Abstract
Kretschmann-based surface plasmon resonance sensor utilizing chromium and gold nanofilms is ideal for label-free biomedical sensing. In this work, Taguchi’s L9 orthogonal array method was used to optimize the effects of three control factors and noise factor, which are the incident optical wavelength, chromium and gold nanofilm thicknesses, and their root-mean-square surface roughness, on the performance of the Kretschmann-based surface plasmon resonance sensor. The control factors were varied at three levels for a novel multi-response optimization of the Kretschmann-based surface plasmon resonance sensor for the minimum reflectivity, the full-width-at-half-maximum, and the sensitivity of 3% glucose detection, executed using Lumerical’s two-dimensional finite-difference time-domain method. Using Taguchi method, the best control factor setting in air was A3B2C2 corresponding to 785 nm optical wavelength, 0.5 nm chromium, and 50 nm gold layer thickness, respectively, with minimum reflectivity of 0.0017%, full-width-at-half-maximum of 0.4759°, and glucose-sensing sensitivity of 106.73°·RIU−1. The detection accuracy and quality factor were 0.01 and 224.26 RIU−1, respectively. It was also indicated that chromium nanofilm thickness of 0.5–3 nm and its root-mean-square surface roughness has a negligible factor effect compared to other control factors. Taguchi method’s factor effect analysis showed that for chromium layer thickness of 1–3 nm, the minimum reflectivity values are predominantly determined by the gold layer thickness with 75% factor effect, followed by optical wavelength with 11%. Factor effect of full-width-at-half-maximum is determined by optical wavelength (57%), followed by gold layer thickness (38%). Sensitivity is 88% determined by optical wavelength and 10% determined by gold layer thickness. The Kretschmann-based surface plasmon resonance glucose sensor with the best glucose-sensing sensitivity was at optical wavelength of 632.8 nm with a higher sensitivity value of 163.415°·RIU−1 but lower detection accuracy and quality factor values of 0.001 and 24.86 RIU−1, respectively, compared to near-infrared wavelength of 785 nm. In conclusion, finite-difference time-domain and Taguchi method is suitable for multi-response optimization of control and noise factors of Kretschmann-based surface plasmon resonance sensors.
Keywords
Introduction
Nowadays, surface plasmon resonance (SPR)-based sensors are receiving profound attention for its fast response and high accuracy to measure biomolecular interaction in real time and label-free environment. 1 –4 Even in the ongoing COVID-19 pandemic, SPR-based biosensors have emerged as a promising and alternative solution for the diagnosis of the lethal virus. 5 The inclusion of localized SPR and metamaterial effects are also popular sensing alternatives. 6,7 SPR sensing is strongly dependent on the type of thin metal film with different refractive index (RI) and layer thickness in the nanometer scale. Gold (Au) and silver (Ag) are the metal nanofilms that are frequently utilized for marketable SPR sensors due to their steadiness and optical behavior. On the other hand, copper (Cu), aluminum, and Ag are excessively vulnerable to oxidation and thus debase the effectiveness of Kretschmann-based SPR (K-SPR) sensors. Iron, nickel, and chromium (Cr) are theoretically predicted with high losses and are impractical to be used in industrial devices. 8 –10 Recently, Trejo-Tzab et al. introduced a very simple way to prepare a new nanocomposite comprising N-TiO2-x/Au/Ag for potential SPR sensor applications. 11 This work is an extension of our previous work on K-SPR sensors where we have extended the current work to include root-mean-square (RMS) surface roughness of the SPR layers as noise factor as well as optimizing multi-responses of the full-width-at-half-maximum (FWHM), minimum reflectivity (R min), and sensitivity of the K-SPR sensor. 12
As in previous work, Taguchi method was used in this study to identify the optimum parameters that will produce a robust high-sensitivity multi-response Au nanofilm K-SPR glucose sensor. Taguchi method is a well-known instrument to improve efficiency during research and innovative work. It reduces the number of tests immensely and increases the efficiency using a unique design of orthogonal arrays (OAs) to consider the control factors with only a few number of trials. It helps to identify the impact of numerous control and noise factors on the quality characteristics of the device. Besides that, the signal-to-noise ratio (SNR) in Taguchi method assists product designers or manufacturers to easily identify the optimal combinations of the targeted design parameters. Many examples on the usage of Taguchi method in the design and development of various nanoscale optical devices can be found in the literature. 13 –16
Ideally, every fabricated device is expected to have a smooth, uniform, and perfect thin film surface. In reality, there exists a device performance setback due to thin film surface roughness. 17 –19 In nanophotonics, a smooth surface is ideal for producing quality devices because surface roughness induces scattering and reflection which can interfere with optical performances. 20,21 Some researchers have used Taguchi method to investigate the optimal parameters on surface roughness and have produced robust designs in various fields. 22 –25 To the best of our knowledge, the surface roughness and multi-response output of nanophotonic devices have never been studied before. 12,26 Therefore, in this work, a novel approach was undertaken where the surface roughness of Au and Cr nanofilms of the sensor design was taken into consideration to optimize the multi-response output of the K-SPR sensor. This can be achieved due to the uniqueness of Taguchi method in predicting the best combination of process or control factor levels, insensitive to noise factors. Moreover, almost all devices have multi-responses to be considered before they can be recommended as good sensors. In contrast to the conventional way, many researchers have reported the use of Taguchi method to optimize multi-response design for numerous applications due to its simplicity and efficiency. 27 –34 Taguchi method can combine these multi-responses at an optimal range of parameters to maintain the quality and robustness of a device design in the field of nanomaterial and nanotechnology.
Previously, the ideal thickness of Au at 50 nm 35 –38 demonstrated the most sensitive K-SPR-based sensor irrespective of whether a nanofilm Cr adhesion layer exists or not. Therefore, the thickness of 50 nm Au as the metallic material has a strong effect on the K-SPR sensor. On the other hand, Ag and Cu K-SPR sensors exhibit lower sensitivity. But these K-SPR sensors were only studied when the visible wavelength was at 670 nm. 39 For further investigation, the visible wavelength of 632.8 nm was studied in this work along with the near-infrared (NIR) incident light of 785 nm. 40 –42 Previous studies on Au nanofilm K-SPR sensors have showed a decrease in SPR resonance angle and FWHM with increasing optical wavelength. Hence, longer optical wavelengths (>750 nm) provide the largest dynamic range to measure RI changes caused by analyte adsorption to the sensor surface. Larger resonance angle shifts and sensitivity were also observed for shorter optical wavelength such as at 600 nm, but the resonance angle shift for aqueous media at this visible wavelength was beyond the measurable angular range of the K-SPR instrument. Longer wavelengths such as 785 nm were also proven to produce K-SPR sensors with optimum detection accuracy (DA) and quality factor (Q-factor) and allow thicker sensing layers to be measured and used for sensing. 43
In this work, the RMS surface roughness of the Cr adhesion layer and Au thin film was designated as noise factor and multi-responses of R min, FWHM, and sensitivity were considered simultaneously to produce a robust multi-response Cr/Au K-SPR sensor, analyzed in mediums of air, water, and glucose and subsequently optimized using Taguchi method.
Methodology
Materials
In this work, three materials were considered; glass prism or borosilicate glass (BK7), Cr, and Au. The optical properties of these materials depend on the incident wavelength that propagates through them. Table 1 shows the value of the RI for the real, n, and extinction coefficient, k, for the thin films at different wavelengths based on the BioNavis database. The RI for air is 1. The thickness of the prism (BK7) is fixed accordingly to match the wavelength of 785 nm as the platform while Cr and Au were changed according to the experiment.
Optical properties of the K-SPR sensor materials at different wavelength.
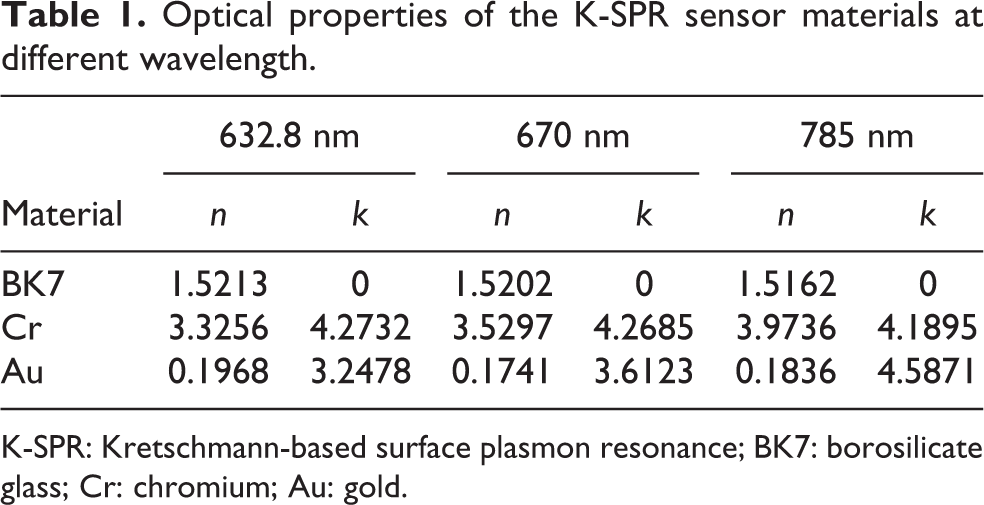
K-SPR: Kretschmann-based surface plasmon resonance; BK7: borosilicate glass; Cr: chromium; Au: gold.
Two-dimensional finite-difference time-domain simulation
The numerical analysis of various parameters was carried out using two-dimensional (2D) finite-difference time-domain (FDTD) simulation from Lumerical. The conformal mesh technology was used for mesh refinement method. This meshing algorithm was set as “conformal variant 1” that is available for all materials including metals and the minimum mesh step is 0.01 nm. The maximum mesh override region was set as 0.05 nm (dx) and 0.1 nm (dy) for x and y directions, respectively. The range of mesh accuracy from low to high is 1–8, and simulations were executed at high accuracy of 6 with simulation time of 1500 fs. The wavelength of the incident optical light, λ, was considered as 632.8, 670, or 785 nm. This incident light propagated into the prism, Cr adhesion layer, and Au nanofilm layer and is finally refracted to the air. Since the physical adhesion of Au nanofilms to BK7 glass prism is low, Cr nanofilm is deposited as an adherent between them. 44 –46 The range of Cr layer thicknesses evaluated in this work was 0.5–3 nm, while the Au nanofilm thicknesses were 40–60 nm. The metal adhesion thickness of 0.5 nm has been reported to be the best for Au-based thermoplasmonic applications. 47 The adhesion layer thickness of below 1 nm produces a robust K-SPR sensor without performance deterioration. 48 The presence of a 1.2 nm thick Cr nanofilm will reduce the plasmon propagation distance to one-fifth of the total propagation as compared to a setup without the presence of Cr at the Au and silicon dioxide interface. 49 In designing the optimum model of the Au K-SPR sensor, Taguchi method was applied to design the experiment. The graphical illustration of the Au K-SPR sensor layers is shown in Figure 1. The resonance angle, θ, in the K-SPR curve is described as an angle at which a sharp dip or R min takes place in the optical reflection spectra. Lower R min and narrower FWHM values are preferred due to the emergence of a deeper resonance peak for an effective detection of the K-SPR sensor. The results in FDTD calculation are governed by the Fresnel equation that calculates the reflectivity using transfer-matrix method.

Graphical illustration of the Au K-SPR sensor based on Kretschmann configuration. Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Taguchi method
Taguchi method was utilized to optimize the control and noise factor variations on the K-SPR sensor. The control factors and its levels are listed in Table 2. According to Taguchi method, for three control factors, the suitable OA is L9 as constructed in Table 3. Therefore, L9 OA which consists of nine experiments was used to investigate the impact of three control factors on R
min, FWHM, and glucose sensor sensitivity which contributed to the multi-response performance of the Au-based K-SPR sensor.
12,26
The aim of this nine-row experiment is to obtain a lower R
min value as well as a narrower/smaller FWHM and optimized using Taguchi’s SNR of Smaller-the-Better. The SNR (Smaller-the-Better),

Control factors and their levels for Taguchi’s L9 experiment for K-SPR sensor.

K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium; Au: gold.
Taguchi’s L9 orthogonal array for K-SPR sensor optimization.
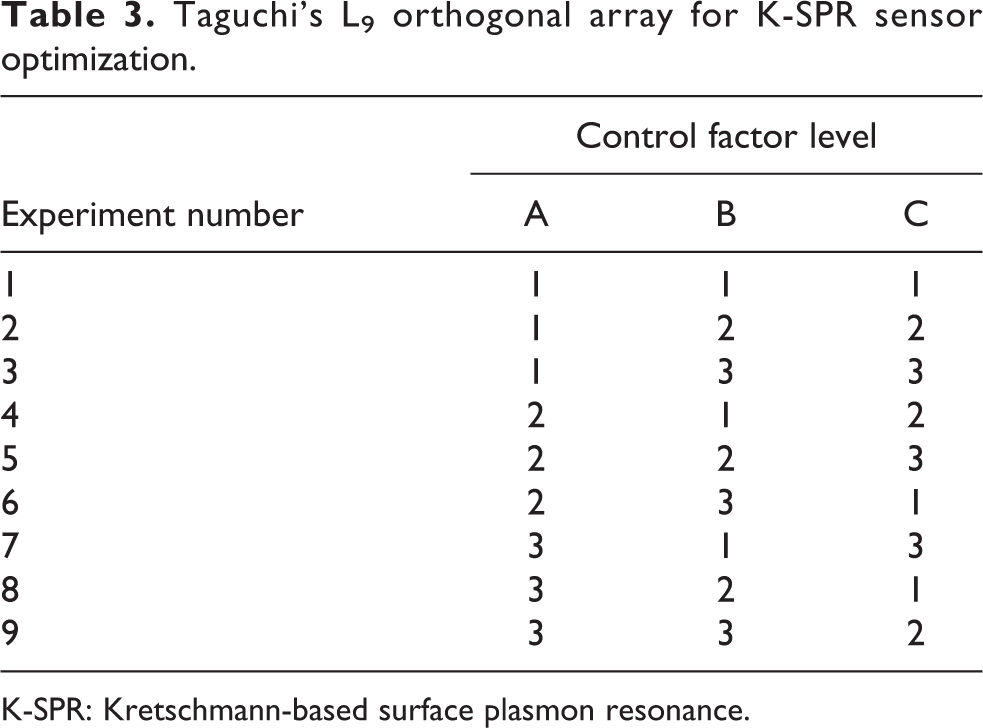
K-SPR: Kretschmann-based surface plasmon resonance.
K-SPR measurement
Upon Taguchi method optimization, SPR Navi 200 L instrument purchased from Bionavis Finland and available at IMEN-UKM, was used to run the experimental work based on the best setting parameters. The instrument employed the Kretschmann configuration, similar to the illustration given in Figure 1. The wave number of the incident light is enhanced to be equal to the excited surface plasmon by coupling them via a prism. The interface between the BK7 glass slide and the BK7 prism contains an index matching gel to match the RI and eliminate air gaps.
Results and discussion
FDTD simulation of L9 OA
Firstly, the FDTD simulation was run based on the L9 OA as shown in Table 3. It simulated the Maxwell’s equation with ultrafine setting and mesh override for high reliability and accurate results. Figure 2 shows the smooth Au K-SPR sensor performance of the L9 OA. These K-SPR curves occur when the wave vector of the electromagnetic waves from surface plasmons (SPs) is equal to the wave vector of the incident light source. The SPs are the collective free electron density that oscillates on surfaces of metal contact with dielectric media. The R min and FWHM of the K-SPR curves of the nine experiment runs in Figure 2 were measured and summarized in Table 4. Before optimization of Taguchi method, experiment 2 showed the highest SNR of 60.00 dB corresponding to the lowest R min of 0.001%. Meanwhile, experiment 7 showed the highest SNR of 6.66 dB corresponding to the narrowest FWHM of 0.4647°.

Reflectance intensity versus incidence angle of smooth Au nanofilm K-SPR sensor in air for nine experiments in the Taguchi’s L9 OA. Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; OA: orthogonal array.
R min and FWHM of smooth Au nanofilm K-SPR sensor in air.
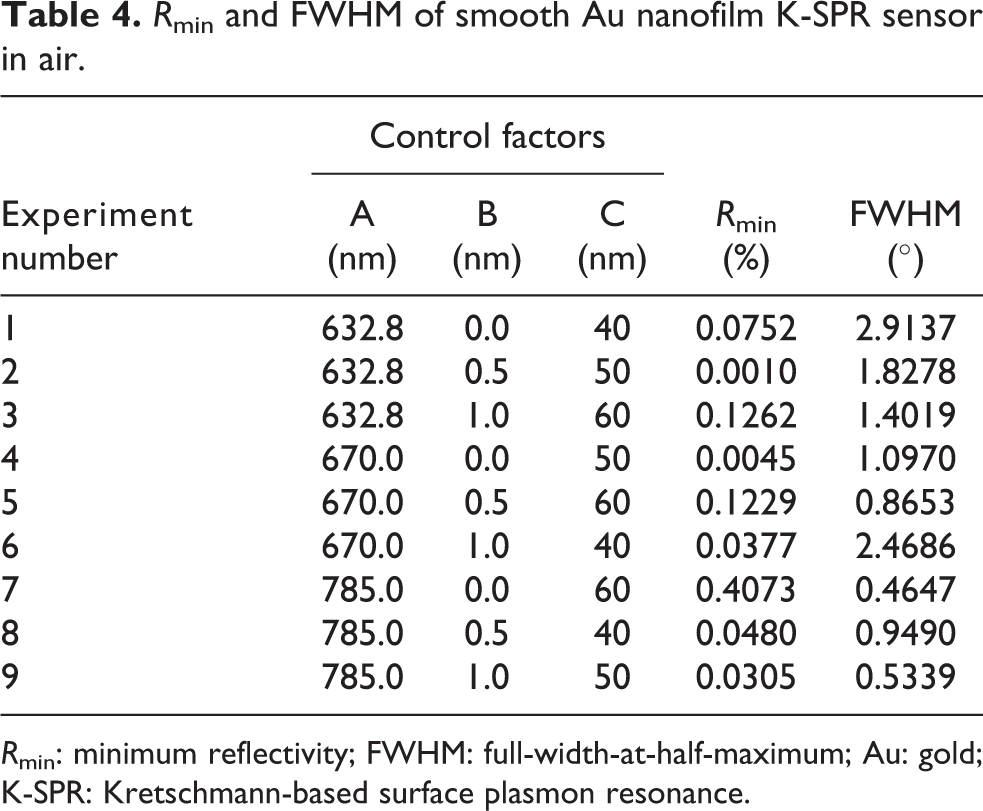
R min: minimum reflectivity; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Taguchi method analysis of Au nanofilm K-SPR sensor in air
After nine experimental runs, the next step is to determine the most significant control factor that affected the Au nanofilm K-SPR sensor response in air medium. The average SNR for each control factor and their levels were calculated and shown in Figure 3 (R min) and Figure 4 (FWHM), respectively, as main FE plots of the SNR. Referring to Figure 3, the best factor combination for R min is A2B2C2 which is 670 nm of wavelength, 0.5 nm thick Cr nanofilm, and 50 nm thick Au nanofilm. Meanwhile, the best factor combination for FWHM is A3B1C3 which is 785 nm of wavelength, 0 nm thick Cr nanofilm, and 60 nm thick Au nanofilm, as shown in Figure 4.

FE plot for SNR (Smaller-the-Better) of R min for smooth Au nanofilm K-SPR sensor in air. R min: minimum reflectivity; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; FE: factor effect; SNR: signal-to-noise ratio.

FE plot for SNR (Smaller-the-Better) of FWHM for smooth Au nanofilm K-SPR sensor in air. SNR: signal-to-noise ratio; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; FE: factor effect.
Next, ANOVA was conducted to identify which control factors significantly influence the performance of the Au K-SPR sensor. The FE of ANOVA for R min and FWHM are combined and presented in Table 5. To find the best setting from these two responses for the Au K-SPR sensor, the value of FE for both R min and FWHM were compared. The higher FE percentage from either the R min or FWHM in Table 5 was chosen as the best setting for the Au K-SPR sensor. For the first factor A, which is the optical wavelength, 67% of the A3 factor level, equivalent to 785 nm optical wavelength, affects the FWHM and it is a more dominant factor than the 7% FE of A2 corresponding to the 670 nm wavelength for R min. The thickness of Cr nanofilm is pooled and deemed negligible because its FE is 1% for both R min and FWHM. Lastly, for the third factor, which is the Au nanofilm thickness, 90% of the FE of C2 (50 nm thick Au nanofilm) affects the R min in a much more significant manner than the 31% FE of C3, corresponding to 60 nm thick Au nanofilm for the FWHM response. Therefore, multi-responses from R min and FWHM can be combined as one best setting which is A3BXC2, shown in Table 6. The thickness of Cr which is BX shows a small value of FE for both R min and FWHM and can be considered as pooled or negligible with little effect on both the R min and FWHM responses of the K-SPR sensor.
Result of ANOVA for R min and FWHM for smooth Au nanofilm K-SPR sensor in air.

ANOVA: analysis of variance; R min: minimum reflectivity; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; FE: factor effect; Cr: chromium; Au: gold.
The best setting of control factors for smooth Au nanofilm K-SPR sensor in air.
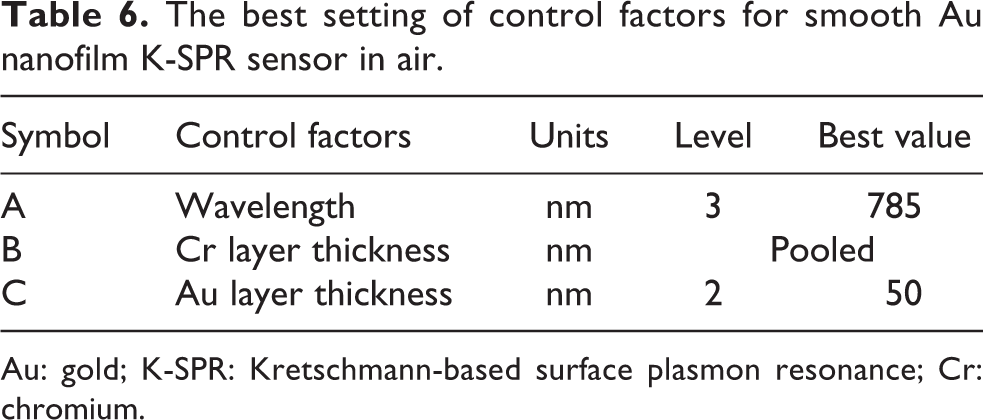
Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium.
Confirmation run of the best setting, A3B2C2 for multi-response Au K-SPR sensor in air
Confirmation run is the closing procedure in the design of experiment process. The objective of the confirmation run is to certify the results obtained during analysis stage. 14 Once the optimal level is selected, the final simulation was conducted utilizing the final best level setting of control factors as suggested by Taguchi method as shown in Table 6. Based on the table, Cr layer thickness is pooled. As a result, the best setting is limited to the wavelength which is 785 and 50 nm of Au layer thickness. Referring to the L9 OA in Table 3, experiment 9 matched this setting which is A3B3C2 and can be used again as the confirmation run.
Referring to Table 7, A3B3C2 is the best setting after optimization which can produce the optimum value for both R min and FWHM of the Au K-SPR sensor in air. Prior to the optimization approach, control factor setting of A1B2C2 or A3B1C3 only showed either the lowest R min or narrowest FWHM, respectively. This indicates that Taguchi method can be used to optimize multi-response experiments to obtain one set of best settings of design parameters and achieve insensitivity to noise factors.
SNR upon confirmation run after optimization of smooth Au K-SPR sensor in air.

SNR: signal-to-noise ratio; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; R min: minimum reflectivity; FWHM: full-width-at-half-maximum.
Besides that, according to Taguchi method, the thickness of Cr showed a small influence for both R min and FWHM which can be considered as pooled and negligible. Therefore, the confirmation runs for pool analysis were also conducted for A3B1C2 and A3B2C2. The wavelength is fixed at 785 nm, the Au nanofilm thickness is 50 nm, and the Au nanofilm surface roughness is assumed to be 0. The fabrication tolerances or Cr adhesion layer is 0.5–2 nm according to the LayerSolver of BioNavis’s manual. In this section, we considered the Cr thickness lower than 1 nm because it should be as thin as possible. 47,48 Therefore, the Cr layer thickness was varied from 0 nm to 0.5 nm to 1 nm. The results for pool analysis and the confirmation of Cr layer thickness effect are shown in Figure 5 and Table 8. The best combination run of R min and FWHM is at A3B2C2 which is 0.0017% and 0.4759°, respectively. This K-SPR sensor showed a better performance so far compared to other related works. 26,35 –37,51,52

Dependency of Cr layer thickness on Au K-SPR sensor in air. Cr: chromium; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Cr layer thickness effect on smooth Au K-SPR sensor in air.

Cr: chromium; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; R min: minimum reflectivity; FWHM: full-width-at-half-maximum.
Furthermore, all the values for both R min and FWHM of 0, 0.5, and 1 nm Cr layer thicknesses are within the predicted range of Taguchi method analysis. As a result, these simulated experiments can be run using 0 nm Cr layer thickness for analysis so that a lot of time could be saved although it produces a larger R min and FWHM but within the range of Taguchi method prediction. Therefore, the experiments can be run without considering the fine mesh of Cr layer thickness in FDTD simulation because the wavelengths of 785 nm and 50 nm thick Au nanofilm are more dominant in this case. This can save simulation time from several hours to several days.
Taguchi method analysis of Au nanofilm K-SPR sensor in air including its RMS surface roughness
In this section, two additional measurements of the Au nanofilm surface roughness were introduced as noise factor levels, as shown in Table 5. 14 In the previous section, the Au nanofilm was assumed to be smooth where the surface roughness of the Au nanofilm was assumed to be 0 (level 1 of the noise factor). Two more measurements of R min and FWHM that consist of two rough Au nanofilms were measured again from the K-SPR curve according to L9 OA shown in Table 9 for level 2 (1.26 nm) and level 3 (2.52 nm) and then converted to SNR (Smaller-the-Better), based on equation (1). The results of these SNR values are summarized in Tables 6 and 7. The rough surface of the Au nanofilm is located at the bottom of Figure 1 where the RMS surface roughness values were modified accordingly in the FDTD software to reflect different roughnesses. Zhitao et al. reported that upon annealing, the RMS surface roughness of Au nanofilms differed from 1.26 nm at different incremental temperatures where the surface becomes smoother and the resonance angle, θR, of K-SPR curve was red-shifted because higher thermal annealing temperatures diffuse the Au atoms further into the surface. The R min values did not show any obvious trend, whereas the FWHM increased when surface roughness increased. In particular, the surface roughness is caused by the isolated island of metal on the surface of the Au nanofilm. 52,53
Au nanofilm noise factor levels for K-SPR sensor in air.

Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; RMS: root mean square.
The performance characteristics of the Au K-SPR sensor can be predicted based on the experimental run with the highest SNR values as shown in Tables 10 and 11. Experiment 4 shows the highest average SNR of 39.03 dB corresponding to the lowest R min. Meanwhile, experiment 7 shows the highest average SNR of 7.62 dB which corresponds to the narrowest FWHM. Upon optimization, the R min and FWHM were observed to be 30.72 dB and 5.46 dB, respectively (referring to Tables 10 and 11). These numbers are well within the value that was predicted in the Taguchi analysis. For R min, 30.72 dB is within the predicted range of 32.38 ± 4.99 dB and closely near to the center value. Meanwhile, for FWHM, 5.46 dB is also within the predicted range which is 5.69 ± 1.90 dB.
Average SNR of R min for rough Au nanofilm K-SPR sensor in air.

SNR: signal-to-noise ratio; R min: minimum reflectivity; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Average SNR of FWHM for rough Au nanofilm K-SPR sensor in air.
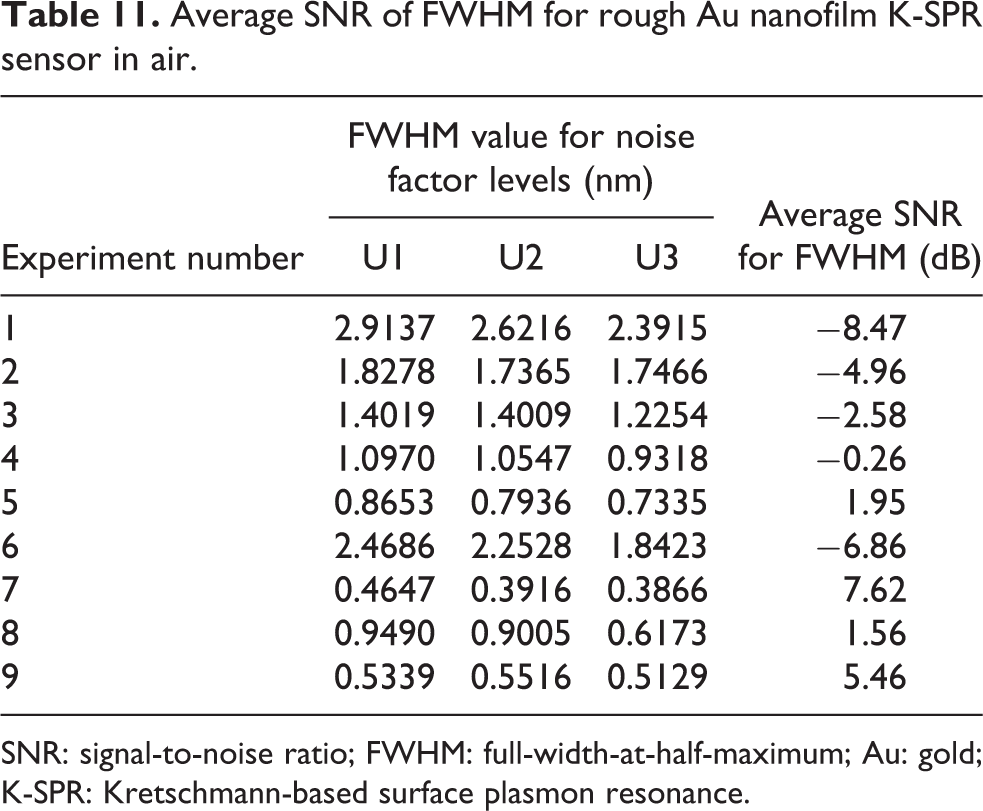
SNR: signal-to-noise ratio; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Referring to Table 12, the effect of the Au nanofilm surface roughness on the resonance angle was analyzed. Overall, the resonance angle is slightly blue-shifted and decreased with the increment of Au nanofilm RMS surface roughness from 0 nm to 2.52 nm. The R min and FWHM values also decrease with increment in the Au nanofilm surface roughness. Previous work suggests that if the dielectric constant of Au nanofilm is fixed as in our FDTD simulations, the resonance angle will decrease when surface roughness or gratings are introduced to the metallic layer. 54 The increase in surface roughness increases optical absorption which further enhances the SPR field. This is also verified from the results obtained by Velinov et al. where when the surface roughness of the Au nanofilm increased from 0.6 nm to 0.73 nm, the θR, R min, and FWHM values of the K-SPR curve decreased. 55 However, further increment of Au nanofilm surface roughness will eventually degrade the R min values due to dampening of the surface plasmon waves. 56 Therefore, the K-SPR Au sensor is sensitive to the Au nanofilm surface roughness but can be optimized using Taguchi method to produce a robust design. 57
Effect of the Au nanofilm surface roughness on K-SPR sensor in air.
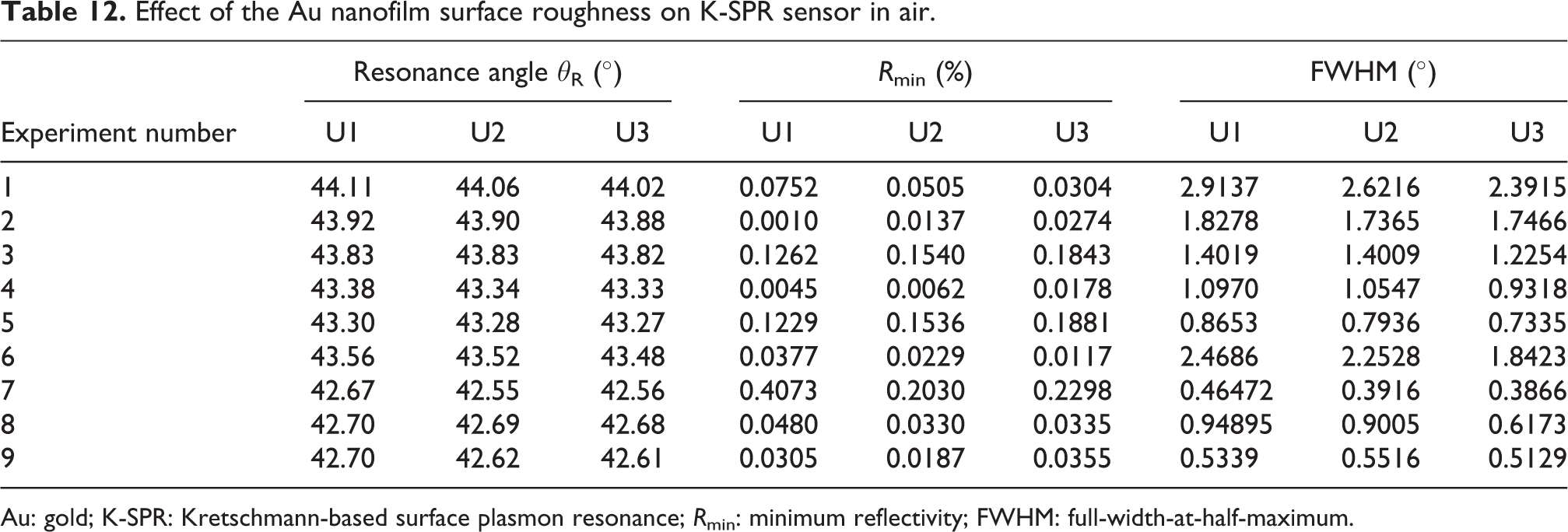
Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; R min: minimum reflectivity; FWHM: full-width-at-half-maximum.
Experimental results of best setting parameters using Taguchi method for multi-responses of Au K-SPR sensor in air
The K-SPR sensor measurement in air using BioNavis SPR Navi 200 L instrument was conducted according to the best setting parameters from Taguchi method. 39 As mentioned before, the best setting is heavily dependent on the wavelength which is 785 and 50 nm of smooth Au layer thickness, but Cr layer thickness is insignificant. Referring to Figure 6, experimental results exhibit a good agreement with FDTD simulation. The resonance angle shows the same value at 42.6° which indicates the important point in sensor sensitivity measurement. The value of R min from the experimental result is slightly higher than the FDTD simulation. And because of this, the FWHM obtained experimentally is slightly narrower than FDTD simulation with a value of 0.3952°. Therefore, the difference in FWHM by FDTD simulation and experimental results is small, which is less than 0.0807° for 0.5 nm Cr layer thickness.

Comparison of experimental result and FDTD simulation of best setting A3B2C2 for the smooth Au nanofilm K-SPR sensor in air with 0.5 nm Cr layer. FDTD: finite-difference time-domain; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium.
Sensitivity of smooth Au-based K-SPR sensor on glucose sensing
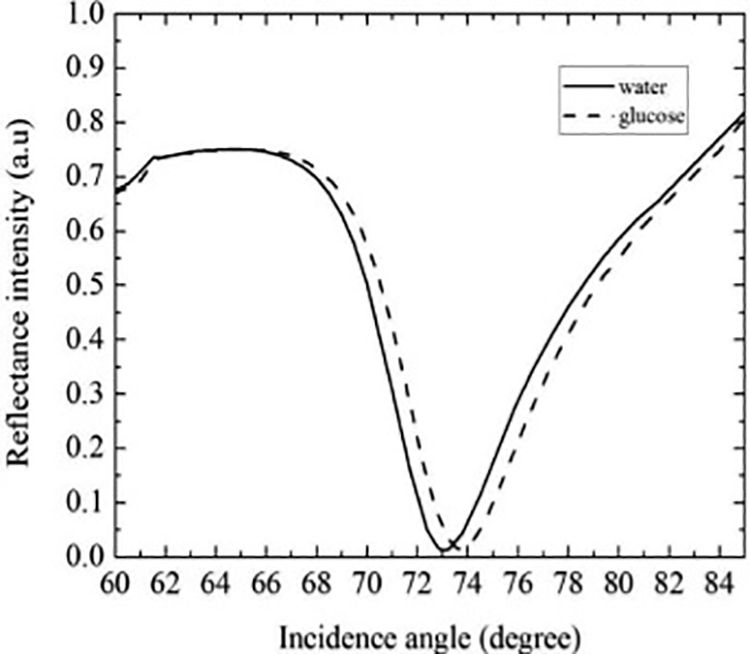
Table 13 and Figure 7 show the K-SPR sensor’s SPR resonance angle and RI value versus increasing glucose concentration where a linear curve is obtained. In the previous section, the best setting of K-SPR sensor was A3B2C2 that optimized the R min and FWHM values for sensing in air. The sensor sensitivity is 106.73°·RIU−1 for glucose concentrations of 1–5% corresponding to RI values of 1.3301, 1.3318, 1.3335, 1.3352, and 1.3369 at optical wavelength of 785 nm. These RI values were obtained based on the linearity of RI glucose concentration, y = 0.0017x + c, as disclosed in Menon et al. 3 The RI value of water of 1.3284 was obtained from the BioNavis SPR Navi 200 L LayerSolver datasheet. The sensitivity of this K-SPR sensor is higher than that reported in the studies of Gan et al., 58 Kushwaha et al., 59 and Rahman et al. 60 but lower than our previous work 61 –63 due to the usage of 2D materials with higher RI values that enhance SPR signal amplification. In summary, the Taguchi-optimized K-SPR glucose sensor of A3B2C2 achieved high DA of 0.01 and Q-factor of 224.26 RIU−1. A high Q-factor for the K-SPR sensor implies that the plasmons are strongly confined.
SPR resonance angle and RI versus increasing glucose concentration (%) at optical wavelength of 785 nm for Taguchi best combination of A3B2C2 of the K-SPR sensor.

SPR: surface plasmon resonance; RI: refractive index; K-SPR: Kretschmann-based surface plasmon resonance.

Refractive index versus increasing glucose concentration (%) at optical wavelength of 785 nm for Taguchi best combination of A3B2C2 of the K-SPR sensor. K-SPR: Kretschmann-based surface plasmon resonance.
Glucose-sensing sensitivity and effect of Cr adhesion layer thickness with roughness on smooth Au-based K-SPR sensor
In the previous section, the Cr adhesion layer was investigated with a thickness of 0–1 nm. For further investigation, the sensitivity of the Au K-SPR sensor with thicker Cr layer of 1.0–3.0 nm with an additional 0.1% RMS surface roughness to detect glucose was also analyzed using the L9 OA as depicted in Table 14. Figure 8 shows the reflectance intensity versus incident angle of Au nanofilm K-SPR in water. These SPR curves were calculated based on the RI of water from BioNavis datasheet which are 1.3319 at 632.8 nm, 1.3309 at 670 nm, and 1.3284 at 785 nm wavelengths, respectively. The value of R min and FWHM of Au nanofilm K-SPR in water from Figure 8 is summarized in Table 14. The lowest R min occurred at experiment 4, while the narrowest FWHM was achieved in experiment 7. Referring to Figure 8, the R min or the depth of the SPR spectrum is believed to depend on the metal layer thickness where 50 nm Au nanofilm displayed close to zero reflectance intensity although at different incident light. Therefore, 50 nm thick Au nanofilm absorbs almost 100% of the incident light and excited the surface plasmon polaritons. In addition, R min of 40 and 60 nm thick Au nanofilm showed higher reflectance intensity compared to 50 nm thick Au nanofilm for the same incident wavelength. Also, the K-SPR resonance angle and FWHM are highly dependent on the incident wavelength.
R min and FWHM of Au nanofilm K-SPR sensor in water.

R min: minimum reflectivity; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; RMS: root mean square.

Au nanofilm K-SPR sensor in water for nine experiments in the Taguchi’s L9 OA when Cr layer thickness is 1.0–3.0 nm + 0.1% RMS surface roughness. Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; OA: orthogonal array; Cr: chromium; RMS: root mean square.
Table 15 shows the sensitivity of K-SPR when the thickness of Cr based on L9 OA in Tables 4 and 14 are 0–1.0 nm and 1.0–3.0 nm with 0.1% RMS surface roughness. The RI values of 3% glucose at 632.8 nm, 670 nm, and 785 nm are 1.336, 62 1.3351, and 1.3335, respectively. The RI values were obtained based on the linearity of RI glucose concentration, y = 0.0014x + c, as disclosed in Menon et al. at 670 nm. 3 Compared to the sensing in air, the K-SPR sensor showed an increment in sensitivity values for thicker Cr layers for glucose sensing. As expected, higher glucose detection sensitivity was achieved at lower visible wavelength of 632.8 nm compared to the longer NIR wavelength of 780 nm. These values of sensitivity were then converted to SNR (Larger-the-Better) based on equation (2). Referring to Figures 9 and 10, the best factor combination for glucose-sensing sensitivity is A1B3C3, which is 632.8 nm of wavelength, 1 and 3 nm thick Cr nanofilm, and 60 nm thick Au nanofilm.
Sensitivity of Au K-SPR sensor for 3% glucose detection for different thicknesses of Cr layer.

SNR: signal-to-noise ratio; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium; RMS: root mean square.
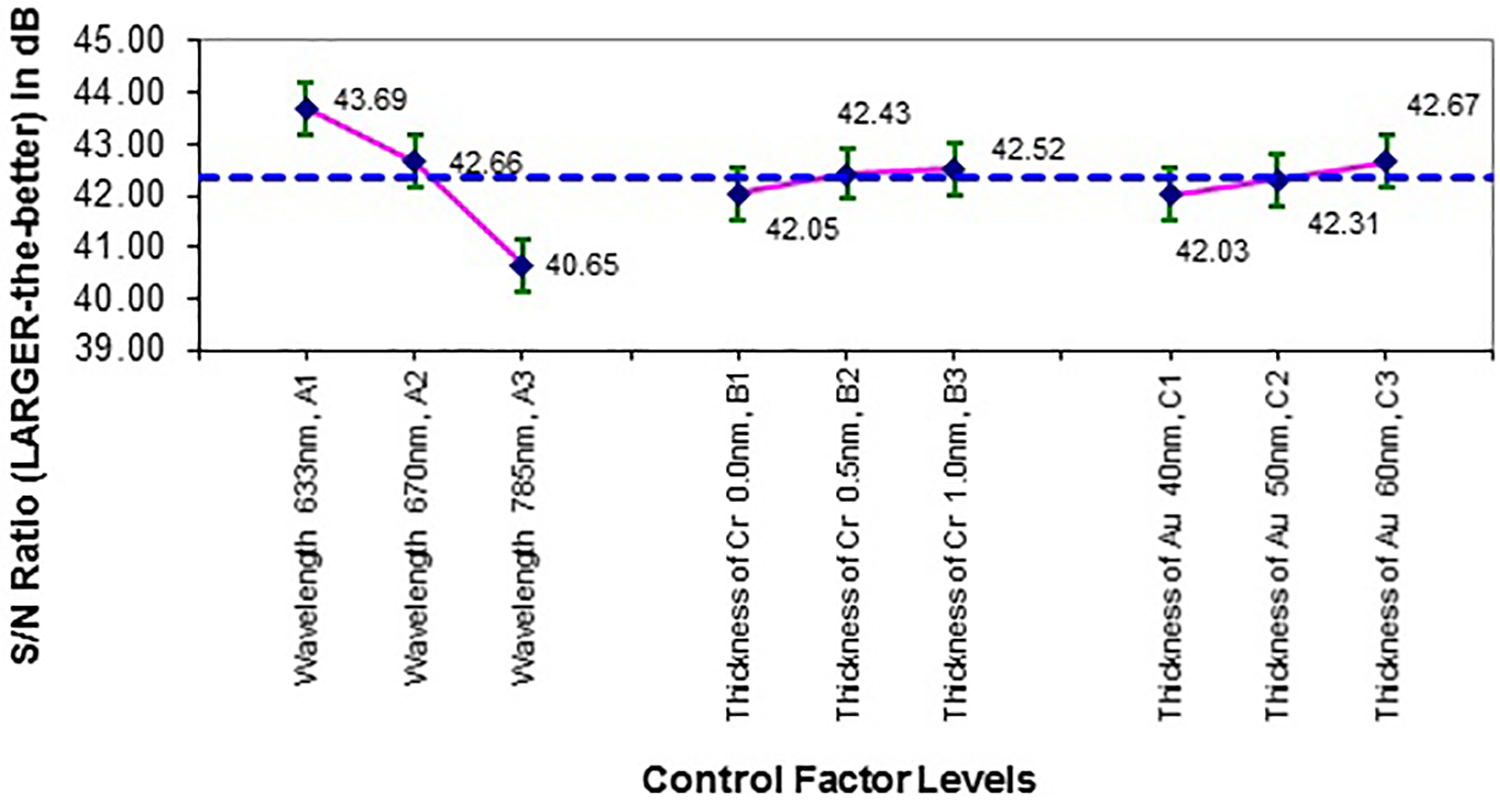
Factor effect plot for SNR (Larger-the-Better) of sensitivity for smooth Au nanofilm K-SPR sensor when the Cr layer thickness is 0–1 nm and without any surface roughness for 3% glucose detection. SNR: signal-to-noise ratio; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium.

Factor effect plot for SNR (Larger-the-Better) of sensitivity for smooth Au nanofilm K-SPR sensor when the Cr layer thickness is 1–3 nm with 0.1% RMS surface roughness for 3% glucose detection. SNR: signal-to-noise ratio; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; Cr: chromium.
Table 16 compares the FE plot for 3% glucose detection sensitivity of the Au nanofilm K-SPR sensor for 0–1 nm thick Cr layer and 1–3 nm thick Cr layer with 0.1% RMS surface roughness. Compared to sensing in air, the optical wavelength is now the dominant factor versus Cr and Au nanofilm thickness for glucose detection sensitivity performance. K-SPR sensing sensitivity is strongly dependent on the wavelength by more than 86% of FE which is A1 at 632.8 nm. However, referring to Figure 8, the SPR curve for 632.8 nm is broad and a sensitive photodetector is needed to measure the reflectivity in the experimental measurement. Also, the dynamic range for resonance angle shift measurement at 632.8 nm is limited (nearing 80°) compared to 670 and 785 nm wavelength. The Au layer thickness has a significant effect of 10% to the sensitivity when a thicker or more than 1 nm thick Cr nanofilm adhesion layer is used. This is because when thicker Cr adhesive layers are used, the atoms interact with the Au atoms as well as the incident optical light and change the effective dielectric constant of the Cr/Au hybrid metallic layer. However, the Cr layer thickness ranging from 0 nm to 3 nm with surface roughness has a negligible FE of 2% on the sensitivity measurement. Hence, the Cr adhesive layers should remain as thin as possible in the development of K-SPR sensors.
Result of ANOVA for sensitivity of smooth Au nanofilm K-SPR sensor for 3% glucose sensing.

ANOVA: analysis of variance; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; FE: factor effect; Cr: chromium; RMS: root mean square.
The FE values of ANOVA for R min, FWHM, and sensitivity are combined and presented in Table 17 for the detection of 3% glucose. To find the best setting from these three responses for the Au K-SPR sensor, the values of FE were compared. The highest FE percentage from either the R min or FWHM or sensitivity in Table 17 was chosen as the best setting for the Au K-SPR sensor. For the first factor A, which is the optical wavelength, 86% of the factor level A1, equivalent to the 632.8 nm optical wavelength, affects the sensitivity and it is a more dominant factor than the 11% FE of A2 corresponding to the 670 nm wavelength for R min and 57% FE of A3 corresponding to the 785 nm wavelength for FWHM. The thickness of Cr nanofilm is pooled and deemed negligible for all three responses. Lastly, for the third factor, which is the Au nanofilm thickness, 75% of the FE of C2 (50 nm thick Au nanofilm) affects the R min in a much more significant manner than the 39% FE of C3 corresponding to 60 nm thick Au nanofilm for the FWHM and 10% FE of C3 corresponding to 60 nm thick Au nanofilm for the sensitivity response. Therefore, multi-responses from R min and FWHM can be combined as one best setting which is A1B1C2, as shown in Table 18.
Result of ANOVA for R min, FWHM, and sensitivity for Au nanofilm K-SPR sensor (1–3 nm Cr layer with 0.1% roughness).

ANOVA: analysis of variance; R min: minimum reflectivity; FWHM: full-width-at-half-maximum; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; FE: factor effect; Cr: chromium.
The best setting of control factors for Au nanofilm K-SPR sensor for glucose-sensing sensitivity.

Au: gold; K-SPR: Kretschmann-based surface plasmon resonance; RMS: root mean square; Cr: chromium.
Lastly, the confirmation run of best setting for Au K-SPR sensor to detect 3% glucose was simulated using A1B1C2 based on Table 18. Figure 11 shows the SPR curve for glucose sensing by incident light of 632.8 nm, 1.0 nm + 0.1% RMS surface roughness, and 50 nm thick Au nanofilm. After the optimization approaches, the R min, FWHM, and sensitivity were observed to be 0.012558%, 6.5719°, and 163.415°·RIU−1, respectively. These numbers are well within the value that was predicted in the Taguchi analysis. For R min, 0.012558% or 38.02 dB is within the predicted range of 44.54 ± 17.58 dB; for FWHM, 6.5719° or −16.35 dB is within the predicted range of −15.79 ± 2.58 dB; and for sensitivity, 163.415°·RIU−1 or 44.27 dB is within the predicted range of 44.12 ± 0.99 dB which is closely near to the center value. In summary, the Taguchi-optimized K-SPR glucose sensor of A1B1C2 at visible wavelength of 632.8 nm achieved higher sensitivity but lower DA of 0.001 and Q-factor of 24.86 RIU−1. Therefore, Taguchi method can be used to optimize multi-response experiments and providing insensitivity to noise factors.
FDTD simulation of best setting A1B1C2 for 3% glucose sensitivity of Au K-SPR. FDTD: finite-difference time-domain; Au: gold; K-SPR: Kretschmann-based surface plasmon resonance.
Conclusion
In conclusion, the optimum parameters in realizing a robust design with multi-response Cr/Au nanofilm K-SPR sensor were successfully achieved using FDTD and Taguchi method. R min, FWHM, and glucose detection sensitivity responses of the Cr/Au nanofilm K-SPR sensor were optimized simultaneously. The best setting of control factors was identified to be A1B1C2 equivalent to 632.8 nm incident light, 1.0 nm Cr nanofilm thickness, and 50 nm Au nanofilm thickness. R min is dominantly dependent on the Au metal layer thickness where 50 nm thick nanofilm displayed near-zero reflectance intensity even at different optical wavelengths. The presence of increasing Au nanofilm RMS surface roughness was found to affect the R min and FWHM values. However, the FE of Cr nanofilm thickness and RMS surface roughness was deemed to be negligible on the K-SPR sensor sensitivity for lower Cr thicknesses. As Cr thickness increases, the FE of Au nanofilm thickness becomes significant as thicker Cr/Au nanofilms will have a different effective RI. Meanwhile, the resonance angle and FWHM is highly dependent on the optical wavelength. Taguchi optimization produced a Cr/Au nanofilm K-SPR sensor with R min of 0.012558%, FWHM of 6.5719°, and glucose-sensing sensitivity of 163.415°·RIU−1 at 632.8 nm but with lower DA and Q-factor of 0.001 and 24.86 RIU−1, respectively, compared to 785 nm wavelength. Hence, Taguchi method is highly recommended because of its capability to optimize control and noise factors in an effort to design robust multi-response sensors in nanotechnology.
Footnotes
Authors’ note
A portion of the findings within this article was previously presented at the NanoMITe Annual Symposium & Nanotechnology Malaysia Annual Symposium (NanoSym), Putrajaya, 2019.
Acknowledgment
The authors would like to thank the Institute of Microengineering and Nanoelectronics (IMEN), Universiti Kebangsaan Malaysia (UKM), the Ministry of Education (Malaysia), and Universiti Teknikal Malaysia Melaka (UTeM) for the support. The authors also would like to thank BioNavis Ltd, Kulim Hi-Tech Sdn Bhd, and Professor Prakash Apte for all the advice, consultation, and training related to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Malaysian Ministry of Higher Education using the AKU254: HICoE (Phase II) MEMS for Biomedical Devices (artificial kidney) and Fundamental Research Grant Scheme (FRGS) with grant code of FRGS/1/2019/STG02/UKM/02/8.
