Abstract
Introduction
Clinical registries are continuous data collection programs that seek to prospectively enrol all individuals with a particular diagnosis, or receiving a particular procedure or service, with the aim of understanding the natural history, trends in clinical practice and patient outcomes (Gliklich et al., 2020). Clinical registries may also be established primarily for research purposes, or to monitor high-risk treatments and provide outcome data to regulatory agencies such as the Therapeutic Goods Administration (Australian Government Department of Health, 2021). Such data may be used to inform national regulatory approval and subsidisation decisions (Ahern et al., 2022). Clinical quality registries (CQRs) are a type of clinical registry that has as its primary aim to monitor and report on processes and outcomes of clinical care for quality assurance and/or quality improvement purposes. When sufficiently mature, CQRs provide benchmarked reports to participating clinicians and/or health care organisations to support shared clinical decision-making and drive clinical improvement (Australian Commission on Safety and Quality in Health Care (ACSQHC), 2024). By monitoring health system outcomes, they can also inform clinical service planning and health care policy. CQRs are an integrated component of a “high-functioning, outcomes-focused” health system (Ahern, 2023: 984).
The Australian healthcare system offers universal public coverage for health care via national and state government funding. This coexists with a substantial private health insurance sector that provides a range of hospital-based and community services, with some private services also requiring consumer co-payment. As of 2023, ~55% of Australians had private health cover (Private Healthcare Australia, 2023). One of the strengths of CQRs in Australia is that they monitor care and outcomes across both public and private systems.
In May 2023, the Australian Department of Health and Aged Care established its first National Clinical Quality Registry Program, contributing $40 million over 4 years to support existing and develop new CQRs (Australian Government Department of Health and Aged Care, 2025), in line with the National Strategy for Clinical Quality Registries and Virtual Registries 2020–2030 (Australian Government, 2020). The Australian government also collaborates with activities aligned to the national registry strategy including a Clinical Registries Special Interest Group (SIG). This group comprises representatives (academics, clinicians, researchers) from nearly 100 Australian registries and is led by Monash University, the South Australian Health and Medical Research Institute (SAHMRI) Registry Centre and the Australian Clinical Trials Alliance (ACTA, 2023).
The ACSQHC (n.d.) lists more than 100 self-identifying clinical registries in its Register of Clinical Registries. This register publishes individual registry name, condition, website, contact, year established, description, lead organisation, ethics approval site, reporting process, patient-reported outcome measures (PROMs) or patient-reported experience measures (PREMs) and participating sites. However, important additional information relating to registry functions, activities and outcomes is not collected. The clinical registries listed by the ACSQHC (2025) have been self-nominated and have not been independently approved nor endorsed as CQRs. As such, the broader term “clinical registries,” will be used to describe the registries surveyed in this study.
The aims of this study were to provide contemporary national data regarding the characteristics, activities, operational costs and outputs of CQRs in Australia. The findings from this study intend to also be used to inform a future study for the development of a maturity framework for CQRs.
Method
Study design
The study design was a cross-sectional survey of Australian clinical registries.
Survey development
A rapid review of Australian and international literature regarding clinical registry characteristics, attributes, maturity models and frameworks was undertaken to inform the development of the survey. Existing registry frameworks that were identified included Blumenthal’s (2019) Registry Maturational Framework, and a registry framework specific for device registries (Sedrakyan et al., 2022). A number of previous surveys were also identified that described specific clinical registry characteristics or outputs for Australian registries, including those by Evans et al. (2011) and Hoque et al. (2017, 2018).
The Organising Committee of the 2023 Australian Registry Annual Scientific Meeting (ASM) led the development and supported the implementation of the Australian Clinical Registry Survey during the period from August 2023 to October 2023. The survey was designed to separately describe registry characteristics (inherent qualities or processes) and registry outputs/outcomes (registry products/end goals), with the future aim of determining registry characteristics associated with high levels of outputs/outcomes.
Key items for the proposed survey were identified from the literature review and reviewed by the project team to ensure that items and response options were relevant for the contemporary Australian context. Initially, 33 registry characteristics and 32 registry outputs were listed (65 questions), which were refined following feedback from the Organising Committee to 20 characteristics and 12 outputs (32 questions), to ensure that the survey could feasibly be completed within 5–8 minutes. Item responses for registry characteristics included both single and multiple response questions, with responses to registry outputs/outcomes being binary (yes/no) only. The survey was piloted by Organising Committee members prior to finalisation. Piloting confirmed the expected duration of time to complete the survey and highlighted a few opportunities to make minor revisions to the wording of questions. Committee members were asked to complete the survey again upon finalisation of the survey questionnaire.
Final clinical registry characteristics and outputs/outcomes were categorised into eight domains each (Box 1). The survey was online only and implemented using the Qualtrics platform.
Registry characteristics and output/outcome domains in the Registry Survey.

Survey population
The survey aimed to capture all currently operating clinical registries in Australia. Eligibility criteria included clinical registries that were associated with Monash University, SAHMRI Registry Centre and the ACTA Clinical Registry SIG (~90 clinical registries in 2023), as well as clinical registries on the ACSQHC’s register that were not identified via these sources. Eligible registries were identified as (1) clinical registries that, (2) collected data from patients in Australia (this could be as part of an international registry) and (3) were multisite (i.e. not single site) activities.
Distribution
The survey was distributed via existing communication channels (email distribution lists) including to ACTA Registry SIG members, Monash University and SAHMRI Registry Centre internal contact lists.
An initial email including a direct link to the survey was sent, with two reminders over a period of 3 weeks. Preliminary findings were presented at the 2023 Clinical Registries ASM, Melbourne, 20 October 2023. Following the meeting, the survey was also sent to any new clinical registries on the ACSQHC’s list that were not identified previously to obtain further responses. In total, 107 identified registries were contacted regarding the survey between 21 September 2023 and 22 January 2024.
The email invitation recommended that the survey be completed by a senior member of each registry. It also asked for the name of the registry to be included so that any duplicate responses could be identified. Names of personnel completing the survey were not collected. Participation was entirely voluntary.
Data management
Survey data were extracted from Qualtrics via Excel and stored on a secure drive at Monash University. After removing duplicate responses from the same registry, the dataset was de-identified for analysis. Data were analysed using R software (RStudio: integrated development environment for R, Posit Software, PBC, Boston, MA, USA) by an experienced data analyst. Descriptive statistics were generated and reported in frequencies and percentages.
Ethics approval
The project was approved by Monash University Low Risk Ethics Committee (approval number 40056). Survey methods and results were reported using Improving the Quality of Web Surveys: The Checklist for Reporting Results of Internet E-Surveys (CHERRIES; Eysenbach, 2004).
Results
Response rates
Seventy-five responses were received of which eight were removed for the following reasons: one duplicate, two unnamed registries (to avoid duplication), four single-site activities and one module of another registry, all of which did not meet the definition of a standalone registry. Three further surveys were incomplete (discontinued after a few questions) and removed from analysis, resulting a total of 64 completed surveys, providing a registry response rate of 60% (64/107). Completeness of each of the 32 individual questions was very high. Of the Registry Characteristic Questions, eight questions had one missing response, one question had two missing responses, and one question had three missing responses (noted as footnotes in the tables). Regarding the completeness of the Registry Output/Outcome questions, eight questions had one missing response.
Descriptive results are presented according to clinical registry characteristic and outcome domains, with the first survey question being Registry Name, followed by the following nineteen reported questions (Tables 1–5).
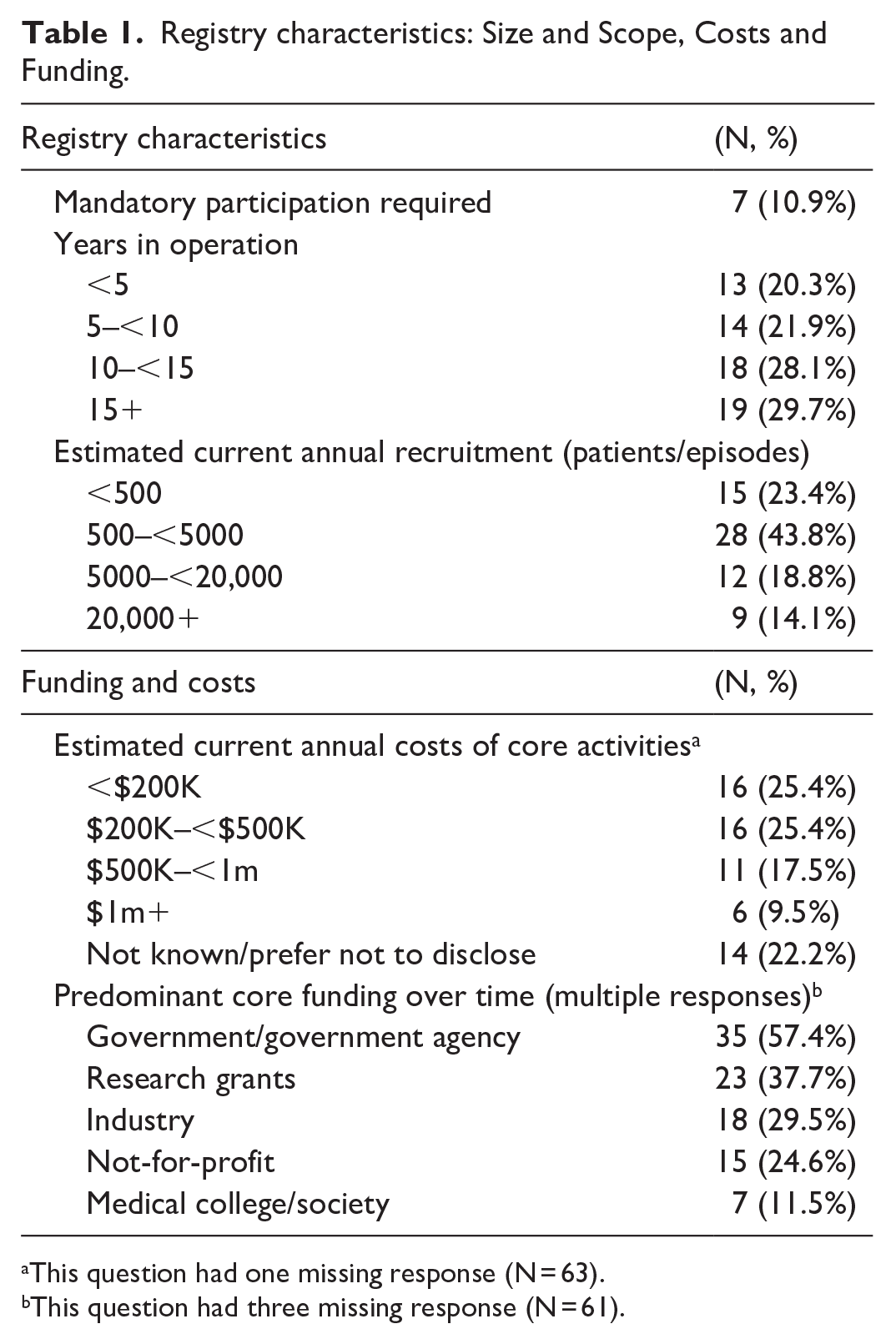
Registry characteristics: Size and Scope, Costs and Funding.
This question had one missing response (N = 63).
This question had three missing response (N = 61).
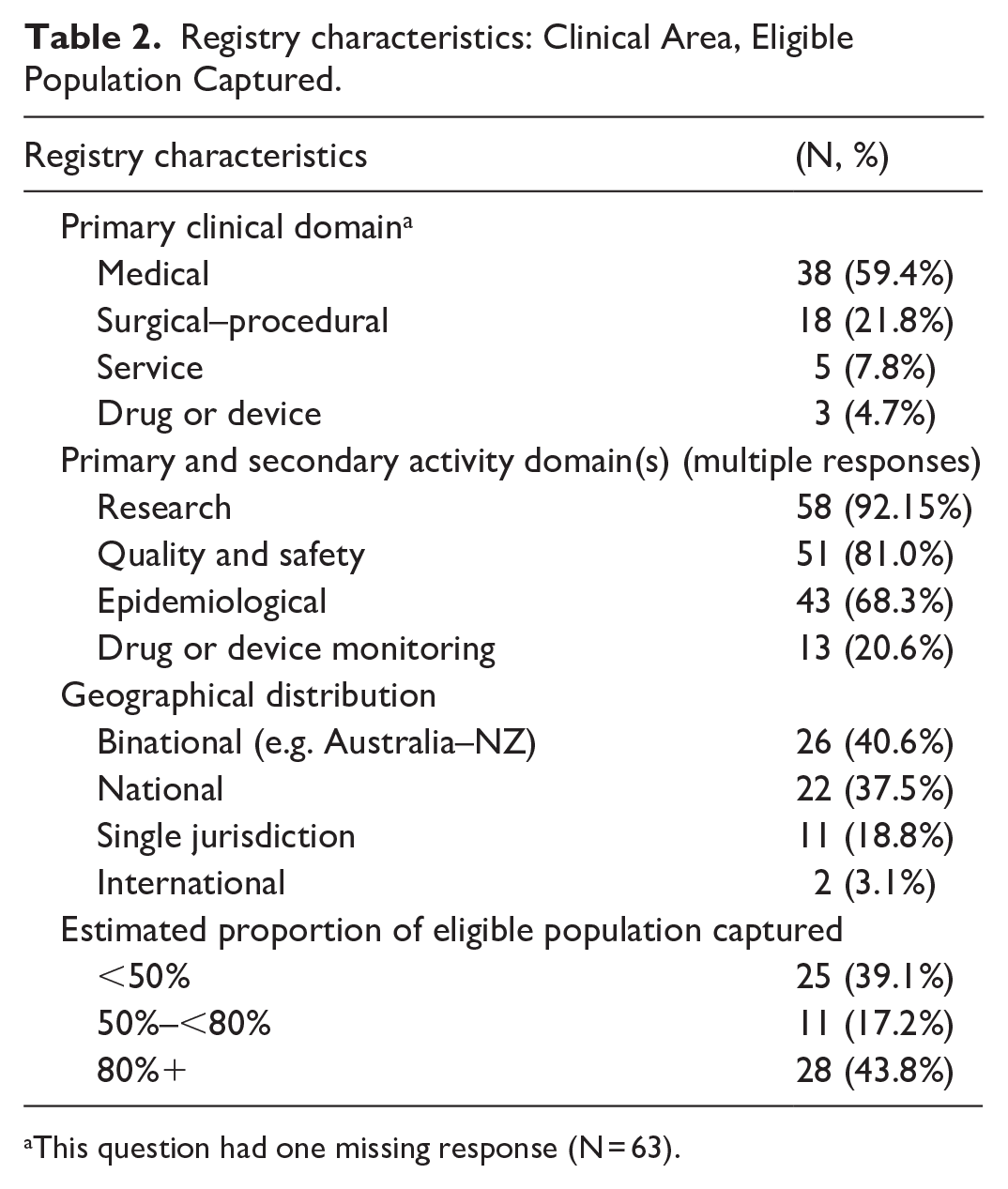
Registry characteristics: Clinical Area, Eligible Population Captured.
This question had one missing response (N = 63).
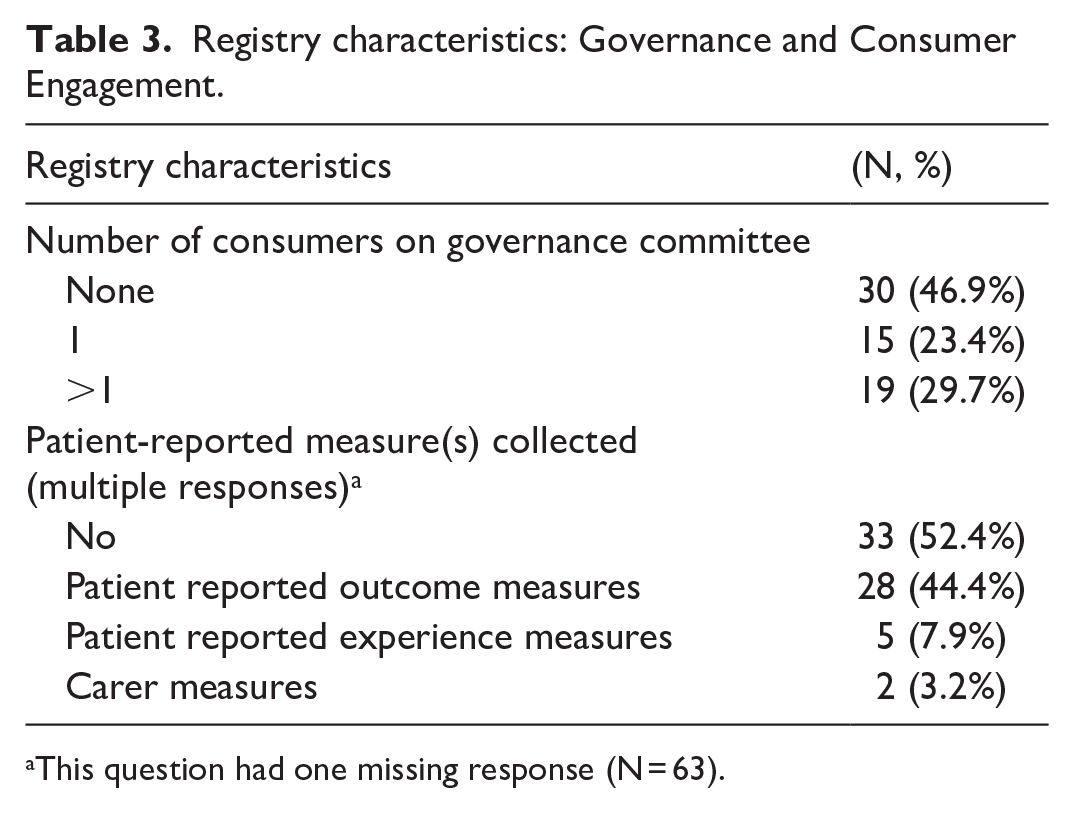
Registry characteristics: Governance and Consumer Engagement.
This question had one missing response (N = 63).
Registry characteristics: Data Collection and Access.

This question had one missing response (N = 63).
This question had one missing response (N = 63).
This question had one missing response (N = 63).
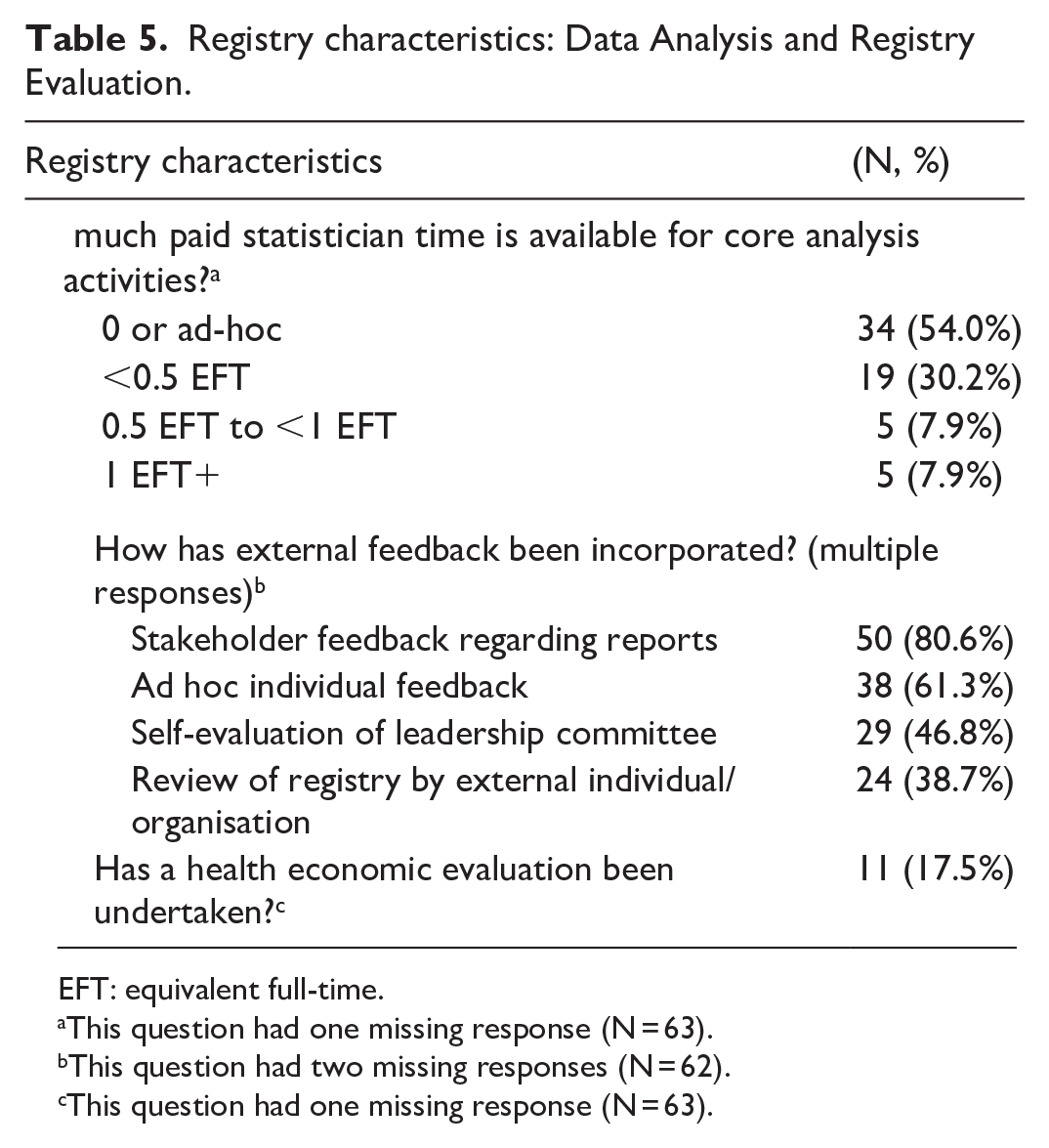
Registry characteristics: Data Analysis and Registry Evaluation.
EFT: equivalent full-time.
This question had one missing response (N = 63).
This question had two missing responses (N = 62).
This question had one missing response (N = 63).
Approximately 90% of registries relied on voluntary provider participation. Seven clinical registries (10.9%) had mandatory participation (e.g. were notifiable diseases or procedures). Of these, four were state-based (cancer, birth defects, ambulance), and three were national (transplant, in vitro fertilisation, other) registries. Fifty-eight percent (N = 37) of registries had been in operation for ⩾10 years. Seventy-seven percent (N = 49) of registries collected data in relation to at least 500 patients/episodes per year. The highest proportion of registries (44%, N = 28) collected 500–5000 patients/episodes per year, with the least common annual caseload being >20,000/year (14%; Table 1).
Annual running costs were fairly evenly spread among registries, with just over half operating for <$500,000/annum. Only six registries (10%) had annual running costs in excess of $1million/year, noting that over 20% of respondents chose to decline to respond to this question. Of those with more than $1million funding, additional descriptive analysis showed that they had all operated for at least 10 years, were national, binational or international, and half had annual case volumes of over 20,000 episodes/annum. Where multiple sources of registry funding were acquired, the highest proportion were from government/agency funds (57%, N = 35), followed by research grants (38%, N = 23), industry (30%, N = 30%) and not-for-profit organisations (25%, N = 15). Medical colleges/societies contributed funding to only 12% (N = 7) of registries. Approximately 60% of registries relied on a single source of funding, usually this was government or government agency funding (60%) or research funding (23%).
Nearly 60% of registries (N = 38) identified their primary clinical area as medical, 28% (N = 18) as surgical/procedural, 8% (N = 5) as clinical service (e.g. intensive care units) registries, and 5% (N = 3) as drug or device monitoring registries. Within these clinical areas, 92% (N = 58) reported undertaking research, 80% (N = 51) performed quality and safety activities and 68% (N = 43) reported information for epidemiological purposes; while 20% (N = 13) performed drug and device monitoring, even if this was not identified as their primary aim. Nearly 80% (N = 48) of registries were either national or bi-national (Australia and New Zealand). Forty-four percent of registries (N = 28) captured ⩾80% of their available population; while ~40% (N = 25) had a case ascertainment of <50%. Just over half (53%, N = 30) of surveyed registries had consumer representation on their main governance committee, with 23% (N = 15) having a single consumer, and 30% (N = 19) having more than one. Fewer than half (44%, N = 28) of the registries collected patient-reported measures, with 8% (N = 5) collecting PREMs and two registries (3%) collecting carer measures. Approximately two-thirds of surveyed registries collect identified patient data. Duration of follow-up data collection was variable, with 35% collecting outcomes beyond 2 years, and 38% collecting follow-up data from 30 days to 2 years.
Over 80% of registries reported relying on clinicians or their support staff to provide at least some of the data; 46% also utilise data collectors, and 33% incorporate data linkage into their core data collection. Core registry data are collected via multiple methods – the most common (70%) is via direct entry into the registry database; 64% of registries extract some data from electronic databases, and 56% collect some data from medical records. Thirty-five percent use paper forms. Fifty-five percent of registries provide real time data access to clinicians; however, one-third do not provide any direct access to clinicians.
Eighty-four percent of registries have <0.5 full-time equivalent (FTE) of statistician time for core registry activities (e.g. reporting); of these 54% have no funded time or ad hoc time only. A majority of registries had received stakeholder feedback regarding registry reports (81%) or had received ad hoc feedback (61%). Less commonly, registries undertook a governance self-evaluation (47%), or had undergone an external review (39%). Eighteen percent have had a health economic evaluation of their core activities. The 12 registry outcome results (yes/no responses only) are provided below (Table 6).
Registry outcomes (N = 64).

These questions had one missing response (N = 63).
In summary, of the 64 clinical registries: The vast majority provided data upon request (97%), and produced regular academic publications (83%). The majority provided annual reports (69%) and site benchmarked reports (64%). More than half supported quality improvement programs (60%), monitored adherence to clinical standards (58%), informed policy (52%) and were financially sustainable (52%). Less than half supported clinical trials (49%); supported shared patient–clinician decision-making (40%), were integrated with existing data frameworks (21%) or provided drug/device regulatory reporting (18%).
Discussion
This 2023 survey is the first since a 2018 study to comprehensively evaluate the key characteristics, activities and outcomes of clinical registries in Australia. Given the rapid evolution in the sector and ongoing development, updated information on the number and scope of clinical registry operations is important to understand current coverage, gaps and opportunities to monitor effective and efficient health care. These data provide insights into the strengths, challenges and opportunities for improvement across the Australian clinical registry sector.
Importantly, over 90% of registries that responded indicated they operate on a voluntary basis and require the participation of clinicians, hospital staff and patients to provide the data needed for registry outputs and outcomes. Often, this data collection is not specifically funded, particularly when data are entered by clinical staff. Nevertheless, more than 50% of registries had been in operation for at least 10 years, suggesting that the value proposition of many registries is sufficient to maintain significant clinician buy-in and stakeholder support. At a time of increasing implementation of hospital electronic medical records and clinical systems, this ongoing clinical support of registries also suggests that they remain a useful and reliable source of information regarding quality of care and outcomes in the hospital setting (Barbazza et al., 2021, McDonough, 2020). Nevertheless, limited registry funds could produce more high-quality outputs if hospital participation and data collection were mandatory and technologically enabled (Brown et al., 2022, Duplancic et al., 2019).
Nearly 80% of clinical registries are national or binational (associated with New Zealand), and recruit thousands of patients/episodes of clinical care annually. More than half of the registries surveyed have annual operating costs of <$500,000/annum, while those with greater funding tended to be longer-standing and recruit higher patient volumes. Most funding for clinical registry activities are spent on data acquisition, including obtaining the necessary ethics and hospital governance approval processes (Barnett et al., 2016), data management and data analysis activities for reporting.
Despite increasing interest in secondary use of registry data, only 16% of clinical registries have at least 0.5 FTE of statistician time allocated to core registry data analysis. Yet statistical expertise is fundamental for ensuring registry reports and publications are accurate, valid and provide reliable information to providers and stakeholders. Many stakeholders contribute to funding clinical registries. The largest funder is the public sector, including funding from both national and jurisdictional governments and government agencies, followed by not for profits including medical societies and colleges. Industry contributes funding to approximately one-third of clinical registries surveyed, and the value proposition to industry is clear when a registry can provide pre-or post-marketing information in relation to a novel device or drug. However, not all registry-related clinical conditions have a potential industry funder. Given the primary role of registries in monitoring clinical outcomes and health system performance, including in relation to surgical procedures predominantly performed in the private sector, there is a significant imperative for the private healthcare sector to contribute to core registry funding. This is a common funding model in Europe as used by the Dutch Institute of Clinical Auditing (Beck et al., 2020).
Most registries identified their primary clinical domain as medical, with only 5% identifying as drug or device-focused. Yet nearly 20% of registries reported undertaking post-market drug or device monitoring, indicating their broader scope and impact as clinical registries become more mature. Despite this, clinical registries in Australia are generally underutilised in supporting health technology assessment, and given the high volumes of granular data collected, there are further opportunities to embed clinical registry data in regulatory decision-making (Scott, 2017).
Eighty-one percent of registries reported that they undertook quality and safety monitoring activities, suggesting that the majority of clinical registries surveyed may meet the ACSQHC’s definition of being a CQR. Further, 92% of clinical registries stated that they undertook research activities. This may be in conjunction with quality and safety monitoring, but there are also likely to be a proportion of clinical registries that have research as their primary function. This aligns with the high proportion of registries that produce regular academic publications (82.5%).
Approximately two-thirds of registries collect identified patient data, enabling more accurate data management (e.g. reduced risk of duplicate data entries), collection of PROMs, follow-up data and data linkage with other datasets. However, collection of highly sensitive patient data has implications for registry operations, with legislation mandating data storage on highly secure infrastructure, increasing registry costs. Managing registries is an increasingly risky business that requires high levels of data governance and cybersecurity expertise. The gradual implementation of the Individual Health Identifier in health services may be useful in reducing the risk of breach of personal information held by registries and increase automated data exchange between clinical systems (Australian Government OAIC, 2025).
This survey also highlights the broad range of outputs of many Australian clinical registries. Ninety-seven percent have processes for making data available upon request, with this usually being via a formal registry application process. Over 80% of registries produce academic outputs, to support evidence-based practice in clinical care. The majority of registries provide annual (69%) and regular local hospital/provider reports (64%), with registry maturity being a factor in whether they produce site reports. Subanalysis shows that 61.5% of registries of <5 years of operation and 84.2% of registries of over 15 years of operation produce annual reports, while 38.5% of registries <5 years and 84.2% of registries over 15 years producing site reports. These reports comprise important comparative information regarding processes and outcomes of care. Many registries also reported that they support a range of quality, policy and planning activities at the local or system level. There is evidence of planning and review activities being undertaken among many clinical registries, with a majority of registries incorporating feedback regarding their activities into their future planning.
The volume of individual registry outputs largely relates to registry maturity, with less mature registries less commonly using registry data or infrastructure registry data to support patient–clinician decision-making, integration of data into government data frameworks or provision of drug/device reports to regulators. Approximately 50% of registries have sustainable funding. This suggests that many registries have nurtured meaningful stakeholder relationships and attract ongoing investment. However, a significant number of clinical registries have insecure funding, many continuing year by year as in-kind endeavours by passionate clinicians, researchers and not-for-profit organisations.
This 2023 survey shows growth and development of the registry sector since previous Australian surveys of clinical registries were undertaken. The most recent prior national survey of CQRs was undertaken by Hoque et al. (2018) in 2015–2016, with 34 participating clinical registries. At that time, half the surveyed registries had been operational for at least 10 years; this had increased to 58% by 2023. Funding sources for clinical registries have also broadened over the last 10 years. In the 2018 study, 70% of registries were publicly funded, compared to 57% in 2023. Previously, just over one-quarter of clinical registries received funding from research grants, compared with 38% in 2023, with other funding sources including industry and not for profits also increasing substantially. Notably, the production of site reports by registries has increased significantly during this time, from 44% in 2015–2016 to 64% in 2023.
This study identifies some areas of potential improvement for registries. Just over half of surveyed registries involved consumers in their governance structures, as recommended by the 2024 ACSQHC’s Framework (ACSQHC, 2024). Registries with existing partnerships with consumer support groups and not-for-profit organisations lend themselves more easily for potential involvement of consumers than others. However, as with other healthcare and quality improvement activities, the consumer perspective should be an important focus for all registries. The survey reported that less than half of clinical registries collected patient-reported measures. Collecting PROMs or PREMs can be challenging to registries that are lacking resources, although digital methods of communication can reduce the required costs. Approximately one-third of registries do not report real-time data to health care providers for use; however, nearly all registries provide data on request. This enables providers to access to their own registry data for clinical audit, academic or continuing professional development purposes and is a significant additional local benefit to participating clinicians and health services that should be implemented where possible.
Strengths and limitations
The strengths of this study included the development of a contemporary survey, which was informed from the literature and developed with input from multiple experienced registry staff members. The study was able to leverage existing professional networks and publicly available information to engage with the registry sector, as reflected by a high response rate, and largely complete individual survey responses. This study therefore provides contemporary data regarding the current characteristics and outputs of clinical registries in Australia, with the data generated from this survey now available for further registry-related research. Unfortunately, there is currently no independent, objective assessment process for clinical registries to be designated as CQRs in Australia. The ACSQHC’s register includes registries that were self-reported and may or may not meet objective criteria for CQRs. Given this, this survey noted that 80% of registries self-assessed as undertaking quality and safety activities. Without objective criteria, it is not possible for us to stratify the results by registries that were and were not CQRs. However, recently the ACSQHC has developed quality standards and a checklist for CQRs to support CQR self-assessment in line with the Australian Framework for CQRs (ACSQHC, 2024). Additionally, the voluntary nature of participation in the survey could cause selection bias towards registries with time and resourcing to contribute their responses. Further, there were three (4%) clinical registries that commenced but did not complete the survey, with those initial questions not being included in the survey results. The survey is intended to be undertaken annually (with minimal changes) to enhance understanding of the impact of support and funding provided by the Australian Government’s Registry Program (Australian Government Department of Health and Aged Care, 2025) on clinical registry outcomes.
Conclusion
A national survey of clinical registries in Australia highlights that the vast majority undertake both quality and safety monitoring as well as academic activities to achieve impact. Often using multiple funding sources, and with support by participating clinicians, hospitals and patients, clinical registries provide academic and quality improvement outputs, as well as providing their data or reports to stakeholders for a range of safety and performance monitoring and research purposes. Many clinical registries have been operating for at least a decade, highlighting their ongoing contribution to the Australian health system. While there are some opportunities for improvement, Australian clinical registries are well positioned to increase their impact with enhanced national support, profile and community awareness. The willingness of the registry sector to participate in registry-related research will allow further investigation into factors associated with registry maturity and overall contribution to Australian health care.
Footnotes
Acknowledgements
We thank all members of the organising committee of the 2023 ASM for their assistance in the development and distribution of the survey. Most importantly, we thank all those participating clinicians, hospital and clinic support staff and patients who contribute their time to the work of Australian clinical registries.
Accepted for publication May 10, 2025.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Funding for this study was obtained from the Australian Department of Health and Aged Care, which also supported the 2023 Registry Annual Scientific Meeting (ASM).
