Abstract
Two processes of physical liquid phase stripping and chemical redox reduction were used to obtain graphene sheets. Fourier transform infrared spectroscopy and Raman spectroscopy test methods were used to compare and analyze the structure and disorder of graphene. The obtained graphene was modified with oleic acid and stearic acid. The dispersion stability of graphene as a lubricating oil additive was investigated by natural sedimentation method and spectrophotometry. The tribological properties of the graphene dispersion were investigated by a four-ball friction and wear tester. Scanning electron microscope and energy spectrometer were used to characterize and analyze the microscopic morphology and composition of the worn surface. The results showed that the modified liquid phase stripping graphene demonstrated the best anti-wear and anti-friction properties of the dispersion, the lowest friction coefficient is 0.0677, and the average friction coefficient is reduced by about 26%.
Introduction
Friction is inevitable when two entities are in contact under relative motion. 1 Friction causes wear and even failures in mechanical equipment. Therefore, the effective reduction of friction and the control of wear are important considerations in the performance of mechanical equipment and even in the improvement of the national economy. The use of lubrication is an effective method of controlling friction and wear in mechanical equipment. The traditional method is to add liquid lubricating oil as a lubricant to achieve the effect of anti-wear and anti-friction. However, traditional lubricants have begun to show their limitations in the case of microscale friction. 2 –4 Since two-dimensional material has a high specific surface area and is easily adsorbed on the contact surface, the direct contact of the friction pair is avoided, which is expected to bring new application opportunities to the conventional technology and engineering fields. Furthermore, modern technologies such as nanotribology play an increasingly important role.
Since 2004, Andre Geim and Kostya Novoselov 5 of the Department of Physics and Astronomy at the University of Manchester in the United Kingdom have prepared a single layer of graphene for the first time by using transparent tape to repeatedly paste and tear Highly Oriented Pyrolytic Graphite. This single-layer carbon nanomaterial with a theoretical thickness of only 0.335 nm has become another research hot spot. Following fullerenes and carbon nanotubes, it has attracted widespread attention from scientists in the fields of physics, chemistry, and materials. Related theoretical calculations and experimental research studies are emerging in endlessly. Graphene has a hexagonal honeycomb two-dimensional grid structure composed of sp 2 -hybridized single-layer carbon atoms with higher thermal conductivity than carbon nanotubes and diamonds, while maintaining the original planar carbon six-membered ring conjugated crystal of graphite structure. Graphene has remarkable mechanical, electrical, and thermal properties, as well as excellent nanoscale effects and low density. 6 On the microscopic scale, the van der Waals force between the graphene layers makes graphene an ideal candidate for ultra-lubricity, 7 –9 and can be used to establish a promising solid lubricant without friction sliding. 10
The lubrication efficiency of graphene materials depends on its composition, 11 graphene layer number, 12 morphology, 13 and experimental conditions. 14,15 For example, multilayer graphene films impair adhesion and friction. 16 This film has a coefficient of friction comparable to that of bulk graphite. According to Shin et al. 17 the coefficient of friction of the epitaxial graphene microfilm remained at a level of 0.03 regardless of the number of layers. In the study of Huang et al. 18 a critical load was detected under which the wear of the single-layer graphene disappeared. The wear system has a low coefficient of friction of about 0.01.
The mass production of graphene is the core of promoting the industrialization of graphene, and one of the key problems is the preparation method of graphene. Existing preparation methods can be divided into physical, chemical, organic chemical synthesis and doping methods. 19,20 This article mainly studies the dispersion stability and anti-wear and anti-friction properties of graphene based on physical and chemical methods in 500N base lubricant (viscosity index 99 mm2 s−1). Although graphene can effectively reduce friction and wear, insolubility in the medium and tendency to form agglomerates are the two major problems encountered while dealing with graphene as nanofiller, 21 which will lead to the weakening of the interaction between the graphene and polymer interface. 22 At present, it is a more effective method to modify the surface of graphene 23 to improve the stability of the dispersion. Therefore, the surface of graphene was modified by oleic acid and stearic acid, 24 the graphene dispersion with lubricating oil as the solvent, in order to test the dispersibility and tribological properties.
Experimental materials and methods
Material characterization
The physical liquid phase stripping process involves directly dispersing the expanded graphite into a solvent, rapidly heating to 1000°C or more to remove the surface oxygen-containing group, adding it to the solvent, and then stripping by ultrasonic wave, microwave, heating, gas flow, and electrochemical methods. A graphene dispersion was obtained, which was then subjected to centrifugal drying to obtain a graphene sheet. 25
Chemical redox is the gradual addition of KMnO4 26 to a reaction vessel containing H2SO4 and NaNO3. With constant stirring, the reaction mixture is thickened into a paste. Thereafter, distilled water is slowly added to the reaction slurry. After the temperature is raised, excess distilled water and hydrogen peroxide are added, and then reduced to graphene by hydrazine hydrate. The graphene powder was obtained by drying using ultrasonic waves.
As shown in Figure 1, the scanning electron microscopy (SEM) images of physical graphene (Figure 1(a)) and chemical graphene (Figure 1(b)) under scanning electron microscope have obvious sheet structure and wrinkles. There are more graphene layers in darker locations in the image, and relatively fewer graphene layers in lighter locations.

SEM images of physical graphene (a) and chemical graphene (b) magnified ×2500 under scanning electron microscope. SEM: scanning electron microscopy.
The Fourier transform infrared spectroscopy could confirm the presence of the graphene. Figure 2 shows the infrared spectrum graphs of the physical method graphene and the chemical method graphene, respectively, from which it can be seen that the vibration peaks of the water molecules corresponding to 3489.65 and 3488.27 cm−1, 2922.41 and 2923.01 cm−1 correspond to C=O, 1634.53 and 1632.69 cm−1 correspond to C=C, 1080.31 and 1095.87 cm−1 correspond to C–O. The 1557.64 cm−1 at the left corresponds to C–C. In the figure on the right, 1464.89 cm−1 and 1384.66 cm−1 correspond to the functional groups –OH and C–OH, respectively, which shows that there are still a small amount of unreduced oxygen-containing functional groups and adsorbed water molecules remaining in the chemical graphene. The results show that the physical method graphene has high purity graphite than the chemical method graphene.
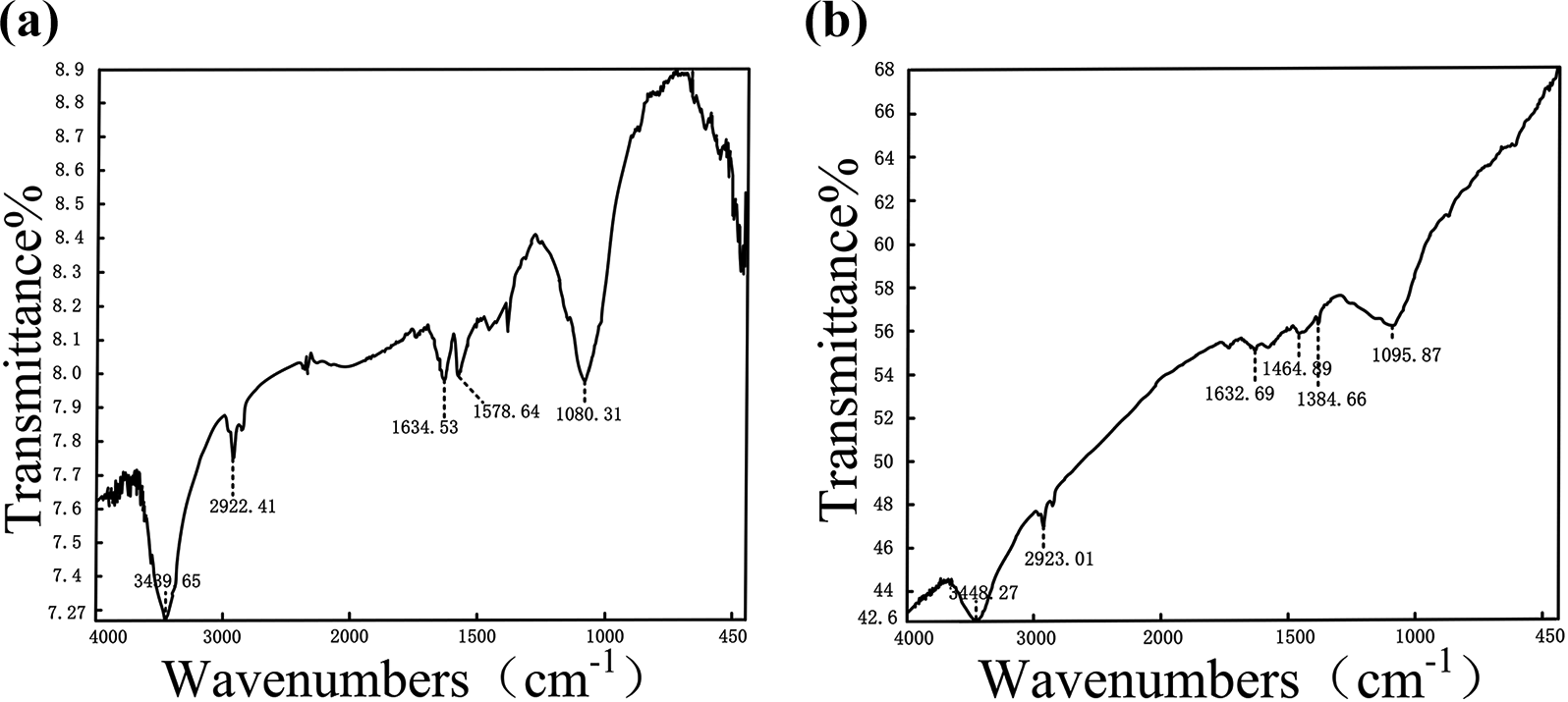
Infrared spectra of physical method graphene (a) and chemical method graphene (b).
For graphene research, it is crucial to determine the number of layers and quantify the disorder. Figure 3 shows the Raman spectra of two graphenes. As shown in Figure 3(a), there are three peaks, and the positions of these three peaks are at 1349 cm−1, 1582 cm−1, and 2692 cm−1, respectively. There are two peaks in Figure 3(b), the positions of these two peaks are at 1342 cm−1 and 1597 cm−1, respectively. The G peaks in both graphenes are relatively narrow, which indicates that the carbon exhibits a hexagonal arrangement and is sp2 hybrid. The D peak comes from the defects in the carbon of the graphene skeleton and the vibration caused by the irregular arrangement of the edge carbon. These carbons are sp3 hybrids. However, the G′ peak in Figure 3(a) of the physical method graphene indicates that there are interlayer stacking of carbon atoms in the graphene sample, and the physical method has fewer graphene layers. And the intensity of the D peak is weaker than that of the G peak, indicating that the disorder of atoms in the graphene by physical method is relatively low.

Raman spectra of physical graphene (a) and chemical graphene (b).
Preparation of graphene as oil-based additive dispersion
In order to solve the problem of agglomeration of graphene in lubricating oil, the surface modification was carried out in this test. After mixing oleic acid 5 ml and stearic acid 3 g, the mixture was sonicated for 30 min and the temperature was set to 30°C. A total of 80 ml of 500N base lubricating oil was placed in a 100 ml three-necked flask, 0.1 g of graphene was added, and heated to 80°C in a stirrer (the optimum temperature for oleic acid–stearic acid modification was 80°C). After 5 min in a constant temperature water bath, the modifier was instilled at a constant rate with a micropipette and stirred for 30 min at a rate of 20 r s−1.
The solution obtained above was transferred to an ultrasonic machine, the temperature was set to 30°C, and ultrasonication was performed multiple times. That is, the modified graphene dispersion is obtained. The dispersion stability of the modified graphene was analyzed by natural sedimentation method and spectrophotometry (Shimadzu Enterprise Management (China) Co., Ltd.UV-2550 UV-VIS spectrophotometer).
Tribological test
The test used an MRS-10A microcomputer to control the four-ball friction and wear test machine, testing the pure lubricating oil, physical modified and unmodified graphene dispersion, and chemical modified and unmodified graphene dispersion. Experimental conditions: 392 N load, room temperature, spindle speed with 600 r min−1 for 60 min. During the test, data were collected every 10 s and a total of 360 sets of data were obtained. After the test, the experimental steel balls were washed several times with absolute ethanol and air-dried in a cool place.
Test results and analysis
Evaluation method of dispersion stability
Dispersion stability, that is, both dispersion and stability. 27 Dispersibility refers to the characteristic that the graphene additive can be uniformly dispersed in a lubricating oil in a small volume under a certain dispersion condition. Stability refers to a characteristic that the graphene additive in the lubricating oil maintains stable suspension for a long time with a certain particle size.
Through comparative analysis (Figure 4), for the chemical graphene, when the sedimentation time is 6 days, the dispersion begins to stratify, and the color of the lubricating oil changes significantly, and significant white floes began to appear at the delamination .With the extension of the settling time, the apparent precipitation of the chemical graphene dispersion is serious. The physical method graphene dispersion is basically stable in color and remains black after standing for 20 days.

Dispersion of graphene in chemical process (left) and physical method (right) in base oil.
The ultraviolet–visible spectra of the two graphene dispersions are shown in Figure 5. The curves of different dispersions in the absorbance range of 300–800 nm showed a consistent change trend, that is, as the settling time increased, the absorbance gradually decreased. The absorption value of the physical method graphene dispersion is higher than that of the chemical graphene, indicating that the physical method graphene is more easily dispersed in a solvent and has higher dispersion stability. This may be due to the structural damage of the chemical graphene in the preparation process.

Curve of absorption value of two graphene dispersions with sedimentation time.
In addition, this experiment studied the variation of the absorption value of graphene dispersion with wavelength. It can be seen in Figure 6 that the absorption value of graphene dispersion gradually stabilizes with increasing wavelength. For the physical method graphene dispersion, the absorption value tends to be stable at the wavelength of 400 nm notably from the third day. The absorption value on the first day is higher than the other days in the wavelength range. The change curve of the chemical graphene dispersion every day is close at 400 nm, the absorption value tends to be stable.
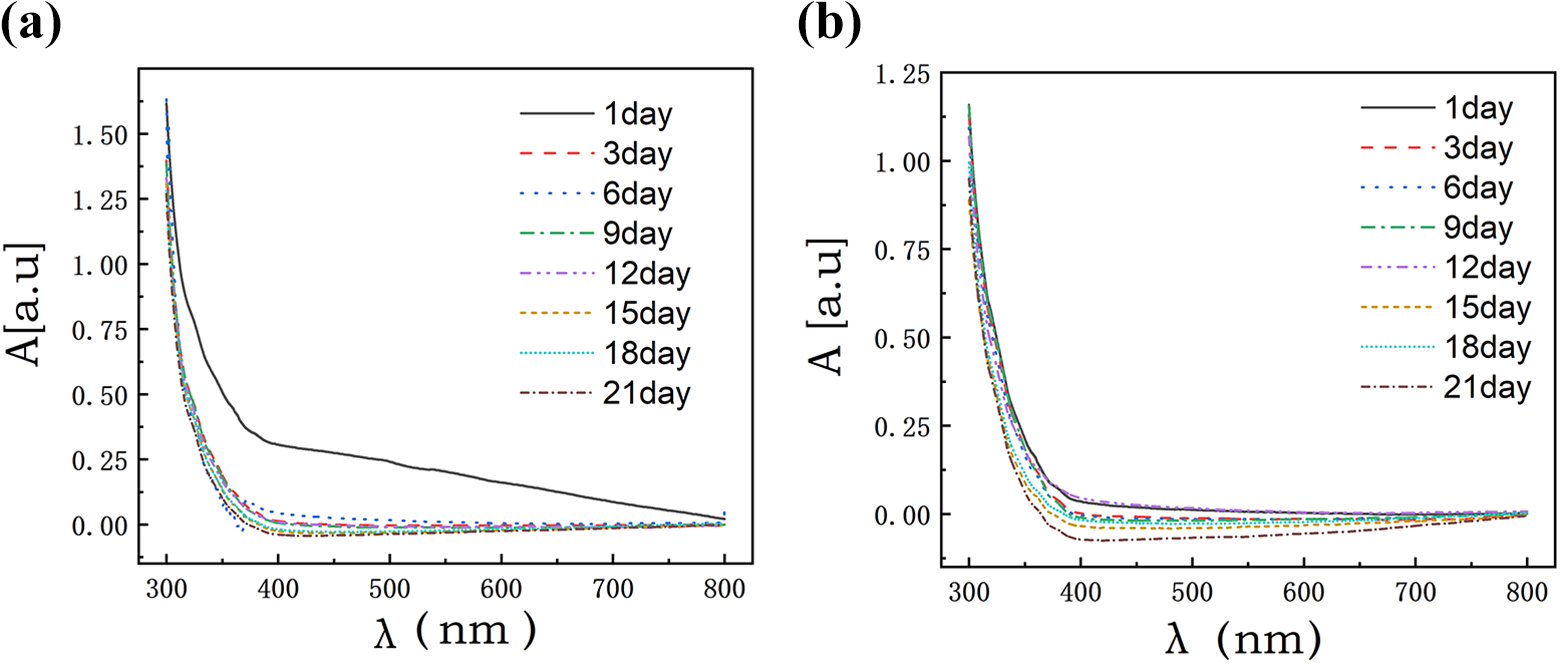
Curve of absorption of physical graphene (a) and chemical graphene (b) with wavelength.
Tribological test results analysis
The lubrication mechanism of graphene materials has been the subject of active research. Theoretical studies 28 show that the formation of lubricating film is related to the chemical adsorption of graphene nanosheets on the iron surface. Under the action of the mutual movement of the upper and lower friction pairs, slippage occurs between the layered nanosheets. The graphene acts directly on the surface of the substrate, and then undergoes a tribochemical reaction with the metal substrate to form a lubricating film of the carbonaceous material, thereby preventing the direct contact of the friction pair and reducing the friction and wear of the material.
According to Marchetto et al., 29 in the friction between steel and steel, the ethanol dispersion of graphene acts as a lubricant and the coating is unstable. After evaporation of the solvent, the graphene sheets no longer move, which results in an increase in the coefficient of friction. Novikova et al. 22 studied the effect of multilayer graphene oxide dispersion on the friction pair in glycerin. When the additive concentration was 0.05 wt%, the ultra-low friction coefficient was obtained. In our work, we studied the lubricating effect of the unmodified and modified graphene dispersions under a load of 392 N. Figure 7 shows the coefficient of friction of a pure base oil with both modified and unmodified graphene dispersions as a function of time. As the friction factor is affected by factors such as the magnitude and speed of the normal load, the stiffness and elasticity of the friction pair, the sliding speed, the geometric characteristics of the friction surface contact, and the physical properties of the surface layer during the sliding process, irregular friction vibration occurs.

(a) The curve of friction coefficient of pure base oil and two modified physical graphene dispersions with time. (b) The curve of friction coefficient of pure base oil and unmodified and modified physical method graphene dispersion with time. (c) The curve of friction coefficient of pure base oil and unmodified and modified chemical graphene dispersion with time.
It can be clearly seen from the figure that the friction coefficient of the modified graphene is greatly reduced. On the contrary, it can be seen from Figure 7(b) that the friction coefficient of the unmodified dispersion is higher than that of the pure base oil. This is because the unmodified and unbroken graphene particles have agglomerated. As the friction progresses, the agglomerates act as abrasive particles to make the lubricating oil film discontinuous, which in turn causes an increase in wear. During the preparation process, the unmodified chemical graphene dispersion liquid has its structure destroyed, and the surface of the graphene oxide obtained in the intermediate contains certain oxygen-containing groups, which can confer surface activity, and so on. Special characteristics are easier to contact with lubricating oil than unmodified physical graphene particles. The modified physical graphene in this test showed a good lubricating effect and remained stable throughout the test. The average friction coefficient was maintained at about 0.07, which was reduced by about 26%, and the lowest friction coefficient reached 0.0677.
Based on the above test results, the increase in the friction coefficient at the beginning of the experiment is related to the running-in process. The initial roughness of the friction metal surface causes a temporary increase in friction until the surface flattens and becomes smooth, thereby reducing friction. Compared with pure base oils, graphene as a lubricating oil additive can more effectively improve the wear resistance of lubricating oil. It is speculated that the mechanism of action is as follows: For pure base oil, in the initial stage of friction, the oil film is not uniformly dispersed between the friction pairs, which may lead to direct contact of the friction pair, as the friction coefficient is large and the lubricating oil increases with time. An oil film is formed between the friction pairs to protect it, so the friction coefficient at this stage is maintained in a relatively stable state. When a certain amount of unmodified graphene is added to the base oil, the oil film still plays an important role in the rubbing process. The difference is that the graphene sheets added in the oil can easily enter between the contact faces and form a protection layer. The graphene debris formed during the rubbing process can also fill the wear scar on the contact surface and bear part of the load, thereby achieving the effect of reducing wear and increasing load-carrying capacity. However, graphene particles and metal wear debris from the agglomeration make the contact surface rough, which reduces the wear resistance.
Wear surface characterization
According to the investigation of wear marks by SEM and energy-dispersive spectroscopy (EDS), the wear surface characteristics and anti-wear mechanism of the graphene dispersion on the steel ball are further revealed. Figure 8(a), (b), (c), (d), and (e) shows the wear spots on the surface of the steel ball after the pure lubrication, unmodified physical method graphene dispersion liquid, modified physical method graphene dispersion liquid, unmodified chemical method graphene dispersion liquid, and chemical graphene dispersions, respectively. Figure 8(a-1) to (e-1) shows magnified wear scars, and Figure 8 (a-2 to e-2) and (a-3 to e-3) shows the points and regions selected by the EDS spectrogram, respectively. After the friction test, the wear marks of the steel ball are clear traces of wear spots, obvious plowing effects, deformation marks, and covered by many scratches. Figure 9 shows a histogram of the diameter of the wear spot. It is more intuitive to see that the diameter of the wear scar on the surface of the steel ball under the action of the modified graphene dispersion after the modification is 8.0% lower than that on the surface of the steel ball under the action of pure base oil, compared with that in the unmodified physical graphene The diameter of the wear spot on the surface of the steel ball decreased by 10.7% under the action of the graphene dispersion. It can be seen in Figure 8(a-1) to (e-1) that the SEM micrograph of the wear trace shows that there are many deep grooves and pits in the wear trace. Dark areas are clearly visible on the friction surface, which may be due to the formation of rubbing film graphene particles. By EDS analysis inside the worn surface (Figure 8(a-2 to e-2) and (a-3 to e-3)), it was confirmed that the film formation was confirmed to be an increase in the carbon concentration on the worn surface. This result proves that an increase in graphene particles leads to an increase in lubrication, and the modified graphene particles can be stably adsorbed on the friction surface, migrate on the friction surface to form a physically deposited film, and provide lower shear strength, resulting in interlayer sliding, thereby improving the wear resistance of the friction pair surface.

(a to e) SEM micrographs and EDX spectra of steel surface wear trajectories of pure base oil, unmodified and modified physical graphene, and unmodified and modified chemical graphene lubricated steel, respectively. SEM: scanning electron microscopy; EDX: energy-dispersive spectroscopy.

Surface wear diameter of steel with pure base oil, unmodified and modified physical method graphene, and unmodified and modified chemical graphene.
The chemical redox process is mainly carried out by chemical oxidation to form a carboxyl group and a hydroxyl group at the edge, and an oxygen-containing group such as an epoxy group or a carbonyl group is intercalated between the layers. This process can enlarge the graphite layer spacing, and the graphene oxide is obtained by external force stripping, further reducing the graphene. The product obtained by this method has high yield and simple equipment, but the reaction takes a long time and the damage to the graphene structure is serious.
Graphene prepared by physical liquid phase stripping is an intrinsic graphene material without any physical and chemical properties, so it has high quality and is suitable for theoretical research. This is also an important factor in the tribological performance of graphene dispersions obtained by physical methods over chemical methods.
Conclusion
In this experiment, the effects of two different preparation processes on the dispersion stability and tribological properties of graphene dispersion were studied. The results show that the modified physical method graphene dispersion has the best anti-wear and anti-friction performance, with the lowest friction coefficient. The lubrication effect under the scanning electron microscope is the most obvious. This is because the graphene oxide surface obtained by the redox method to obtain graphene contains certain oxygen-containing groups. These oxygen-containing groups give special characteristics such as surface activity. The resulting graphene has large structural defects and contact areas. The redistribution of interfacial atoms and the reconstruction of the outermost surface layer as well as the in situ formation of interfacial nanostructures usually occur, affecting their lubricating properties.
Since the stability of the lubricating state is highly correlated with the characteristics of the environmental gas, it is very important for the development of nanotribology to study the tribochemical interaction between the carbon-metal surface and gaseous substances. An in-depth study of the chemical reactions occurring on the surface and interface during the friction and wear of materials is of great significance for exploring the wear mechanism of materials at the microscopic or molecular and atomic scales.
Footnotes
Author contributions
YL and YD contributed equally to this work.
Authors’ note
YL and YD are co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
