Abstract
We report on the controlled enhancement of the UV emission from ZnO nanoparticles synthesized via hydrothermal sol-gel routes. Various characterization techniques were used in this study to determine the crystal structure, defect formation, morphology and size of the ZnO nanoparticles. Firstly, the crystallinity of the ZnO nanoparticles was assessed by X-ray diffraction. The role of the precursor quantities on their morphology and bandgap states was investigated via transmission electron microscopy and photoluminescence spectroscopy, respectively. Subsequently, the impurity content and related bonds were evaluated by Fourier-transform infrared spectroscopy. In this work, the ZnO nanoparticles manifest a sharp UV emission along with a subdued green emission in the visible region. Conjoining ZnO with carbon nanotubes (CNT) suppressed the visible emission, as well as blueshifted and intensified the UV emission by ∼5-fold. These results suggest that CNT are effective in passivating the surface states of ZnO nanoparticles.
Introduction
ZnO is a semiconductor material that has attracted interest for a wide range of applications, such as solar cells, 1 light emitting diodes2,3 and other optoelectronic devices.4,5 Its most significant properties are a wide bandgap of 3.36 eV along with a high exciton binding energy of 60 meV at room temperature. 6 ZnO is therefore considered suitable for a variety of short-wavelength devices, viz. Ultraviolet (UV) light emitting diodes, UV lasers and tuneable UV photodetectors.7,8 For the preceding devices, nanostructures, especially nanoparticles of ZnO have been widely studied.9,10 However, obtaining pure UV emission from nanoparticles of ZnO is challenging due to the large quantities of surface and volume defects they harbour. These defects tend to augment the visible emission at the detriment of the UV emission. Their photoluminescence (PL) spectrum is composed of two main emission peaks: a near band-edge emission (NBE) in the UV region and defect level emission (DLE) in the visible region. The NBE originates from the recombination of free excitons due to band-to-band transitions, whereas the DLE arises from surface or volume defect states, such as oxygen vacancies (Vo) and zinc interstitials (Zni). 11 In effect, surface defects or impurities trap a large number of photogenerated electrons. Furthermore, the NBE diminishes when the particle size reduces or the surface to volume ratio increases, whereupon the DLE dominates the emission spectrum owing to a higher amount of surface defects.12–15 Therefore, strategies to enhance the NBE consist of supressing defect formation during synthesis or post-growth.16,17 Several, methods have been investigated to that end, which include surface state modification of ZnO by TiO2 and Al2O3 capping layers; hydrogenation18–20; annealing under N2 and O221–23; controlling defects during synthesis itself24,25; and conjoining them with metal nanoparticles displaying surface plasmon resonance.26,27
Organic and inorganic materials have witnessed enhancements in physical, chemical and structural properties when combined with CNT. For instance, CoFe2O4-CNT present higher electromagnetic shielding, while as Sn–Ag–Cu composites benefit from better structural and electrical properties in solder joints for certain concentrations of CNT.28,29 Similarly, for flexible electronics, polyaniline-CNT hydrogels have demonstrated excellent electrical conductivity owing to their high dispersion in hydrogel-based matrices. 30 Furthermore, combining CNT with rubber increases the resistance-strain response of the composite. 31 CNT are produced by several different synthesis routes such as chemical vapour deposition, electric arc discharge and laser ablation. 32 Due to their inertness, functionalizing CNT, that is, attaching functional organic moieties to the walls of the CNT is a prerequisite to combining them with other organic and inorganic materials. Functional moieties include amine, ester, carboxyl, carbonyl and hydroxyl groups.32,33 Sonication is also a viable method of activating the CNT walls and rendering them receptive to inorganic nanoparticles.15,34–36
In a previous work, the influence of hydrated precursors on the surface defects of ZnO nanoparticles synthesized via non-aqueous sol-gel routes was investigated. The results demonstrated that the hydrates present in ZnO tend to enhance the NBE and diminish the green emission component of the DLE due to improved oxidation of ZnO during synthesis. 37 However, even though hydrated precursors are efficient at suppressing volume-related defects, surface-related defects nevertheless persist. Subsequently, the ZnO nanoparticles were combined with carbon nanotubes (CNT) in order to passivate residual defect states, 38 which then enhance the UV emission. Lastly, enhancing the UV emission from ZnO-CNT nanohybrids could also be beneficial for other applications, including photocatalysis, 39 charge separation, 40 field electron emission, 41 photoresponse, 42 gas or electrochemical sensors43,44 and other optoelectronic devices. 45
This manuscript reports on the effect of varying NaOH and Zn precursor ratio on the properties of ZnO nanoparticles prepared via hydrothermal sol-gel routes. In addition, these ratios provide an approach to controlling the size of the nanoparticles and consequently, the formation of surface defects. Moreover, by combining the as-synthesized ZnO nanoparticles with CNT, it is further possible to reduce the residual surface states in order to obtain a notable enhancement in the UV emission. The optical properties are discussed in terms of the synthesis conditions, crystal structure, chemical properties and morphology of the samples.
Materials and methods
Synthesis
ZnO
All the chemicals used for the syntheses of ZnO nanoparticles were of analytical reagent grade. In order to prepare a 0.05 M solution, 219.5 mg of zinc acetate dihydrate (Zn(CH3CO2)2.2H2O) (99.9%, Aldrich) was dissolved in 20 ml of aqueous ethanol (70%) in a beaker placed in a water bath. The solution was maintained at 600C under continuous magnetic stirring until a transparent solution was obtained. Further, 0.075 M, 0.10 M, 0.125 M and 0.15 M solutions of NaOH (99.9%, Aldrich) in 20 ml of aqueous ethanol (70%) were prepared and added drop wise to 0.05 M precursor solutions to obtain 1:1.5, 1:2, 1:2.5 and 1:3 M ratios, respectively. Thereafter, the mixtures were maintained at 600C for 2 h and cooled to ambient temperature. After cooling, white ZnO precipitates settled at the bottom of the reaction vessel. The resulting solutions containing ZnO nanoparticles were then centrifuged at 4500 r/min for 6 min and dried for 24 h in air at 600C. This resulted in an agglomeration of ZnO nanoparticles in the form of a porous pellet, which is typical after drying nanoparticles synthesized via sol-gel and hydrothermal synthesis routes. These pellets were thereafter gently crushed using a pestle and mortar to obtain very fine nanopowders of ZnO nanoparticles. Nanopowders consist of well-dispersed ZnO nanoparticles that are necessary to create ZnO-CNT hybrids.
ZnO-CNT nanohybrids
For the preparation of ZnO-CNT nanohybrids (16.66 wt%), 2 mg of NANOCYL NC7000 multi-walled carbon nanotubes (MWCNT) were mixed with 10 mg of ZnO nanoparticles synthesized with precursor to NaOH molar ratios of 1:1.5 and 1:3. Absolute ethanol was added to the mixture and the solution was sonicated for 5 min. Thereafter, the ZnO-CNT mixtures were dried in air at 600C for 24 h. The resulting ZnO-CNT pellets were further crushed to obtain a very fine black powder.
Characterization
X-ray diffraction patterns were collected in Bragg–Brentano geometry using a Bruker D8 Discover diffractometer (Bruker AXS, Germany) with CuKα1 radiation (λ = 0.15,406 nm) selected by a Ge (111) monochromator and LynxEye detector. Transmission electron microscopy (TEM) was carried out on a JEOL 2010 LaB6 TEM (JEOL, Japan) operating at 200 kV in TEM mode and providing a point-to-point resolution of 1.9 Å. Fourier-transform infrared (FTIR) spectrometer (Nicolet is 10 Thermo Scientific, Germany) was cooled with liquid nitrogen for FTIR measurements in the range of 540–4000 cm−1. The optical absorbance of ZnO and ZnO-CNT nanohybrids were determined using a NANOCOLOR UV-VIS II spectrometer (MACHEREY-NAGEL, Germany) in 200–900 nm region. The bandgap of ZnO and ZnO-CNT nanohybrids were subsequently calculated with Tauc plots. PL spectroscopy was carried out at room temperature with an excitation wavelength of 365 nm of a LSM-365A LED (Ocean insight, USA) with a specified output power of 10 mW. The emission was collected by FLAME UV-–Vis spectrometer (Ocean optics, USA) with spectral resolution 1.34 nm. Optical images of ZnO were taken under UV lamp ZLUV220 with an excitation source of 365 nm.
Result and discussion
List of ZnO samples and ZnO-CNT nanohybrids synthesized in this work. The quantity of the Zn(CH3CO2)2.2H2O precursor was fixed at 0.2195 g.

Structure and morphology
XRD patterns of the ZnO samples are shown in Figure 1. The peaks (100), (002), (101), (102) and (110) correspond to the hexagonal wurtzite structure (a = 3.25 Å and c = 5.20 Å) of ZnO (JCPDS, Card Number 36–1451).46,47 The spectra also indicate the presence of unreacted precursor Zn(CH3CO2)2.2H2O (marked with *). The precipitation of ZnO nanoparticles are described by the following chemical reactions
48
: XRD patterns of ZnO samples Z–1, Z–2, Z–3 and Z–4. The peaks marked with (*) indicate the presence of unreacted precursor.
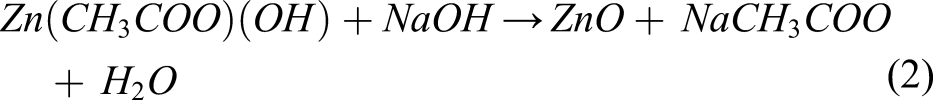

The reactions of equations (1) and (2) indicate that the formation of ZnO nanoparticles depends on the quantity of NaOH that controls the reaction kinetics. 49 Varying the pH via the NaOH content is commonly used in non-aqueous or hydrolytic sol-gel routes for ZnO nanoparticle synthesis. In hydrothermal synthesis, increasing the quantity of NaOH for a fixed concentration of Zn(CH3CO2)2.2H2O precursor, increases the pH of the solution and the reaction kinetics, which in turn, decreases the particle size and modifies the surface defects. Additionally, the presence of Zn(CH3COO) (OH) suggests an incomplete reaction, which can therefore be attributed to an insufficient quantity of NaOH. The XRD pattern of sample Z–1 exhibits the most intense diffraction peaks of the unreacted precursor followed by Z–2. A less intense peak is also visible for the sample with the highest quantity of NaOH, that is, Z–4. On the other hand, for sample Z–3, this peak is the least intense. Co-workers usually calcine the ZnO sample in order to remove impurities and unreacted precursors. 48 However, calcination changes the defect states of ZnO. Our aim is to evaluate the properties of the as-synthesized ZnO nanoparticles.

List of particle sizes estimated from the Scherrer equation with their corresponding shapes obtained from TEM.

TEM analysis on the as-synthesized ZnO nanoparticles confirmed their actual size and shape. ZnO-CNT samples were not characterized by TEM, as conjoining ZnO with CNT does not produce any changes to shape, size or morphology of the nanoparticles as shown in previous studies.34–36 Figure 2 provides an overview of the sizes and morphologies of ZnO nanoparticles summarized in Table 2. Different nanoparticle morphologies are visible, including hexagonal, rectangular, needle-like and spherical (Table 2). Hexagonal, rod and rectangular shaped ZnO nanoparticles are obtained for sample Z–1 synthesized with the lowest NaOH (Figures 2(a) and (b)). On increasing the NaOH quantity, ZnO nanoparticles of Z–2 display a needle-like morphology (Figure 2(c)) along with several hexagonally shaped nanoparticles (Figure 2(d)). An additional increase in the NaOH quantity in Z–3 produced smaller nanoparticles (Figures 2(e) and (f)) with variable shapes. The highest NaOH quantity of Z–4 led to the smallest nanoparticles (Figures 2(g) and (h)). The decrease in the particle size with increase in NaOH quantity is attributed to an increase in the reactivity of the precursors. The reaction consists of a rapid conversion of Zn(CH3COO)2.2H2O to an intermediate phase of Zn(CH3COO) (OH) and then to ZnO, with relatively low aggregation with a reduced average particle size.
51
On the other hand, the transformation in the shape of the nanoparticles can be attributed to an excess of hydroxyl ions in the solution due to increase in the NaOH quantity. These additional hydroxyl ions are adsorbed on the surfaces of the ZnO nanoparticles, blocking their growth along the c-axis and in turn, leading to an unselective and uncontrolled hydrolysis nucleation.
52
With the increase of NaOH quantities, the ZnO nanoparticles no longer follow their preferential growth along the c-axis and are transformed into nanoparticles with smoother facets, for example, Z–4. TEM images of the as-synthesized ZnO samples. Overview images: (a) Z–1 (c) Z–2 (e) Z–3 and (f) Z–4. High magnification TEM images: (b) Z–1 (d) Z–2 (f) Z–3 and (h) Z–4.
In Figure 3, the FTIR spectra provide information on various vibrational bonds produced during the synthesis of ZnO, ZnO-CNT nanohybrids and pristine CNT. The band located between 700-500 cm−1 corresponds to the Zn-O stretching bond, which confirms the formation of ZnO.
53
The vibration band of ZnO downshifts from ∼680 cm−1 to 640 cm−1 for increasing NaOH quantities, suggesting changes in the morphology of ZnO nanoparticles, analogous to TEM results.
52
The broad band near 3300 cm−1 corresponds to OH-stretching, originating from water and NaOH.
54
The OH-stretching band appears to be less intense and narrower for sample Z–1, but more intense and broader for samples Z–2, Z–3 and Z–4. This variation in band broadness and intensity is attributable to unequal quantities of OH− functional groups chemisorbed on the ZnO nanoparticle surfaces. In addition, the OH− functional groups bonded to Zn, thart is, the Zn-OH bonds
55
are located between 1000 and 800 cm−1. Bands between 3000 and 2800 cm−1 correspond to CH-stretching of alkane groups. Furthermore, the bands situated between 1600 and 1100 cm−1 comprise of several acetate groups (-COOH).
56
These groups are adsorbed on the surfaces of the ZnO nanoparticles during synthesis. In particular, the bands located between 1600 and 1500 cm−1 and 1500 and 1400 cm−1 correspond to symmetric and asymmetric stretching vibrations of the C=O bond, respectively.53,57,58 The broad peak at around 1350 cm−1 corresponds to C–O bonds. With variations in the NaOH quantities, the vibration peaks of C=O and C–O bonds manifest changes in their band intensities, owing to morphological changes of the nanoparticles. The bands (marked as *) between 1300 and 1100 cm−1 are attributed to the carboxylate (COO−) functional group originating from unreacted Zn(CH3CO2)2.2H2O precursor, analogous to XRD results. FTIR spectra of ZnO nanoparticles (labelled Z–1, Z–2, Z–3 and Z–4), ZnO–CNT nanohybrids (labelled Z–1–CNT and Z–4–CNT) and pristine CNT.
The FTIR spectra of ZnO-CNT nanohybrids, that is, samples Z–1–CNT and Z–4–CNT contain similar bonds to the as-synthesized ZnO samples. However, a broad peak at 2130 cm−1 is present for the ZnO-CNT samples. This peak is also visible for pristine CNT and can therefore be safely attributed to CO2 peak. 59
Optical properties
The bandgaps of ZnO and ZnO-CNT hybrids were calculated through UV–Vis absorption spectroscopy followed by Tauc plots presented in Figure 4. The absorption properties of ZnO-CNT are similar to the as-grown ZnO nanoparticles. In fact, MWCNT are prone to bundling, supressing their optical signals. In addition, they absorb in the infrared region owing to Van Hove singularities. Therefore, no contribution to the near band absorption of ZnO is expected.
36
The bandgaps of these ZnO samples range from 3.16 eV to 3.29 eV. Bandgaps of 3.27 eV have also been observed for ZnO samples with a high concentration of surface defects.
37
These variations in the bandgap are a result of syntheses via sol-gel routes .60,61 The absorption spectra of the ZnO samples revealed a sharp shoulder at 3.3 eV, stretching down to 2 eV–2.5 eV. However, shoulders related to defect level absorption were absent. For samples Z–1–CNT and Z–4–CNT in Figures 4(a) and 4(d), (a), broader absorption shoulder starting at 3.3 eV and extending below 2 eV is visible. The presence of metallic MWCNT therefore has an effect on the near infrared absorption of the hybrid samples as explained above. In both ZnO-CNT samples, a slight decrease in bandgap is observable, suggesting modifications to specific defect states investigated in the next section by PL spectroscopy. Tauc plots of samples (a) Z–1 and Z–1–CNT, (b
The emission properties of the as-synthesized ZnO and ZnO-CNT nanohybrids were examined through room temperature PL measurements. The 365 nm (3.4 eV) excitation source was used to incite band-to-band transitions in ZnO and ZnO-CNT samples with bandgaps between 3.17 eV and 3.29 eV. The PL spectra of Figure 5 manifest typical PL emission characteristics of ZnO nanoparticles comprising of the NBE and DLE. It is noteworthy that the unreacted acetate precursor does not produce any PL emission under 365 nm excitation; the PL emissions emanating from the samples therefore originate from the ZnO nanoparticles alone. Additionally, the ratio of the NBE to DLE provides a qualitative measure of the crystallinity of the ZnO samples; a higher ratio signifies a lower number of intrinsic defects. PL emission spectra of samples (a) Z–1, (b) Z–2, (c) Z–4 (d) Z–1–CNT, (e) Z–3 and (f) Z–4–CNT. Vertical marked dashed lines indicate major emission components. The insets are optical images taken under 365 nm illumination.
The major emission wavelengths are deconvoluted with the Gaussian function in Figure 5. In general, all the samples exhibited similar PL emission tendencies; however, differences in quantum yield are observed with variations in NaOH quantities. For example, the quantum yield of the NBE for sample Z–4 is ∼50% lower than Z–1. This suggests that the increase in NaOH tends to create non-radiative defect centres within the ZnO nanoparticles, which in turn lower the UV emission intensity. Furthermore, the increase in the DLE or a lower NBE to DLE ratio also supports the presence of surface defects. Other factors, such as shape, size and post-synthesis treatment also modify surface defects. 37 The varied morphologies and size distributions of the ZnO nanoparticles have therefore an important influence on the NBE to DLE ratios. However, for samples combined with CNT, that is, Z–1–CNT and Z–4–CNT, a ∼5-fold increase of the UV emission intensity is observed along with a slight blueshift compared to Z–1 and Z–4, respectively. These preceding modifications clearly suggest that sonication is effective in creating ZnO-CNT nanohybrids. The sonication time of 5 min was optimized in other studies by the authors,15,36 where the mechanism behind nanoparticle decoration on CNT was also explained. Defective areas of CNT, including kinks, bends or zones where C-C bonds break down, are active sites for anchorage of nanoparticles. The advantage of sonicating CNT in ethanol is 2-fold. Firstly, ethanol allows a homogeneous dispersion of CNT; secondly, sonication breaks down C-C bonds on the outer walls of the CNT, facilitating their decoration with nanoparticles.
The NBE to DLE ratios are the highest for these CNT-based samples, confirming a notable reduction or passivation of defects. The UV emission peaks for ZnO samples in Figure 5 lie between 393 nm and 397 nm and correspond to the transitions from Zn shallow donor levels to the valance band.47–49 Only samples Z–1 and Z–4 were combined with CNT as they were synthesized with the lowest and highest quantities of NaOH. These samples also present the lowest and highest bandgaps from Tauc plots, the largest and smallest particle sizes from TEM study and the least to most intense DLE. Therefore, understanding the effect of surface passivation by CNT for these two samples will help elucidate the mechanism of UV emission enhancement as a function of synthesis parameters. For ZnO-CNT nanohybrids, the blueshifted UV emission peaks situated between 383 nm and 386 nm correspond to the band-to-band transitions of ZnO. This implies that the defects in these samples are surface related and therefore, CNT clearly play a role in their passivation, whereupon they turn optically inactive. On the other hand, Z–4–CNT still manifests a hint of DLE even though its intensity has largely reduced. The insets of Figure 5 display optical images of the ZnO and ZnO-CNT samples under 365 nm UV light illumination. A violet-blue hue corresponding to surface and bulk Zn interstitials mainly emanates from the ZnO samples. The emission is the most significant for sample Z–3 (inset of Figure 5(e)) and less intense for other samples (insets of Figure 5(a), 5(b) and 5(c)). On the other hand, ZnO-CNT samples emanate only in the UV; in the insets of Figures 5(d) and (f), visible PL emission is absent.
A more detailed analysis was carried out for the DLE component of samples Z–1, Z–4, Z–1–CNT and Z–4–CNT in Figure 6. The DLE is a combination of optically active point defects, such as oxygen vacancies (VO), zinc vacancies (VZN), oxygen interstitials (Oi) and antisites (OZN), zinc interstitials (Zni), as well as their complexes.
62
It is noteworthy to mention that Z–1–CNT did not produce any visible luminescence. On the other hand, Z–4–CNT manifested a clear decrease in the visible luminescence. The Gaussian deconvolution of sample Z–4 in Figure 6, provides the most significant emission peaks, such as the green emission at ∼2.2 eV, yellow-orange emission at ∼2.1 eV and red emission at ∼1.95 eV. DLE of the samples is dominated by a yellow-green emission in addition to a less significant red component at 1.95 eV. Besides, the highly intense UV emission of the ZnO-CNT samples overwhelms the DLE, which is therefore not discernible in the PL photographs in the insets of Figure 5 (c) and 5(f). The yellow-green component is related to Vo; in particular, Vo+ at 2.5 eV due to volume related oxygen vacancies is absent or negligible in our samples. The use of aqueous ethanol has therefore ensured an appropriate oxygenation of ZnO leading to the suppression of Vo+. However, the emission at ∼2.2 eV is related to the surface oxygen vacancies or Vo++. In this study, it is the major emission component of the DLE. PL was carried out in air ambient; therefore, the likelihood of adsorption of OH− and O2 is high. Chemisorbed oxygen species provoke an upward band bending of ZnO, boosting electron-hole pair separation and in turn, delaying excitonic recombination. Subsequently, the surface trapped holes tunnel to deep levels i.e. to Vo+ and convert them into Vo++. Eventually, an electron from the conduction band then recombines with the Vo++ and emits the DLE at 2.2 eV. On conjoining with CNT, the adsorption of O2 and OH− is hindered; thereupon, the upward band bending is reduced. Thus, the tunnelling of holes to deep levels is suppressed and the probability of NBE increases.
63
Visible PL emission of samples Z–4–CNT, Z–4 and Z–1. The dashed curves are the Gaussian deconvolution of the most significant peaks of sample Z–1.
Furthermore, variations in the ZnO nanoparticle morphologies engender changes in their defect states. Rod-like or bean-like ZnO tend to harbour lower amounts of Vo, while as in spherical ZnO nanoparticles, it is the major emission component of the DLE. 64 On the other hand, hexagonal ZnO is made up of six facets: four polar and two non-polar. 65 The latter has tendencies to adsorb oxygen species from air. However, polar facets are either O or Zn terminated and therefore harbour related point defects. Cubic or rectangular shaped ZnO has two polar and two non-polar surfaces. 66
Additionally, the size of Z–4 nanoparticles was the smallest, implying the highest surface to volume ratio among the as-produced ZnO samples. The increase in the surface to volume ratio therefore explains the more intense 2.2 eV emission for sample Z–4. On combining with CNT, the Vo++ emission significantly recedes (Z–4) or disappears (Z–1). Furthermore, the red emission at 1.95 eV also diminishes for both ZnO-CNT samples. This emission has been regarded as transitions related to Zni, usually observed in ZnO samples synthesized in oxygen rich conditions.67,68 The yellow-orange emission at around 2.1 eV relates to the transitions from Zni to Oi. 69 The residual DLE of Z–4–CNT is likely due to the incomplete passivation of the ZnO surface. The peak intensities of the NBE for samples Z–1–CNT and Z–4–CNT are at ∼383 nm and ∼385.5 nm, respectively. Therefore, a difference of ∼2.5 nm between the peak NBE intensities exists for these two samples. Since the Z–4 nanoparticle sizes are smaller, signifying that their surface is larger, a higher quantity of CNT would therefore be necessary to ensure optimum surface coverage. The latter would ascertain passivation of surface defects and a further blueshift of the NBE for Z–4–CNT.
Conclusion
We have demonstrated a feasible route to control the UV emission from the ZnO nanoparticles via the suppression of both volume and surface defects. The ZnO nanoparticles were prepared via hydrothermal sol-gel routes by varying Zn precursor to NaOH ratios. In effect, the quantity of NaOH was instrumental in determining the kinetics of the ZnO precipitation reaction. Furthermore, the morphology and sizes of the ZnO nanoparticles are dependent on these ratios. Various morphologies of ZnO nanoparticles i.e. hexagonal, rectangular, needle-like and spherical were obtained as a result of the synthesis conditions. TEM study revealed that for the highest NaOH quantity generated the smallest ZnO nanoparticles. Additionally, the use of aqueous ethanol supported the synthesis of well-oxygenated ZnO nanoparticles as volume related defects were essentially absent in the PL emission spectra. In all cases, the ZnO nanoparticles manifested an intense UV emission and a negligible surface related DLE. The DLE was suppressed and the NBE was blueshifted and further amplified ∼5-fold on conjoining with CNT.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been supported by the European Regional Development Fund project grant number TK134 “Emerging orders in quantum and nanomaterials” EQUiTANT, EMÜ Astra project EMBio “Value-chain based bio-economy”, and Eesti Maaülikool (EMÜ) Bridge Funding grant number P200030TIBT. We acknowledge use of the Norwegian national resource centre for X-ray diffraction, scattering and imaging (RECX), Research council of Norway project number 208896.
