Abstract
In the current study, a hybrid mesoporous material infused with metallic oxide nanoparticles, MCM-48 with TiO2 nanoparticles, has been developed for potential application in water treatment. MCM-48 belongs to a family of mesoporous siliceous materials—M41S, in which MCM-48 has a unique three-dimensional cubic symmetry structure. Using this hybrid approach, it will allow the polluted water to pass through the strong yet high permeable structure of mesoporous material and to allow enough time for the pollutants to react with the metallic oxide nanoparticles infused on the porous structure so that the polluted water can be treated without introducing secondary pollutants. A modified hydrothermal method has been developed to synthesize three samples of this hybrid material with TiO2 nanoparticles of 15, 50, and 300 nm in diameter, respectively. The microstructures before and after the infusion of nanoparticles are characterized using scanning electron microscope and X-ray powder diffraction. Under the current study, the synthesized samples are tested for their capability of removing organic dye and trace metals. The results have shown that while all three hybrid materials have shown over 80% adsorption rate for organic dye, the sample synthesized using 300 nm TiO2 nanoparticle has shown the highest adsorption rate. Similarly, the highest adsorption rate for most trace metals tested here also occurs in the sample made with 300 nm TiO2 nanoparticle. Coincidently, the sample prepared with 300 nm TiO2 nanoparticle has a much larger internal surface area and smaller average pore size compared to the two other samples, which may lead to the higher adsorption rate of trace metals and organic dye tested here. This study has presented a hybrid mesoporous material that can be potentially used for pollutants removal in water treatment. Future study is needed to investigate how to reuse this hybrid mesoporous material and to obtain a further understanding of the mechanism affecting the micro structures of the hybrid material.
Introduction
Water pollution is a fundamental and critical problem for human health and the environment, and it remains one of the main threats and challenges humanity faces today. 1 –3 Pollution loading from point and non-point sources continues to have significant impacts on our receiving waters, that is, rivers, streams, and lakes, despite massive public investments in drainage infrastructure (i.e. sewer systems and treatment plants) and the implementation of several federal and state regulations. The World Water Council estimates that by 2030, 3.9 billion people will live in regions characterized as “water scarce.” In addition to overall water shortage, poor water quality is near crisis in many parts of the world. Across the United States, thousands of waters are listed as impaired waters by a wide variety of pollutants. Based on a recent list, as of mid-2009, the national list encompassed over 43,000 impaired waters with over 73,000 impairments. 4 The pollutants including sediments, nutrients, metals, and pathogens are the most common pollutants included on state lists and the top ten listed impairments account for over 75% of the total listings in the nation.
More than ever, existing fresh water resources need protection and new water resources must be developed to meet the growing demand for clean water. This will require better water treatment technology. Developing an efficient and effective water pollution restoration solution, which can remove the pollutants in water in a more effective and economically sustainable way, is critical to help business, federal facilities, local governments, and tribes meet environmental regulatory requirements. 3 –5 Together with the developed industrialization is the environmental pollution. A large amount of toxic organic and inorganic compounds from many industries and agriculture is excreted into the soil, air, water, food, and eventually they will be consumed by human. Those compounds take a long time to decompose and they can harm human health if being exposed in an extended period. Heavy metals in drinking water are a big concern because they are toxic and can cause death. These metals can get into water through corrosion of the pipes and plumbing system, erosion of natural deposits, or runoff from old paints. Some of the effects heavy metals cause to human are damages to skin, brain, lungs, circulatory system, kidneys, livers, and eventually leading to cancer or death. 2,6,7
While there are many polluted water pretreatment and remediation technologies available today, most of them very often are costly and/or time-consuming, particularly pump-and-treat methods. Recent advances suggest that many of the issues involving water quality could be resolved or greatly ameliorated using nanostructure materials resulting from the development of nanotechnology. 3,8 –17 Nanostructured materials have two key properties that make them particularly attractive for water pollution treatment: (1) on a mass basis, they have much larger surface areas than bulk particles; (2) they can also be functionalized with various chemical groups to increase their affinity toward target compounds. The pollutants removal using nanostructured materials provides an alternative approach. One key bottleneck to the applications of nanotechnology to water purification will be the availability of feasible approaches that can provide copious quantities of nanomaterial at economically viable prices 13,16 without introducing additional pollutants into the treated water.
Mesoporous materials have pore size from 2 to 50 nm. They are used as adsorbents for environmental contaminants. 18 –23 In 1992, Mobil oil’s scientists discovered a family of mesoporous siliceous materials—M41S. Typical members of the M41S family are MCM-41, MCM-48, and MCM-50. MCM-41 has a one-dimensional hexagonal structure, resembling a honeycomb network. MCM-48 has a three-dimensional (3-D) cubic symmetry structure, and the structure of MCM-50 is two-dimensional stabilized lamellar. Because of the large surface area and a highly ordered and uniformly porous structure, MCM material is a good adsorbent and a catalyst for acid-catalyzed reactions and petroleum refining process. 18 –27 MCM can be used to break down organic matters such as oxidation of cyclohexene with H2O2, photocatalytic reduction of CO2 and H2O, and peroxidative oxidation of methyl methacrylate and styrene. This current work focuses on MCM-48 because MCM-48 has a 3-D channel system compared to the one-dimensional hexagonal MCM-41 and two-dimensional lamellar MCM-50, which can lead to an effective molecular trafficking of pollutants and an enhanced pollutants removal efficiency.
TiO2 nanoparticle (NP) is used widely for its photocatalytic activity in oxidation/reduction reactions. TiO2 NP can degrade some organic compounds such as benzene, phenolic compounds, and pesticides. It can also disinfect bacteria like Escherichia coli and remove organic dyes. 28 –37 TiO2 is also a good removal material for heavy metals, especially chromium (Cr), arsenic (As), cadmium (Cd), copper (Cu), and lead (Pb). The ability to remove toxic compounds of TiO2 NP together with the ability to adsorb on the large surface area and uniform pores of MCM-48 can be combined to make a great material for water purification. Previous studies have shown that MCM-41-TiO2 can remove Cr and MCM-48 has been studied to remove As in drinking water. 14,38
In this study, MCM-48 infused with TiO2 NP is synthesized by a modified hydrothermal technique. 26,27,39 Three varied sizes of TiO2 NP (15 nm, 50 nm, and 300 nm) are used for the synthesis with MCM-48 and then tested for their adsorptions of heavy metals and organic components.
Materials and methods
Synthesis of the hybrid MCM-48 infused with TiO2 NPs
Materials
TiO2 NP sizes 15 nm, 50 nm, and 300 nm were purchased from the US Research Nanomaterials, Inc., Houston, TX, and NH4OH with 28 to 30% NH3 basis, cetyltrimethylammonium bromide (CTAB), and tetraethoxysilane (TEOS) were purchased from Sigma-Aldrich Chemistry, St. Louis, MO, USA.
Experimental procedure
Synthesis of MCM-48 infused with TiO2 NP was performed using a two-step procedure: (1) MCM-48 is synthesized using the modified hydrothermal technique, and (2) TiO2 NPs of three varied sizes were infused into MCM-48.
Step 1: MCM-48 was synthesized hydrothermally in a water bath at 40–50°C; 50 mL of deionized (DI) water was added to a 250-mL beaker immersed inside the water bath; 11.077 g CTAB was added and stirred vigorously for 30 min. After that, 9.6 mL TEOS and 3 mL NH4OH were added and sealed. The sealed bottle was then heated at 90°C for 72 h. Afterward, the bottle was removed from the oven and the product formed was washed thoroughly with water and dried overnight at room temperature (25°C). The dried sample was calcined at 600°C for 5 h to decompose and remove the organic surfactant.
Step 2: The TiO2 source was infused into the MCM-48 structure through a modified impregnation method. During the impregnation process, 3 g TiO2 NP was dispersed in water (250 ml) using an ultrasonicator. The impregnation of the calcined and dried MCM-48 (3.5 g) was carried out by adding it into the TiO2 solution. Then it was stirred for 2 h with 10 min ultrasonication at every 30-min interval. The mixture was then filtered and washed thoroughly with water and dried at room temperature overnight. The sample was transported into a calcination oven and kept at 200°C overnight. The procedure was repeated for all TiO2 NP samples. To distinguish between the three solutions, they were labeled as MT15, MT50, and MT300, which correspond to MCM-48 infused with 15 nm, 50 nm, and 300 nm TiO2 NP, respectively.
Characterization of organic dye and trace metal contaminants removal
Two groups of contaminants were tested here, and an analysis was done using an inductively coupled plasma-mass spectrometer (ICP-MS) from PerkinElmer Pure Plus—NexION 300D model and a Clarus 600/560 D gas chromatography-mass spectrometer (GC-MS). For each test, an equal amount (6 g) of the three hybrid mesoporous samples (MT15, MT50, and MT300) along with the pure MCM-48 are used here to ensure the experimental consistency. The filtration was performed using a standard vacuum filtration system in which a filter paper of 2 μm pore size was used for all samples. 1. Dye solution: It was prepared by dissolving 0.1 ml of 1% (m/v) methylene blue dye into 100 mL of DI water. Then the dye solution was divided into 10 glass vials of solution at 10 mL each and labeled as “DS#1, 2, 3,….” The 10 vials of solution were then filtered through each sample (MT15, MT50, MT300, and pure MCM-48, respectively) and collected into another 10 vials labeled as “DS_AF#1, 2, 3,…” afterward. Color of filtrates were collected and compared with the initial solution, meanwhile the concentration of the filtered solutions was analyzed using GC-MS. The dye removal efficiency,%R, was expressed as a percentage as follows
where Ci and C are the initial and final dye concentrations, respectively. 2. Trace metals: 0.5 mL of 100 mg/L instrument calibration standard 2 from PerkinElmer Pure Plus (trace metals and minerals: aluminum, antimony, As, barium, beryllium, Cd, calcium, Cr, cobalt, Cu, iron, Pb, magnesium, manganese, molybdenum, nickel, potassium, selenium, silver, sodium, strontium, thallium, tin, titanium, vanadium, and zinc, each component at 100μg/ml) was added into 497 mL of DI water. For each synthesized material, 90 mL of the spiked solution was prepared and divided into six 15 mL vials labeled as “TM#1, 2, 3,….” The 10 vials of solution were filtered through each MCM sample (MT15, MT50, MT300, and pure MCM-48, respectively) and collected into another 6 vials labeled as “TM_AF#1, 2, 3,…” afterward. The collected filtrates were then analyzed for trace metals with ICP-MS. The trace metal removal efficiency,% MX, was expressed as a percentage as follows
where Xi and X are the initial and final concentrations of trace metal X, respectively.
Results and discussion
Structural characterization results
The MCM-48 material thus formed was characterized using a Hitachi SU-70 field emission gun scanning electron microscope (SEM). The result is shown in Figure 1 and the operation conditions are listed as well.

SEM image of an ordered area of a calcined MCM-48 sample. SEM: scanning electron microscope.
It can be seen from Figure 1 that the calcinated MCM-48 sample shows a spherical structure on the surface and the average feature of the structure was estimated to be around 1 µm. This narrow pore size distribution and its regular porous structure makes MCM-48 as an idea matrix to immobilize catalytically active NPs.
The structures of the hybrid mesoporous materials, MCM-48 infused with TiO2, were characterized using the same SEM and the result is shown in Figure 2.

SEM image of an ordered area of calcined MCM-48 infused with TiO2 samples: (a) MT15, (b) MT50, and (c) MT300. SEM: scanning electron microscope.
It can be seen in Figure 2 that the infused TiO2 NP has changed the porous structure of the hybrid material and the size of pores is much smaller compared to pure MCM-48. It can also be seen here that the size of TiO2 NP used here can affect the structure and porous size of the hybrid material, in which the larger the TiO2 NP is the smaller the porous structure is formed.
To further quantify its porous structure, nitrogen adsorption experiments were carried out for all the hybrid mesoporous samples (MCM-48 infused with TiO2) at 77 K on a Micromeritics™ surface area and porosity analyzer, and the results are listed in Table 1. In Table 1, the Brunauer–Emmett–Teller equation was used to calculate the specific surface area from the adsorption data, the total pore volume was calculated from the amount of nitrogen adsorbed, and the pore size distribution was calculated by analyzing the N2 isotherm using the Barrett–Joyner–Halenda (BJH) method and by applying the BJH method. 18,21 –23,26,27,38 –42 All calculation is performed using the software provided by the analyzer.
Surface area and porosity of hybrid MCM samples (MT15, MT50, and MT300).

X-ray power diffraction experiments were carried out using a Bruker C2 Discover diffractometer with Cu radiation, XYZ stage, and a two-dimensional high-resolution detector at room temperature. The short-range angle regions were scanned from 2θ = 2°–10° with a step size of 0.02°. The X-ray powder diffraction (XRD) patterns for the hybrid mesoporous MCM-48 with TiO2 compared with pure MCM-48 are given in Figure 3. Compared to pure MCM-48, the hybrid material, MCM-48 with TiO2, peaks at around 2.7°(2θ) which confirms that after the impregnation procedure the structure of the silica matrix is well maintained. It can also be seen here that the peak intensity of the hybrid material is lower than the pure MCM-48, which is since the uptake of molecules inside the pores reduces the scattering contrast between pore wall and pore, thus leading to a decrease in peak intensity. 39,43,44

XRD patterns of a calcined MCM-48 infused with TiO2 (MT300) sample compared with pure MCM-48. XRD: X-ray powder diffraction.
Results for dye removal
The results of three hybrid samples for dye solution removal are shown below. Figure 4 shows a comparison of the dye solution before and after one filtration using the MT300 sample and it can be seen clearly that the concentration of dye has been significantly reduced after the first filtration.

Comparison of dye solution: (a) initial solution, and (b) after one filtration, respectively (MT300 is used here for demonstration).
The efficiency of three hybrid MCM-48 with TiO2 samples is compared and summarized in Figure 5. The pure MCM-48 sample is also included for comparison purpose. It can be seen here that while all three samples have shown over 80% adsorption rate for the blue dye, the MT300 sample has the highest adsorption efficiency at 1 filtration and the highest overall efficiency after 10 runs of filtration tested here. Most of the samples tested here have shown a peak or near-peak performance at six runs of filtration, which suggests a maximum adsorption rate has been achieved. Meanwhile, the pure MCM-48 sample has shown a lower adsorption rate compared to the three hybrid materials and it reached the maximum capacity at the fifth test. The results here have suggested that the hybrid material of MCM-48 with TiO2 has an improved adsorption efficiency and capacity compared to the pure MCM-48. It may be attributed to the fact that the TiO2 NP itself can remove organic dye in addition to the increased surface area. 29 –31,33 –35,37,38,45 –50
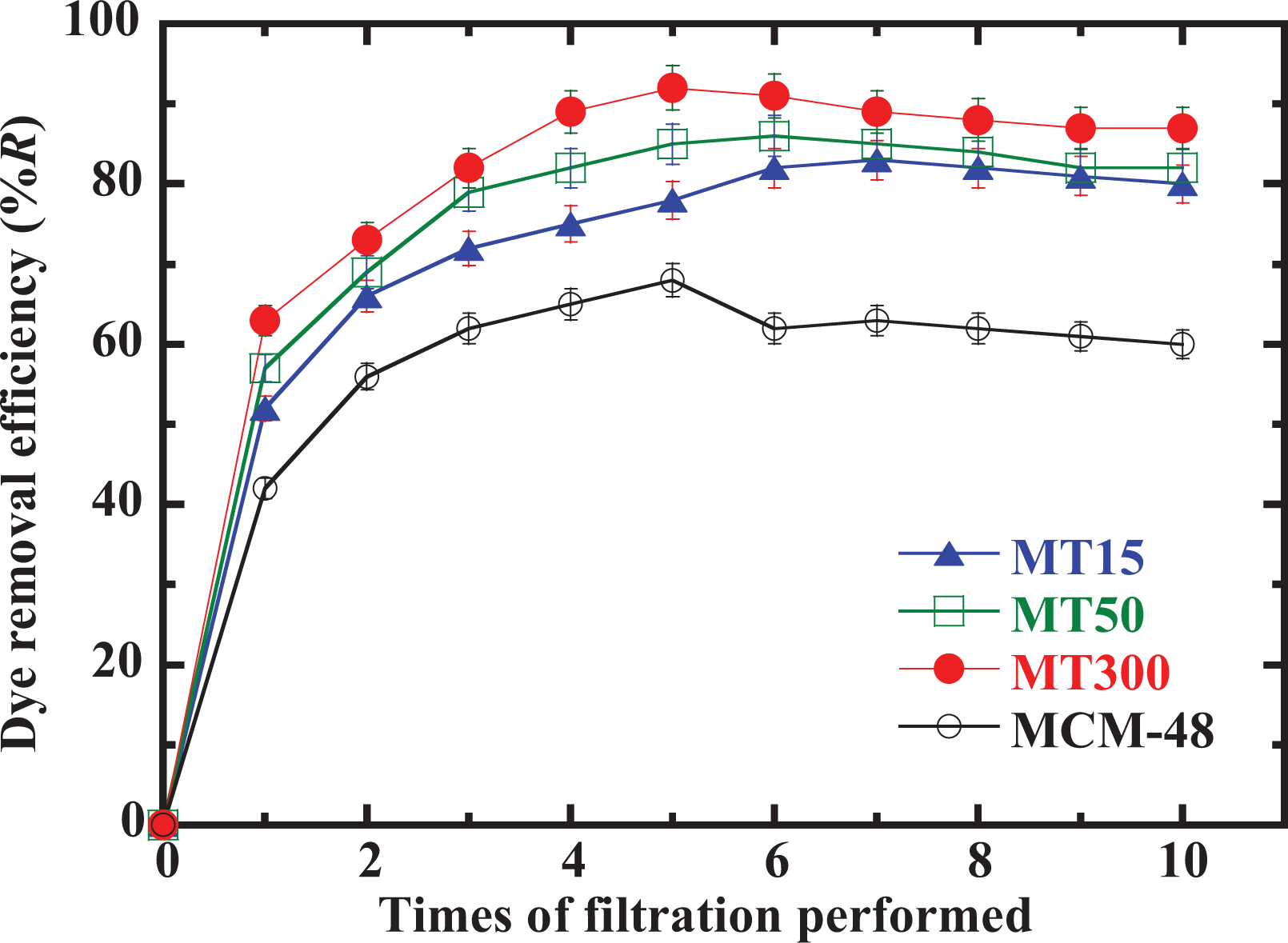
Dye removal efficiency (%R) with time of filtration tested for three hybrid samples and pure MCM-48.
Results for trace metals removal
Following the procedures listed under the “Material and methods” section, the results of the three hybrid samples for trace metals removal were obtained using ICP-MS, and the results are shown in Table 2. For each hybrid sample, six tubes of filtered solution were collected and the sum of the adsorption rate from the six tubes gives the total adsorption rate of each sample regarding that trace metal. The results have been compiled in Figure 6, in which the removal efficiency for each trace metal using the three hybrid samples has been calculated using equation (2) and included as well.
Adsorption data of hybrid MCM-48 with TiO2.

Cr: chromium; As: arsenic; Cd: cadmium; Cu: copper; Pb: lead.

Total adsorption and adsorption efficiency of heavy metals on MCM-48-TiO2 with three NP sizes 15 nm, 50 nm, and 300 nm (results of pure MCM-48 are included here for comparison purpose). NP: nanoparticle.
It can be seen here that all hybrid samples have shown a higher adsorption rate compared to the pure MCM-48, in which all three hybrid samples have shown a higher adsorption rate for As, Cu, Cd, and Pb compared to Cr, while the MT300 sample has the largest adsorption for all the trace metals tested here with an overall 80% efficiency for As, Cu, Cd, and Pb. Meanwhile, the MT15 sample has the lowest adsorption rate among the three hybrid samples. In addition, the maximum amount of heavy metals that each hybrid sample can adsorb cannot be determined at this movement as none of them has reached its maximum rate. The maximum adsorption of the material for certain metal can be determined based on whether there is negative value of amount adsorbed during each filtration, and no negative value has been observed under current test conditions.
It also seems that the size of TiO2 NP has a positive impact on the adsorption rate of trace metals tested here. It may be attributed to the much larger internal surface area for the MT300 sample compared to the other two samples as well as the pure MCM-48 as shown in Table 1. The smaller average pore size may also help to keep the spiked trace metals solution longer inside the mesoporous structures during each filtration test, which may lead to a higher adsorption rate. In addition, the TiO2 NP itself has also shown the capability to remove trace metals.
Conclusions
The current study has shown a promising hybrid mesoporous material infused with metal oxide NPs, namely “MCM-48 with TiO2 NP,” can be synthesized using modified hydrothermal methods for water treatment. This material has shown a good adsorption capability for organic dye and trace metals: While using this hybrid material, it can achieve over 80% adsorption rate for organic dye and most trace metals tested here. It has also shown that the size of TiO2 NP has a significant impact on the overall adsorption rate and the sample made with the largest NP size tested here has shown the highest adsorption rate for both organic dye and trace metals. While its mechanism is not completely clear yet, it is believed that the infused TiO2 NPs on the mesoporous structure and the resulted larger internal surface area are the two main contributing factors. It is also observed here that the size of TiO2 NP used to form the hybrid material has a positive impact on the adsorption rate of trace metals and organic dyes as tested here. Future study is needed to investigate how to reuse this hybrid mesoporous material and to obtain a further understanding of the mechanism affecting the micro structures of the hybrid material.
Footnotes
Acknowledgments
The authors want to thank the lab technicians (Yacov Assa and Sebhat Tefera, Lab Technicians at Water Resources Lab, the University of the District of Columbia) for constructive discussion and helping with the ICP-MS experiment and Clarus 600/560 D GC-MS. The authors also want to thank Dr Bao Yang’s group at the University of Maryland, College Park, for helping with the X-ray powder diffraction (XRD) and Micromeritics™ surface area and porosity analyzer experiment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the DC Water Resources Research Institute (DCWRRI) seed grant and the National Institute of Food and Agriculture under project #DC0022016.


