Abstract
Tags are attached to salient information during the wake period, which can preferentially determine what information can be consolidated during sleep. Previous studies demonstrated that spindles during non-rapid eye movement (NREM) sleep give priority to strengthening memory representations with tags, indicating a privileged reactivation of tagged information. The current meta-analysis investigated whether and how spindles can capture different tags to consolidate declarative memory. This study searched the Web of Science, Google Scholar, PubMed, PsycINFO, and OATD databases for studies that spindles consolidate declarative memory with tags. A meta-analysis using a random-effects model was performed. Based on 19 datasets from 18 studies (N = 388), spindles had a medium effect on the consolidation of declarative memory with tags (r = 0.519). In addition, spindles derived from whole-night sleep and nap studies were positively related to the consolidation of memory representations with tags. These findings reveal the shared mechanism that spindles are actively involved in the prefrontal-hippocampus circuits to consolidate memory with tags.
Introduction
In the past two decades, breakthroughs in memory research indicate that newly acquired information can be stored in a long-term memory network during sleep (Cowan et al., 2021; Diekelmann & Born, 2010; Gais & Born, 2004; Inostroza & Born, 2013; Marshall & Born, 2007; Marshall et al., 2020; Schönauer, 2018; Stickgold & Walker, 2013). Human sleep consists of two distinct stages, non-rapid eye movement (NREM) and rapid eye movement (REM) sleep (Deliens et al., 2013; Squire, 2004). According to the sequential hypothesis, the sequence of NREM and REM stages in cyclical succession supports memory consolidation (Gais et al., 2000). An alternative theory assumes that the consolidation of declarative memory relies on NREM sleep, while REM sleep supports non-declarative memory consolidation (Maquet, 2001). Although there are subjective debates over the precise role of different sleep stages in memory consolidation, it is generally agreed that NREM sleep plays an essential role in declarative memory consolidation (Peyrache & Seibt, 2020).
Recently, much attention has been paid to specific oscillations during NREM sleep (Farhadian et al., 2021; Jegou et al., 2019; Petzka et al., 2022; Peyrache & Seibt, 2020). Neocortical slow oscillations (SO; 0.5–1 Hz), thalamocortical spindles (11–16 Hz), and hippocampal sharp wave-ripples (SWR; 200 Hz) are hallmarks of NREM sleep (Peyrache & Seibt, 2020). The active system consolidation theory postulates that precise temporal interactions of SOs, spindles, and SWRs may facilitate the transfer of information from the hippocampus to the neocortex (Rasch & Born, 2013; Staresina et al., 2015). SOs upstates trigger the emergence of spindles, which in turn promotes the transfer of information replayed during hippocampal SWRs, leading to long-term consolidation in the neocortex (Klinzing et al., 2019). This assumption indicates an active process of memory consolidation during NREM sleep.
Among these oscillations during NREM sleep, spindles are known for involvement in neural plasticity processes and declarative memory consolidation (Kumral et al., 2022; Peyrache & Seibt, 2020; Sun et al., 2020; Ulrich, 2016). At the cellular level, spindles have been shown to induce synaptic plasticity change (Marshall et al., 2006), thereby consolidating newly acquired information. At the systems level, numerous studies found that spindles are related to activation in the hippocampus and cortex (Bergmann et al., 2012; Schabus et al., 2007), hippocampal-cortical functional connectivity (Andrade et al., 2011; Cowan et al., 2021), and memory reactivation (Antony et al., 2019; Bergmann et al., 2012; Cowan et al., 2020; Schabus et al., 2007; Schönauer, 2018). However, the sleeping brain does not equally preserve the recently encoded information due to the limited storage capacity (Cowan et al., 2021). Instead, sleep does exert its beneficial influence on declarative memory consolidation selectively. Emerging studies demonstrated that sleep spindle during NREM may be a compelling candidate in the selective consolidation of declarative memory.
The role of sleep in consolidating memory with tags
Tags are set during or near the learning event that can designate specific traces of more meaningful and valuable information within an event to selective consolidation. Numerous tags, such as novelty, reward, and future relevance, have been found to enhance memory strength by inducing activation of the medial temporal lobe and oscillatory activity (Gruber et al., 2013; Kishiyama et al., 2009; Wilhelm et al., 2011). Likewise, the cellular mechanism reveals how memory with tags is selectively consolidated. When reacting to tags, neuromodulators are released to alter synaptic plasticity in the hippocampus, which can favor memory with tags into a stable form of long-term storage (Lisman & Grace, 2005; Shohamy & Adcock, 2010). Therefore, in the peri-encoding wake period, tags attached to memory representations provide the initial selection for discriminatory consolidation (Stickgold & Walker, 2013).
During subsequent sleep, tags may be utilized to consolidate memory selectively. According to the selective memory consolidation model, encoded information tagged as future relevant would undergo consolidation during sleep (Stickgold & Walker, 2013). In support of this model, several studies have highlighted a selective consolidation during sleep for memory tagged with future actions (Barner et al., 2017; Scullin et al., 2019), highly rewarded memory (Fischer & Born, 2009; Michon et al., 2019), and the “remember” but not “forget” items in directed forgetting lists (Saletin et al., 2011; Scullin et al., 2017). Critically, when encoding to-be-remembered and cued-to-be-forgotten items, individuals show different degrees of activation in the hippocampus, which predicts subsequent the magnitude of differential offline sleep-dependent consolidation (Rauchs et al., 2011). This hippocampal activity during awake can serve as a signal for the consolidation of tagged memory during subsequent sleep. Furthermore, recent studies found that specific oscillations during NREM sleep, like spindles, could govern differential retention of memory with tags (Blaskovich et al., 2017; Igloi et al., 2015; Saletin et al., 2011; Studte et al., 2017). However, the precise neural mechanisms of specific oscillations during sleep remain unclear.
Indeed, existing studies use targeted memory reactivation (TMR) to exogenously trigger spindles and manipulate memory processing during sleep, which can selectively enhance memory (Cairney et al., 2018; Cox et al., 2014; Rihm et al., 2014). TMR is a paradigm in which individuals learn memory items accompanied by auditory or olfactory stimuli. Then, presenting these stimuli during sleep can reactivate the paired memory items in a noninvasive way (Hu et al., 2020). In the work of Rasch et al. (2007), individuals learned the spatial location task in the presence of an olfactory cue. Compared with the control group delivered with the odorless vehicle, memory performance improved when the same olfactory is re-presented during slow wave sleep. However, Rasch et al. (2007) did not reveal how exogenous manipulation by TMR evokes the brain’s endogenous consolidations. Olfactory and auditory-induced memory reactivation during NREM sleep has been additionally shown to induce an increase in spindle activity and enhance memory performance (Cairney et al., 2018; Cox et al., 2014; Rihm et al., 2014). Interestingly, an auditory-induced increase in spindle activity could be reliably decoded as information about the learned material (Cairney et al., 2018). These studies indicate that sleep spindles exogenously modulated by TMR provide neuronal conditions favorable for the reactivation of memory traces. However, whether endogenous spindles can spontaneously and selectively consolidate memory is still unknown.
The role of spindles in consolidating memory with tags
Declarative memory consolidation in a selective way may point to the relevance of sleep spindle activity during NREM sleep. Specifically, using scalp EEG recordings, studies showed that spindle numbers in post-learning sleep were larger than in the control night (Clemens et al., 2005). Notably, the modulation of spindles is learning-dependent, as pre-sleep learning evokes an increase of spindles in learn-related brain regions (Bergmann et al., 2012; Jegou et al., 2019). Spindle topographies at the group level varied with different types of declarative memory tasks. Spindle numbers over the left frontocentral region were associated with verbal memory consolidation (Clemens et al., 2005), while parietal spindle numbers were related to the consolidation of visual-spatial memory (Clemens et al., 2006). However, as both studies did not measure brain activity during learning, the link between pre-sleep learning patterns and post-learning spindle topography is conjectural. Another study found that topographies of post-learning spindles were related to learning patterns measured by alpha power, thereby promoting memory consolidation (Petzka et al., 2022). The degree to which endogenous sleep spindles tracked learning patterns reflects that spindles per se could be selectively expressed at the learning-related cortex. Given that, spindles may facilitate the independent reactivation of memory traces in the task-related regions corresponding to awake learning.
When memory traces are attached to tags, sleep spindles may recognize memory with tags and facilitate the reactivation of the tagged circuit. In a word-pair association task, the experimental group was informed to receive a future test, while the control group was not. During the post-learning sleep, spindle counts were on average higher in the experiment than in the control group. Importantly, individuals with more spindles consolidate more items only in the experimental group (Wilhelm et al., 2011). This finding reflects that spindles can recognize tags of previously encoded memory traces. In another study, Saletin et al. (2011) asked individuals to perform a directed forgetting paradigm, in which an instructional cue to either “remember” or “forget” was followed by a word. They found that fast spindles at the left parietal cortex can predict the efficiency of sleep in differential reaction to to-be-remembered and to-be-forgotten items. Moreover, spindles were positively related to to-be-remembered items, yet were negatively associated with to-be-forgotten items. Additional source analysis identified a loop of recurring brain activity in a network of prefrontal, medial-temporal, and parietal in the presence of spindles. Consistent with previous neuroimaging studies, inferior prefrontal, superior parietal, and medial temporal lobes can promote successful differential remembering and forgetting (Anderson et al., 2004; Nowicka et al., 2011). It seems that, when coinciding with tagged circuits, spindles may resonate more strongly to facilitate the reactivation of memory representation with tags.
Indeed, sleep spindles preferentially consolidate memory processing attached to tags. There is ample evidence that spindles showed significant relationships with retention of high reward (Igloi et al., 2015; Studte et al., 2017), future relevance (Fillmore et al., 2021; Stickgold & Walker, 2013; Wilhelm et al., 2011), instruction to be remembered memory (Blaskovich et al., 2017; Saletin et al., 2011), association memory (Sopp et al., 2018; Studte et al., 2015), abstract (McCreesh, 2017; Schmidt et al., 2006) and semantic-related encoded items (Schreiner et al., 2021; Sopp et al., 2018; Studte et al., 2015; Van Der Helm et al., 2011), but not low-reward, future irrelevance, instruction to be forgotten, item-memory, concrete, and unrelated semantic items. An EEG-fMRI study further found that spindles correlated with functional connectivity between the hippocampus and caudate nucleus for high-reward items, but not low-reward items, indicating a privileged replay of memory tagged with high reward.
Sleep type as a moderator between spindle and memory consolidation
As a regular sleep pattern, whole-night sleep is distinct from nap in several aspects (Devine & Wolf, 2016). The length of whole-night sleep reported in young adults approximately ranges from 7.5 h to 8.5 h (Carskadon & Dement, 2005), while naps commonly last between 30 and 90 min (Dinges, 1989), which results in different sleep cycles between whole-night sleep (i.e., approximately four to six sleep cycles) and nap (i.e., one sleep cycle). Notably, although there are differences in sleep cycles, whole-night sleep and nap include NREM sleep, in which hippocampal memory representations are particularly replayed (Diekelmann & Born, 2010; Inostroza & Born, 2013). In addition, according to interference theories, retroactive interference during NREM sleep is reduced (Berres & Erdfelder, 2021). From the consolidation and interference perspective, NREM sleep can benefit memory. Furthermore, specific oscillations during NREM sleep, such as sleep spindles, matter most (Cordi & Rasch, 2021). When directly comparing the spindle from whole-night sleep and nap, previous research found that whole-night sleep showed higher spindle density, but lower spindle activity and amplitude (Van Schalkwijk et al., 2019). However, whether the role of spindles in overnight sleep and nap is consistent lacks systematical evidence.
Taken together, tags are initially established in the peri-encoding period and subsequently captured during sleep. Spindles during NREM sleep can consolidate declarative memory with tags. However, it remains unclear whether different tags can index spindles to stabilize memory items into long-term retention and how spindles can capture different tags to complete consolidation. In addition, the present meta-analysis attempted to explore whether the role of spindles in overnight sleep and naps is consistent. Therefore, this meta-analysis could outline the role of spindles in the consolidation of declarative memory with different tags and investigate the moderate effect of sleep type on memory consolidation with tags.
Methods
Literature review
The present meta-analysis was exhaustive to select articles in a systematic search and comply with the latest Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Page et al., 2021). The meta-analysis registered in the PROSPERO database (CRD42023478570). We searched relevant literature reviews on Web of Science (core), Google Scholar, PubMed, PsycINFO, and OATD, following these terms: “spindles” OR “memory consolidation” OR “memory recall” OR “memory retention” OR “declarative memory.” For those unpublished works, we use the same terms and combinations to search on the databases OATD.org (open access theses and dissertations). Microsoft Excel recorded the final search results and eliminated duplicates.
Inclusion criteria
Literature reviews that met the following criteria were included: (1) original article published from the inception (2000) to January 1, 2023; (2) participants were healthy adults; (3) assessing declarative memory; (4) research investigating endogenous sleep spindles recorded by the polysomnography; (5) studies manipulated different cues during encoding; (6) studies reporting the correlation coefficients between spindle and memory with different cues or the change of spindle activities in different cue; (7) English language and peer-reviewed studies.
Exclusion criteria
Literature reviews were excluded if (1) studies are abstracts, case reports, editorials, letters, narrative reviews, opinions, position statements, and systematic reviews and meta-analyses; (2) infancy, children, adolescent, and the aging; (3) with a known psychological/psychiatric disorder; (4) animal studies; (5) studies exclusively investigating the effect of sleep manipulations (e.g., pharmacological agents, exploring transcranial direct current stimulation, and target memory reactivation); (6) studies investigating the relationship between spindles and declarative memory consolidation at the cell level; (7) without salient cues during encoding; (8) data necessary for correlational coefficients were not available (Figure. 1).
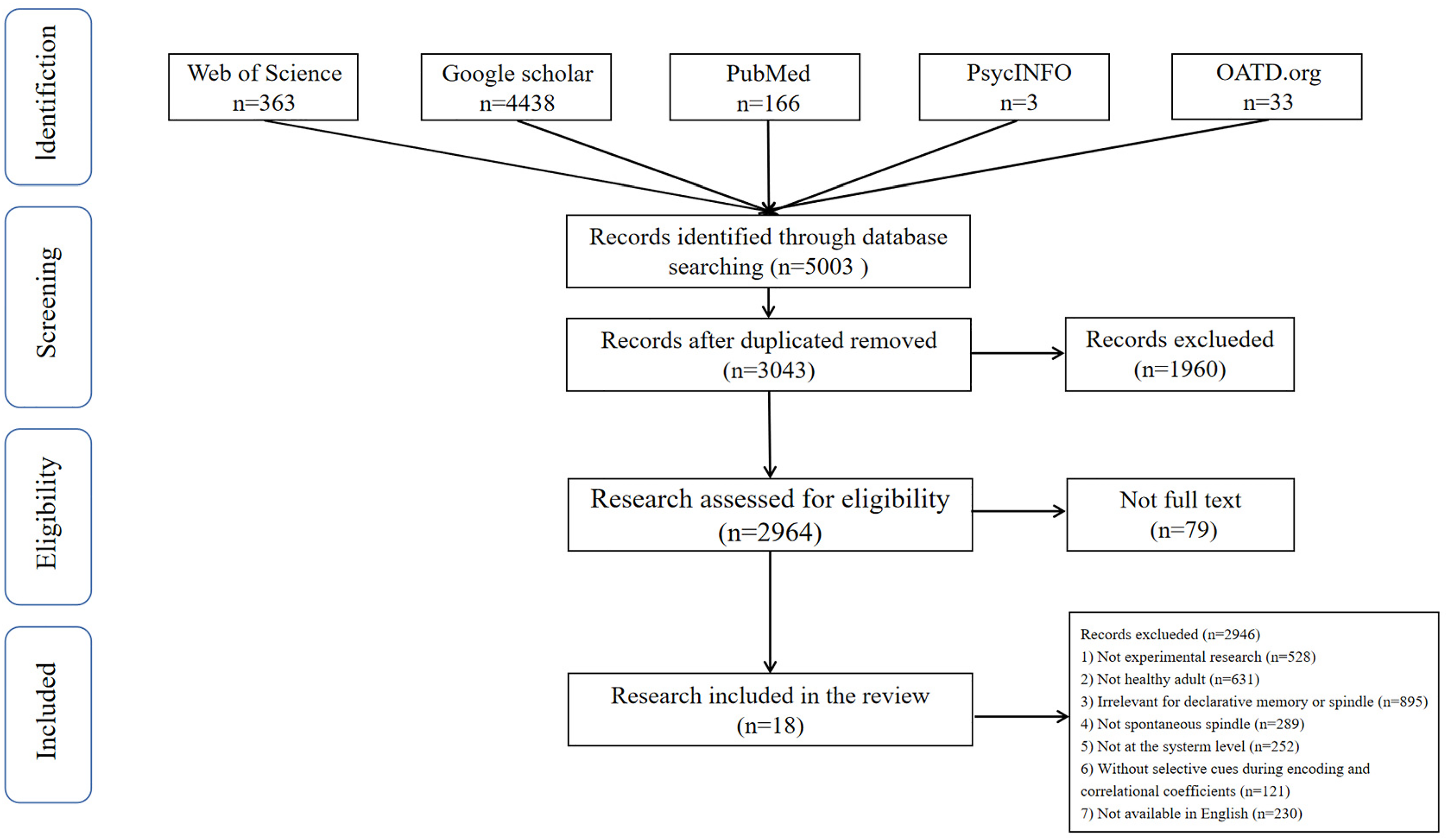
PRISMA flow diagram of study inclusion.
Data extraction
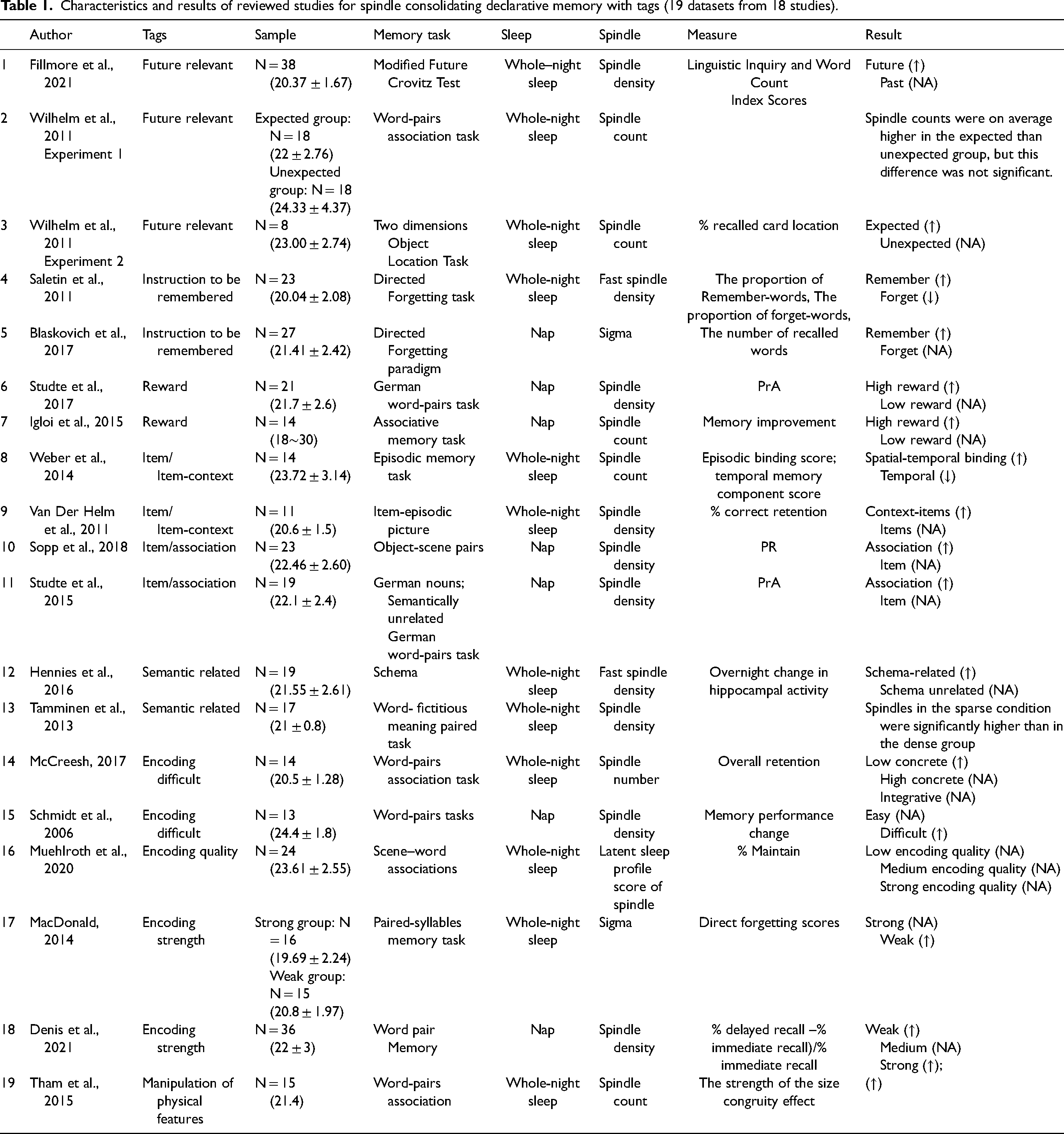
The data extraction protocol meets the standard of PRISMA guidelines (Page et al., 2021). Two independent researchers searched studies and decided which should be included according to the inclusive and exclusive criteria. There were no main discrepancies, like whether to include a study that did not report accurate age but only reported the age range. Given that the age range reported by the study is within adults, both researchers decided to include it. After screening, the following information was extracted: author name(s), tags, year of publication, sample size (age information), memory task, sleep type (whole-night or nap), spindle index, memory measure, and results relevant to this review (Table 1). Considering that Wilhelm et al. (2011) had two experiments, we split their research into two datasets.
Characteristics and results of reviewed studies for spindle consolidating declarative memory with tags (19 datasets from 18 studies).
Quality assessment and risk of bias
The Joanna Briggs Institute (JBI) checklist was used to assess the quality and risk of bias of the research included in the meta-analysis. Due to the methodologically rigorous development by expert committees and accessibility to thorough user-guidance documentation, the JBI checklist was chosen. The score from the JBI checklist ranged from 5 to 8 in the current meta-analysis.
Statistical analyses
Random-effects meta-analyses were conducted with Comprehensive Meta-Analysis Version 3.0 (Biostat, Englewood, NJ). Based on 19 datasets, we computed summary effect sizes for the effect of sleep spindles on declarative memory with tags. The analysis used funnel plots and Egger’s test of intercept (Egger et al., 1997) to measure publication bias. Ninety-five percent confidence intervals are employed to estimate the statistical significance.
For the meta-analyses, between-study heterogeneity was indicated by Cochran's Q and the I2 statistic. While the Q statistic provides information on the significance of the heterogeneity of effect sizes, the I2 index is a measure of the magnitude of the extent of true heterogeneity, versus sampling error, between studies. Percentages of 25%, 50%, and 75% indicate low, medium, and high heterogeneity, respectively (Higgins et al., 2003). A sensitivity analysis was conducted to evaluate the robustness.
Subgroup analyses were conducted to examine the possible moderator of effect size. Although the meta-analysis is low heterogeneous (see Results for more details), we identified sleep type (whole-night sleep, nap) that could potentially explain the heterogeneity in effect sizes. The present meta-analysis used subgroup analyses to quantify the effect size of spindles from whole-night and nap studies on memory with tags separately.
Results
Study characteristics
Nineteen datasets were included in this meta-analysis (Table 1) published from 2006 to 2021, in peer-reviewed journals. A total of 388 participants were tested across all studies, with each individual study sampling around 20 participants on average. The mean age was 21.86 years. Twelve datasets assessed memory performance following the whole-night sleep, and seven datasets following the nap. All studies used polysomnography (PSG) to measure the sleep spindles of participants. Nineteen studies investigated the spindles consolidated declarative memory with tags.
The methodological quality of the included studies
Among 19 datasets, the average JBI quality score was 7.11 (range from 5 to 8 points; Supplementary Table 1).
Primary outcome: Spindles effect on memory with tags
Table 2 showed that the Q statistic was significant (p < .05), indicating that the effect sizes in the present meta-analyses were heterogeneous. According to Borenstein's view of I2 (Borenstein et al., 2009), I2 in the meta-analysis of tags was 39.767, indicating that 39.77% of the observed variation in the association of spindles and memory with tags was caused by the real differences. The results of the heterogeneity analysis supported that a random-effects model should be adopted.
Assessing tags of heterogeneity.
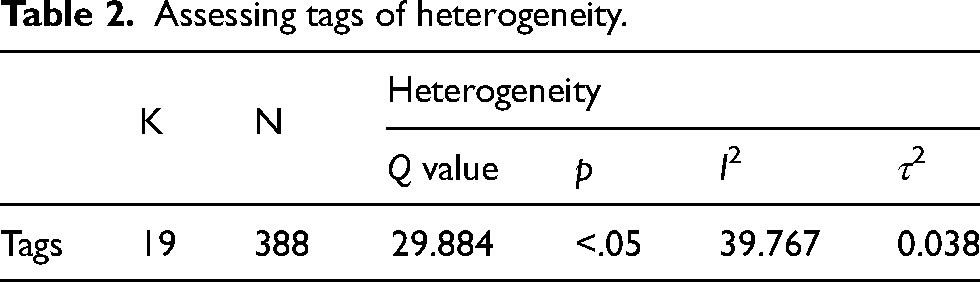
Publication bias was observed (Egger's test: p > .05) and is illustrated in a funnel plot whereby effect sizes of studies were plotted against their standard errors (Figure 2(A)). Sleep spindles were significantly related to tag memory, and the effect size is medium (r = 0.519, p < .001; Figure 2(B)). A sensitivity analysis was conducted. We found that the overall effect of spindles remained significant, indicating the stability of the results (Figure 2(C)).
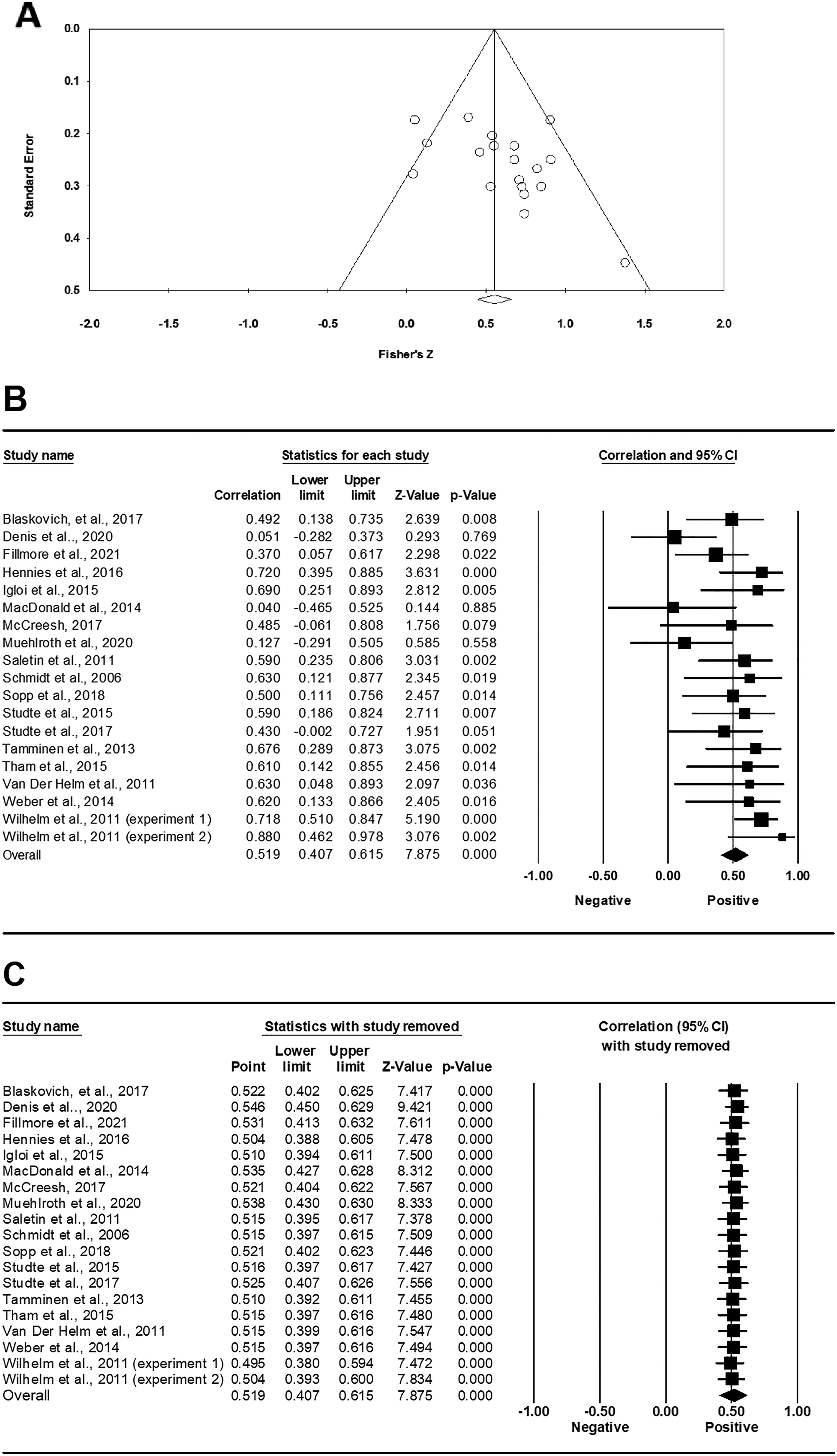
(A) Funnel plots of correlational coefficients between spindles and memory with tags. (B) Forest plot of correlational coefficients between spindles and memory with tags. (C) Sensitive analysis between spindles and memory with tags.
Secondary outcome: The moderator of sleep type
Subgroup analyses were conducted to quantify the effect size of spindles from the whole-night sleep (n = 12) and nap (n = 7) datasets separately. Spindles from whole-night sleep datasets were significantly related to memory consolidation with tags (r = .552, p < .001). The effect of spindles from nap datasets was also significantly related to memory consolidation with tags (r = .460, p < .001).
Discussion
The present meta-analysis investigated the effect of spindles on the consolidation of declarative memory with tags during NREM sleep. Based on 19 studies, we found that spindles had a significant medium benefit on consolidating declarative memory with tags. This finding verified the selective consolidation model that favors the existence of proactive memory processing during sleep. Specifically, we revealed that spindles, one of the specific sleep oscillations, could be spontaneous to consolidate memory with tags. Finally, this meta-analysis took different tags into consideration and speculated a shared mechanism that spindles may be actively involved in the prefrontal-hippocampus circuit to consolidate memory items with tags.
The meta-analysis found a positive relationship between sleep spindles and the consolidation of declarative memory with tags. The possible mechanism is that spindles facilitate the reactivation of memory representation with tags in the hippocampus. Memory consolidation relies on “reactivation,” whereby patterns of neuronal population activity representing previous encoded events spontaneously reactivate (Peyrache & Seibt, 2020). In particular, there is a privileged reoccurrence of neural patterns corresponding to memory items with tags during sleep (Sterpenich et al., 2021). The present meta-analysis demonstrated that spindles during NREM sleep could be spontaneous to favor the reactivation of memory representations with tags. Compared to other stimuli in the same learning episode, information with tags evokes more spindle activities and higher spindle densities in post-learning sleep (Tamminen et al., 2013; Wilhelm et al., 2011). The increased spindle activities trigger the increased hippocampus ripple activities (Mölle et al., 2006), which have been associated with the reactivation of the learning experience (Joo & Frank, 2018). Through repeated reactivation, memory items with tags can be stabilized in the hippocampus and then facilitated their redistribution into the neocortex for long-term consolidation. TMR showed a similar mechanism, which exploits spindle-mediated information processing to strengthen selectively specific memory representations (Cairney et al., 2018). These findings argue against the opportunistic theory that sleep passively protects memory from interference (Mednick et al., 2011). Indeed, an active selection of memory with tags favors an active role of sleep in memory consolidation. Given that, the differential consolidation indicated that spindles could actively prioritize consolidating declarative memory with tags rather than passively protecting against interference.
Another key question is how different tags can be captured by spindles to realize consolidation. Dovetailed with the selective memory model (Stickgold & Walker, 2013), different tags, such as future relevance, reward, and instructions to be remembered, are attached to memory items in the peri-encoding period. This initial selection distinguishes recently encoded information into memory items with relevant and irrelevant tags, which initially accomplishes discriminatory retention. During sleep, relevant tags are subsequently utilized to selectively consolidate the paired memory items (Stickgold & Walker, 2013). This model supports the possibility that different tags can index spindle-mediate spontaneous reactivation. Critically, it is inevitable to cast doubts as to whether the different kinds of relevant tags listed in the selective model are comprehensive. According to the tag-and-capture model, relevant tags consist of encoded-based tags and retroactive tags. Encoded-based tags, presented as salience cues such as the reward, directly pair with memory items during encoding. Retroactive tags originate from an experience signaling the relevance of preexisting information before encoding (Frey & Morris, 1998; Redondo & Morris, 2011), such as the shared structure or overlap. Processing information with different tags is bound to functions of the prefrontal cortex. Inhibitory control of the prefrontal cortex can process desired memory traces to suppress competing memories (Anderson & Hulbert, 2021). In this way, different tags can help the paired memory items form better by evoking higher activation of hippocampal activity (Adcock et al., 2006; Rauchs et al., 2011; Rugg et al., 2012), thus enabling sleep-dependent memory processes to optimally consolidate salient information. During sleep, spindles may be actively involved in the prefrontal-hippocampus circuits, in which the top-down different tags sourced from the prefrontal cortex are utilized to reactivate the bottom-up item memory information from medial temporal areas, like the hippocampus (Shimamura, 2011). Spindles over the prefrontal lobe resonate more strongly when coinciding with those tagged circuits, which may trick the hippocampus by inducing what looks like a spontaneous reactivation of the specific memory items in the cortex and in turn influence what the hippocampus replays. In this way, spindles can capture different tags to consolidate memory.
Cellular consolidation may also provide a mechanism that the positive relationship between spindles and the consolidation of memory with tags reflects synaptic strengthening that processes on memories tagged. During encoding, tags supported the conversion of early phase long-term potential into late-phase, a better situation favorable to plasticity (Frey & Morris, 1998). Such tags ensure memory specificity by capturing the plasticity-related products of the activated memory representations, but not others, which creates the optimal conditions for consolidation (Payne & Kensinger, 2018). The allocation of tags may be manifest in tagging the newly acquired memory traces that promote their access to SWS-sleep-dependent consolidation (Marshall & Born, 2007). Spindles provided optimal conditions for the synaptic consolidation of traces (Petzka et al., 2022). Animal studies found that spindles could alter neural plasticity, which spindle-like firing patterns provoke long-term potentiation in pyramidal cells (Rosanova & Ulrich, 2005). Spindles capture synaptically tagged circuits via increased dendritic activity and the influx of Ca2+ to complete the tag-and-capture cycle, which could strengthen memories with tags in the neocortex.
This meta-analysis found that spindles from whole-night sleep and naps were consistently related to memory consolidation with tags. This result indicated that the number of sleep cycles and the characteristics of spindles could not influence the spindles-consolidation of declarative memory with tags relationships in both sleep types. In line with previous meta-analysis, naps showed a similar consolidation effect to whole-night sleep (Berres & Erdfelder, 2021; Hokett et al., 2021; Kumral et al., 2022). When further investigating the specific sleep oscillations, studies found that sleep duration has no moderate effect on the relationship between spindle and memory consolidation (Kumral et al., 2022). Our result further found that spindles could facilitate the consolidation of memory representations with tags regardless of sleep type. Taken together, the current findings might suggest that spindle plays a general role for consolidating declarative memory with tags, regardless of sleep type and duration.
Limitations and future studies
The meta-analysis revealed that spindles could be spontaneous to capture the tag created during the awake period and consolidate memory with tags. However, our meta-analysis did not distinguish the role of spindles in N2 and slow wave sleep, as previous research indicated the differential involvement of spindles during N2 versus slow wave sleep on declarative memory consolidation (Dehnavi et al., 2019). Future studies should further clarify the differential role of sleep spindles on memory processing during sleep.
Second, our meta-analysis did not examine the possible moderators of spindle activities. The meta-analysis, conducted by Kumral et al. (2022), found a moderator effect of spindle types between spindles and memory consolidation, with the most significant effect sizes for spindle power and frequency. In this perspective, future studies can conduct the moderator analysis of spindle power and frequency in the sleep spindle-memory consolidation with tags relationship.
Third, the present study only included research among adults. However, due to the different physiological proportions of spindles, it is hard to generalize to the other age groups. Specifically, from infancy to adolescence, sleep spindle density and length changes with maturation (Scholle et al., 2007), while age-related reduction in spindles occurs among the aging (Wilson et al., 2012). It remains unclear whether the physiological change in spindles can influence the consolidation of declarative memory with tags. Given that, future studies need to clarify the role of spindles in consolidating declarative memory with tags across different age groups.
Conclusion
The current meta-analysis found that spindles during NREM sleep have a significant medium benefit on the consolidation of declarative memory with tags. Based on tags created during the wake period, spindles from both whole-night sleep and nap can consolidate declarative memory with tags. This finding extends the selective consolidation model and provides insight into the role of specific oscillations during sleep in exerting differential consolidation. Such processing benefits individuals by spontaneously strengthening important and valuable memories into long-term consolidation during sleep.
Supplemental Material
sj-docx-1-pac-10.1177_18344909241226761 - Supplemental material for Sleep spindles consolidate declarative memory with tags: A meta-analysis of adult data
Supplemental material, sj-docx-1-pac-10.1177_18344909241226761 for Sleep spindles consolidate declarative memory with tags: A meta-analysis of adult data by Peiyao Chen, Chao Hao and Ning Ma in Journal of Pacific Rim Psychology
Supplemental Material
sj-docx-2-pac-10.1177_18344909241226761 - Supplemental material for Sleep spindles consolidate declarative memory with tags: A meta-analysis of adult data
Supplemental material, sj-docx-2-pac-10.1177_18344909241226761 for Sleep spindles consolidate declarative memory with tags: A meta-analysis of adult data by Peiyao Chen, Chao Hao and Ning Ma in Journal of Pacific Rim Psychology
Footnotes
Author contributions
Peiyao Chen: Data curation, formal analysis, methodology, visualization, writing-revising original draft; Chao Hao: formal analysis, revising original draft; Ning Ma: conceptualization, funding acquisition, resources; writing-review and editing, supervision, validation.
Availability of data and materials (data transparency)
The data that support the findings of this study are available on request from the corresponding author. The meta-analysis was not preregistered.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for publication
The authors confirm that the work described was original research that has not been published previously, and is not under consideration for publication elsewhere, in whole or in part. All the authors listed have approved the manuscript that is enclosed.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic and Applied Basic Research Foundation of Guangdong Province (grant number 2023A1515011873).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
