Abstract
Silver nanostructures (silver nanopaerticles e.g AgNPs and silver nanoplates e.g AgNPls) of various sizes and shapes were prepared using different reducing agents in ionic liquid media. The ionic liquid 1-butyl-3-methylimidazolium tetrafluoroborate, which acts both as a cosolvent and a surfactant, was used to synthesize smaller and more ordered silver nanostructures. Spherical nanoparticles (silver NPs) were prepared using sodium borohydride as a reducing agent. On the other hand, plate-shaped silver nanostructures (silver NPls) were prepared using hydrazine hydrate as a reducing agent. The as-synthesized silver nanostructures were characterized by scanning electron microscopy, powder X-ray diffraction, field-emission scanning electron microscopy, transmission electron microscopy, X-ray photoelectron spectroscopy, ultraviolet–visible spectroscopy, and Brunauer–Emmett–Teller analysis. The resulting materials were applied for the photodegradation of methyl orange under visible light irradiation. Because of their large surface area, the silver NPs showed better photocatalytic activity compared with that of Ag NPls.
Introduction
Water pollution is a considerable threat to human health. Water is one of the most vital resources for humans and the ecosystem. 1 Currently, various industries such as the textile, food, cosmetics, and pharmaceutical industry produce large amounts of wastewater, which is extremely harmful to humans and the environment. 2 Methyl orange (MO) is a good representative of anionic azo dyes, which does not decompose under ambient conditions and is usually resistant to classical biological treatment. Conventional physicochemical techniques such as adsorption, coagulation, and reverse osmosis are the most frequently used to remove MO from aqueous solutions. However, these methods can merely accumulate MO and cannot convert the dye into harmless compounds. 3 –6 Hence, the development of an efficient method for MO degradation is a scientific problem of considerable interest.
The representative approach was to synthesize nanostructures with well-defined sizes and shapes. 7 –9 Most of the previous researchers preferred to synthesize nanomaterials with the smallest possible sizes because such nanomaterials have a larger surface-to-volume ratio than do bulkier nanomaterials. 10 This in turn not only reduces the amount of catalyst used but also provides many active sites for transition-metal catalysis. Many researchers have also attempted to optimize the morphology and composition of nanomaterials, thereby achieving high catalytic activity and good selectivity. 11 Lee et al. reported platinum nanocatalysts with different shapes to control the selectivity in the hydrogenation and isomerization of many unsaturated hydrocarbons. 12 Our group reported on manganese oxide nanocrystals with uniform sizes and various morphologies as a highly active catalyst for the reaction between 2-hydroxyacetophenone and 1,2-diaminobenzene to afford the corresponding quinoxaline derivative, which is a versatile heterocyclic compound. 13
Silver (Ag) nanomaterials have unique properties such as stability, low cost, antibacterial activity, and high speculative open circuit potential and hence can be widely used as catalysts. Furthermore, Ag nanomaterials with various sizes and shapes show enhanced catalytic activity in reactions such as the oxidation of organic matter, epoxidation of alkenes, and photocatalytic reactions. 14 –17 Representative photocatalytic reactions are those involving the degradation of methylene blue (MB), rhodamine B, and MO, which are well-known water pollutants. Generally, Ag nanomaterials influence dye molecules because of the plasmon resonance property of Ag. 18 Rodriguez-Cabo et al. reported the photocatalytic degradation of MO, MB, and rhodamine B using a silver chloride nanocatalyst synthesized from the corresponding bulk material in an ionic liquid. 19 Cui et al. reported the efficient photocatalytic degradation of rhodamine B by graphitic carbon nitride sputtered with Ag nanoparticles. 20 Very recently, our group reported the reduction of nitroarenes by PdO/ZnO@mSiO2 hybrid nanocatalysts. 21 Xu et al. emphasized on the photocatalytic degradation of 4-chlorophenol by Ag/AgBr/TiO2/GO. 22 Cui et al. reported on the facile preparation of silver halide nanoparticles on visible light photocatalysts. 23
Interestingly, as the size of Ag NPs changes during the reaction, their unique properties dissipate and change as well. 24 Accordingly, mutual contact between the Ag NPs and their aggregation renders them unstable. 25 For this reason, many surfactants such as cethyltrimethylammonium bromide, poly(vinylpyrrolidone), polyvinyl alcohol, D-sorbitol, perfluoromethyl vinyl ether, gelatin, polyacrylamide, and ionic liquids are used. 26 –29
Traditionally, an ionic liquid is an environmentally friendly solvent owing to its excellent characteristics such as low volatility, nontoxicity, noncombustibility, high conductivity, high thermal stability, and good solubility for organic substances. Furthermore, ionic liquids are well known to be a preventer, which fairly hinders aggregation and agglomeration of nanomaterials. 30 Moreover, an ionic liquid comprises large-sized positive ions with high polarizability, which permits the stabilization of metal nanoparticles via strong coordination to low-valent transition-metal nanomaterials during the synthesis and catalytic process. 31 Here, we have reported the synthesis of Ag nanostructures with different sizes and shapes (Ag NPs and Ag NPls) prepared using various reducing agents in ionic liquid media as well as their photocatalytic activity in the degradation of MO.
Experiment
Reagents
The reagents were purchased from Sigma Aldrich Chemical Company Limited (USA), Tokyo Chemical Company Limited (Tokyo, Japan)) and Acros Chemical Company Limited (USA) respectively.
Synthesis of Ag NPs
Silver nitrate (9.8 mg, 0.06 mmol) in 1 mL of methanol (MeOH) was taken in a 25-mL Schlenk tube and sonicated at room temperature until complete dissolution. Sodium borohydride (NaBH4; 25 eq) dissolved in 1 mL of MeOH was added to 2 mL of 1-butyl-3-methylimidazolium tetrafluoroborate (BMIM·BF4). After dissolution, this solution was rapidly added to the first solution, and the mixture was stirred well at RT for 0.5 h. The precipitate was collected by centrifugation, washed with MeOH (3 × 10 mL), and vacuum dried. The as synthesized material was denoted as Ag NPs.
Synthesis of Ag NPls
Silver nitrate (9.8 mg, 0.06 mmol) in 2 mL of MeOH was placed in a 25-mL Schlenk tube and was sonicated at RT until complete dissolution. Hydrazine hydrate (H2N–NH2·H2O; 25 eq) dissolved in 1 mL of MeOH was added to 2 mL of BMIM·BF4. After dissolution, this solution was rapidly added to the first solution, and the mixture was stirred well at RT for 12 h. The precipitate was collected by centrifugation, washed with MeOH (3 × 10 mL), and vacuum dried. The as-synthesized material was denoted as Ag NPls.
Photocatalytic activity
The photocatalytic activities of the as-prepared samples were evaluated using them in the photodegradation of the MO under visible light irradiation. In the photoreaction experiment, the in situ catalysts were dispersed into 50 mL of MO aqueous solution (2.5 mg/L). Prior to irradiation, the dispersion was kept in the dark under magnetic stirring for 1 h to establish an adsorption–desorption equilibrium state. A xenon (Xe) lamp (200 W) was used as the light source. To monitor the photodegradation, 3 mL of dispersion was taken out from the reaction system at time interval of 30 min and then monitored by ultraviolet–visible (UV-Vis) spectroscopy through the absorbance change.
Materials characterization
The prepared samples were characterized by X-ray powder diffraction (XRD, Rigaku D/MAX-RD, 12 kV at KAIST, South Korea), X-ray photoelectron spectroscopy (XPS, Rigaku D/MAX-RB, 12 kV at KAIST, South Korea), scanning electron microscopy (SEM, FEI Quanta, 15 kV at KAIST), field-emission SEM (FESEM, Magellan 400, 15 kV at KAIST), and transmission electron microscopy (TEM, JEOL JEM-2100F microscope, 200 kV, KAIST). The surface area was calculated using the Brunauer–Emmett–Teller (BET) equation. The UV-Vis spectra were recorded on a spectrometer (Shimadzu UV-1800).
Results and discussion
Figure 1 shows the SEM images of the Ag nanostructures prepared using silver nitrate with different reducing agents. The resulting Ag NPs reduced by NaBH4 shows spherical morphology. Figure 1(a) shows an image of the spherical Ag NPs. The ionic liquid, which acts both as a cosolvent and a surfactant, aided the synthesis of smaller and more ordered Ag NPs. In order to determine the role of the ionic liquid, Ag NPs were synthesized without the ionic liquid in the presence of NaBH4 as the reducing agent, as shown in Figure 1(b). The SEM image clearly shows that the average diameter of Ag NPs prepared without the ionic liquid is approximately 98 nm, which is twice that of the Ag NPs prepared using the ionic liquid. Furthermore, in order to verify the effect of the reducing agent, we used hydrazine hydrate instead of NaBH4 at the same temperature and reaction time. When hydrazine hydrate was used, the Ag NPs formed were in the shape of plates with a broad size distribution, as shown in Figure 1(c). 32

Scanning electron microscopic images of the synthesized Ag nanostructures (a) Ag NPs (Ionic liquid (IL))+NaBH4), (b) Ag NPs (NaBH4), and (c) Ag NPls (IL+NH2–NH2.H2O). Ag: silver; NaBH4: sodium borohydride.
Generally, longer reaction times lead to the formation of larger plates when the other experimental conditions are kept unchanged. Hence, the Ag nanostructures were synthesized at reaction times of 4, 8, and 12 h, respectively; the images are shown in Figure 2. Figure 2(a) depicts the SEM images of the irregular-shaped Ag NPls with a thickness of about 45 nm and width up to 3 µm. When the reaction time was increased from 4 h to 8 h, Ag NPls with small sizes and imperfect shapes were formed (Figure 2(b)), possibly due to the agglomeration of some plates. From its rounded angles, it was hard to notify as an immaculate hexagonal plate. The imperfect hexagonal plates had a thickness of approximately 1.6 µm and area of 4.9 µm. When the reaction time increased from 8 h to 12 h, absolute hexagonal Ag NPls were formed with straight edges, as shown in Figure 2(c). To evaluate the influence of ionic liquid in the synthesis of Ag nanostructures using hydrazine hydrate, the synthesis was carried out using hydrazine hydrate for 12 h without ionic liquid. In that case, Ag NPls of irregular size and thick shape were found in Figure 2(d). These results demonstrate that the ionic liquid works as a surfactant to enhance the stability of catalysts. Also, ionic liquid has an influence on the shape evolution of Ag NPls.

Scanning electron microscopic images of Ag NPls with different reaction times. (a) 4 h (b) 8 h, (c) 12 h, and (d) 12 h, IL free. Ag: silver.
When using NaBH4 and an ionic liquid at a reaction time of 0.5 h, spherical nanomaterials were obtained. Figure 3(a) and (b) shows the TEM images of the Ag NPs formed using NaBH4. The FESEM images in Figure 3(c) and (d) show that the Ag NPs are perfectly spherical, with an average size of 43 nm. Figure 3(e) shows the TEM image of Ag NPls.

(a) and (b) TEM images, (c) FESEM image, (d) size distribution of Ag NPs, and (e) TEM image of Ag NPls. TEM: transmission electron microscopy; FESEM: field-emission scanning electron microscopy; Ag: silver.
Ag NPs were ascertained by distinctive peak observed at 413 nm. The specific Ag surface plasmon resonance (SPR) band was observed at 400–450 nm λ which has been shown in Figure 4. XRD analysis was performed to investigate the crystallized phases and detect any traces of impurities in the prepared materials. The results are presented in Figure 5(a). The Ag phase synthesized using hydrazine hydrate and NaBH4 was characterized by the peaks located 2θ = 37.97°, 44.29°, 64.66°, 77.46°, and 81.44°, which corresponded to the crystal reflections of the (111), (200), (220), (311), and (222) planes of Ag, respectively. These planes could be indexed to the crystal faces of the face-centered cubic Ag(0) nanoparticles (Crystal Structure Database-ICSD/Code 64,994), and no impurities were detected in the synthesis Ag nanostructures.

UV-Vis absorption spectra of Ag NPs. UV-Vis: ultraviolet–visible; Ag: silver.

(a) XRD pattern of Ag nanostructures, (b) XPS data of Ag 3d (Ag NPls), and (c) XPS data of Ag 3d (Ag NPs). XRD: X-ray powder diffraction; XPS: X-ray photoelectron spectroscopy; Ag: silver.
XPS was performed to qualitatively analyze the elemental composition and valence of the synthesized materials. Figure 5(b) and (c) shows the X-ray photoelectron spectra of the Ag nanostructures. The binding energies of the Ag nanostructures synthesized using hydrazine hydrate appeared at 373.71 and 367.74 eV, which corresponded to Ag0 3d3/2 and Ag0 3d5/2, respectively. Similarly, the binding energies using NaBH4 appeared at 373.85 and 367.85 eV, which corresponded to Ag0 3d3/2 and Ag0 3d5/2, respectively. The XPS results confirmed that the synthesized nanomaterials were in the Ag0 oxidation state. Because of the SPR, the Ag nanostructures can be applied as a photocatalyst for the degradation of dyes.
To confirm the photocatalytic activity of the synthesized nanostructures, the degradation of MO was performed under natural sunlight as well as under visible light (Xe lamp) irradiation, as shown in Figure 6(a) and (b). The SPR effect leads to strong absorption in the visible light region. 33 The absorption peak at 464 nm progressively decreased with increasing irradiation time (Figure 6(a)). On the other hand, very slow degradation of MO was observed while using Ag NPls (Figure 6(b)). Figure 6(c) shows the images of the MO solution at 0 min and after the whole degradation in the presence of the Ag NPs prepared using NaBH4. With increasing the irradiation time, the orange color faded, indicating the degradation of MO. In contrast, the color was same as before and a slight decrease in the absorption peak was observed when using the Ag NPls as catalysts even after irradiation under sunlight and visible light for 2 h. Thus, it was concluded that the synthesized Ag NPs are more efficient and active as photocatalysts compared to the Ag NPls. It was also necessary to perform the degradation study without any catalyst. Figure 6(d) shows the time-dependent UV-Vis spectra of MO without any catalyst. Here, with increasing the irradiation time, the color of MO was same with only a slight decrease of absorption was observed. It explains that, without the catalyst, no degradation of MO will be possible.
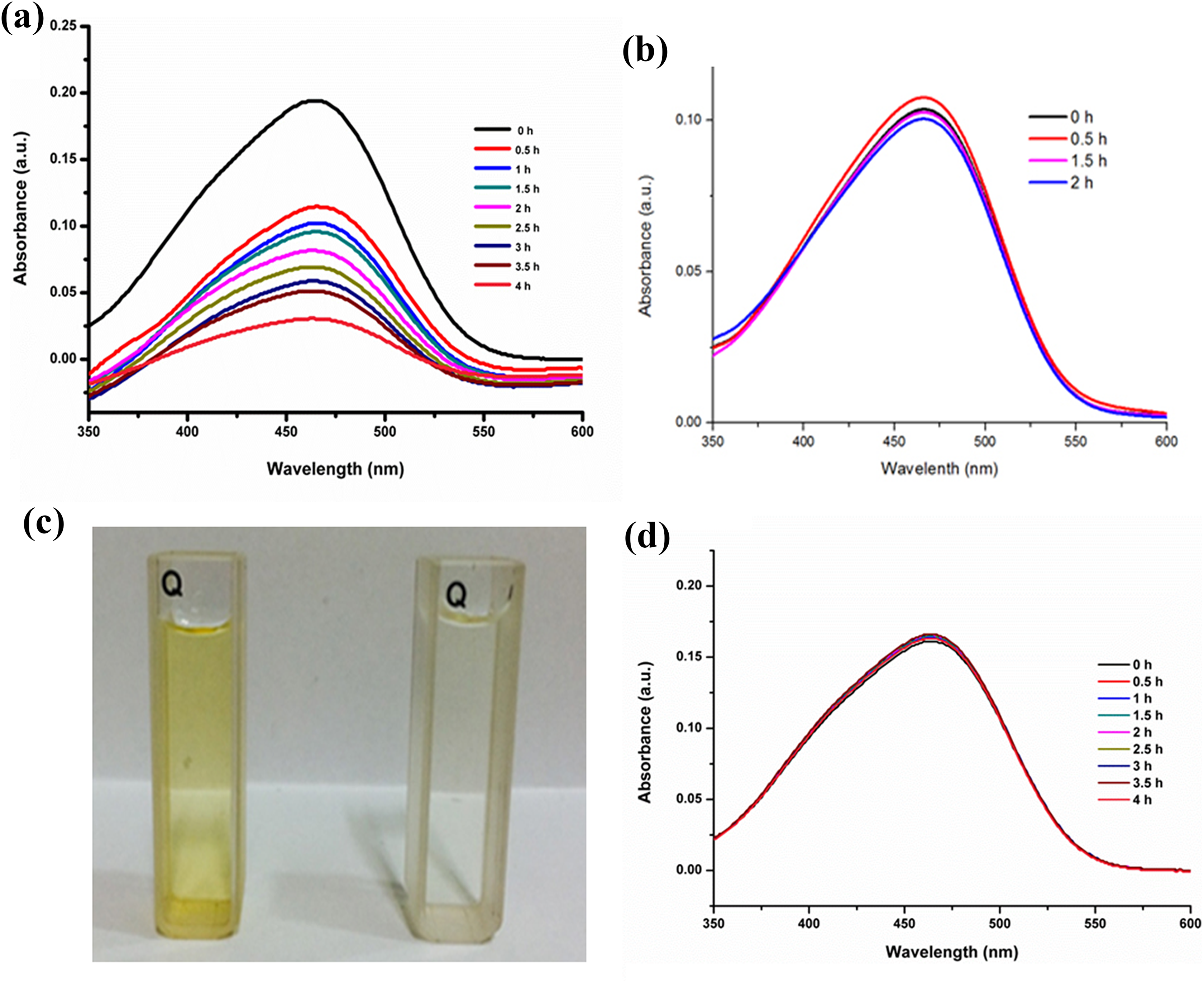
Time-dependent UV-Vis spectra of MO using Ag nanostructures. (a) Ag NPs, 0.5 h and (b) Ag NPls, 12 h. (c) Images of MO solution at 0 min (left) and after 4 h (right) in the presence of Ag NPs. (d) Time-dependent UV-Vis spectra of MO (without catalyst). UV-Vis: ultraviolet–visible; Ag: silver; MO: methyl orange.
Figure 7(a) shows the C/C 0 versus time interval curve for the degradation of MO. We did the same experiment for MO degradation to find the reproducible data. From these, we have plotted the C/C 0 versus time interval curve including error bar. Figure 7(b) shows the kinetics for MO degradation. It can be noted that the experiment in the absence of photocatalysts showed almost no MO degradation, implying that the self-photolysis of MO is negligible when irradiated with visible light. However, in the presence of Ag NPs nanostructures, a vast degradation took place with irradiation of visible light. Such result indicates that the effective degradation occurs because of the presence of Ag NPs.

(a) The curve of C/C 0 versus time interval including error bar for the degradation MO in the presence of Ag NPs and (b) kinetics of MO degradation in the presence and absence (photolysis test) of catalyst. MO: methyl orange; Ag: silver.
There are several factors which impacts on the photocatalytic performance of Ag NPs. Firstly, surface morphology, such as particle size and agglomerate size, is an important factor to be considered in photocatalytic degradation process because there is a direct relationship between organic compound and surface coverage by photocatalyst. The spherical Ag NPs were expected to demonstrate promising catalytic capability, especially when the spherical morphology can facilitate the optimum charge migration without any diffusion limitations of reactant molecules. 34 Secondly, effective catalytic activity of the Ag nanostructures for the photodegradation of MO under solar light irradiation is dependent on a large BET surface area. As shown in Table 1, the Ag NPs synthesized using NaBH4 had a surface area of 7.54 m2/g, which was three times larger than that of the Ag NPls synthesized using hydrazine hydrate as the reducing agent. As a result, the Ag NPs show better photocatalytic activity compared with that of the Ag NPls. The large surface area provides many binding sites where the MO molecules can interact with the Ag NPs during the catalytic reaction.
Specific surface area of the synthesized Ag nanostructures.

Ag: silver; BET: Brunauer–Emmett–Teller.
aDetermined by BET analysis.
The possible photodegradation mechanism of the as-prepared Ag nanostructures is as follows: When Ag NPs absorb visible light, the surface electrons from the outermost sp band are excited to a higher energy state due to SPR effect. These electrons are readily accepted by the oxygen (O2) molecules to form oxygen radicals (O2 −) which attack and degrade the MO dye molecules. Also, the holes generated in the 5sp orbital are filled by the acceptance of electrons from the adsorbed photosensitized dye molecule, thereby degrading the MO dye molecule. Absorption of UV irradiation by the Ag NPs causes the excitation of electrons from the 4d orbital to 5sp orbital. This interband transition leads to excitation of many photogenerated electrons. These excited electrons interact with the oxygen molecules to form oxygen radicals (O2 −) and the hydroxyl ion to form hydroxyl radical (OH−). Thus, formed radicals attack the dye molecule adsorbed onto the surface of the Ag NPs and bring about the degradation of the dye. In addition to the degradation of the dyes by the radicals, the holes generated in the d orbital of the Ag NPs accept electrons from the adsorbed dye molecule leading to further degradation of the MO dye. Thus, the Ag NPs are known for absorption of whole of the light spectrum due to SPR effect and the interband transition of 4d electrons to 5sp band. This mechanism of photocatalysis by UV and visible light irradiation by Ag NPs in the degradation of MO dye is presented in Figure 8. We have compared the photocatalytic activity of our materials with some reported catalysts and the comparison data have been shown in Table 2.

Schematic diagram illustrating the proposed degradation mechanism of MO over plasmonic Ag nanostructures. MO: methyl orange; Ag: silver.
Comparison data showing the photocatalytic activity of Ag NPs with other catalysts.

MO: methyl orange; Xe: xenon; Cu: copper; Cu2O: cuprous oxide; AgCl: silver chloride; Ag: silver; Ag3PO4: silver phosphate.
Apart from the photodegradation study, the recycling ability of a photocatalyst is also an important factor. Figure 9 shows the recyclability image of the synthesized Ag NPs during MO degradation. The reusability of the synthesized Ag NPs for the degradation of MO was investigated until four consecutive cycles. After completion of one catalytic cycle, a fresh solution of MO dye was added to the reaction mixture. The degradation percentage decreases from first cycle to fourth cycle. This may be because of the loss of catalyst while taking solution from the reaction mixture for UV. The degradation percentage was calculated using the following equation


Reusability study for the degradation of MO by Ag NPs. MO: methyl orange; Ag: silver.
where C 0 denotes the initial concentration and Ct represents the concentration of MO at time t.
Conclusion
We successfully synthesized Ag nanostructures (Ag NPs and Ag NPls) with different sizes and shapes in ionic liquid media. The shapes of these materials were controlled using different reducing agents (NaBH4 and H2N–NH2.H2O). Here, the ionic liquid, which acts as a cosolvent as well as a surfactant, resulted in the formation of smaller and more ordered Ag nanostructures. The resultant Ag nanostructures exhibited good photocatalytic activity in the oxidation of MO during water purification under natural sunlight and visible light (Xe lamp) irradiation. In addition, good recyclability was obtained until four consecutive cycles.
Footnotes
Acknowledgments
Sungkyun Park would like to thank the National Research Foundation of Korea and the Kora Basic Science Institute for funding the work. Hyesu Park and Shamim Ahmed Hira have an equal contribution but in the proof copy.
Declaration of conflicting interests
The author’s declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2017R1A4A1015533 and NRF-2017R1D1A1B03036303). The work of Sungkyun Park was supported by NRF (2018R1D1A1B07045663) and Kora Basic Science Institute (KBSI grant C38529).
