Abstract
Docosahexaenoic acid is a omega-3-fatty acid which together with other long-chain omega-3-fatty acid known to have protective effect against various diseases including hypertension, myocardial infarction, Alzheimer disease, and cancers. Poor bioavailability owning to limited aqueous solubility limits its effective therapeutic delivery. Self-nanoemulsifying drug delivery systems are known to enhance the systemic absorption of poorly bioavailable lipophilic bioactive/therapeutics compounds. The purpose of this work was to investigate the potential of self-nanoemulsifying drug delivery systems produced by spontaneous nanoemulsification to enhance the oral bioavailability of docosahexaenoic acid. Initially, the screening of oil, surfactant, and cosurfactant was carried out by determining the miscibility and emulsifiability of the component with docosahexaenoic acid. Docosahexaenoic acid-containing self-nanoemulsifying drug delivery systems were prepared using Capryol 90, Tween 20, and polyethylene glycol 200 due to their excellent miscibility and emulsifiability with docosahexaenoic acid. Docosahexaenoic acid-containing self-nanoemulsifying drug delivery systems’ droplet size, size distribution, and zeta potential were found to be 111.5 ± 4.2 nm, 0.269 ± 0.05 nm, and −23.53 ± 2.9 mV, respectively. The in vitro drug release and ex vivo absorption studies showed better in vitro release and intestinal absorption as compared to docosahexaenoic acid aqueous dispersion. In vivo studies demonstrated a significant increase (p < 0.001) in the oral bioavailability of docosahexaenoic acid-containing self-nanoemulsifying drug delivery systems in comparison to a docosahexaenoic acid aqueous dispersion. This indicated the potential of self-nanoemulsifying drug delivery systems as an effective unit dosage form for the oral delivery of docosahexaenoic acid.
Keywords
Introduction
Fish oils are rich in omega-3-fatty acids and have been assigned various health benefits for their use. Docosahexaenoic acid (DHA) and eicosapentaenoic acid are the major constituents of fish oil 1 and are polyunsaturated fatty acids. As both are essential fatty acids, their role in nutrition is unquestionable. 1 They are structural components of membranes and have a definite role in development and growth. Essential fatty acids cannot be produced in the body and need to be delivered to the body through diet. 1,2 DHA is one of the essential omega-3-fatty acids and plays an important role in improving cardiovascular disorders, psychiatric disorders, cognitive activities, rheumatoid arthritis, and age-related neurodegenerative diseases. 2,3 The clinical applications of DHA are hindered due to its low water solubility and oral bioavailability. 4 DHA showed poor organoleptic properties due to its oily nature and fishy odor. 4
Soft gel capsules are the most common formulation of DHA-containing fish oil. The administration of omega-3-fatty acids in emulsions results in better bioavailability than in when it is administered as capsules. 5 More reports are available on the applications of fish oil emulsion. 6 –8 However, the emulsifier must be cautiously selected due to oxidative and physical instability. 1 Libinaki and Gavin 1 showed that co-delivering tocopheryl phosphate mixture (TPM) and DHA significantly enhanced the bioavailability of DHA, suggesting the potential use for commercially available TPM + DHA fortified products. 1 This requires further clinical studies to determine the feasibility of adding TPM to improve the bioavailability of other omega-3-fatty acids.
Recently, the application of nanotechnology such as micelles, liposomes, nanoparticles, microemulsions, and nanoemulsions has attracted attention for improving oral absorption and bioavailability. 9 Microemulsions and nanoemulsions are emulsion-based delivery systems, having some unique properties that benefit their application in drug delivery including simplicity of preparation, and their ability to improve the physical stability of natural oils. 10 –14 Both systems have a small particle size (less than 200 nm) and have a lipophilic core and a hydrophilic shell; nevertheless, nanoemulsions are thermodynamically unstable systems but can be designed to be kinetically stable, whereas microemulsions are thermodynamically stable. 15,16 Nanoemulsions can be prepared by high- or low-energy methods. High-energy methods use specialized mechanical devices to breakdown the bigger droplets into small droplets using ultrasonication, high pressure homogenization, and microfluidiziation. 15,16 Abbasi and coworkers developed flaxseed oil nanoemulsions using ultrasonication 17 ; this nanoemulsion has the potential to protect omega-3-fatty acids and to improve broiler meat. 17 Low-energy methods such as emulsion phase inversion, phase inversion temperature, and spontaneous emulsification can spontaneously form very small droplets as a result of controlled changes in the solution or environment conditions. 15,16 The interest in low-energy methods for certain applications is increasing because of their simple production methods, lower manufacturing costs, and ability to create smaller particle sizes compared with high-energy methods. 15,16 An oil phase is one of the components of any nanoemulsion. Thus, DHA fatty acid could be easily miscible with an appropriate oil system and employed as the oil phase itself if we consider a nanoemulsion formulation. Various researchers have developed fish oil nanoemulsions using the spontaneous emulsification method. 15,16
Self-nanoemulsifying drug delivery systems (SNEDDS) are anhydrous nanoemulsions (without aqueous phase) which are specifically designed for oral administration. They consist only of oil and S mix (mixture of surfactant and cosurfactant) phase and produce ultrafine droplets on contact with a gastrointestinal medium, showing improved oral absorption and increased bioavailability of lipophilic bioactive ingredients. 18,19 SNEDDS have added advantages including long-term stability, ease of preparation, and high drug loading. 20 Puri and coworkers developed DHA-containing SNEDDS using olive oil, mixtures of Span 80 and 20 as surfactants, and Tween 80 and propylene glycol (PG) as cosurfactants for improved palatability, dispersibility, and bioavailability. 4 The optimized SNEDDS was further evaluated for estimation of brain concentration using gas chromatography. Following the administration of DHA-containing SNEDDS, there was a 2.6-fold increase in the brain concentration of DHA as compared to that achieved following administration of a marketed DHA formulation. 4 However, there are limited reports on DHA formulations on oral bioavailability enhancement of DHA in an efficient and acceptable manner.
Therefore, the aim of our current investigation was the development of an improved formulation of DHA-containing SNEDDS having a new composition and high loading to increase the oral absorption and bioavailability of DHA, as determined using liquid chromatography–mass spectroscopy (LC-MS).
Materials and methods
Materials
DHA ethyl ester (purity > 98%, light yellow to colorless clear liquid, molecular weight: 356.5 g/mol, acid value mg KOH/g: 2 max, peroxide number: 40 max) was purchased from Sigma Aldrich, St. Louis, Missouri, USA. Tween 80, Tween 20, sorbitan monolaurate (Span 20), polyethylene glycol 200 and 400 (PEG 200 and 400), and PG were purchased from Merck, Kenilworth, NJ, USA. Capryol 90 (propylene glycol monocaprylate), Lauroglycol 90 (propylene glycol monolaurate), Labrasol, and Transcutol HP (diethylene glycol monoethyl ether) were obtained from Gattefosse (Saint-Priest, France). All other reagents used in the study were of analytical grade.
Formulation design and optimization of DHA-containing SNEDDS
Preparation of DHA-containing SNEDDS
To screen the suitable formulation components for the development of DHA-containing SNEDDS, the miscibility of DHA as a lipid phase was determined in various oils such as Capryol 90, castor oil, soybean oil, isopropyl myristate (IPM), Lauroglycol 90, as well as its emulsifiability in various surfactants and cosurfactants (Tween 20, Tween 80, Labrasol, Solutol HS15, PEG 200, PEG 400, Transcutol HP, and PG). An oil showing good miscibility and that formed a clear solution was selected. Surfactants and cosurfactants having good emulsifiability with DHA were screened further based on their maximum emulsifying ability. 21,22 SNEDDS were prepared by the low-energy spontaneous emulsification method. Pseudo-ternary phase diagrams were constructed to determine the concentration of oil, S mix (mixture of surfactant and cosurfactant), and aqueous phase. An aqueous titration method was followed which works on the process of spontaneous emulsification. Aqueous titration was carried out by mixing the S mix and oil phase in ratios of 9:1, 8:1, 7:1, 6:1, 5:1, 4:1, 3:1, 2:1, 1.5:1, 1:1, 0.66:1, 0.43:1, and 0.25:1 in glass vials at room temperature. 21 Phase diagrams were constructed for S mix ratios of 4:1, 3:1, 2:1, 1:1, 1:2, 1:3, and 1:4. 21 The specific compositions of oil, S mix, and water were chosen from phase diagram study and homogenously mixed through vortexing to obtain clear isotropic system in form of nanoemulsion. The different combinations of optimized SNEDDS were sealed in transparent glass bottles and further evaluated for dispersibility, thermodynamic stability, and rate of self-emulsification.
Thermodynamic stability study
To check whether the selected formulation composition generates stable nanoemulsions, the designed SNEDDS formulations were subjected to thermodynamic stability studies, which comprised the heating–cooling cycle, freeze–thaw cycle, and centrifugation tests. 23 Physical stability was continuously monitored over a time period. Various aspects like phase separation, precipitation, and turbidity were observed at room temperature. 24 Heating–cooling cycle: Storage of SNEDDS at 4°C and 45°C at each temperature were studied for six cycles, each of not less than 48 h. Centrifugation study: Selected formulations were centrifuged at 3500 r/min for 30 min. Freeze–thaw cycle: SNEDDS samples were stored between −21°C and +25°C for three cycles, each of not less than 48 h.
Dispersibility study
The study was carried out in a USP rotating paddle apparatus at 50 r/min. The experiment was performed by the dropwise addition of the formulation to 500 mL of distilled water and 0.1 N HCl at 37 ± 0.5°C in the dissolution vessel. The performance of the SNEDDS was assessed using a reported grading method. 22 The experiment was performed in triplicate to observe the consistency of the formulation. Grade A is characterized by the rapid formation of nanoemulsions within 1 min with a clear or bluish appearance. Grade B is characterized by rapidly forming, slightly less clear nanoemulsions that have a bluish or white appearance. Grade C is characterized by bright-white emulsions with a formation time near to 2 min. Grade D is characterized by a slightly oily, grayish-white emulsion. 22
Determination of self-emulsification rate
The emulsification rate of all identified formulations from phase behavior studies was determined in triplicate. This test was carried out in a USP apparatus II with a paddle speed of 50 r/min in 500 mL water at 37°C. One milliliter of SNEDDS was added slowly to the dissolution medium and the time required for the disappearance of SNEDDS was recorded. 22
Characterization of selected SNEDDS formulation
Determination of droplet size, polydispersity index, and zeta potential
Droplet size, polydispersity index (PDI), and zeta potential of designed SNEDDS formulations were determined using a Zetasizer ZSP (Malvern Instruments, UK) after 100 times dilution in distilled water. 25
Refractive index
The refractive index of designed SNEDDS formulations was determined using an Abbe’s type refractometer, Rudolph Research Analytical, NJ, USA (Brookfield Engineering Laboratories, Inc., Middleboro, MA, USA) at 25 ± 0.5°C.
Viscosity
The viscosity of the selected SNEDDS formulations was determined at 25 ± 0.5°C using Brookfield cone and plate rheometer.
Transmission electron microscopy
The size and morphology of the prepared SNEDDS formulation were studied using transmission electron microscopy (TEM) (JEOL JEM1010, Tokyo, Japan). Before analysis, the diluted SNEDDS (1:100) was deposited on a carbon-coated copper grid and examined at 120 kV High-Contrast/High-Resolution Digital TEM JEM1010.
In vitro drug release
In vitro release was determined for selected formulations (OFA1c, OFA2c, OFA3c, and OFA4c—see Table 1 for the composition of the various formulations), using a dialysis bag technique. The test was performed using dissolution apparatus II (at 100 r/min and 37 ± 0.5°C) in 500 mL simulated intestinal fluid (pH 6.8). One milliliter of SNEDDS (containing 10 mg DHA) or DHA aqueous dispersion (10 mg DHA dispersed in 1% v/v Tween 20 as a control) formulations were placed in the dialysis bag. Aliquots of 1 mL samples were withdrawn at regular time intervals (15, 30, 60, 90, 120, 240, 300 and 360 min) from the media and the released DHA was estimated using a reported LC-MS method with slight modification (LC-QqQ-MS, Agilent Technologies, Santa Clara, CA, USA). 26 The mobile phase consists of 0.1% ammonia solution and acetonitrile (10:90, v/v), and the flow rate was set at 0.5 mL/min. The injection volume used was 10 μL. The MS conditions were as follows: gas flow rate, 11 L/min; gas temperature, 330°C; nebulizer pressure; 35 psi, and capillary voltage, 4100 V. Multiple reaction monitoring (MRM) transitions were applied applying negative ionization mode; DHA, m/z 327.3.0–283.20, fragmentor 80 eV, collision energy 20 eV. Linearity of the assay method was verified within the concentration range of 50–8000 ng/mL. The retention time was found to be 1.64 min (Figure 1). The release of DHA from SNEDDS formulations was compared with the DHA aqueous dispersion.
Composition of various SNEDDS formulations obtained from phase behavior study.
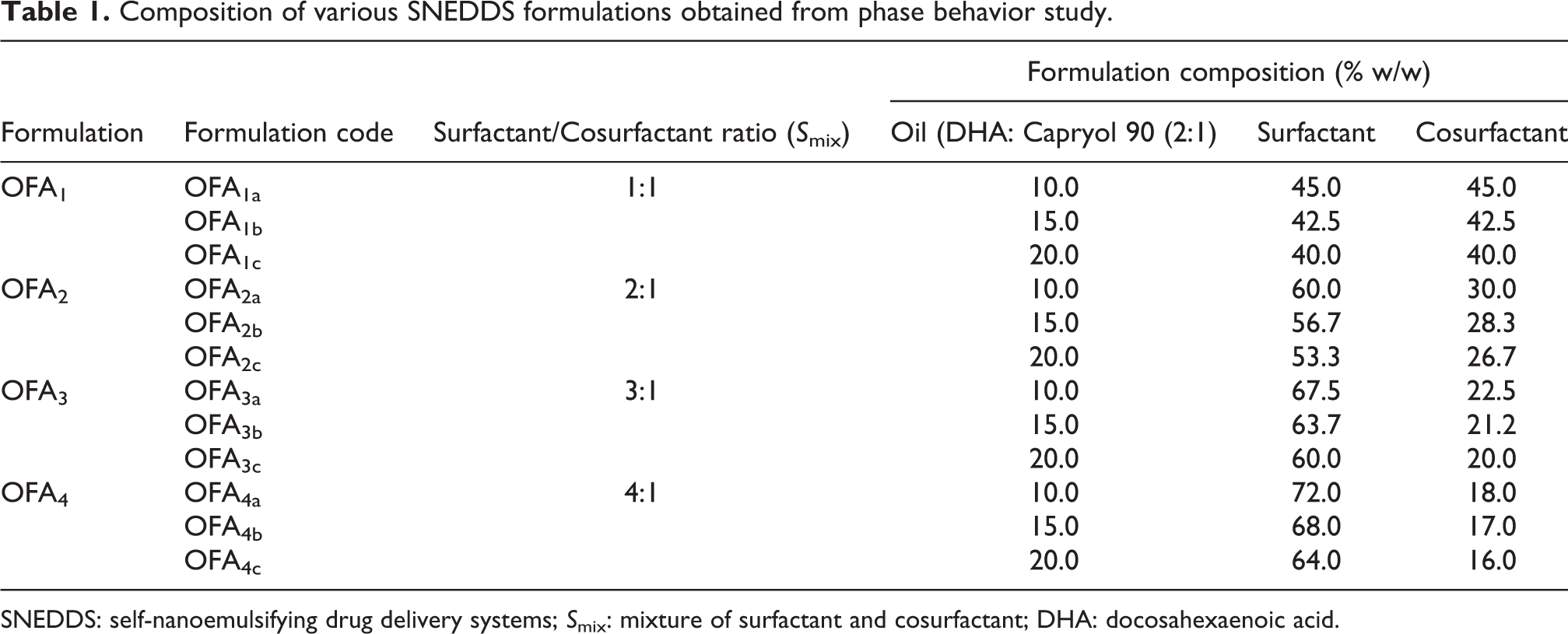
SNEDDS: self-nanoemulsifying drug delivery systems; S mix: mixture of surfactant and cosurfactant; DHA: docosahexaenoic acid.

MRM chromatograms of standard DHA. Quantitation was performed with transitions at m/z 327 → 283 for DHA. The retention time was found to be 1.64 min. MRM: multiple reaction monitoring; DHA: docosahexaenoic acid.
Ex vivo absorption study: Non-everted gut sac method
The non-everted gut sac method was used to determine the ex vivo absorption of selected formulations (OFA1c, OFA2c, OFA3c, and OFA4c) using albino Wistar rats, weighing between 200 and 220 g. 27,28 The studies were conducted to compare the permeability coefficient (P app) of developed DHA-containing SNEDDS formulation and DHA aqueous dispersion. The jejunum segments were excised carefully from the rats and washed 5–7 times with Krebs-Ringer solution (KS) under continuous aeration. One end of the jejunum segment was occluded using a thread to make an empty gut sac and 1 g weight was tied to the end of the gut sac assembly. After setting the assembly, the gut sac was filled with 1 mL of SNEDDS (containing 10 mg DHA) or DHA aqueous dispersion (10 mg DHA dispersed in 1% v/v Tween 20 as a control) formulations and placed inside the bath containing 100 mL of KS and the temperature of the gut sac bath and the medium was maintained at 37 ± 0.5°C. The samples were withdrawn at 15, 30, 45 and 60 min and were analyzed for DHA content by LC-MS. The P app was determined using the following equation 29 :
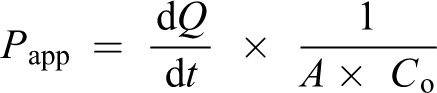
where dQ/dt is the slope of the linear region plot of DHA permeated versus time, A is the surface area of tissue (cm2), and C o is the initial concentration of DHA (µg/mL) in the donor compartment.
In vivo study
The animal study was approved by the Research Ethics Committee (PH-107-41) Faculty of Pharmacy, King Abdulaziz University. Wistar rats (200–220 g) of either sex were allocated to two groups. The group I animals received oral standard DHA aqueous dispersion (containing 100 mg/kg). DHA aqueous dispersion was prepared by dispersing DHA in 1% v/v Tween 20 as a control. Group II animals received DHA-containing SNEDDS (equivalent to 100 mg/kg) with the help of an oral feeding needle. The rats were anesthetized using diethyl ether and blood samples (0.25 mL) were withdrawn from the tail vein of the rat at 0 (pre-dose), 0.5, 1.0, 1.5, 2, 4, 8, 12 and 24 h. The plasma was separated by centrifugation at 5000 r/min for 30 min and stored at −21°C until analysis. DHA was extracted from the plasma samples using ethyl acetate. The residue obtained was reconstituted with the mobile phase. The average concentration of DHA in plasma at the respective sampling time was observed.
Statistical analysis
The statistical analysis was performed using an independent samples t-test for comparison between two groups, and one-way analysis of variance for multiple comparison using post hoc Tukey’s multiple analysis. The results are expressed as mean ± SD. Statistical significance was considered when p < 0.05.
Results and discussion
Formulation design and optimization of DHA-containing SNEDDS
The oil system Capryol 90 (>2500 mg/g) exhibited very good miscibility with DHA and formed a clear solution as compared to other oils screened; it was therefore used as the oil phase (with DHA and Capryol 90 in ratio of 2:1) for the development of DHA-containing SNEDDS. The other oil systems studied, including castor oil (95.33 ± 5.13 mg/g), soybean oil (151.66 ± 3.05 mg/g), IPM (503.66 ± 4.04 mg/g), and Lauroglycol 90 (1006.33 ± 4.72 mg/g) were to be unsuitable due to their low miscibility and its high lipophilicity. The results for the screening of surfactants and cosurfactants are shown in Figure 2. Tween 20 had the highest capacity to emulsify the DHA-containing oil phase, being able to emulsify more than 100 µL; it was more effective than Tween 80. The emulsification of DHA-containing oil phase in Labrasol (caprylocaproyl macrogol glyceride) was comparable with that in Tween 80. PEG 200 showed more capacity to emulsify DHA-containing oil phase than PEG 400. PG had the least capacity for emulsifying the DHA-containing oil phase, with a value of around 20 µL. Therefore, Tween 20 and PEG 200 were selected as the surfactant and cosurfactant respectively and designated as S mix for the phase behavior study.

Volume of oil phase emulsified in various surfactants and cosurfactants.
The phase behavior study was carried out using the DHA-containing oil phase, Tween 20 and PEG 200 using the aqueous titration method. A considerable region of formation of nanoemulsion could be observed in the pseudo-ternary phase diagram (Figure 3). Among the different S mix ratios, the highest area of the nanoemulsion region was observed for 3:1. This confirmed that the selected S mix ratio and the selected S mix components can favorably lower the interfacial tension to facilitate the formation of a nanoemulsion. 30

Pseudo-ternary phase diagram of S mix 3:1 (blue shaded area indicates nanoemulsification region and circle indicates nanoemulsion formation during aqueous titration). S mix: mixture of surfactant and cosurfactant.
The composition of various selected SNEDDS obtained from the phase behavior study are shown in Table 1. Three formulations having different S mix ratios (4:1, 3:1, 2:1, and 1:1) were selected for further evaluation. The oil concentrations used for screening of formulations for each S mix ratio were 10%, 15%, and 20%.
All the selected nanoemulsion formulations passed all the tests for thermodynamic stability (Table 2). Thus, the selected formulations are stable and can successfully overcome the stress produced by cycles of centrifugation, heating–cooling, and freeze–thaw.
Observation of different compositions of SNEDDS for thermodynamic stability, dispersibility efficiency, and rate of emulsification.

SNEDDS: self-nanoemulsifying drug delivery systems.
In the dispersibility tests (Table 2), all the selected formulations rapidly form SNEDDS within 1 min and were clear or bluish in appearance. The results thus support the rapid formation of SNEDDS and the spontaneous nanoemulsification process. The results demonstrated that the selected SNEDDS formulations are capable of dilution in gastric fluid. 22 This is a critical step favoring enhancement in bioavailability and subsequent therapeutic efficiency.
As in the dispersibility test, the emulsification test showed that all the formulations were emulsified in less than 1 min (Table 2). On close examination of the data in Table 2, there appears to be a correlation between the relation of the S mix ratio and the emulsification rate. The emulsification time was the lowest for the S mix ratio of 1:1 and the highest for the S mix ratio of 4:1. Thus, the emulsification time followed a regular pattern in the order 1:1 < 2:1 < 3:1 < 4:1. Thus, it may be inferred that a higher concentration of surfactant in the S mix increases the emulsification time. When we further analyze the pattern of emulsifying time for each S mix ratio, higher oil concentrations result in a definite increase in emulsifying time. This observation is seen in all S mix ratios. In the selected formulations of the S mix ratio 1:1, the value of surfactants and cosurfactant is the same. Here also, an increase in the emulsifying time is seen for higher concentrations. Thus, this is a clear indication of the fact that the concentration of oil also influences the emulsifying rate. 11,31
Characterization of selected SNEDDS formulation
The results of droplet size, PDI, and zeta potential are shown in Table 3. The droplet size was well below 200 nm for all S mix ratios. From the droplet size of the S mix ratio 1:1, where the same concentrations of surfactant and cosurfactant were used, it can be inferred that a higher oil concentration leads to a higher droplet size. A similar effect of oil concentration could be seen for all other S mix ratios. 32 On comparing the droplet size resulting from the different S mix ratios, we observed that with an increase in surfactant concentration, the droplet size of the nanoemulsion decreased, because the amount of adsorbed surfactant at the oil–water interface of the droplet increases, thus reducing the interfacial tension between immiscible liquid, allowing the formation of small droplets. 33 However, when a S mix ratio of 4:1 was used the droplet size increased compared with the other three S mix ratios; this could be due to the presence of excess surfactant concentration causing penetration of excess water into the bulk of the oil phase resulting in the breakdown of interfacial tension and ejection of the droplet into the bulk of the aqueous phase. 33 The PDI was dependent on the concentration of oil, surfactant, and cosurfactant. From the PDI values of nanoemulsions having a S mix ratio 1:1, it is apparent that increasing the oil concentrations lead to lower PDI values (Table 3). A similar effect of oil concentration on the PDI can be seen for all other S mix ratios. Moreover, similar to the response obtained for droplet size, the PDI value was higher for the OFA4c when compared with that of the OFA3c. The zeta potential values were negative in sign for all the selected formulations, indicating a negative surface charge for the droplets. Similar to the effect of oil concentration on droplet size and PDI, an increase in oil concentration was found to increase the zeta potential. 32,33 This effect was more pronounced when the oil concentration was changed from 15% to 20%. Unlike for droplet size and PDI, the S mix ratio had no notable effect on the value of zeta potential. The average droplet size, PDI, and zeta potential of the optimized DHA-containing SNEDDS are shown in Figure 4.
Characterization of different compositions of DHA-containing SNEDDS.

SNEDDS: self-nanoemulsifying drug delivery systems; DHA: docosahexaenoic acid; PDI: polydispersity index; ZP: zeta potential; cP: centipoise; SD: standard deviation.

Average droplet size and zeta potential of optimized SNEDDS. SNEDDS: self-nanoemulsifying drug delivery systems.
The value of the refractive index of the SNEDDS samples is selected from different locations of the nanoemulsification region. The range of refractive index was found to be 1.40 to 1.49. There was no significant difference between the mean refractive index values of different formulations; this indicates the isotropic nature of the chosen formulations. 34
The results of viscosity determination are given in Table 3. Fish oil has high viscosity (around 20 cP) when compared to water. 35 Thus, it is obvious that higher oil content increases the viscosity of SNEDDS and this is supported by the viscosity data shown in Table 3. The S mix ratio also influences viscosity, with higher S mix ratios resulting in increased viscosity, with the high viscosity of both surfactant and cosurfactant contributing to this. 32 Thus, both oil concentration and S mix ratio were found to influence the viscosity of SNEDDS.
Based on the characterization results, four formulations, one from each S mix ratio, were selected for further studies. Thus, OFA1c, OFA2c, OFA3c, and OFA4c were selected for studies on in vitro release and ex vivo absorption.
The TEM image of the optimized formulation (OFA3c) (Figure 5) showed small droplet size near 100 nm and this agreed with the droplet size determined for the OFA3c formulation using the Zetasizer. Spherical morphology was observed for the droplets. Thus, the observed morphology was typical of reported SNEDDS formulations. 30

TEM image of the optimized SNEDDS formulation. TEM: transmission electron microscopy; SNEDDS: self-nanoemulsifying drug delivery systems.
In vitro drug release
The in vitro release profile (Figure 6) shows that the release of DHA was significantly (p < 0.01) greater and more rapid for all the SNEDDS compared than the DHA aqueous dispersion. This type of behavior is well known for SNEDDS, which show much greater release than the pure drug. 36,37 This demonstrates the importance of SNEDDS in increasing the bioavailability of DHA.

In vitro release profile of DHA using dialysis bag technique from different SNEDDS formulations in phosphate buffer pH 6.8. DHA: docosahexaenoic acid; SNEDDS: self-nanoemulsifying drug delivery systems.
The formulation OFA4c showed the greatest and most rapid release of DHA compared with the other formulations. The percentage cumulative release of DHA from the SNEDDS formulation in 6 h was found to be maximum for OFA3c (83.43 ± 3.25%) and OFA4c (88.32 ± 1.5%). However, there was no statistically significant difference (p ≥ 0.05) in release behavior between OFA3c and OFA4c at all-time points. On further close examination, the rank order of drug release at almost all time points was OFA4c > OFA3c > OFA2c > OFA1c. Thus, it can be concluded that a higher S mix ratio favors enhanced in vitro drug release. The in vitro release profile appears to be inversely correlated with the droplet size of the formulations; the rank order of the droplet sizes was OFA4c < OFA3c < OFA2c < OFA1c. Thus, the droplet size has a major contribution to the in vitro drug release, with higher droplet size leading to reduction in vitro drug release. Similar behavior of SNEDDS has been reported elsewhere. 22
Ex vivo absorption study: Non-everted gut sac method
The P app determined by the ex vivo absorption using non-everted gut sacs followed a similar pattern to that seen from the in vitro release profile. The rank order of P app for the different formulations was OFA4c (7.10 ± 0.56 × 10−6 cm/s) > OFA3c (6.97 ± 0.43 × 10−6 cm/s) > OFA2c (3.92 ± 0.52 × 10−6 cm/s) > OFA1c (2.16 ± 0.12 × 10−6 cm/s) (Figure 7); the P app of DHA aqueous dispersion was only 1.84 ± 0.26 × 10−6 cm/s. Thus, a higher S mix ratio favors enhanced ex vivo permeation. However, there was no statistically significant difference (p ≥ 0.05) in P app between OFA3c and OFA4c. The P app was also clearly related to the droplet size of the various formulations; thus, a lower droplet size favored higher values for the P app. 37,38 The high permeability of formulations with a higher S mix ratio could be due to higher DHA release from the SNEDDS. The observed results were in good agreement with the in vitro release profile of DHA from the SNEDDS. Based on the results of in vitro release and ex vivo absorption studies, formulation OFA3c was selected as the optimized formulation due to satisfactory droplet size, small PDI, higher zeta potential, and less use of surfactant compared to OFA4c. The final optimized formulation (OFA3c) was further studied in vivo.

P app of different SNEDDS formulations by non-everted gut sac method. P app: permeability coefficient; SNEDDS: self-nanoemulsifying drug delivery systems.
In vivo study
Figure 8 shows the plasma concentration of DHA at various times after the oral administration of DHA-containing SNEDDS or DHA aqueous dispersion and the corresponding calculated pharmacokinetic data are presented in Table 4. The C max and AUC0→t for the DHA-containing SNEDDS formulation were significantly (p < 0.01) greater than those for the DHA aqueous dispersion. The relative bioavailability of DHA from the SNEDDS formulation was found to be 332 ± 32% relative to the bioavailability of DHA from an aqueous dispersion. The time taken to achieve the maximum plasma concentration of DHA (t max) was also markedly reduced for DHA-containing SNEDDS relative to the DHA aqueous dispersion (Table 4). Thus, these data indicate that the formulation DHA-containing SNEDDS successfully enhanced the bioavailability of DHA. The in vivo results were in good agreement with the in vitro DHA release and ex vivo DHA permeation results. The increase in bioavailability could be due to small droplet size leads to better absorption. 22

Comparative concentration–time profiles of DHA in plasma after oral administration of DHA-containing SNEDDS and DHA aqueous dispersion. DHA: docosahexaenoic acid; SNEDDS: self-nanoemulsifying drug delivery systems.
Pharmacokinetic parameters of DHA after oral administration of DHA aqueous dispersion and SNEDDS (Mean ± SD, n = 4).a

DHA: docosahexaenoic acid; SNEDDS: self-nanoemulsifying drug delivery systems; SD: standard deviation.
a Control (DHA aqueous dispersion).
Conclusion
The optimized DHA-containing SNEDDS consist of Capryol 90, Tween 20, and PEG 200 as the oil, surfactant, and cosurfactant and showed outstanding self-emulsification characteristics along with enhanced dissolution or dispersibility rate. Moreover, significantly higher ex vivo absorption of DHA was observed which finally corroborated in the in vivo pharmacokinetics data. The relative bioavailability of DHA from the SNEDDS was found to be 3.32 times higher in comparison to a DHA aqueous dispersion. These findings are remarkably significant in the drug delivery research of DHA or other omega-3-fatty acids. It is known that SNEDDS being an aqueous free novel formulation system help in improving the drug stability. This is exceptionally important in case of polyunsaturated fatty acids (DHA or other omega-3-fatty acids) delivery as they are highly sensitive to hydrolysis and oxidation. Moreover, improved bioavailability would allow to reduce the dose and daily intake of omega-3-fatty acids. It is also important to consider here that formulation and manufacturing of SNEDDS does not require expensive setup compared to the existing omega-3-fatty acids formulations. Therefore, if we consider these facts the DHA-SNEDDS would be more economical compared to commercially available supplements. A potentially stable SNEDDS for DHA would be more appealing compared to exiting conventional formulations and could be established as an effective adjuvant therapy in various diseases.
Footnotes
Acknowledgements
The authors, therefore, acknowledge with thanks the DSR technical and financial support. The authors are thankful to Professor Brian L. Furman (University of Strathclyde, Glasgow, UK) for critical reading of the manuscript and language editing.
Author contributions
Nabil A Alhakamy and Shadab Md conceptualized the ideas; Khaled Hosny, Javed Ahmad, Sohail Akhter, and Adel F Alghaith performed the data curation; Javed Ahmad, Ahmed Kammoun, and Adel F Alghaith contributed to formal analysis; Nabil A Alhakamy, Sohail Akhter, Hani Asfour, and Shadab Md worked in investigation; Javed Ahmad, Ahmed Kammoun, and Shadab Md helped in development of methodology; Nabil A Alhakamy helped in project administration; Khaled Hosny and Adel F Alghaith provided resources; Sohail Akhter supervised the work; Shadab Md contributed to writing—original draft; Hibah M Aldawsari, Hani Asfour, and Mohammed W Al-Rabia helped in writing—review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah (grant no. RG-5-166-40).
