Abstract
Graphene is expected to be a potential device material for sensor applications due to its high charge mobility and high sensitivity to adsorbates. This article reviews the first-principles density-functional study that clarifies gas adsorption effects on graphene layers doped with boron and nitrogen atoms. We show adsorption effects of not only common gas molecules but also environmentally polluting or toxic gas molecules on stabilities and structural properties of graphene layers and carbon nanotubes. We also show physical properties induced by the adsorption of the gas molecules and discuss the possibility to detect these gas molecules.
Keywords
Introduction
Graphene, a two-dimensional carbon (C) atomic layer with a hexagonal network, exhibits various unique and excellent properties. 1 –7 Among them, one of the most remarkable properties is that graphene shows extremely high charge mobility. 3 Furthermore, graphene as well as carbon nanotubes (CNTs) is also sensitive to adsorbates. 8 –11 For example, graphene and CNTs can detect nitrogen dioxide (NO2) and ammonia (NH3) gases with fast response time and high sensitivity at room temperatures. 8,9 Thus, graphene is a good candidate for device materials to be used in high-performance sensor applications due to the high carrier mobility as well as the high sensitivity to adsorbates.
One of the most effective methods to tune electronic properties of graphene and CNTs is to dope with heteroatoms. Moreover, substitutional doping with heteroatoms can often enhance the reactivity of nanomaterials. The boron (B) and nitrogen (N) atoms are good dopants for C-based nanomaterials. There are so far a large number of reports on B- and N-doped graphenes and CNTs, since B, C, and N elements possess similar structural properties. 12 –25 The B-doped graphene and CNTs have been successfully synthesized through chemical vapor deposition and electrothermal reactions. 24 –26 The p-type field-effect transistors are realized using the B-doped graphene. 26 It is reported theoretically that CO molecules are adsorbed on B-doped (8,0) nanotubes, while those are not adsorbed on the undoped ones. 27
On the other hand, experimental observations by X-ray photoelectron spectroscopy revealed two major N-defect bonding configurations in N-doped graphene as well as N-doped CNTs; one is the substitutional N defect where the C atom is simply replaced by the N atom, and the other is the pyridine-type N defects where the N atoms are located around atomic vacancies. 12,22,28,29 Theoretical calculations suggested that the structural variety of N defects in graphene and CNTs provides various electronic properties such as p-type and n-type conduction properties. 30 –32 The N doping into graphene as well as CNTs improves the adsorption energies of hydrogen atoms. 33,34 It is also reported that the N-doped graphene with transition metals might provide hydrogen storages. 35 Furthermore, the N-dopant sites in the N-doped graphene behave as chemical reaction centers. 36 Thus, the graphene doped with B and N atoms not only offers rich electronic properties but also is expected to be useful device materials in sensor applications.
In this article, we review the first-principles density-functional study of the adsorptions of environmentally polluting and common gas molecules on B-doped and N-doped graphene layers. The section on “Structural properties and stabilities” reports the adsorption effects of various gas molecules on the stabilities and the structural properties of B- and N-doped graphene layers and CNTs. The next section on “Physical properties” discusses several physical properties for detecting gas molecules. Finally, we summarize this article together with outlook in the “Concluding remarks” section.
Structural properties and stabilities
In this section, the adsorption effects on the stabilities and the structural properties of the environmentally polluting and common gas molecules (CO, CO2, NO, NO2, H2, H2O, and NH3) to the B- and N-doped graphene layers and CNTs are reported.
Energetics
Table 1 summarizes the adsorption energies (Ea) and the distances (d) between molecules and dopant atoms for the adsorption of various molecules on the B(N)-doped graphenes and CNTs. For adsorptions of CO and CO2 molecules, the adsorption energies of B(N)-doped graphene monolayers as well as bilayers take relatively small values and the distances between the molecules and the dopant atoms are considerably long (d > 2.5 Å). On the other hand, in the case of the B-doped (8,0) CNT, the adsorption energy for the CO molecule (|Ea| = approximately 0.8 eV) is much larger than the adsorption energies of the B(N)-doped graphene monolayers and bilayers, and the distance between the CO molecule and the B atom in the (8,0) CNT is relatively short (d = approximately 1.5 Å). 27 Thus, the CO and CO2 molecules are not chemically but rather physically adsorbed on the doped graphene monolayers and bilayers, while the B-doped (8,0) CNT can bind chemically with the CO molecule. It should be noticed that the adsorption energies for the monolayer graphenes are overall smaller than those of the bilayer graphenes. This is because the adsorption energies for the bilayer graphene and the CNTs with CO and H2O molecules are obtained within the local-density approximation, 37 whereas those for the monolayer graphenes and the CNTs with NH3 and NO2 molecules are within the generalized gradient approximation, except for the pyridine-type defects. 38
Adsorption energy Ea (eV) and distance d (Å) between molecule and B(N) atom for gas molecules adsorbed on B(N)-doped MLG and BLG and CNTs.
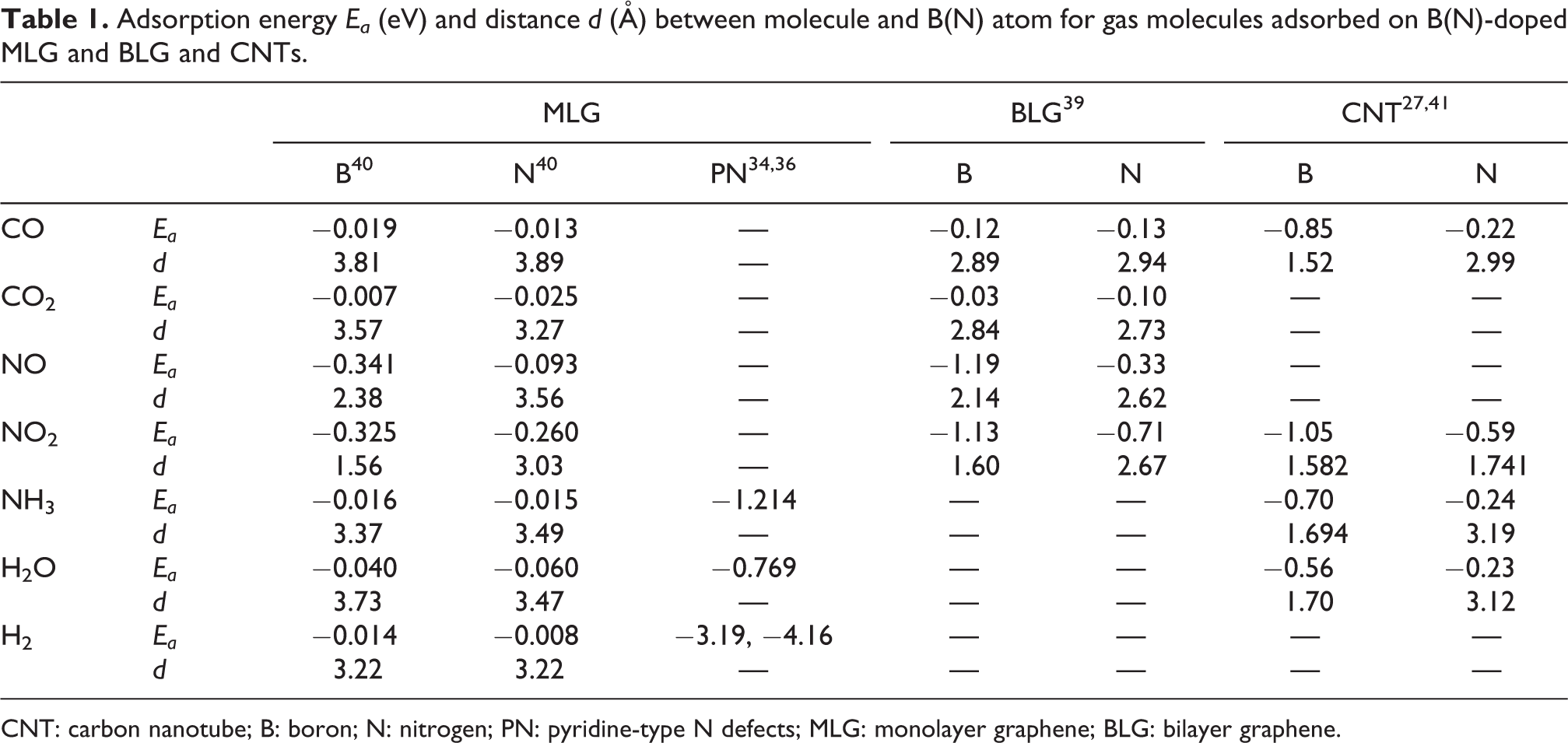
CNT: carbon nanotube; B: boron; N: nitrogen; PN: pyridine-type N defects; MLG: monolayer graphene; BLG: bilayer graphene.
In the case of NO and NO2 molecule adsorptions, the adsorption energies for the B-doped graphene monolayers and bilayers are relatively larger compared with those for the CO and CO2 molecules on the B(N)-doped graphene monolayers and bilayers. The distances between the NO (NO2) molecule and the B-doped graphene layers are relatively short. 39,40 Therefore, the NO and NO2 molecules can bind chemically on the B-doped graphene monolayers and bilayers. For the N-doped cases, these two molecules are physically adsorbed with the relatively small adsorption energies as well as the very long distances between the molecule and the dopant atom (d > 2.6 Å). On the other hand, the NO2 molecule is chemically adsorbed on the N-doped (10,0) CNT. 41,42 It is interesting that the adsorption energies are enhanced with the curvature effects of nanotubes.
The H2, NH3, and H2O molecules are physically adsorbed on the B-doped as well as the N-doped monolayer graphenes with small adsorption energies and long distances between the molecules and the dopant atoms. 40 In the case of the B-doped CNTs, NH3 and H2O molecules can be adsorbed chemically, while, for the N-doped CNTs, these molecules are physically adsorbed with the small adsorption energies and the long distances. 33,41 The H2, NH3, and H2O molecules can chemically bind to the pyridine-type N defects in the monolayer graphenes with very large adsorption energies. 34,36
Structure
The chemical adsorption of the gas molecules would give rise to the structural deformation of graphene layers. When the NO2 molecule is adsorbed on the B-doped graphene bilayer, the B atom in the graphene moves outward from the planar sheet (Figure 1(b)). Thereby, the binding distance between the B atom and the O atom in the NO2 molecule becomes very short. The structural deformation of the B atom is also reported in the B-doped monolayer graphene. 40 On the other hand, in the case of the NO molecule adsorption, the B atom still resides in the planar sheet (Figure 1(a)). 39 The short binding distance for the NO2 adsorption gives rise to the strong chemical bonds between the NO2 molecule and the B-doped graphene layer compared with the NO molecule adsorption.

Optimized atomic geometries of the B-doped bilayer graphene with (a) NO molecule, (b) NO2 molecule, (c) the trimerized, and (d) the tetramerized pyridine-type defected graphenes with H2 molecules, and the pyridine-type defected graphene with (e) NH3 and (f) H2O molecules. 36 Reproduced with permission from, 39 copyright 2016 the Elsevier, and from, 34 copyright 2014 the American Institute of Physics.
For the H2 adsorptions on the pyridine-type defects, the H2 molecule is dissociatively adsorbed on the trimerized as well as the tetramerized pyridine-type defects in the graphene (Figure 1(c) and (d)). In both the cases, the chemical dissociation of the H2 molecule takes place without a reaction barrier, that is, the H2 molecule near the pyridine-type defect can dissociate into two H atoms and eventually bind chemically with two different N atoms in the pyridine-type defect. 34 In the case of adsorptions of the NH3 molecule on the pyridine-type defects, the NH3 molecule dissociates into the H atom and the NH2 molecule. For the H2O molecule adsorption, the dissociative adsorption takes place into the H atom and the OH molecule as well. On the other hand, for the adsorption of these three molecules on the substitutional N defects, the dissociative adsorption does not occur. 40 Unlike the substitutional N defect, the pyridine-type defect exhibits the high reactivity for the H2, NH3, and H2O molecules. Therefore, graphenes with the pyridine-type defects would be useful as not only sensors but also catalysts.
Physical properties
This section reports several physical properties arising from the chemical adsorption of the gas molecules.
Charge transfer
When the molecules are adsorbed chemically to the doped graphene layers and CNTs, the charge transfers are induced between the adsorbed molecules and the graphene layers (CNTs) due to the orbital hybridizations. Table 2 lists the charge transfers between adsorbed molecules and doped graphene layers (CNTs). By the NO molecule adsorption, the electrons move from the NO molecule to the B-doped bilayer graphene with approximately 0.03 electrons. 39 On the other hand, in the case of the NO2 molecule adsorptions, it is interesting that the electrons also transfer from the B-doped graphene monolayer as well as bilayer to the NO2 molecule. For the B-doped and N-doped CNTs, the electrons also move from doped CNTs to the NO2 molecule. 41 For the adsorption of the NH3 molecule, electron transfers occur from the NH3 molecule to the B-doped CNT. However, in the case of the N-doped CNT, the electron transfer does not occur between the NH3 molecule and the N-doped CNT, because the NH3 molecule is not chemically but physically adsorbed to the N-doped CNT with the long binding distance (see Table 1). Thus, the NO and NH3 molecules behave as electron donors, whereas the NO2 molecule acts as electron acceptors.
Charge transfer between molecules and B-doped MLG, BLG graphenes, and B(N)-doped CNTs.a

CNT: carbon nanotube; B: boron; N: nitrogen; MLG: monolayer graphene; BLG: bilayer graphene.
aThe positive and negative values mean electron transfers from molecules to doped graphene layers (CNTs) and from doped graphene layers (CNTs) to molecules, respectively.
Work function
The adsorption of the molecules would change the work functions of graphene layers. The work functions of the pristine graphene monolayer and bilayer are approximately 4.5 eV, which agree with experimentally observed results. 43 –45 When the B atom is doped into bilayer graphene, the work function increases to approximately 5 eV, since the number of the valence electron of the B atom is less by one compared to the C atom. On the other hand, the work function of the N-doped bilayer graphene decreases below 4 eV, since the N atom has one extra electron than the C atom. When the NO2 molecule is adsorbed to the B-doped bilayer graphene, its work function further increases up to beyond 5 eV (Figure 2(a)). 39 In the case of the adsorption of the NO2 molecule, the work function diminishes below that of the pristine graphene. The variation in the work function induced by the adsorption of different types of adsorbates is mainly caused by the charge transfer. In the case of the NO2 molecule, electrons move from the B-doped graphene layer into the NO2 molecule. On the other hand, in the case of the NO molecule, there exists the electron transfer from the NO molecule into the B-doped layer. The charge transfer between the molecule and the graphene layers leads to the shift in the Fermi energy and should modify the electronic structures and the transport properties of the graphene layers. 41,46 –51
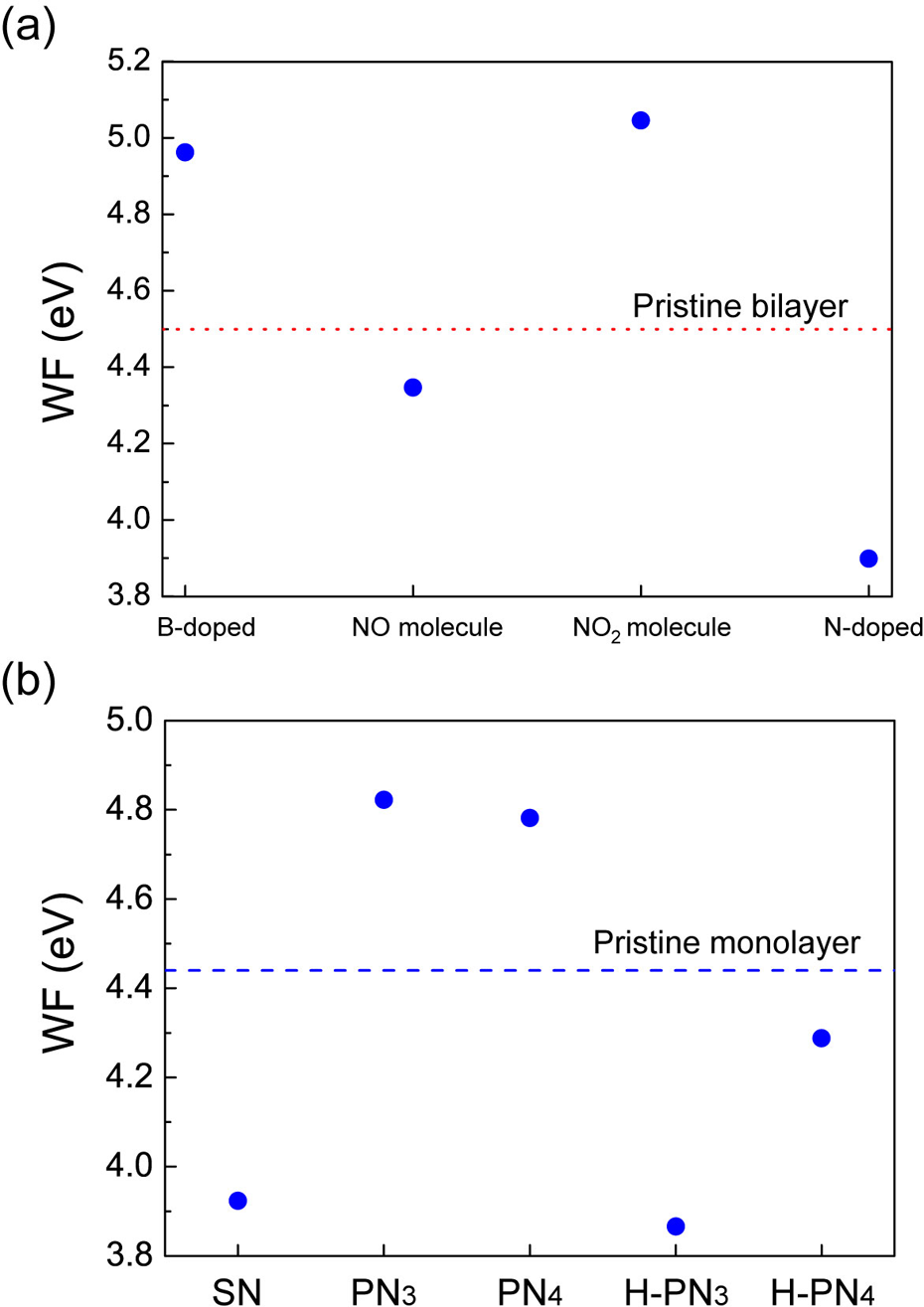
Work functions (WFs) of (a) doped bilayer graphenes with and without NO and NO2 molecules and (b) various N defects in monolayer graphene with and without the H2 molecules. The horizontal lines denote WFs of (a) the pristine bilayer graphene and (b) the pristine monolayer graphene. Reproduced with permission from, 39 copyright 2016 the Elsevier and from, 34 copyright 2014 the American Institute of Physics.
The monolayer graphene with pyridine-type N defects exhibits unusual behaviors in work functions. 34 When the N atom is substitutionally doped to the graphene (SN), the work function decreases as discussed above. On the other hand, when the trimerized (PN3) as well as the tetramerized pyridine-type (PN4) defect is formed in the graphene, 52 it is interesting that the work functions increase, since the pyridine-type defects in the graphene could induce the acceptor states (see Figure 2(b)). When the H2 molecules are adsorbed to the trimerized as well as the tetramerized pyridine-type defect in the graphene, their work functions decrease dramatically below that of the pristine graphene (H-PN3 and H-PN4). The work function varies depending on the p-type and n-type conduction properties. 34,53,54 In addition, the work function depends on the dopant concentration.
Scanning tunneling microscopy
Scanning tunneling microscopy (STM) is a powerful tool to observe the surface electronic structures of semiconductors and metals at atomic levels. 55 –61 Hence, not only the types of adsorbates but also the atomic configurations of adsorbed molecules could be identified using the STM methods. When the B atom is doped into the graphene layers, a triangle-shaped hillock is observed around the B atom in the STM image of the B-doped bilayer graphene (Figure 3(a)), 53,54 and it is also observed experimentally. 62 When the NO molecule is adsorbed, two ring-like corrugations appear above the NO molecule (Figure 3(b)). 39 For the case of the NO2 molecule adsorption, the STM image has large and small spherical-shaped protrusions above the NO2 molecule (Figure 3(c)). Thus, the B atom and the NO molecule as well as the NO2 molecule are observable using the STM methods.

STM images of B-doped bilayer graphenes (a) without adsorbate, (b) with NO molecule, and (c) with NO2 molecule. The STM images are generated at the bias voltage of +0.5 eV. Reprinted with permission from, 39 copyright 2016 the Elsevier. STM: scanning tunneling microscopy; B: boron.
Concluding remarks
This review reported the first-principles density-functional study that clarifies the gas adsorption effects on the atomic structures, the stabilities, and the physical properties of the graphenes doped with B and N atoms. The B-doped graphene can bind chemically with the NO and NO2 molecules, while the CO and CO2 molecules are not chemically adsorbed. The H2, NH3, and H2O molecules are not chemically bound to the substitutionally N-doped and B-doped graphenes as well, whereas those molecules can bind chemically with the pyridine-type N defective graphenes. The formation of chemical bonds between the adsorbed molecules and the graphenes produces the charge transfers, and the electronic structures of the graphenes are modified significantly.
The work function varies depending on the types of the adsorbates, and therefore, the adsorbed molecules on the graphenes could be detected by gauging the work function. The STM method could identify the types of adsorbates as well as the atomic configurations of adsorbed molecules. From the viewpoint of the practical sensor applications, it is one of the effective ways to examine the variation in the electrical conductance of the graphenes induced by the adsorption of the gas molecules and it is an important issue of future work.
The theoretical analysis based on the first-principles electronic-structure calculations suggests that the B-doped graphenes are candidate materials for detecting the NO and NO2 molecules, and the pyridine-type N defects in the graphenes are also important for detecting various common gas molecules. Furthermore, the pyridine-type N defects would be useful as catalysts and chemical reaction centers.
Footnotes
Author note
Computations were partly done at the Institute for Solid State Physics, the University of Tokyo, and at the Cybermedia Center of Osaka University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by MEXT Elements Strategy Initiative to Form Core Research Center through the Tokodai Institute for Element Strategy, JSPS KAKENHI grant number JP17K05053 and grant number JP26390062.
