or PEG-b-poly(aspartate diethyltriamine) (PEG-DET) followed by crosslinking reduced infarct volume by 50–60%. 103,104
Even though SOD converts SOX into H2O2, there is evidently a therapeutic benefit in reducing SOX levels at the expense of increasing H2O2 levels, by reducing mitochondrial oxidative stress and fragmentation. 105 Catalase scavenging of H2O2 by itself or in tandem with SOX scavenging has also shown positive effects in reducing acute brain injury. Catalase delivery via PLGA polymer or polyion complexed micelle nanoparticles has been explored in vitro, but in vivo application has been limited. 106,107 Zhang et al. observed a reduction in infarct size with cross-linked dendrigraft poly-L-lysine (DGL) nanoparticles containing cis-aconitic anhydride-modified catalase after MCAO in mice, with further infarct reduction when nanoparticles were decorated with N-acetyl-proline-glycine-proline tripeptides with a high affinity for neutrophils, which enhanced BBB penetration. 108 SOD and catalase loaded separately into PLGA nanoparticles were administered after tissue plasminogen activator (tPA) in a thromboembolic stroke rat model, resulting in reduced number of caspase-positive cells, neutrophils, and hippocampal swelling, and an increase in nestin-positive neuron progenitor cells. 109 Combinatorial delivery of SOD nanoparticles and catalase nanoparticles offers promise in disrupting neuronal death processes via interruption of SOX-mediated primary damage as well as longer time-scale inflammatory processes caused by H2O2 accumulation. 110 Although enzyme-encapsulating nanoparticles are promising for combating excitotoxicity with their precise and continuous catalytic functions, enzyme delivery still faces many challenges including poor hydrophilic macromolecule loading in hydrophobic matrices, and enzyme deactivation from high-energy mixing and organic/aqueous interfaces during formulation processes. 111 –114 Researchers are exploring other antioxidant-capable nanoparticle strategies that circumvent some of these challenges.
ROS scavenging antioxidant-mimetic materials as nanoparticles
Nanoparticles themselves may be composed of therapeutic materials that exhibit antioxidant capabilities. 115,116 Cerium oxide nanoparticles of 4 nm diameter exhibit SOD- and catalase-mimetic activity, and can scavenge NO, and peroxynitrite. 117 They have been shown to reduce infarct size by over 50% after MCAO in rats at 0.5 mg/kg and 0.7 mg/kg doses. 118 Interestingly, higher concentrations of 1 mg/kg and 1.5 mg/kg provided no significant change in infarct volume compared to no treatment. 118 Capping cerium oxide nanoparticles with zeolitic imidazolate framework-8 further improved ROS scavenging ability. 119 Cerium oxide nanoparticles have been covered in-depth in other reviews. 117,120,121 Platinum nanoparticles are also SOD- and catalase-mimetics and result in reduced infarct size in the cortex after transient MCAO in mice. 116 Yttrium oxide nanoparticles have similar antioxidant effects as cerium oxide nanoparticles in in vitro cell culture and could warrant further exploration. 122
Fullerene nanoparticles, which are carbon allotropes consisting of 60 carbons in a spherical arrangement, have also been investigated for their free radical scavenging capabilities of neutralizing hydroxyl radicals, SOX, and NO by transferring radicals into stable conformations within the fullerene nanoparticle. 123 –125 Hydroxylated fullerene nanoparticles reduced neuronal death in the CA1 hippocampal region after MVI in rats. 126 Vani et al. applied hydroxylated fullerene nanoparticles before or after MCAO in rats, yielding a 53% or 81% reduction in infarct volume, respectively, a reduction in malondialdehyde levels, and rescuing of reduced glutathione (GSH) content and SOD activity. 127 Carboxyfullerene nanoparticles decreased cortical infarction and prevented GSH depletion and lipid peroxidation, but also had adverse effects including writhing and even death in rats after MCAO and BCCAO. 124 Fullerenes can also produce pro-oxidant SOX and singlet oxygen in the presence of UV light, which could lead to adverse effects if fullerene nanoparticles localize in skin epithelium. 125 This UV-induced radical producing capability warrants further study of fullerene nanoparticles in the presence of other biologically relevant radical-generating conditions such as free iron.
PEG-functionalized hydrophilic carbon clusters (PEG-HCCs) consisting of 40 nm by 2–3 nm carbon nanotubes have also been explored for excitotoxic and immunomodulation applications due to their SOD-mimetic and hydroxyl radical scavenging properties. 128,129 Interestingly, PEG-HCCs are inert towards NO and peroxynitrite, holding potential as selective ROS scavengers that avoid NO scavenging and consequent vasodilatation and blood flow interference. 128 PEG-HCCs can also load hydrophobic molecules into their hydrophobic core for combinatorial therapy. 130 PEG-HCCs administered during reperfusion of transient MCAO rats under hyperglycemic conditions resulted in a 42% reduction in infarct size, and reduced edema and hemorrhage. 131 PEG-HCCs furthermore normalized NO and SOX levels and restored cerebral blood flow after controlled cortical impact (CCI)-induced TBI in rats. 132 With self-regenerating ROS-specific rapid antioxidant capacity, PEG-HCCs warrant further investigation as a viable excitotoxicity therapeutic for stroke and TBI applications. 130 Due to the non-biological origin of antioxidant-mimetic material nanoparticles and their relatively recent application in the neurological disease fields, further research is also needed to assess their long-term biocompatibility, clearance and pharmacokinetics, and maximum tolerated dosing before translation to clinical trials can be achieved. 133,134
Broad-acting antioxidant-loaded nanoparticles
While the antioxidant-nanoparticle strategies covered thus far have focused on specific scavenging of SOX and/or H2O2, there are also several broad-acting antioxidants that have utilized nanoparticle delivery to improve therapeutic accumulation at the target site to enhance efficacy. Erythropoietin (EPO) exerts its neuroprotective effects for acute brain injury by chelating iron to reduce radical formation, modulating inflammation in multiple brain cell types, and acting as a neurotrophic agent. 135 –137 EPO delivery within PLGA nanoparticles has been shown to reduce infarct volume in a perinatal rat model of HI at 10 times lower doses than recombinant EPO. 138
Curcumin is a small molecule found in the culinary spice turmeric that has broad-acting anti-inflammatory and antioxidant effects. Curcumin has been associated with activation of the antioxidant response element pathway in mice after closed-skull impact-induced TBI, ∼50% infarct volume reduction after MCAO in rats, and microglial polarization towards the anti-inflammatory state after distal MCAO in mice. 139 –141 However, curcumin’s direct application for acute brain injury is suboptimal due to its hydrophobicity and consequent low aqueous solubility. Curcumin incorporation within PLGA-PEG nanoparticles permitted curcumin penetration across the BBB, diffusion effectively to disease sites, and a decreased infarct size in neonatal HI rats while free curcumin provided no significant therapeutic efficacy. 41 Curcumin-loaded within solid lipid nanoparticles administered orally to rats after BCCAO also improved neurological scoring by 79% and restored SOD, GSH, and catalase levels to sham control levels. 142 Curcumin-loaded N-isopropyl acrylamide (PNIPAM) nanoparticles administered intranasally after MCAO in rats restored grip strength, locomotor activity, glutathione peroxidase, glutathione reductase, SOD, and catalase activity, and reduced lipid peroxidation and neuronal loss by ∼40%. 143
Multiple other antioxidants have also shown promise for acute brain injury when incorporated into nanoparticle platforms. Melanin exhibits broad antioxidant activity against SOX, H2O2, hydroxyl radical, peroxynitrite, and NO. 144 Injection of PEGylated melanin nanoparticles before MCAO injury in rats resulted in ∼50% infarct reduction and showed no immediate toxicity in preliminary in vitro and in vivo studies. 144 N-acetylcysteine (NAC) is a precursor to GSH and therefore an antioxidant and free radical scavenger, as well as a pleiotropic anti-inflammatory agent. 145 Once internalized into cells, NAC is hydrolyzed to release cysteine which is then used for GSH production. 145 NAC-conjugated dendrimers (D-NAC) administered to neonatal mice that underwent permanent unilateral carotid artery ligation with mild hypoxia improved white matter myelination. 146 Adenosine provides neuroprotection by binding to inhibitory adenosine A1 receptors to hyperpolarize neurons and reduce glutamate release, mitigating downstream excitotoxic processes. 147,148 The hydrophilic small molecule adenosine is readily metabolized and cleared in the bloodstream, but when conjugated to squalene and formed into nanoparticles, these adenosine complexes significantly improved neurologic deficit scores after MCAO in mice. 149 The natural polyphenol antioxidant resveratrol is water insoluble with a short half-life in its free form. 150 Resveratrol-loaded poly(N-vinylpyrrolidone)-b-poly(∊-caprolactone) (PVP-b-PCL) polymeric nanoparticles reduced infarct volume by 30–40%, malondialdehyde levels, and neuronal apoptosis after transient MCAO in rats. 150 The flavanone glycoside antioxidant hesperidin suffers from poor BBB passage and therefore bioavailability. 151 Hesperidin nanoparticles increased GSH, catalase, and total protein levels, and decreased infarct volume and malondialdehyde levels after BCCAO in rats. 151 Broad-acting antioxidants are promising in mitigating ROS damage in acute brain injury, and many also exhibit broad anti-inflammatory properties as well, which may further improve therapeutic efficacy. However, because pleiotropic drugs act via multiple neuroprotective mechanisms, gleaning broader understanding of which specific mechanisms to target from these studies to combat excitotoxicity is limited.
Our review of anti-excitotoxicity and antioxidant therapeutic efficacy has focused on survival, infarct volume reduction, and oxidative stress markers at the molecular and cellular scales. Fortunately, physical improvement overall correlated well with behavioral outcomes across antioxidant-nanoparticle studies. 98,116,131,138,142 For example, infarct reduction occurred alongside improvements in hind limb flexion, the ability to walk straight, and noise sensitivity upon administration of SOD-loaded PLGA nanoparticles after transient MCAO in rats. 98 However, slight impairment in forelimb movement and blinking reaction remained. 98 Platinum nanoparticles and PEG-HCCs improved performance on the Bederson exam, encompassing forelimb strength, stability from pushing, and circling behavior, alongside infarct reduction after transient MCAO in rats. 116,131 Nanoerythropoietin in a neonatal HI rat model, platinum nanoparticles after MCAO in rats, and curcumin-loaded solid lipid nanoparticles after BCCAO in rats improved performance on the Rotarod test alongside infarct reduction. 116,138,142 Curcumin-loaded solid lipid nanoparticles also improved Morris water maze and elevated plus maze performance to closer to that of sham injury scores. 142
Pitfalls of translating anti-excitotoxic therapies
There remains a distinct lack of therapeutics targeting excitotoxicity in routine clinical use, despite having been described as a key component of the pathophysiology of acute neurological injury mechanisms for several decades. A large part of this lack of successful translation is likely due to the issues outlined above—the requirement of more targeted or stable delivery of therapeutics to the site of injury. However, a number of other obstacles or common problems exist in the translational pipeline that should be addressed as new therapeutics are developed. These include the timing of the therapeutic, adequate control of confounding physiological factors in preclinical studies, and the number and heterogeneity of animal models assessed before moving to clinical trials.
The timing of therapies with respect to the specific pathophysiological processes that occur after injury is a crucial component of successful clinical translation. This is likely to be particularly important for therapies that directly target the accumulation of EAAs such as glutamate. For instance, in piglet models of perinatal HI brain injury, a small increase in extracellular glutamate is seen during the insult itself as a result of primary energy failure, followed by a decrease after resuscitation until a significant increase as secondary energy failure occurs 6–12 h later. 152 Preclinically, therapeutic hypothermia (TH) is significantly neuroprotective in a wide range of acute brain injuries, at least in part due to its ability to suppress the release of EAAs. 153 –155 However, TH has only shown clinical success after perinatal asphyxia, and must be initiated within 6 h of the injury or earlier for maximum benefit. 156
Similarly, the NMDA-R antagonist xenon augments hypothermic neuroprotection in rat and piglet models of perinatal asphyxia when given at a concentration of 50% and initiated within 3 h of resuscitation. 157 –159 Yet, the TOBY-Xe trial found no benefit of adding xenon to TH for infants after birth asphyxia, with significant confounders including that median time of xenon onset was 10 h (range: 4.0–12.6 h), and that it was given at a concentration of 30%. 160 Overall, the data therefore suggest that any directly anti-excitotoxic therapy must be in place as soon as possible after reperfusion. 156 This may be because of ongoing excitotoxicity that occurs even in the absence of measurable increases in extracellular glutamate. For instance, despite the relatively delayed rise in glutamate after injury, others have shown that an “excitotoxic index” consisting of relative levels of glutamate, aspartate, and glycine to γ-aminobutyric acid (GABA) begins to increase almost immediately after resuscitation in a piglet model of perinatal HI brain injury. 161 In the clinical setting, it is likely that any anti-excitotoxic therapeutic should therefore be at high local concentrations within 3–6 h of the initial injury, and this must be incorporated into the design of both preclinical and clinical studies. Importantly, therapies that target downstream mitochondrial dysfunction, neuroinflammation, oxidative stress, and delayed neurodegeneration, which continue to occur over hours to days, may have an extended therapeutic window as long as they can be reliably delivered to the site of injury. 162,163
The history of anti-excitotoxic therapies, particularly when used in rodent studies, is significantly confounded by issues with temperature regulation because hypothermia, which is known to be neuroprotective, spontaneously occurs after brain injury and is rarely adequately controlled. 156,164 –166 Additionally, glutamate signaling plays a dominant role in thermoregulation and maintenance of normal temperature, with pharmaceutical inhibition of glutamate signaling generally associated with loss of cold-evoked temperature responses (e.g. brown fat thermogenesis), and subsequent hypothermia. 167 –169 In both animal models and humans, early spontaneous hypothermia is a common occurrence after global brain injury, with greater decreases in core temperature generally seen with a greater extent and severity of injury. 166,170 –173 In preclinical models at least, this spontaneous hypothermia can result in neuroprotection, with enforced periods of temperature regulation required after injury to standardize the degree of injury seen. 166,174 However, in one analysis of studies assessing neuroprotective strategies in adult rodent models of both global and focal ischemia, only around 30% of studies did temperature measurements after the injury procedure. 164 A more recent analysis of preclinical studies examining neuroprotective agents in perinatal HI injury found similar results. 165
In their seminal paper in 2006, O’Collins et al. described “1,026 Experimental Treatments in Acute Stroke,” the vast majority of which had not been translated to clinical trials. 175 Anti-excitotoxic therapies provided, on average, about 25% neuroprotection, which was a similar magnitude to thrombolysis, the current standard of care for acute ischemic stroke. The general lack of successful translation of anti-excitotoxic therapies in this setting may either be due to problems with timing relative to injury, or that the majority of benefit from directly anti-excitotoxic therapies, for instance those that regulate glutamate release or signaling, result in hypothermia that is not controlled for in the preclinical setting. 164,165 Hypothermia has not been shown to be robustly beneficial after stroke, TBI, or global brain ischemia (e.g. after cardiac arrest) in pediatric or adult populations. 153,155,173 However, rigorous temperature management is commonplace in hospitalized adults after global and focal acute brain injuries due to the benefit of preventing hyperthermia (fever). 153,173,176,177 Therefore, more evidence is still required to examine whether directly anti-excitotoxic therapies will have their place in routine clinical use by providing neuroprotection above and beyond their effects on thermoregulation. Although promising, antioxidant-nanoparticle therapies will also necessarily undergo the same rigor of evaluation before clinical translation.
Barriers to clinical translation of nanotechnologies for acute brain injury
As with the majority of putative therapies developed for treating neurological disorders, any lack of success in the clinical setting could potentially be determined earlier by developing an adequate preclinical pipeline. 178 This was the goal of the STAIR (Stroke Therapy Academic Industry Roundtable) criteria when they were developed more than two decades ago. To fully meet the criteria, a therapy had to be successfully tested: i) in two or more laboratories, ii) in two or more species, iii) in animals at a disease-appropriate life stage, iv) in both sexes, v) in both temporary and permanent models of ischemia, vi) at least 1 hour after reperfusion, vii) at two or more doses, viii) using a clinically relevant mode of delivery, ix) using both histological and behavioral outcomes, and x) with outcomes at least 4 weeks after injury. Unfortunately, these criteria, as well as the ARRIVE (Animal Research: Reporting of In Vivo Experiments) criteria for design and reporting of preclinical studies, are still rarely applied today. 178
In addition to these criteria, any use of nanotechnology to transport therapeutic cargo must also include the necessary controls to account for the nanoparticle vehicle and any component of the nanoparticle that could result in toxicity, off-site effects, or added therapeutic benefit. For nanoparticles encapsulating a therapeutic, this typically involves adding treatment groups for the empty nanoparticle and the free drug. For therapeutics chemically linked to nanoparticles, controls might also need to include the nanoparticle with and without the linker chemistry. Further complicating preclinical studies, the use of nanoparticles to deliver therapeutics should decrease the necessary dose of the therapeutic; yet, this difference in dose between the therapeutic nanoparticle and the therapeutic in free form introduces an additional variable. Inevitably, the more multifunctional the nanoparticle delivery system, the more controls are needed to account for the potential effects of each component of that system.
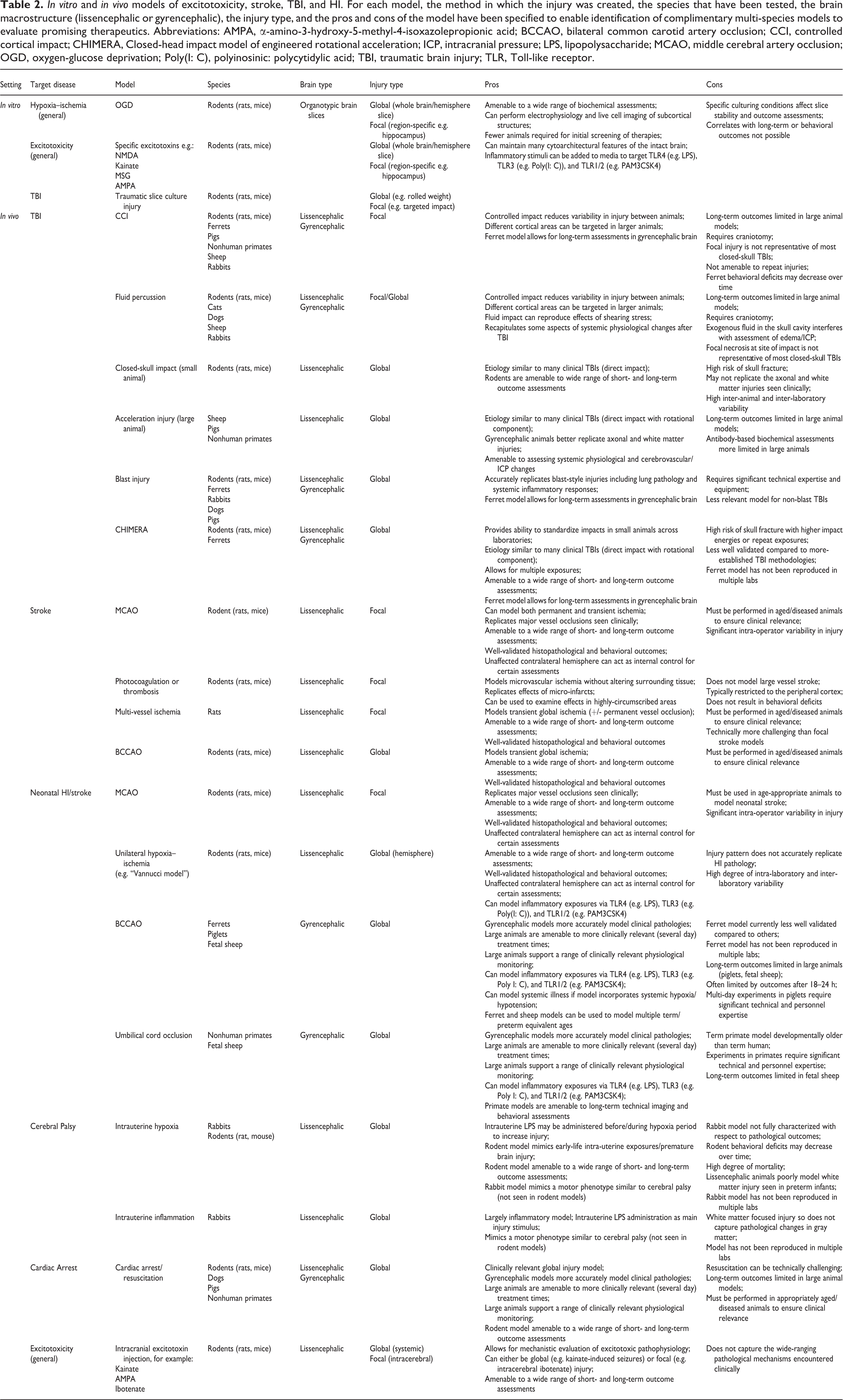
While nanotechnologies might increase drug bioavailability and reduce dosing needs over the longer term, initial preclinical studies often require more experimental groups. Meeting these needs increases costs substantially, especially when scaling nanotherapeutic formulation methods to test in multiple animal models with increasingly larger species. By not testing therapies in multiple animal models, including both rodent species and larger gyrencephalic animals (pigs, sheep, dogs, nonhuman primates, etc.) at multiple different time points in relation to the initial injury, the preclinical pipeline is unlikely to capture the heterogeneity of acute brain injury populations seen clinically. Preclinical work often focuses on reproducibility of injury to maintain statistical power while using small group sizes, and without testing a therapy in the variety of settings outlined in the STAIR criteria, the likelihood of failure in the clinic will remain high. 178 Any anti-excitotoxicity therapy must achieve target site delivery at the right dose at the right timing of injury. Therefore, a robust preclinical pipeline including a combination of complementary models (Table 2) is paramount in the translation of nanotherapeutics for the treatment of excitotoxicity and oxidative stress.
In vitro and in vivo models of excitotoxicity, stroke, TBI, and HI. For each model, the method in which the injury was created, the species that have been tested, the brain macrostructure (lissencephalic or gyrencephalic), the injury type, and the pros and cons of the model have been specified to enable identification of complimentary multi-species models to evaluate promising therapeutics. Abbreviations: AMPA, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; BCCAO, bilateral common carotid artery occlusion; CCI, controlled cortical impact; CHIMERA, Closed-head impact model of engineered rotational acceleration; ICP, intracranial pressure; LPS, lipopolysaccharide; MCAO, middle cerebral artery occlusion; OGD, oxygen-glucose deprivation; Poly(I: C), polyinosinic: polycytidylic acid; TBI, traumatic brain injury; TLR, Toll-like receptor.
Conclusions and looking forward
Delivering drugs that act specifically at diseased target sites at the appropriate dose at the right time is crucial for neurotherapeutics given the sensitivity and precision of brain function. Antioxidant intervention by scavenging injury-associated SOX and other ROS provides alleviation of multiple subsequent neuronal death pathways. Many studies have already shown the therapeutic potential of SOD-loaded nanoparticles or SOD-mimetic platforms, as well as various other antioxidants. However, despite promising inhibition of excitotoxic damage, further work needs to be performed to identify the ideal intervention point for therapeutic efficacy.
Understanding the mechanisms involved in excitotoxicity and oxidative stress can assist the nanoparticle drug delivery field in identifying promising points of intervention. Therapeutic development to inhibit ROS production can be guided by pinpointing the threshold at which ROS overwhelms antioxidant defenses and whether antioxidant uptake would enhance therapeutic effect. Determining whether excessive SOX elevation is primarily generated by NOX after glutamate receptor activation, by respiring mitochondria, or by xanthine oxidase after reperfusion, or whether the SOX source even matters, would better inform how to target the downstream pathology. However, it is imperative to remember that excitotoxicity is not the only disease hallmark at play. Inhibition of ROS in neuroinflammatory pathways could also assist in preventing delayed neurodegeneration that can arise days, months, or even years later. 163,179 Since neuronal excitation and inflammation play roles in survival and growth, there is risk of injuriously over-inhibiting basal neuronal function or over-scavenging ROS and interrupting cell signaling. 40,179 –181
Preclinically, most antioxidant-nanoparticle therapies achieved no greater than 60% reduction in infarct size on ischemia/reperfusion, HI, or TBI injury in vivo. It remains to be elucidated whether ∼60% reduction is a practical limit, or whether additional strategies could further reduce damage. Therapeutic advances achieving greater than 60% infarct reduction could consist of increasing drug concentrations at injured sites, using targeting ligands for improved accumulation or cell-specific uptake at the target site, optimization of the timing of administration, or applying combination therapies with anti-inflammatory agents, pro-regenerative growth factors, or stem-cell based therapies. Furthermore, despite extensive preclinical evidence of antioxidant-nanoparticle efficacy in acute brain injury, research must comply with STAIR and ARRIVE criteria before translation to clinical studies. Finally, though excitotoxicity is a primary mechanism of brain damage in stroke, TBI, and HI, the process may be present in the majority of neurological diseases. 14 Therefore, even when excitotoxicity is not the primary mechanism of pathology, neurological diseases more broadly could benefit from the development of antioxidant-nanoparticle therapies to reduce neuronal injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of General Medical Sciences (Grant #R35GM124677).
