Abstract
Alexithymia is a subclinical personality disorder characterized by difficulties in identifying and expressing one's own emotion. Individual differences in alexithymia are influenced by both genetic and environmental factors. However, the interplay between these factors and their influences on alexithymia are unclear. Considering that oxytocin plays important roles in emotion processing and that parental bonding influences the development of alexithymia, we explored the associations between OXTR polymorphisms and alexithymia and examined whether the potential associations are moderated by parental bonding. To this end, we genotyped the OXTR polymorphisms (rs53576 and rs1042778) and scored alexithymia and parental bonding with the Toronto Alexithymia Scale and the Parental Bonding Instrument. Results indicated that: (1) the characteristics of alexithymia were negatively associated with parental care and positively with parental overprotection; (2) the OXTR rs53576 was significantly associated with the characteristics of alexithymia, such that the AA genotype was associated with fewer difficulties in identifying feelings and describing feelings than the AG/GG genotypes; and (3) the OXTR rs1042778 interacted with parental care in alexithymia with the vantage sensitivity model: the GG genotype was related to less severity of alexithymia than the AG/GG genotypes only in individuals with higher parental care. Overall, these findings suggest that the OXTR is related to alexithymia and that the quality of parental care influences the relationship. However, considering that the evidence from this study is weak, more research is needed to understand the roles of OXTR in alexithymia.
Keywords
Introduction
Alexithymia is a subclinical personality disorder mainly characterized by difficulties in identifying and expressing one's own emotions and lack of fantasizing emotional experiences (Bagby et al., 1994). It has been shown as a transdiagnostic risk factor of psychiatric diseases (Paniccia et al., 2018; Ricciardi et al., 2015) and a predictor of interpersonal functions and quality of life (Solati et al., 2018; Zhang et al., 2021). Research suggests that alexithymia is influenced by both genetic and environmental factors (Jorgensen et al., 2007; Terock et al., 2021). However, the interplay between these factors and their influences on alexithymia are unclear.
Alexithymia is related to the deficits in self-awareness (the ability to recognize and understand one's own emotions and thoughts) and emotion processing (Loiselle & Dawson, 1988; Panayiotou et al., 2020). Studies have indicated that individuals with high alexithymia exhibit low self-disclosure and private self-consciousness (Loiselle & Dawson, 1988; Panayiotou et al., 2020). In addition, individuals with high alexithymia show impaired emotion monitoring and perception, deficient auditory emotion processing, and reduced affective reactivity (Pollatos & Gramann, 2012; Rosenberg et al., 2020; Wang, Chen, et al., 2021; Wang, Goerlich, Luo, et al., 2022; Wang, Goerlich, Xu, et al., 2022). Inspired by the links of self-awareness and emotion processing with alexithymia, it is possible that genetic predispositions to self-awareness and emotion processing contribute to the development of alexithymia.
Oxytocin plays important roles in self-awareness and emotion processing. It has been shown that intranasal oxytocin administration, a method of testing pharmacological effects of exogenous oxytocin, can facilitate emotion regulation (Koch et al., 2019; Xin et al., 2020) and the differentiation of self from others (Pfundmair et al., 2018; Zhao et al., 2020), and can also increase one's willingness to verbally share painful emotions (Lane et al., 2013) and reduce the accuracy of interoception and the link between interoceptive and exteroceptive cues (Betka et al., 2018; Quattrocki & Friston, 2014). Moreover, the OXTR polymorphisms, such as rs53576 and rs1042778, are associated with emotion suppression and emotional experiences (Isgett et al., 2016; Kim et al., 2011). Additionally, strong evidence shows that brain regions with highly expressing oxytocin receptors partly overlap with the brain structures and neural correlates of alexithymia (Boccia et al., 2013; Rokicki et al., 2022; van der Velde et al., 2013; Xu et al., 2018). Based on these findings, we investigated whether the function of oxytocin, measured as the OXTR polymorphisms, is associated with the characteristics of alexithymia.
The rs53576 and rs1042778 of the OXTR regulate the level of plasma oxytocin, the expression of oxytocin receptors (Akdemir et al., 2020; Feldman et al., 2012; Rybicka et al., 2021), and the interactions between oxytocin and dopamine systems (Chang et al., 2014). Besides their links with social cognition and psychiatric disorders, these polymorphisms are also related to self-related cognitive and emotional processes. For example, the G allele of the OXTR rs53576 is related to higher levels of self-compassionate mindfulness and psychological resources than the A/A (Saphire-Bernstein et al., 2011; Wang et al., 2019). Notably, studies have attempted to build the links of these polymorphisms with alexithymia. One study found that a haplotype including the OXTR rs53576 was not related to alexithymia in patients with obsessive-compulsive disorder (Koh et al., 2015), while another study found that the GG genotype was related to higher risk of alexithymia among heathy individuals with a history of insecure attachment (Schneider-Hassloff et al., 2016). These findings suggest that the OXTR polymorphisms may be associated with alexithymia and that the quality of parental bond may modulate these associations.
Parental bonding (i.e., the emotional and behavioral styles of parents interacting with children) impacts children's emotions and behaviors (Tani et al., 2018). Research has consistently shown that higher parental care, characterized by warmth, responsiveness, and sensitivity to a child's needs, is associated with less severity of alexithymia in offspring. In contrast, higher parental overprotection, characterized by excessive concern and a lack of trust, is associated with more severity of alexithymia (Fukunishi et al., 1997; Thorberg et al., 2011). However, a recent study showed that neither parental care nor overprotection could predict alexithymia (Romeo et al., 2020). Given that the OXTR polymorphisms can modulate the relationships between parental bonding and behavioral outcomes (Cataldo et al., 2021; Truzzi et al., 2018), the relationship between parental bonding and alexithymia may be moderated by the OXTR polymorphisms.
In summary, this study examined the associations between the OXTR polymorphisms and alexithymia and tested whether parental bonding moderates the potential associations. To understand the genetic–environmental interplay, we considered the interactions with three theoretical perspectives: - diathesis–stress model, - vantage sensitivity model, and -differential susceptibility model (Roisman et al., 2012). The diathesis–stress model posits that individuals carrying a risk allele (an allele of a gene that is associated with an increased risk of a particular trait) are vulnerable to aversive environments. The vantage sensitivity model suggests that individuals with a susceptible allele (an allele of a gene that is associated with increased sensitivity to favorable environments) may exhibit more adaptive responses to favorable environments. In addition, the differential susceptibility model suggests that individuals with a plastic allele exhibit more adaptive responses to favorable environments and more maladaptive responses to unfavorable environments (Roisman et al., 2012). Due to limited evidence, we were unable to provide specific hypotheses on which of these models may apply in this study.
Participants and methods
Participants
We conducted a priori power analysis to determine a required sample size for detecting the genetic–environmental effect. The analysis with the G*Power 3.1 indicated that a sample of 1,053 samples would achieve a power of 90% (1−β = 0.90) at a significance level of 0.05 (two-tailed α = 0.05) when a genetic–environmental interplay has a large expected effect size (r2 = 0.01; Border & Keller, 2017; Duncan & Keller, 2011). Considering the uncertainty in expected effect size, we set the sample size at 120% of the estimation. Finally, we invited 1,205 college students (827 females, mean age = 21.55 ± 2.788 years). Each participant received ¥10 for participation. The study was conducted in accordance with the Declaration of Helsinki, and was approved by the ethics committee of our university.
Alexithymia assessment
A Chinese version of the 20-itemed Toronto Alexithymia Scale was used to assess alexithymia (Bagby et al., 1994; Tam & Wong, 2012). This scale consists of three subscales: difficulty in identifying feelings (DIF), difficulty in describing feelings (DDF), and externally orientated thinking (EOT). The DIF taps individuals’ difficulty in distinguishing their emotions from physical sensations of emotional arousal. The DDF measures individuals’ difficulty in verbalizing their emotions. The EOT assesses individuals’ preference for describing external events rather than their internal emotional experiences. Each item was rated on a 5-point Likert scale, with “1” = strongly disagree and “5” = strongly agree. The Cronbach's alphas for the DIF, DDF, EOT, and total scale were 0.83, 0.78, 0.69, and 0.83, respectively.
Parental bonding assessment
A Chinese version of the 25-itemed Parental Bonding Instrument (PBI) was used to assess parental styles (Parker et al., 1979; Yang et al., 2009). This instrument assesses individuals’ perceptions on parents’ care and overprotection during their first 16 years. This scale consists of two subscales. The care subscale measures the extent to which parents accepted and supported a child, and the overprotection subscale measures the extent to which parents controlled and overly concerned about a child. For these items, participants rated the extent to which their mother/father exhibited these attitudes and behaviors (“0” = very unlike, “3” = very like). The PBI was administered separately for mother and father, and the care and overprotection scores for each parent were combined to create a single index of parental care and a single index of parental overprotection. The Cronbach's alphas for the care and overprotection subscales were 0.89 and 0.78, respectively.
Genotyping
Genomic DNA was extracted from 3–5 pieces of hair of each participant. Primers of 5′−ATCACTGGGTCACTCAA−3′ and 5′−AACATCTGTCAGGAGGT−3′ were designed for the OXTR rs53576. A 231 bp PCR fragment was produced with the procedures as described in a previous study (Gong et al., 2017). The PCR fragment was incubated with restriction enzyme BamHI at 37°C. In addition, the primers of 5′−TCCCAGAATGAAGAAGTAA−3′ and 5′−GGTGATGGCGTATGTTT−3′ were designed for the OXTR rs1042778. A 253 PCR product was incubated with restriction enzyme Van91I at 37°C. In this sample, 22 individuals failed in genotyping at the OXTR rs53576, and 11 individuals failed in genotyping at the OXTR rs1042778. Furthermore, we observed no deviation from the Hardy–Weinberg equilibrium at the OXTR rs53576 (GG = 102, AG = 538, AA = 543; χ2 = 3.756, p = .053), but a significant deviation at the OXTR rs1042778 (GG = 1001, GT = 177, TT = 16; χ2 = 6.17, p = .013).
Statistical analysis
We examined the associations between the OXTR polymorphisms and alexithymia with two statistical approaches (empirical regression analysis and Bayesian linear regression), which could partly balance the proportions of false-positive and false-negative test results (Jong et al., 2019; Qian et al., 2020; Wilcox, 1977). In empirical regression, we treated the genotype as a predictor and controlled sex and age as covariates by using SPSS software. In Bayesian linear regression, we included sex and age as nuisance predictors in null model and included the genotype as a predictor in alternative mode by using JASP in Version 0.16.1. Similarly, the interactions between the OXTR polymorphisms and parental bonding in alexithymia were tested both with a three-step hierarchy regression and a Bayesian linear regression. In three-step hierarchy regression, we included standardized dependent variables (e.g., parental care), standardized independent variables (e.g., the DDF), a mediator M (i.e., genotype), and an interactive effect. The region of significance (RoS) was examined with Roisman's suggestion (2012). In Bayesian linear regression, we included sex, age, genotype, and parental bonding in null model and included these variables and the interaction between genotype and parental bonding in alternative model. The Bayes factor (BF10) provides evidence for the likelihood in favor of alternative hypothesis compared to the null hypothesis.
Results
Relationship between parental bonding and characteristics of alexithymia
We found that the score on parental care was negatively correlated with the DIF, DDF, and total scale, while the score on parental overprotection was positively related to all subscales and the total scale (Table 1). Additionally, we found that the age variable was negatively correlated with the characteristics of alexithymia, and that males scored higher on the DIF, DDF, and total scale than females (ts ≤ −2.146, ps ≤ 0.031). These findings indicated that parental bond and demographic factors, such as age and gender, were related to the characteristics of alexithymia.
Correlations between characteristics of alexithymia and parental bonding.

Note: DIF—difficulty in identifying feelings; DDF—difficulty in describing feelings; EOT—externally orientated thinking; Total—total score on the TAS-20; PC—parental care; PO—parental overprotection; * p < .05, ** p < .01 (two-tailed).
Relationships between the OXTR polymorphisms and alexithymia
Linear regression analysis showed that the OXTR rs53576 was significantly associated with the characteristics of alexithymia (DIF: R² = 0.005, β = −0.068, t = −2.353, p = .019; DDF: R² = 0.004, β = −0.067, t = −2.293, p = .022; total scale: R² = 0.005, β = −0.072, t = −2.476, p = .013). Specifically, the AG/GG were related to higher scores than the AA genotype (DIF: AA vs. AG/GG = 2.54 ± 0.65 vs. 2.63 ± 0.65, t = 2.35, p = .02; DDF: AA vs. AG/GG = 2.65 ± 0.66 vs. 2.74 ± 0.59. t = 2.27, p = .02; total scale: AA vs. AG/GG = 2.60 ± 0.46 vs. 2.67 ± 0.44, t = 2.476, p = .013; Figure 1). To control for the effects of demographic variables (Table 1), we further tested associations by controlling for these variables (Table 2). The hierarchical regression still showed significant associations between the OXTR rs53576 and the measures (DIF: △R² = 0.005, β = −0.071, t = −2.456, p = .014; DDF: △R² = 0.005, β = −0.070, t = −2.426, p = .015; total scale: △R² = 0.006, β = −0.069, t = 0.026, p = .008). Additionally, Bayesian linear regression indicated that the BF10 for the effects of OXTR rs53576 on the DIF, DDF, and total scale were 2.320, 2.097, and 3.595, respectively, which provided anecdotal to moderate evidence for the existing relationships between this polymorphism and the characteristics of alexithymia.
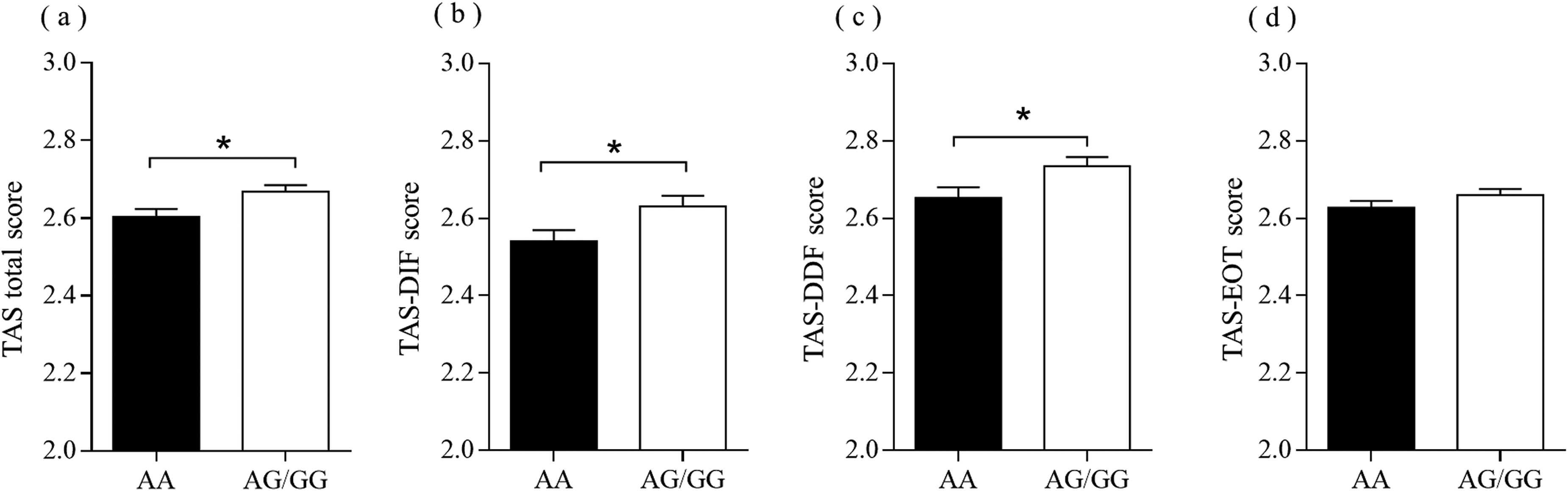
The scores of characteristics of alexithymia between the AA and AG/GG genotypes of OXTR rs53576. (a) total scale: AA vs. AG/GG = 2.603 ± 0.464 vs. 2.669 ± 0.440, t = 2.476, p = .013; (b) DIF: AA vs. AG/GG = 2.541 ± 0.653 vs. 2.631 ± 0.650, t = 2.353, p = .019; (c) Score DDF: AA vs. AG/GG = 2.652 ± 0.655 vs. 2.735 ± 0.592, t = 2.274, p = .023; (d) EOT: AA vs. AG/GG = 2.627 ± 0.408 vs. 2.660 ± 0.413, t = 1.363, p = .173); *p < 0.05.
Relationships between the OXTR polymorphisms and characteristics of alexithymia.

Note: Bold font indicates a significant statistic; DIF—difficulty in identifying feelings; DDF—difficulty in describing feelings; EOT—externally orientated thinking; Total—total score on the TAS-20; PC—parental care; PO—parental overprotection.
In contrast, we did not find any associations between the OXTR rs1042778 and the subscales and total scale, either before or after controlling for demographic variables (Table 2). Moreover, Bayesian linear regression indicated the BF10 only reached 0.249 to 0.341(Table S2 in Supplementary materials), which provides moderate evidence for lack of significant relationships between this polymorphism and alexithymia.
Interactions between the OXTR and parental bonding in alexithymia
We conducted additional analysis to examine the potential gene–environment correlation between the OXTR polymorphisms and parental bonding and found that neither of the polymorphisms was associated with parental care (rs53576: R² < 0.001, β = −0.008, t = −0.287, p = .774; rs1042778: R² = 0.001, β = −0.030, t = −1.043, p = .297) and overprotection (rs53576: R² < 0.001, β = 0.028, t = 0.995, p = .340; rs1042778: R² < 0.001, β = 0.012, t = 0.429, p = .668). Thus, the gene–environment correlation would not increase spurious bias in subsequent analysis.
We conducted a three-step hierarchy regression analysis to examine the potential interactions between the OXTR polymorphisms and parental bonding (Table 3). This analysis showed that the OXTR rs53576 did not interact with either parental care (DIF: β = −0.037, t = −1.282, p = .200; DDF: β = −0.042, t = −1.487, p = .137; EOT: β = −0.057, t = −2.010, p = .045; total score: β = −0.054, t = −1.907, p = .057) or parental overprotection on the measures of alexithymia (DIF: β = −0.037, t = −1.287, p = .198; DDF: β = −0.013, t = −0.471, p = .638, EOT: β = −0.011, t = −0.394, p = .693; total scale: β = −0.027, t = −0.969, p = .333). Moreover, Bayesian linear regression indicated that the BF10 for the OXTR rs53576 interacting with parental care and overprotection reached 0.162 to 0.932 (Table S2 in Supplementary materials). These findings suggested that the OXTR rs53576 may not significantly interact with parental bonding in characteristics of alexithymia.
Hierarchy regressions on the interactions between the OXTR rs53576 and parental bonding in characteristics of alexithymia.

Note: DIF—difficulty in identifying feelings; DDF—difficulty in describing feelings; EOT—externally orientated thinking; PC—parental care; PO—parental overprotection.
We examined the potential interactions between the OXTR rs1042778 and parental bonding (Table 4) and found that this polymorphism significantly interacted with parental care in the DIF (△R² = 0.005, β = 0.068, t = 2.394, p = .017) and total score (△R² = 0.005, β = 0.070, t = 2.498, p = .013). Furthermore, Bayesian linear regression revealed that the BF10 for the interaction between this polymorphism with parental care on the DID and total scale reached 2.669 and 3.162, respectively (Table S3 in Supplementary materials), which provided anecdotal to moderate evidence for the existing interactions. To further understand the nature of these interactions, we conducted simple slope analyses and found that parental care had a greater effect on the DIF and total scale in individuals with the GG genotype (simple slope of the DIF = −0.07, p = .003; simple slope of total scale: = −0.06, p < .001) than those with the TT/TG genotype (simple slope of DIF = 0.06, p = .249; simple slope of total score = 0.03, p = .330). We also conducted a RoS test, which indicated that the interactions between the OXTR rs1042778 and parental care on the DIF and total scale were significant at higher level of parental care (Figure 1: X refers to Z score of parental care within the range of −2 to + 2 SD; DIF: X > 0.279, total scale: X > 0.135), with PoIs of 0.76 and 0.79 for the DIF and total scale. These analyses suggested that this interaction may be consistent with the vantage sensitivity model, given that the crossover points were at the low end of parental care and the significant different lines were at the high end of parental care (Figure 2).

(a) Interaction between the OXTR rs1042778 and parental care in total scale of TAS-20. The GG genotype is related to a lower score than the TT/TG genotypes in individuals with higher parental care of X > 0.135 (X refers to the Z value of parental care within the bounds of −2 to +2 SD). (b) Interaction between the polymorphism and parental care in the score on the DIF. The GG genotype is related to a lower score on the DIF than the TT/TG genotypes in individuals with higher parental care of X > 0.279.
Hierarchy regressions on the interactions between the OXTR rs1042778 and parental bonding in characteristics of alexithymia.
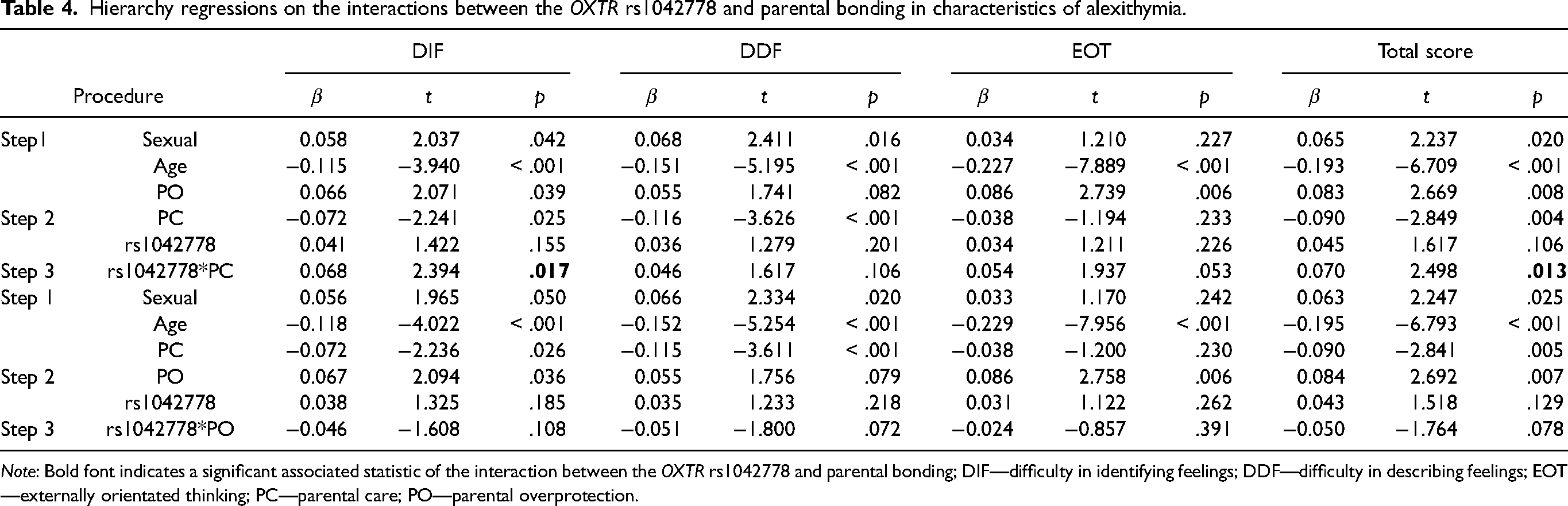
Note: Bold font indicates a significant associated statistic of the interaction between the OXTR rs1042778 and parental bonding; DIF—difficulty in identifying feelings; DDF—difficulty in describing feelings; EOT—externally orientated thinking; PC—parental care; PO—parental overprotection.
On the other hand, we did not find significant interactions between the OXTR rs1042778 and overprotection in the measures of alexithymia (DIF: ΔR² = 0.002, β = −0.046, t = −1.608, p = .108; DDF: ΔR² = 0.003, β = −0.051, t = −1.800, p = .072; EOT: ΔR² = 0.001, β = −0.024, t = −0.857, p = .391; total scale: ΔR² = 0.002, β = −0.050, t = −1.764, p = .078). Furthermore, Bayesian linear regression provided moderate evidence for lack of significant interactions between this polymorphism and parental overprotection (all BF10 ≤ 0.692; Table S3 in Supplementary materials).
Discussion
This study indicated that parental bonding is significantly related to the characteristics of alexithymia. Specifically, parental care is negatively correlated with the difficulties in identifying feeling and describing feelings, while parental overprotection is positively correlated with the characteristics. These results are consistent with a previous meta-analysis indicating a strong relationship between maternal care and alexithymia and a moderate relationship between parental overprotection and alexithymia (Thorberg et al., 2011). These findings suggest that a warm, nurturing, and emotional parental style is a protective factor against the development of alexithymia, while overprotective parenting may be a risk factor. These findings highlight the importance of caregivers adopting appropriate approaches to avoid the abnormal development of offsprings’ self-related emotion processing.
This study found that the OXTR rs53576 is significantly associated with characteristics of alexithymia. Specifically, the GG and AG genotypes are related to higher difficulties of identifying feelings and describing feelings than the AA genotype. These findings are consistent with previous research that the GG genotype of OXTR rs53576 is associated with increased risks of emotion deregulation, trauma-related psychopathology, and disorganized attachment (Bradley et al., 2011; Cao et al., 2020; Costa et al., 2009). Therefore, the association between the G allele and alexithymia may contribute to the allele-dependent effects of the OXTR rs53576 on psychiatric problems. Maybe these results provide new insights into the role of alexithymia in the relationship between the OXTR rs53576 and psychiatric problems.
Inconsistent with the findings that the G allele of OXTR rs53576 is associated with higher sensitivity to other-oriented emotional processes, such as identifying pain in others’ faces and reading minds in others’ eyes (Rodrigues et al., 2009), we found that the G allele is related to weaker abilities in processing self-oriented emotions than the AA genotype. These findings imply that the OXTR rs53576 influences other-oriented and self-oriented emotion processing in opposite effects, possibly due to the roles of oxytocin in shifting attention from the processing of interceptive stimuli to external social cues (Yao et al., 2018), reducing interoceptive accuracy (Betka et al., 2018) and increasing attention allocation to social cues at the cost of reducing top-down control attention (Xu et al., 2019). Additionally, the associations between the OXTR rs53576 and behavioral outcomes are shaped by parental bonding (Cataldo et al., 2021; Truzzi et al., 2018). In contrast, this study showed that the association between this polymorphism and alexithymia is not modulated by parental binding. These findings imply the importance of the OXTR rs53576 in alexithymia. Notably, the GG homozygote than the AA homozygotes is related to higher emotional suppression, smaller hippocampal volumes, and weaker functional connectivity between the right hippocampus and other brain areas in Chinese and Koreans (Kim et al., 2012; Luo et al., 2020), whereas the reversed patterns are demonstrated in Americans (Kim et al., 2011; Tost et al., 2010). Due to such racial differences, the findings should be replicated in Western populations.
This study showed that the association between the OXTR rs1042778 and alexithymia is modulated by a history of childhood parental care. The GG genotype, which is associated with high levels of OXTR transcription and plasma oxytocin (Feldman et al., 2012; Ribeiro et al., 2018), is related to fewer characteristics of alexithymia only in individuals with high parental care. In contrast, there was no significant relationship between this polymorphism and alexithymia among individuals with low parental care. Considering that individuals with the GG benefited more from the positive effects of parental care than those with the TT or TG genotype, the gene–environment interplay should be compatible with a vantage sensitivity model. These findings suggest that the OXTR rs1042778 acts as an endogenous factor that specifically modulates the effects of positive parental experiences on alexithymia.
As compared with previous studies that only addressed the associations between candidate genes and the characteristics of alexithymia (Gong et al., 2014; Kano et al., 2012; Li et al., 2020), this study took into account the effects of parental bonding. Given the vantage sensitivity of the GG of OXTR rs1042778 to positive influences of parental bonding and the advantage of those with the GG genotype in benefiting from kindness training (Isgett et al., 2016), the parental interventional effect on alexithymia would be pronounced in children with the GG genotype. However, these findings are still preliminary, and more research is needed to understand the roles of OXTR polymorphisms in the relationship between parental bonding and alexithymia.
This study has some limitations. First, this study measured alexithymia with the 20-itemed TAS, which has high stability and reliability in both clinical and nonclinical populations. This scale can only assess the cognitive components of alexithymia, while the affective factors such as emotionalizing and fantasizing were not included (Sifneos, 1973; Vorst & Bermond, 2001). To provide comprehensive understanding on the associations between the OXTR and psychometric properties of alexithymia, the Chinese version of the Bermond–Vorst Alexithymia Questionnaire should be used (Wang, Chen, et al., 2021; Wang, Wang, et al., 2021). Second, we tested the associations between genetic polymorphisms and alexithymia with two statistical approaches. However, the issues of multiple tests were not well considered. Considering that the Bayes factors only provided anecdotal evidence, which suggested the links between the OXTR polymorphisms were weak, we should be cautious in explaining the findings. Finally, the OXTR rs1042778 deviated from the Hardy–Weinberg equilibrium, which affected the reliability of the findings.
Conclusion
This study demonstrates that the OXTR rs53576 is associated with alexithymia, and that the association between the OXTR rs1042778 and alexithymia is moderated by parental care. This study provides a novel explanation for the roles of oxytocin in alexithymia.
Supplemental Material
sj-docx-1-pac-10.1177_18344909231154928 - Supplemental material for OXTR polymorphisms and parental bonding modulate alexithymia: The main effects and interaction
Supplemental material, sj-docx-1-pac-10.1177_18344909231154928 for OXTR polymorphisms and parental bonding modulate alexithymia: The main effects and interaction by Wenping Zhao, Yuting Yang, Wenxuan Guo and Pingyuan Gong in Journal of Pacific Rim Psychology
Footnotes
Acknowledgements
We thank Mr. Guomin Shen, Shoumin Xi, and Miss Yong Huang for their assistance in data collection. Performed the experiment: Wenping Zhao, Yuting Yang, Pingyuan Gong. Analyzed the data: Pingyuan Gong and Wenxuan Guo. Wrote the paper: Pingyuan Gong. Designed the study: Pingyuan Gong. This study was supported by the National Social Science Foundation of China grant (20BSH165) awarded to Pingyuan Gong.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the National Social Science Foundation of China (grant number 20BSH165).
Open research data
We have uploaded our data on the Figshare (10.6084/m9.figshare.21802662.v1).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
