Abstract
High concentrations of heavy metals (up to the pollution level) can disturb the normal ecosystems of natural rivers. There are some methods for treating heavy metals in waste waters. Heavy metal treatments save the ecosystem and clean drinking water. We determined a new adsorption method for heavy metals using a special mushroom compost, which shows the adsorption capacity of this biomass. The laboratory methods were the preparation of stock solutions from heavy metals, the adsorptions of heavy metals by special mushroom compost with a new technique (shaking method), the degradation of samples and analytical control measurements by inductively coupled plasma mass spectrometry. The results show the success of the new adsorption method. The heavy metal adsorption capacity of this mushroom compost seems to be useful on waste water treatment plants because waste water contains heavy metal ions above the limit concentrations. The new absorption technology solution also includes a special economic analysis that examines material flows. In the sustainable economic analysis, as the circular economy focuses on material and energy circularity combined with natural resources (circular economic value (CEV)), the key question is how to make the systems currently operating in a linear form circular. The difference between CEV%BAU(mod) = 45.5% and CEV%Scen(mod) = 87.5% is the amount of material which could be recycled at the end of life cycle of the product, which is the same as the primary raw material, and so mushroom compost is a completely recyclable material.
Keywords
Introduction
Literature on heavy metal adsorption techniques
One of the world’s greatest environmental problems is to provide clean and healthy water for people, and treated technological water to industrial companies. The heavy metal contamination (pollution) of live water comes mostly from the industrial sector (e.g. metallurgy, mining, chemical processes, nuclear energy production).
Most of the adsorption methods are efficient and low-cost techniques for treating heavy metals from every type of waste water. 1,2 In adsorption research, the heavy metal adsorption process can be described by Langmuir and/or Freundlich isotherm models. 3 The Freundlich isotherm is useful for modelling the (ad)sorption of metals on any surface with several materials (e.g. biocarbon). 3,4 Although the efficiency of metal sorption described by Langmuir or Freundlich isotherms can be questioned, these models can be useful to describe the maximum adsorption capacity (q max) of heavy metals. 5 The materials used can have a high adsorption capacity in order to achieve the lowest contamination in the treated waste water. 6
The study by O’Sullivan et al. 7 shows the results of heavy metal measurements with inductively coupled plasma mass spectrometry (ICP-MS). Their conclusions were applied in our study, too. They determined the complete process of measurement, from injection to data analyses. The ICP-MS technique is suitable for the measurement of different heavy metal concentrations in natural water samples. Subramanian et al. 8 measured heavy metal and polycyclic aromatic hydrocarbon (PAH) concentrations in agricultural and urban water samples. They determined the relationship between heavy metal concentrations and agricultural produces. The occurrence and the fate of heavy metals (cadmium (Cd), lead (Pb), manganese (Mn), copper (Cu), zinc (Zn), nickel (Ni) and ferrum (Fe)) during the waste water treatment process were investigated on a waste water plant of Thessaloniki (Greece), which is operating in the activated sludge mode. 9 The examples were gathered from various focuses in the plant: (1) influent and gushing of the essential sedimentation point, (2) the emanating of the optional sedimentation point, (3) ooze tests from the essential sedimentation tank and (4) actuated waste water muck tests from the distribution stream. There was an exponential connection between the metal coefficient esteem (logKp) and the suspended focus. 10,11 The larger compost fraction has smaller surface points and worse adsorption properties than the smaller compost fraction. 1,12 They specified the adsorption capacity of compost for Cu, Zn, Ni and chromium (Cr) ions. 13 Other studies have reported the results of heavy metal measurements in waste water samples also. They measured the heavy metal (Cd, Cu, mercury (Hg) and Pb) concentrations in water samples from Thoothukudi and Vizhinjam cities (from India). They also researched the possibility of heavy metal bioaccumulations with Phallusia nigra. The established order of bioaccumulation factors was: Hg-Cu-Cd-Pb. 14
In a new way, many articles present a self-efficient but appropriate eliminate of complex-fixed heavy metals by coagulation process methods. 15,16 This heavy metal removal technique works by the polyelectrolyte flocculation method, followed by centrifugation and filtration methods. 15,17 The mentioned treatment cannot eliminate the heavy metal content of waste water, so it is necessary to include the technique with other treatment techniques, such as precipitation or spontaneous reduction processes. 18 The exchange of ions is a good separation process, which substitutes ions with other ions, which results in a highly efficient removal of metal ions. 19 Based on the relevant literature, sludge production compared to the coagulation treatment process is less effective in the ion exchange process. 20 The chemical precipitation method is a cheaper and more effective technique, and is used in various industries. 21
Research into clinoptilolite adsorbent shows its efficiency as a material for removing heavy metals such as Pb(II), Cd(II), Cr(III), Cu(II), Mn(II), Zn(II) and Ni(II). 22,23 To measure the adsorption efficiency of zeolite clinoptilolite, the toxicity level of this material should be determined by an A. cepa test. 24 This biological and ecotoxicological test can show the application opportunities of zeolite clinoptilolite as an adsorbent material. 24 Aqueous solutions contain several heavy metals in various different concentrations, but all of them can show the Langmuir and Freundlich isotherms. 25 All of the adsorption methods can work with a well-established pH value. The average pH value of working adsorbents is between pH 4 and pH 6. 24 –26 In our study, the adsorbent material (special mushroom compost) works at pH 5.9. Comparing the literature and the data in our experiment, the adsorbent is perfect for heavy metal treatment in terms of its pH value. There is another new method for the adsorption of heavy metal from aquatic solutions. This is the Lewis base removal method. 26 All of the heavy metal ions are soft Lewis acids and have affinity for soft Lewis bases. 26 This soft acid and base correlation leads to the formation of covalent bonds of heavy metal ions and reduces the risk of releasing heavy metal ions in treated water. 26,27 This is the scientific and theoretical basis of this Lewis removal model.
In summary, we can say that heavy metal contamination in the environment is usually treated by physical or chemical methods. There are several methods to remove heavy metals from soils and waters. For example, the pump and treat method is suitable for dealing with heavy metal contamination. Other methods for heavy metal removal from water involve ion exchange or reverse osmosis. 28 –30 Water monitoring often includes procedures for toxic heavy metal testing using in situ methods and chemical and physical laboratory practice. 28 Agricultural activity can increase Cu, Zn, Cd, Cr and Ni pollution. 28,31 Treated waste water is fit for agricultural irrigation but it is important to know the concentrations of metal and heavy metal pollutants in both the original and treated waste water. High values of heavy metals, for example, of Cu, Zn and Ni, appear in waste water. 32 Continuous monitoring of heavy metal accumulation in the environment must be conducted in order to be aware of health risks to humans, and the transport processes occurring in the environment. 31 –33 If we would like to clearly understand the environmental activity and risk associated with several heavy metals several methods can be used, but the most typical method is the BCR sequential extraction procedure (by the European Community Bureau of Reference). 34,35 BCR extraction enables us to determine the order of several heavy metals which are the focus of activity and (ecological) risk: the activity order is Cd > Zn > Cu > arsenic (As) > Pb and the risk order is Cd > Zn > Cu > As > Pb. 34,35 Environmental activity and ecological risk are closely related to each other. Cd has a high ecological risk to the environment, with a potential human toxicity in water. 35 The mobility, availability and persistence of Cu and Zn can be determined by comparing the effect of concentrations and pH changes in a two-step BCR sequential extraction process. 36
The circular economy and the circular material flow concept
Previous waste management concepts are essentially focused on environmentally friendly waste management practice; the main purpose of the circular economy is to prevent waste generation. Avoiding the formation of waste and circulating it is a new concept in the economic evaluation process. The linear processes associated with waste are thus replaced by systems that provide full or partial material circulation. Today, their design and operation is an essential environmental and social requirement. 37 Comparison of linear and circular economic aspects involves two approaches. One is to replace the 3 R strategy with the 9 R strategy, which takes better account of the processes of the business world. This approach can be seen in Figure 1. Another approach is to emphasize the importance of consumer behaviour (a concept that comes from the TerraCycle waste management company). 38 Table 1 shows the two solutions and their groupings. This shared overall approach facilitates the transition from linear structure to circular mechanisms. It is important to mention that in order to develop a circular economic concept outlined in our study, the adaptation of the linear–circular system of relations in Table 1 was necessary.

The 9 R structure of the circular economic system. Source: Based on Cramer. 39
Consumer attitudes linked to linear and circular models.

Source: Authors’ own research, based on Cramer. 39
Switching from a linear economy to circular systems is not a simple task. However, switching involves a key concept: the issue of the product life cycle (or lifetime) and the material resources used for production. In the comparative analysis presented in Table 1, the function of the product and its maintenance over a longer period of time are important for evaluation. In the field of waste management, circular economic systems come to the fore, because instead of disposing of waste, they focus on preventing its creation (declining production volumes) and reducing its volume (reversing solutions). 38,40 Current research approaches, which attempt to measure the level of circularity, are mostly metric methods. There are two mainstream lines in this field; one of them is derived from the study conducted by the international, 41 while the other was created by a single engineer called Maurits Korse, from the University of Twente.
In the following sections we will show the circular economic value (CEV) method based on the two concepts mentioned, which we have successfully applied in our investigations.
Calculations of material flow
The material flow calculation we currently use (CEV) is based on two economic methods: the Material Circularity Indicator (MCI) from the MacArthur Foundation Team and the Korse model. 37,42 The MCI uses the material flows and the possible usefulness of products. The model uses the linear flow index to present material flows. 37 The main point of this index is the identification of primary material sources and the amount of waste which can appear during production. The Korse model uses the amount of recycled material during the description of production (and the amount of primary material resources). 43 The rarity of primary materials (all around the globe) reduces the value of the Korse model. 37 This model used the possible recyclable parts from products at the end of their life cycle. In summary, the MacArthur model presents the material used during production, and the Korse model uses the possible increase in the amount of recycling at the end of production. 37,42,44
Figure 2 shows the structure of the MacArthur model’s material flow, which shows the process of material circulation. 42
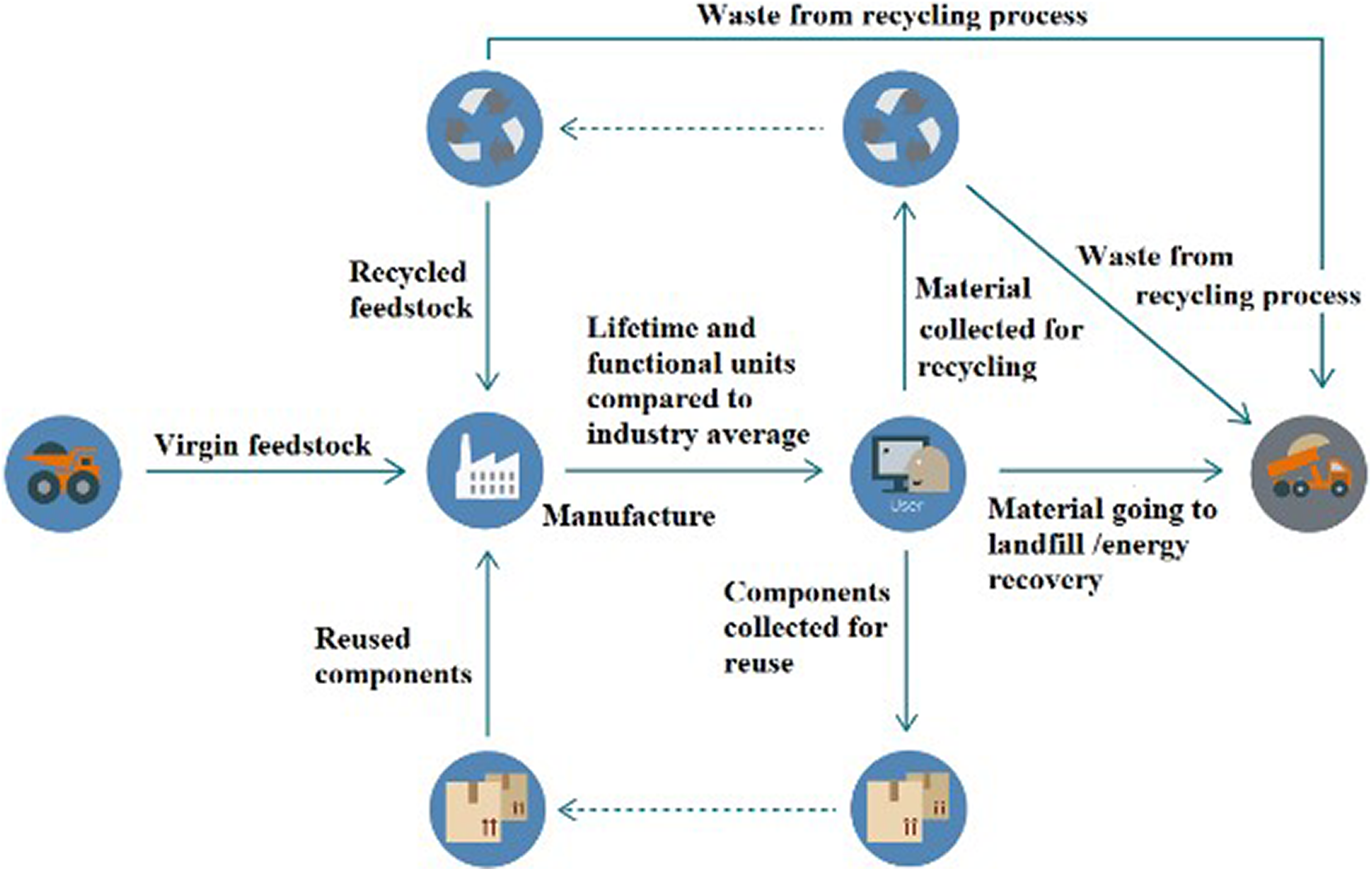
MacArthur’s model of material circulation. Source: MacArthur. 42
The CEV concept
CEV can model the circular economic system by establishing a circular production value. CEV can show the material and energy flows on both sides of the production system. 37 The modern economy focuses on sustainability (i.e. the social, economic and environmental dimensions of the basic modern economic system). 45 The linear structure cannot be used to show the social and natural impacts on the economic system because the linear structure does not focus on reducing the negative aspects of production systems. In the modern economic system, the circular economy focuses on material and energy circularity, combined with natural resources. 37,46 This modern economy can also focus on consumption, because the input and output sides are basic elements of natural ecosystems. 47 This is the main point of the circular economy, because the system must be closed. 48 The product life cycle is the key element in circular economic analyses. All products can reach the end of their life cycles. A possible approach to creating a circular economic production system is extending the life cycle. 49 The end of the life cycle presents a problem in terms of the circularity of materials and the possible methods of ensuring renewable production. 37 The main elements of circular economic models are the extension of the product life cycle and the recycling of products at the end of life cycle. 50 Short life cycle products can be recycled back into the system but this means continuous production with increasing amounts of energy (costs) and environmental pollution. 37 The circular economy uses a production system with a minimal or zero amount of waste. 51 Thanks to this production method, the product can easily be recycled at the end of the life cycle. 52 The background requirement for the circular economy is low-waste production with a structure based on recycling, reproduction and the use of waste.
The whole methodology of environmental (analytical) adsorption measurements, together with a small number of the measured results, has been published by Czikkely and Bálint and Czikkely et al. 53,54 These studies defined the mechanism (the shaking technique, the context of shaking time and adsorption capacity) and confirmed the operability of this special adsorption method. In this article, we publish all the adsorption results from our experiment and calculate the CEV. We have modified the CEV calculation for material flows and determined the circularity of the new heavy metal adsorption technique.
Materials and methods
The first step of our research methodology was a definition of mushroom compost, which seems to be a new working adsorption surface for the treatment of heavy metal contamination. Stock solutions were prepared for adsorption study and the heavy metal concentrations were measured by the analytical chemical control method (ICP-MS equipment). We calculated the CEV of our new adsorption technique in order to describe the circularity of the new system.
Special mushroom compost – the adsorption surface
Mushroom compost samples were used to make all these measurements. The mushroom compost samples derived from a small composting company (Bio Fungi Ltd, Hungary). The mushroom compost is a compost with special properties, which contains some of the most important chemical and biochemical ingredients. The contents of the mushroom compost samples were the following (the measurement results came from the compost production company because they have to measure the contents of their product before sale): calcium content: 3.0%; manganese content: 0.3%; nitrogen content: 0.8%; phosphor content: 0.6%; potassium content: 0.9%; pH value (acidity): 5.9; and dry matter content: 35.0%.
Preparing of stock solutions for laboratory measurements
The different heavy metal elements (list of single, double and triple) of the prepared solutions are shown in Table 2. All of the stock solutions of every single, double and triple element combination were prepared in each concentration (in three replicates): 250 mg/dm3; 500 mg/dm3; 750 mg/dm3 and 1000 mg/dm3. All solutions were prepared with MLQ water (18 MΩ cm−1) from a Milli-Q analytical water preparation system. Sometimes, high concentrations can be found in industrial waste water. This was the reason for the highest measured heavy metal concentrations in our experiment.
List of heavy metal–contaminated stock solutions.

Source: Authors’ own research.
Mn: manganese; Cd: cadmium; Cu: copper.
Development of the adsorption technique
The most important point during the experimental process was to decide which adsorption technique would best fit our heavy metal adsorption research. There are various methods used to treat heavy metals from waste waters by adsorption, including fore sample adsorption by several biomass and chemical adsorbents. 4 All of them use the normal adsorption system: rotation techniques in all dimensions of space. In our study, a new method was used. This is a type of shaking method. This method can also increase the surface size of adsorbent. During the development of the method, all of the main points of the process were determined in order to achieve the best adsorption efficiency. To achieve better adsorption, 10 g of the mushroom compost sample and a 30 cm3 solution of heavy metals were placed into centrifugal tubes. During the operation, the shaking time was 50 min and the rotation velocity was 480/min. The shaking method was developed at the Department of Chemistry and Biochemistry of Szent István University, Hungary. All of the machine settings were used to measure all samples. After application of the shaking method, the solutions were divided into two parts by filtration, and the liquid part was degraded by a microwave machine.
Organic matter degradation for analytical chemical measurement
All samples digested, because the organic compounds failed the correct measurement of heavy metal content. A Milestone (China) Mega 1200 Microwave digestion machine was used to digest the samples. Five cubic centimetre nitric acid and 1 cm3 hydrogen peroxide acid reagents were added to the 5 cm3 shaken sample. The digestion time was 24 min. After digesting, the samples were placed in a water bath for 30 min. In the final stage of the whole digestion programme, the digested sample was diluted to 25 cm3. 55,56
Organic matter degradation for analytical chemical measurement
Measurement with an ICP-MS is an effective analytical method to determine trace elements such as Cd, Cu, Ni, Zn and Mn. The ICP-MS analytical method is sensitive and allows the simultaneous analysis of elements and their isotopes. 57 All of the samples were measured in a 25× diluted solution. The isotope interferences are very important to determine the percentage of heavy metal isotopes in the samples. An ICP-MS machine can measure the heavy metal concentrations in the isotope ratio and the software shows the results of each isotope in order. Beck et al. determined all instruments and parameters of the ICP-MS measurements used. 58 The ICP-MS (Agilent 7500ce – Agilent Technologies, Waldbronn, Germany – equipped with an autosampler) was used at the Institute of Analytical Chemistry, University of Vienna, Austria. The autosampler allows simultaneous measurements of 150 samples. 58
CEV calculations
CEV is a comparison of the newly developed method (alternate scenario) and well-known best practice, that is, business as usual (BAU). 51,46 Equation (1) shows the calculation of CEVs 37

where CEV = circular economic value; M = material flows on input and output sides; and E = energy flows on both sides.
In this study, the new heavy metal adsorption technology (as an alternative scenario – CEVScen) is compared to the BAU technique, which is a catalyst adsorption method (CEVBAU). The original CEV calculations were modified as CEVScen(mod) and CEVBAU(mod). The reason for these modifications was the lack of energy flows in the newly developed technique (alternative scenario) for which the on-field mechanical background of adsorption was not yet prepared. After the preparation of the prototype, the concrete energy flows of the new technique could be determined. In this study, only the material flows were used, on both the input and output sides. The CEV can only be given with material flows as well. The calculation method of the modified CEV is given in equation (2)

Results and discussion
Measurements of best adsorption time
In the analytical system, the adsorption capacity was measured at 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60 and 65 min. Figure 3 shows the changes in adsorption capacity (%) as a function of time (min). The adsorption capacities (%) were measured in a 250 mg/dm3 heavy metal concentration at every point in time. The results show the highest adsorption capacity after 50 min of shaking (the capacity was 98%). The measurements taken at 50, 55, 60 and 65 min showed the same result. According to our results, all of the samples were shaken for 50 min in other experiments.

Measurements of adsorption capacity at different shaking times. Source: Authors’ own research.
Chemical adsorption results for single elements
The solutions containing single elements were subject to the first adsorbing test, before the combinations. The adsorption of 55Mn (Figure 4) was best at the highest concentrations, and the adsorbing capacity did not reach its limit at 750 mg/dm3 and 1000 mg/dm3 concentrations.

Adsorption results for single elements from prepared stock solutions: (a) adsorption of Mn and (b) adsorption of Cd. All of these elements were measured by ICP-MS in isotope structures. Source: Author’s own research. ICP-MS: inductively coupled plasma mass spectrometry; Mn: manganese; Cd: cadmium.
Figure 4 also shows the changes in Cd adsorptions in each concentration. The adsorption of 111Cd and 112Cd produced the same result, and it may be concluded that the adsorption does not depend on the isotope numbers. At 1000 mg/dm3 the adsorped concentration was the lowest. The reason for this value is the ion size of 111Cd and 112Cd, because the largest ions could not be adsorbed perfectly at the highest concentrations, and the adsorption capacity depends on the ion size. This is also the reason for the doubled and tripled heavy metal adsorptions.
Adsorptions of double, combined elements
Other solutions were also prepared, with double elements. The results show the different adsorption capacities, which depend on the doubled element sizes. Some of these elements can stop the adsorption of others because the sizes are also different. The adsorption of the largest sized elements can result in poor adsorption of smaller sized elements. The comparison of adsorption properties from doubled solutions and from single solutions can cause the different adsorption rates in the same original concentration (e.g. the adsorptions of 55Mn were better in single content solutions, and showed a continuous increase). The solutions containing double elements were the following: 55Mn, 63Cu, 65Cu and 55Mn, 111Cd, 112Cd (Figure 5). These two combinations produced unique results because the 500 mg/dm3 concentrations had the highest adsorbing capacity. After this concentration, the adsorption capacity of the mushroom compost was lower.

Adsorptions of heavy metals in double combinations: (a) adsorptions of Mn and Cd and (b) adsorptions of Mn and Cu. All of these combinations were measured by ICP-MS in isotope structures. Source: Author’s own research. ICP-MS: inductively coupled plasma mass spectrometry; Mn: manganese; Cd: cadmium; Cu: copper.
The adsorption of 55Mn, 63Cu and 65Cu was different to that of 55Mn, 111Cd and 112Cd. High concentrations of heavy metals are serious problems when cleaning water (e.g. waste water treatment, drinking water treatment). The adsorption of heavy metals on the surface of mushroom compost is one alternative technology for removing heavy metal pollution from water and waste water. The development of a new shaking method is a new scientific result designed to achieve the best heavy metal adsorption. In this research, special mushroom composts and different heavy metals were used, in different combinations. For the preparation of heavy metal solutions, laboratory methods were used: the shaking method was applied to the adsorption of heavy metals on the surface of the mushroom compost, and the sample degradation and analytical measurements were performed by ICP-MS. Mushroom compost heavy metal adsorption properties can be used for sewage treatment if waste water contains low or high concentrations of heavy metals.
CEV calculation results
The new heavy metal adsorption method developed should not be evaluated only on its analytical chemical elements. CEV calculations are important to determine the economical background of this new technique.
Calculation of an alternate scenario (the new adsorption technique)
The main point of the laboratory and mechanical background of the new adsorption technique is the addition of separate waste water. Consequently, the numbers of waste water samples on the input and the output side are equal. The amount of mushroom compost is different. The amount of mushroom compost input remains in the adsorption system until it can adsorb heavy metals. If it is filled with heavy metals it can be present in the output side as a type of waste (although, from an environmental point of view it is not really waste because after adsorption it can be useful for energetic application, e.g. by pyrolysis). The amount of waste water can easily be determined on the input side, as part of the physical design of the system, given that the proportion of the compost and waste water to be cleaned is already determined. Therefore, the amount of waste water is calculable.
On the output side, we must consider a very important factor: that is, the minimum, but ever-present quantity of materials. This is due to the fact that during the removal of heavy metal – that is, during adsorption – along with the heavy metals, a minimum amount of waste water is added to the compost, as the heavy metal adsorption occurs in an aqueous sample and the maintenance of this aqueous sample is essential for the efficient adsorption of heavy metals. Using equation (2), the following amounts of material were determined (listed in Table 3).
Material amounts (input and output sides) of the alternate scenario.

Source: Authors’ own research.
Choosing the BAU technique for comparison analyses
There are many alternatives to choose from when seeking the best practical method to apply to waste water treatment (e.g. microbiological methods, coagulation and flocculation). All of these heavy metal elimination techniques can be usefully applied in the waste water system, but eliminating heavy metals efficiently and using the system economically and operating in a financially efficient way can be common targets. Our choice of the BAU method was to use the adsorption technique with catalytic material in a chemical approach. In this method, a laboratory prepared chemical material works as the adsorption surface. This method cannot be described with a CEV. The circular-related problems of this method are listed below: It is a chemical-intensive process in the maintenance of the oxidation material. In this case, the chemicals appear as a constant secondary raw material requirement. A catalytic filter is an artificial material which has raw material and energy demands (vs. the technique I developed which has minimal composting raw material and in which the energy required for the process is generated by the microbial process itself during the composting phase). The most important element of the system’s operation is that as a result of metal precipitation chemical rainfall is generated, which can be defined as quasi-waste as it is not recyclable and cannot be returned to the system. Due to the continuous precipitation (e.g. waste), the cleaning process cannot be circulated. We are dealing with a non-circular, linear system, which can only be operated in a sustainable manner by continuously providing primary raw material and from which the resulting waste (a chemical precipitate that contains heavy metals in a complex form) is continuously discharged, which causes clogging and prevents the process from stopping.
In the Table 4 are the raw materials of the input and output sides of the system during optimum operation.
The material amount of BAU on input and output side.

Source: Authors’ own research.
BAU: business as usual.
CEV calculations with material flows and amounts
Based on the CEV formula, the CEV value for the BAU material flows can be written. This is important because this new technology method (i.e. an alternative scenario) can be compared with the selected BAU technique. The full CEV% value for BAU is also part of the weighting of available energy currents for BAU. If CEV%BAU is calculated without the energy balance, the modified value (CEV%BAU(mod)) will be (equation (3))
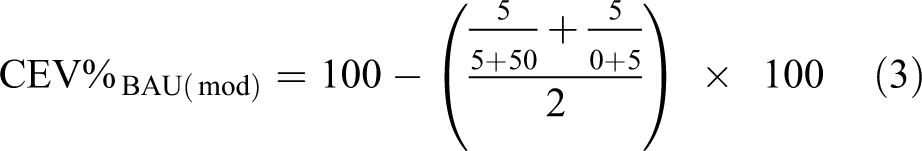
The calculated value is CEV%BAU(mod) = 45.5%. The linearity of BAU is also reflected by energy flows; the number obtained from the modified CEV% formula does not report the total value of the circularity. At the same time, only this modified CEV% value can be compared to the developed new technology, so CEVScen is not yet determined by energy values associated with the mechanical preparation. Calculation of CEV%Scen according to CEV%BAU is based on a modified CEV formula with material flows. So results can be compared only by using the following formula (equation (4))

The result is CEV%Scen(mod) = 87.5%. By comparing the values, we can reach important conclusions. The difference between CEV%BAU(mod) = 45.5% and CEV%Scen(mod) = 87.5% is the amount of material which can be recycled at the end of the life cycle of the product; this is the same as the primary raw material, because mushroom compost is a completely recyclable material. The question of recyclability is important for circularity testing and helps determine the energy balance (energy flows) later. Based on these two modified CEV%mod values, the modified ΔCEVmod formula can also be written (equation (5))

The results of the total circular economic value (ΔCEV) calculation: ΔCEVmod = 42.0% (the CEV calculation results are shown in Figure 6).

CEV calculation results for each technique and ΔCEV. Source: Authors’ own research. CEV: circular economic value; ΔCEV: total circular economic value.
Based on the result, the ΔCEV cannot be identified alone, but there is a significant difference between the BAU method and the developed method in terms of material flows. The difference in BAU is also because the adsorber used for the technique is an artificial chemical, just like the precipitate that is generated as a waste product in the system. However, in the new technique, we are referring to a natural adsorption material that can be produced at any composting site, and which can regenerate and not endanger the end of the life cycle, although it is an energetically usable product. This last point is one of the essential differences between the two techniques.
Possible implementation on a waste water treatment plant
The presented adsorption method developed under the laboratory test. Our results have shown, overall, that our method is suitable for reducing heavy metal pollution affecting some of the city’s major problems, drinking water bases and surface water bodies.
We would like to present a possible practical (use) approach, which is (linking like an alternative route to the existing waste water technology line) daily linked to the main cleaning line and does not replace any of its segments (Figure 7). Since we used special composts during the laboratory research, we concluded that the alternative technology branch should definitely be linked before the biological treatment step. Because the adsorption with compost, a high organic matter content may be present in further waste water amount, typically in the form of water-soluble or water-miscible complex compounds. In order to avoid that the purified and absorbed water does not cause eutrophication, or organic matter accumulation, the biological purification rate should be applied after adsorption.
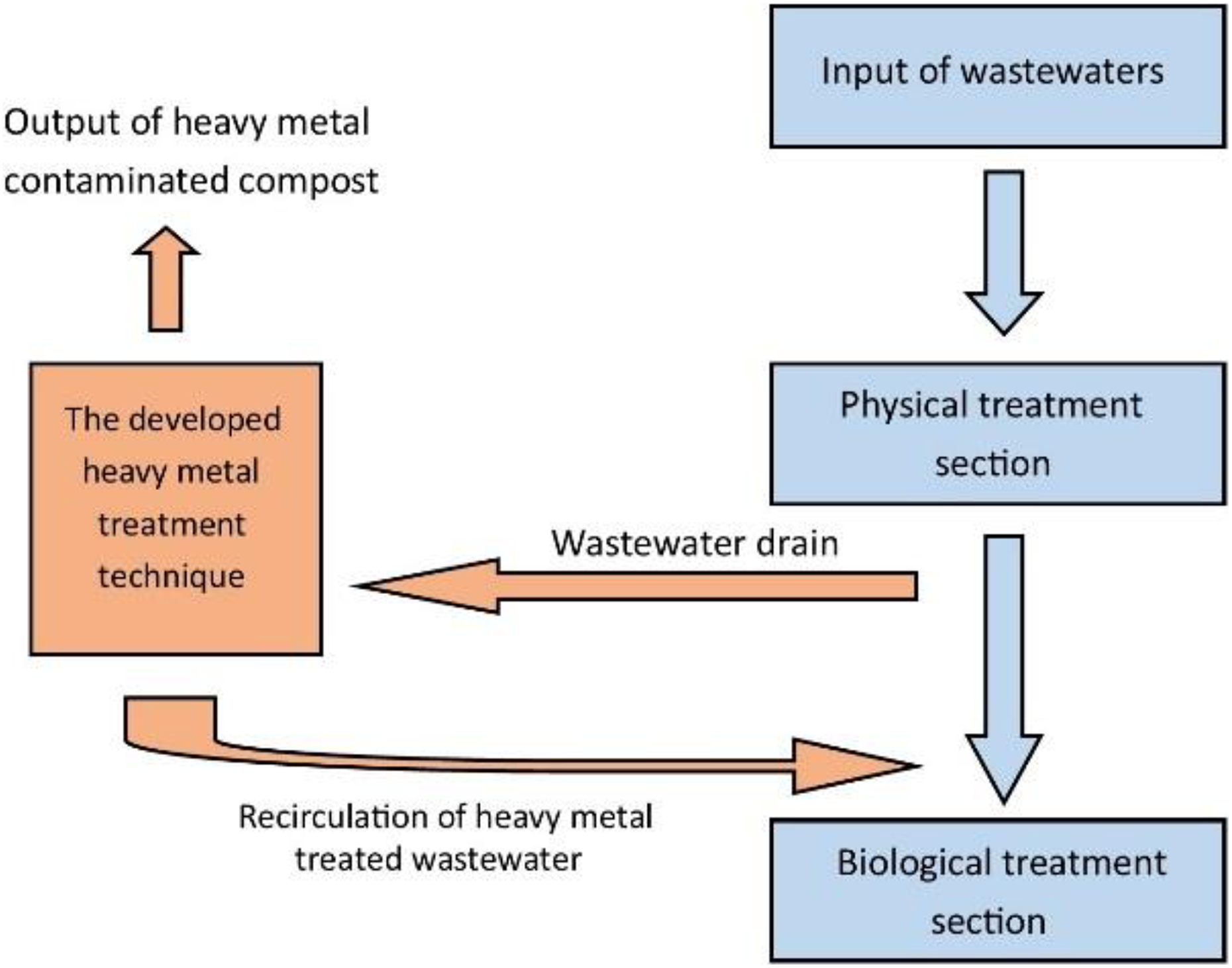
The possible placement of the alternative treatment section. Source: Authors’ own research.
The exact determination of the location of this heavy metal adsorption method is important because of the presence of organic substances in the system due to the compartments of the compost materials.
The energy supply is also an important issue of the system. The operation with biogas seems to be an obvious solution as most domestic waste water treatment plants have biogas production to solve power supply question of waste water treatment plants. As an energy supply, there is also an opportunity based on the production of a compost as an adsorption medium. During the composting process, the heat energy required for the maturation phase is produced by the composting system itself by works of microorganisms. If the spotted compost is produced at a given waste water treatment plant, it is possible to recycle the heat generated by the composting process to provide the energy supply of heavy metal removal technology. At the same time, by examining the possibility in economic terms, the value of the circular economic (CEV) of the developed technology can be further increased.
The heavy metal–contaminated compost could be reused after elimination of their pollution contents. This method means a reusable adsorption material but it could not be a solution many times. The size of the compost surface determines the possible heavy metal adsorption capacity (volume) and a potential size of the reusable adsorption surface. If that size seems to be smaller (adsorption by adsorption), it could be the end of the compost material life. The compost could be used with pyrolysis to heat and energy production.
Conclusions
We have measured the adsorption properties of heavy metals in each concentration, but we have yet to determine the best adsorption capacity by determining adsorption at the highest concentrations. There should be an increase in the concentration up to 1000 mg/dm3, and heavy metals must be involved in other combinations (including triple combinations). We have to develop the shaking method continuously to fit triple heavy metal combinations. We can determine the behaviour of all heavy metals if they are included in the solutions at the right time. This will complement the entire heavy metal adsorption in polluted solutions. Our research has identified a new heavy metal adsorption method that can increase adsorption efficiency by shaking. A special mushroom compost was used as an adsorbent, and we researched the heavy metal adsorption properties of this special compost. Heavy metal solutions were used to determine the adsorption processes. The heavy metal solutions were prepared from Cu, Cd and Mn in single, double and triple combinations and in several concentrations. Following the laboratory practice, we must decide which is the best place for this technology in the waste water treatment chain. The new heavy metal treatment technique developed could be useful in waste water treatment fields. The results of laboratory practice show a hopeful possibility to find a place for the new technique in the waste water treatment system. There are different points in the system that might be the most appropriate, and we would like to suggest one of these. The new technique works with mushroom compost and consequently the treated waste water contains relevant organic matter compounds, so one of the most appropriate points could be before the biological treatment process, because of the organic matter content. There is an opportunity to develop an alternative treatment circle linked to biological purification. The technical modelling of the new adsorption method seems to be an exercise for the future, in order to decide the best technical form in real waste water treatment circumstances. Another future task is the total circular economic analysis of the new system, to help identify the practical and technical parameters of the technique. The analysis should follow the on-field preparation because the mechanical forms can produce other circular parameters.
The CEVs are calculated by modified equations, which are shown in each section. The energy flows cannot yet exactly calculate an alternate scenario because our experiment was laboratory research, and energy amounts can only be given after on-field preparation. The modified CEV equations seem to be suitable to describe the circular system because the material amounts can describe the circularity of the alternative scenario.
Footnotes
Acknowledgements
The authors thank the Szent István University Doctoral School of Management and Business Administration and the Institute of Analytical Chemistry, University of Vienna, Austria, for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
