Abstract
Textile-based piezoelectric nanogenerator generates electrical energy from human motion. Here a novel type of textile-based piezoelectric nanogenerator is reported which is fabricated using the growth of silver-doped zinc oxide on carton fabric. Along with the optical and structural characterization of silver-doped zinc oxide nanorods, the electrical characterization was also performed for silver-doped zinc oxide piezoelectric nanogenerator. The silver-doped zinc oxide piezoelectric nanogenerator was found to generate three times greater power compared to undoped zinc oxide piezoelectric nanogenerator. By applying external mechanical force of 3 kgf and 31 MΩ of load resistance, the silver-doped zinc oxide piezoelectric nanogenerator generated an output power density of 1.45 mW cm−2. The effect of load resistance and load capacitor was determined and optimum values were calculated. The maximum output power was observed at a load resistance of 31 MΩ. The silver-doped zinc oxide piezoelectric nanogenerator was utilized to charge load capacitors and found that maximum energy could be stored at optimum load capacitance of 22 nF in 600 s (1800 cycles). This research may provide the opportunity to design high-output textile-based nanogenerators for practical applications like powering portable devices and sensors.
Keywords
Introduction
Energy harvesting technology obtains electrical energy from the ambient environment such as solar, mechanical, and thermal energy. Energy harvesting has emerged as one of the most promising solutions for the rapidly increasing energy demand. 1 Self-powered energy system is becoming ubiquitous and is receiving great interest, because they reduce the size and weight of electronics devices. Piezoelectric energy is generated by mechanical and vibrational sources which is used to charge portable devices and wireless sensors. 2 Energy harvesting devices can be fabricated by using piezoelectric materials such as zinc oxide (ZnO), indium nitride, gallium nitride (GaN), and cadmium sulfide which has lower output power density for practical application. 3 –6 ZnO is one of the best nanomaterials which has unique piezoelectric, semiconducting, and optical properties due to which it has been used in different piezoelectric devices such as piezoelectric gate diode, piezoelectric field effect transistors, and piezoelectric nanogenerators (PENGs). 7 –10 On the other hand, the synthesis of nanomaterials is at a new stage of development, where it is common requirement to control the properties of the obtained nanostructures, and enables their repeatable application, especially for energy harvesting technology. There are number of reports for controlling size, density, growth temperature, and crystal quality of ZnO which enhanced its applicability in fabrication of PENG. 11 –14 Different approaches have been used for the fabrication of PENGs on nonflexible substrate such as GaN, 15 glass, 16,17 titanium foil, 18 and silicon. 19 –22 However, these materials exhibit poor flexibility and deformation due to which PENGs are facing a challenge in practical applications. In addition, there has been rapid progress on fabricating stretchable or flexible PENGs based on ZnO nanorods. The frequently used flexible substrates are sponge, 23 flexible plastic, 24 polyethylene terephthalate, 25 –27 paper, 28 –30 and textile fabric. 31 Among these, textile materials possess mechanical flexibility, low cost, lightweight, and used for integration into different areas such as clothing accessories. 32
Some researchers have used these smart functional textiles for the fabrication of PENGs. Khan et al. reported ID PENG which was fabricated on conductive textile fabric using hydrothermal method. 31 The output performance of ID PENG has reduced performance due to screening of free charge and parasitic capacitor effect. There is a need to enhance the efficiency of one-dimensional ZnO nanogenerator by modifying its various properties, such as optical, electrical, and piezoelectric, 33,34 which can be accomplished by doping the ZnO with noble metals such as gold, 35 palladium, 36 and silver (Ag). 37 Ag is considered as a good dopant for ZnO due to high solubility, large ionic radii, and less orbital energy, 38 since Ag doping increases the surface defects and optical property that shifts the optical absorption toward visible region. 39 Ag element acts as acceptors and provides a high concentration of charge carriers in ZnO host materials. 40 Fan and Freer reported that Ag act as an amphoteric dopant existing both on substitutional Zn sites and in the interstitial sites. 41 However, there is no report till now on the PENG of Ag-doped ZnO nanorods on cotton fabric.
In this research, a novel method for fabrication of textile-based PENG is reported in which Ag-doped ZnO nanorods on cotton fabric substrate are grown using hydrothermal method and utilized to make PENG. The fabricated PENG was employed to store charge in load capacitor. It was found that reactance of capacitor affects the charging capability of PENG. The load capacitance was optimized at 22 nF for maximum stored energy of 0.5819 nJ which gives a reactance of 31 MΩ.
Experimental details
Materials
All chemical reagents were purchased from Sigma Aldrich (USA, St Louis, Missouri), except zinc acetate dehydrate and zinc nitrate hexahydrate which were purchased from Ajax Fine Chem Pty Ltd (Australia). The chemical reagents used in the fabrication process of the Ag-doped ZnO nanorods were of analytical grade and used without further purification.
Synthesis of pure ZnO and Ag-doped ZnO nanorods
The cotton fabric of 1 cm × 1 cm was ultrasonically cleaned in acetone and deionized (DI) water for 10 min, respectively, and dried in air. Then, the cotton fabric was immersed in 1% 1-dodecanethiol for 2 h to facilitate the subsequent seeding of ZnO nuclei. The ZnO seed layer was deposited using sol–gel process. The sol–gel was prepared using zinc acetate dehydrate (Zn(CH 3COO)2).2H2O) (99.5%) and sodium hydroxide (NAOH; 97%) in methanol. Initially, 0.01 g of zinc acetate dehydrate was dissolved in 50 ml methanol at 60°C and 0.03 g of NAOH was dissolved in 25 ml methanol at 60°, separately. The NaOH-based solution was added drop wise into zinc acetate solution with constant stirring at 60° for 2 h to maintain its transparency. Finally, fabric samples were inserted in sol–gel for few seconds and then annealed at 80°C for 10 min. This process was repeated three times. The nutrient solution was prepared using 0.03 M zinc nitrate (Zn(NO3)2.6H2O; 98%), 0.03 M hexamethylenetetramine (C6H12N4; 94%), and 0.03 M silver nitrate (99.7%) in 1000 ml of DI water. The prepared fabric samples were stuck on an unsinkable substrate and put upside down in nutrient solution. The growth process was carried out for 6 h at 85°C. After 6 h, the fabric samples were dried and annealed for 1 h at 80°C to obtain the fine crystal structure of Ag-doped ZnO nanorods.
Characterizations of pure and Ag-doped ZnO nanorods
The morphological properties of undoped and Ag-doped ZnO nanorods on cotton fabric were studied by scanning electron microscopy (SEM; Hitachi, S-3400N). The compositional studies of the nanostructures were carried out by energy-dispersive spectroscopy (EDS) attached to the SEM. For SEM analysis, cotton fabrics samples were prepared by making its surface conductive with gold sputtering, whereas no preparation step was done before X-ray diffractometer (XRD) analysis. The crystalline structure of the undoped and Ag-doped ZnO was determined by using a X’Pert PRO 3040/60 Philips XRD at an accelerating voltage of 40 kV using copper Kα line (λ = 0.1542 nm). XRD was performed in the 2θ range of 20–70° with a step size of 0.01° and a scanning rate of 0.02 steps s−1. For optical characteristics, the diffuse reflectance spectra (DRS) of the undoped and Ag-doped ZnO nanorods were investigated in the wavelength range of 200–850 nm on a Perkin-Elmer ultraviolet–visible (UV-Vis) diffuse spectrophotometer (National Centre of Physics, Islamabad, Pakistan). The functional group analysis of the nanostructure was performed in the transmittance mode on a Thermo Scientific (model Nicolet 6700) Fourier-transform infrared spectrophotometer (FTIR), in a spectral range of 400–4000 cm−1. The thermal properties of the sample were investigated by using a thermal analyzer (thermogravimetric analysis (TGA); SDT Q600 (TA Instruments, New Castle, Delaware, USA). The sample was heated from approximately 30°C to 800°C at the rate of 10°C min−1 under a nitrogen flow rate of 200 ml min−1.
Device fabrication and its characterization
The fabrication of PENG was achieved by placing thin sheets of copper (Cu; 0.7 µm), one on the top of Ag-doped ZnO nanorods film grown on cotton fabric and other on the bottom of cotton fabric. The fabrication steps of Ag-doped ZnO nanorods PENG are shown in Figure 1. In order to protect the Cu electrode and PENG during measurement, an adhesive tape was applied on the bottom and top sides of the PENG. The size of the Ag-doped ZnO nanorods PENG is 1 cm2. The output voltage and current of the PENG were detected by an oscilloscope (Wave Pro 735Zi; Lecroy, USA).
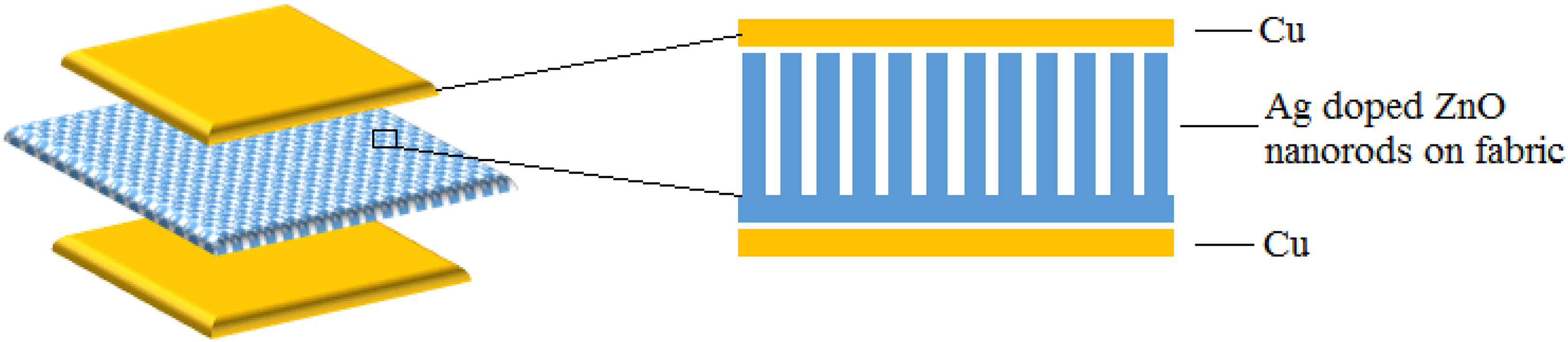
The schematic diagram of fabrication steps of Ag-doped ZnO nanorods PENG. Ag: silver; ZnO: zinc oxide; PENG: piezoelectric nanogenerator.
Result and discussion
Morphological analysis
The morphology of cotton fibers with and without ZnO growth is shown in Figure 2. Figure 2(a) shows the SEM image of the original cotton fibers without ZnO. Figure 2(b) clearly shows the dense growth of nanorods on cotton fibers. It was observed from SEM image that Ag-doped ZnO nanorods have homogenous growth on the entire surface of cotton fibers. The good adhesion between Ag-doped ZnO nanorods and cotton fiber is due to the hydroxyl (OH) group presented in the chemical formula of the cotton. Ag-doped ZnO nanorods can easily bind to OH groups through possibly hydrogen bonding. 42 The average diameter of the Ag-doped ZnO nanorods was found in the range of 85–90 nm, as shown in Figure 2(c).

SEM micrographs of (a) magnified view of original cotton fiber without ZnO growth, (b) magnified image of Ag-doped ZnO nanorods grown on cotton fiber, and (c) Ag-doped ZnO nanorods grown on the cotton fiber at higher magnification. SEM: scanning electron microscopy; Ag: silver; ZnO: zinc oxide.
Compositional analysis
EDS was used to determine the chemical composition of the undoped ZnO and Ag-doped ZnO samples, as shown in Figure 3(a) and (b). The EDS analysis confirms the presence of zinc (Zn), carbon (C), oxygen (O), and Ag elements. There are no detectable traces of any impurity element in EDS profile of pure and Ag-doped ZnO nanorods. The strong signals of O and C in EDS spectra of Figure 3(a) and (b) are due to cotton fabric which consists of a long chain of glucose molecules. 43,44 The EDS spectral peaks of Ag appeared at 3.1 KeV and Zn appeared at 1.0, 8.3, and 8.8 KeV, as shown in Figure 3(b). Also, the strong signal relevant to C at 0.3 KeV and O peak at 0.5 KeV is due to cotton fabric.

EDS analysis of (a) undoped ZnO nanorods and (b) Ag-doped ZnO nanorods. EDS: energy-dispersive spectroscopy; Ag: silver; ZnO: zinc oxide.
Optical analysis
The optical properties of pure ZnO and Ag-doped ZnO were determined by UV-Vis DRS. Figure 4(a) shows the diffuse reflectance spectrum of nanomaterials measured at room temperature in the wavelength range of 245–800 nm. The pure ZnO spectra showed high reflectance about 92% in visible region. Reflectance of Ag-doped ZnO samples decreased by approximately 39% after incorporation of Ag with ZnO. 45

(a) UV-DRS spectrum of ZnO and Ag-doped ZnO nanorods on cotton fabric; (b) Kubelka–Munk diffusive reflectance; and (c) Tauc’s plot for band gap energy estimation of ZnO and Ag-doped ZnO nanorods on cotton fabric. UV: ultraviolet; DRS: diffuse reflectance spectra; Ag: silver; ZnO: zinc oxide.
The Kubelka–Munk function was used to determine the value of optical band gap of pure ZnO and Ag-doped ZnO nanorods, estimated optical absorption edge of nanostructures 46
Where F(R) is the Kubelka–Munk function which corresponds to the absorbance 47 and R is the absolute value of reflectance. The absorption edge was calculated by plotting a graph between F(R) and wavelength (λ) and then extrapolating F(R)-λ of curve to λ-axis at F(R) = 0 corresponds to the absorption edge of pure ZnO and Ag-doped ZnO at 398 and 338 nm, respectively. Alternatively, the band gap energy (E g) of samples was also determined (see Figure 4(c)) via Tauc’s plot by employing the linear correlation between Kubelka–Munk function F(R) and E g 48
where
Structural analysis
The crystalline properties of pure ZnO and Ag-doped ZnO were analyzed by XRD spectroscopy at room temperature in the range of 2

XRD pattern of (a) pure ZnO and Ag-doped ZnO nanorods prepared by hydrothermal method. (b) The enlarged view of (100), (002), and (101) peaks of pure ZnO and Ag-doped ZnO nanorods. XRD: X-ray diffractometer; Ag: silver; ZnO: zinc oxide.
Vibrational analysis
As-synthesized pure ZnO and Ag-doped ZnO nanorods on cotton fabric were analyzed by FTIR, as shown in Figure 6. In the FTIR spectrum, metal oxides exhibit absorption bands below 1000 cm−1, which arise from interatomic vibrations. 54 It was found that the absorption band at around 470 and 719.45 cm−1 was indicated the stretching mode of Zn–O and Ag–O bonds. 55,56 The peak near to 1380.84 and 1450.21 was observed in sample that corresponds to the stretching vibrations of C–O group, which may be due to zinc acetate used in reaction. The peak located at 1721 cm−1 was assigned to (C=O) stretching vibration of cellulose. There is an absorption band located at 2240 cm−1 due to the stretching modes of vibration which formed as a result of binding of O–H group present in the cotton fabric. The absorption peak in the range of 2924.09 cm−1 indicates the stretching mode of C–H in the methyl group. The absorption bands in the range of 3431 cm−1 were observed which indicate the stretching mode of OH group of cotton fabric. 50
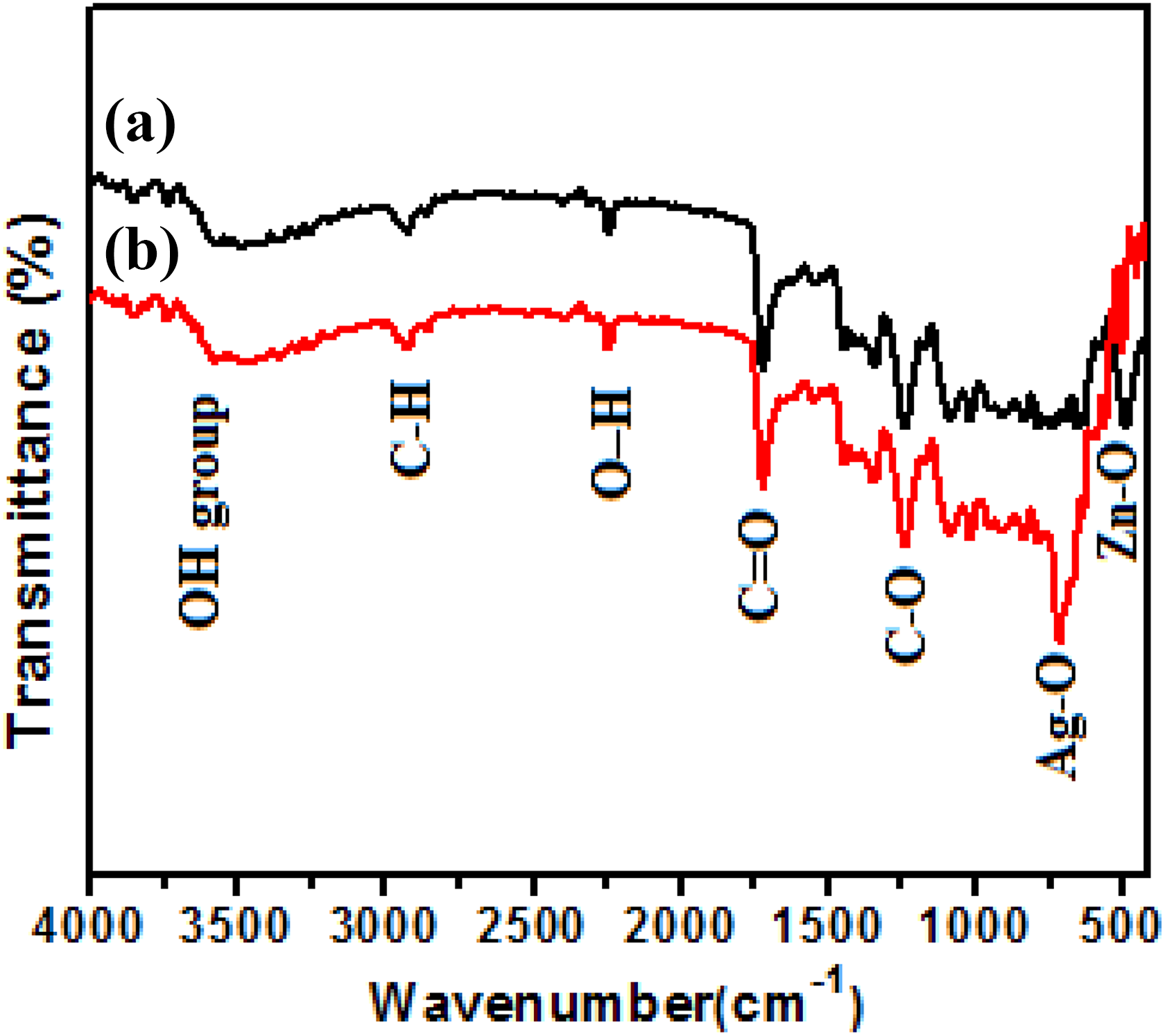
FTIR spectrum of (a) pure ZnO and (b) Ag-doped ZnO nanorods. FTIR: Fourier-transform infrared spectrophotometer; Ag: silver; ZnO: zinc oxide.
Thermal analysis
The thermal properties of pure ZnO and Ag-doped ZnO nanorods on cotton fabric were determined by TGA and derivative thermogravimetry analysis, as shown in Figure 7(a) and (b). As-synthesized fabric sample was heated in a ceramic pan up to 800°C at the rate of 10°C min−1 under a nitrogen atmosphere. In TGA analysis, three main decomposition steps can be observed. In the first step (35–103°C), a small weight loss (5%) and (9%) was observed in pure ZnO fabric and Ag-doped ZnO fabric samples, which was ascribed to the evaporation of moisture from the surfaces of the samples. 57 In the second step (103–500°C), the main weight loss was occurred due to the pyrolysis of cellulose. 58,59 The maximum pyrolysis rate occurred at 430°C and 400°C, with weight losses reaching 63% for the pure ZnO fabric and 41% for Ag-doped ZnO fabric, respectively. In the third step (500–800°C), the weight loss percentages of pyrolysis residue were approximately 7 and 13 mass % for the pure ZnO fabric and Ag-doped ZnO fabric, respectively.

TGA-DTG curves of (a) pure ZnO and (b) Ag-doped ZnO nanorods on cotton fabric. TGA: thermogravimetric analysis; DTG: derivative thermogravimetric; Ag: silver; ZnO: zinc oxide.
Electrical output of a PENG
By pressing the top electrode, undoped ZnO and Ag-doped ZnO nanorods are deformed and create piezoelectric potential along the growth direction. The potential is generated by the relative dislocation of Zn+2 cations and O−2 anions due to the piezoelectric effect in the wurtzite crystal structure. Thus, these ions can neither move nor recombine without releasing the strain. The pure and Ag-doped ZnO nanorods could provide well order synchronization that maximum of the nanorods generate piezoelectric potential at the same time and in the same direction, which ultimately decreases the screening effect and enhances the output of PENG.
When top electrode is pressed by some external force, equal amount of positive and negative piezoelectric potential is produced on the top and bottom side of ZnO nanorods. At this time, electrons will flow from the top electrode to the bottom electrode through external circuit and accumulate at bottom electrode. As the external force is released from the Ag-doped ZnO nanorods, which results in the diminution of piezoelectric potential, at the same time, the electrons flow reversely. Figure 8 shows a schematic illustration of the fabricated PENG.

A schematic illustration of the fabricated PENG. PENG: piezoelectric nanogenerator.
The insulating fabric layer plays an important role in the working mechanism of the PENG by creating a high potential barrier between the top and bottom electrodes. Moreover, its insulating characteristic avoids the electron leakage through internal ZnO nanorods film. The electrical output of PENG was measured by applying periodic external mechanical pulses. In order to confirm that the measured signal originated from the device rather than noise, a switching polarity test of the induced output voltage was performed by reversing the connection.
To investigate the output performance of PENG, a mechanical force was periodically applied. At an applied stress of 3 kgf, the obtained output voltage (peak to peak) and output current (peak to peak) values for undoped ZnO are estimated at 2.28 V and 1.16 µA, respectively, as shown in Figure 9(a) and (b). Similarly at an applied stress of 3 kgf, the Ag-doped ZnO PENG gives 6.85 V and 3.42 µA of output voltage (peak to peak) and output current (peak to peak), respectively, as shown in Figure 9(a) and (b). It is revealed from Figure 9(a) and (b) that the value of positive peaks is much larger than the corresponding negative peaks. This difference in the magnitude of the output peaks occurs due to difference in strain rate during pressing and releasing process. 60,61 From Figure 9(a) and (b), it is clear that the output voltage and current of Ag-doped ZnO nanorods is about three times higher than that of undoped ZnO nanorods. A noticeably enhanced piezoelectric output voltage after Ag doping was mainly associated with the efficient reduction of free charge carrier concentration. Figure 9(c) shows the behavior of output current and voltage as a function of RL . With increasing RL , the output voltage increases while the current decreases. Therefore, the output power of the device is related to RL . The maximum output power of 1.45 mW cm−2 was achieved at 31 MΩ of RL , as shown in Figure 9(d).

Electrical output performance with (a) an output voltage and (b) an output current of the undoped ZnO nanorods and Ag-doped ZnO nanorods PENG. (c) The behavior of output current and voltage against RL (Ag-doped ZnO PENG) and (d) The output power as a function of RL (Ag-doped ZnO PENG). PENG: piezoelectric nanogenerator; Ag: silver; ZnO: zinc oxide.
Charging characteristics of load capacitors using Ag-doped ZnO nanorods PENG
The PENG was utilized to charge a load capacitor

A schematic illustration of the PENG with bridge rectifier to charge the load capacitor. PENG: piezoelectric nanogenerator.
The results of charging different load capacitors are shown in Figure 11. For all the load capacitor values, the voltage obtains the same saturation value, which is similar to resistor–capacitor charging curve, as shown in Figure 11(a). The charging speed is higher for smaller load capacitor, and it reaches to saturation voltage quickly. On the other hand, the stored charges on different load capacitors were calculated by using this relation,

(a) Output saturation voltage of PENG at different load capacitances. (b) Stored charge at different load capacitances. (c) The behavior of output voltage and stored charges against the load capacitance and (d) the maximum stored energy at an optimum CL of 22 nf. PENG: piezoelectric nanogenerator.
The stored charge and output voltage were analyzed as function of load capacitor for 1800 charging cycles, as shown in Figure 11(c). The results indicate that there is inverse relation between output voltage and stored charge. When load capacitor is 1 nF, then voltage on load capacitor is at its saturation value of 369 mV, while the stored charges are equal to 0.369 nC. In contrast, when load resistance is 400 nF, the output voltage is equal to 43 mV, while the stored charges reach at maximum value of 17.2 nC. Figure 11(d) illustrates that maximum stored energy is 0.5819 nJ at the optimum capacitance of 22 nF at 1800 cycles. Figure 11(d) shows the maximum stored energy of 0.5819 nJ at the optimum of 22 nF at 1800 cycles for time duration of 600 s. The numerical calculations show that the reactance of 22 nF capacitance is approximately 31 MΩ at 6 Hz. With reference to Figure 11(d), maximum power was achieved at 31 MΩ which resembles the reactance of at 22 nF.
To verify the above anticipations, the stored energy was determined as a function of load capacitance

Analysis of the optimum load capacitance. (a) Relationship between load capacitance
Conclusion
In summary, the undoped and Ag-doped ZnO nanorods were grown on cotton fabric by hydrothermal method and utilized to fabricate textile-based PENG. Elemental and optical analysis revealed the fine crystal quality and piezoelectric property of Ag-doped ZnO nanorods.
Ag-doped ZnO was found to be a good candidate for PENG compared to undoped ZnO. By applying the same conditions, the Ag-doped ZnO PENG power output was three times greater than undoped ZnO PENG. The maximum output power density of 1.45 mW cm−2 was achieved from Ag-doped PENG at load resistance of 31 MΩ. The effect of load resistance was observed on the output power of PENG and found that at 31MΩ the Ag-doped PENG gives maximum power.
The fabricated PENG was utilized to store charge in load capacitors and found that capacitive reactance also affects the charging capability of PENG. It was observed that the maximum energy could be stored at optimum load capacitance of 22 nF at 1800 cycles. This research work shows the unique PENG charging characteristics, which may provide a pathway to design an integrated PENG energy harvesting system for practical applications.
Footnotes
Acknowledgment
The authors are very grateful to the chairperson, Department of Physics, University of Balochistan and National Centre for physics, Islamabad, Pakistan, for support in the characterization.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


