Abstract
Keywords
Introduction
Almost one-third of perinatal deaths in Australia in 2017–2018 were due to congenital anomalies (CA) (Australian Government Australian Institute of Health and Welfare [AIHW], 2024a), making them nationally the most common cause of perinatal deaths (Australian Government AIHW, 2024b). CAs encompass “a wide range of atypical bodily structures or functions that are present at or before birth” (Australian Government AIHW, 2024b, para. 1). Due to their high prevalence (3–4% of all births, Australian Government AIHW, 2024a), potential lethality (Australian Government AIHW, 2024a) and contribution to chronic disability (Bower et al., 2010b; Thong, 2014), maintaining population health monitoring and surveillance of CAs is considered an essential feature of functional health systems (Stevens et al., 2018). The main purposes of congenital anomaly registers (CARs) are (i) to provide population-based incidence and prevalence figures; (ii) to monitor trends; (iii) to provide insight into potential aetiological factors and (iv) to inform health service planning (Thong, 2014).
Globally, the monitoring and surveillance of CAs at a national or regional level is well-established in middle- and high-income countries (Cardoso-Dos-Santos et al., 2021). Cardoso-Dos-Santos et al. (2020) identified six “international congenital anomaly surveillance collaboration networks” (p. 1), which encompassed 98 CA programmes across 58 countries. These covered all continents except Africa. Historically, two states in Australia, Victoria and Western Australia, separately provided CA data to the International Clearinghouse for Birth Defects Surveillance and Research (ICBDSR), a “voluntary nonprofit international organisation affiliated with the World Health Organization (WHO)” (International Clearinghouse for Birth Defects Surveillance and Research, 2025). National CA data have also been intermittently sent to the ICBDSR.
In today’s rapidly changing health landscape, CARs should be prioritised as essential surveillance tools to safeguard reproductive health. Emerging global threats – including rising temperatures (Haghighi et al., 2021), pandemics, environmental toxins (Spinder et al., 2019) and maternal risk factors (obesity, maternal age) (AIHW, 2024c; Goldsack et al., 2024; Hassold et al., 2007) – underscore the growing need for high-quality, timely CAR systems in Australia. A salutary example was the international Zika virus outbreak in 2015, which caused severe birth defects, including microcephaly and central nervous system anomalies. This epidemic exposed the lack of a functional national surveillance system in Australia. The Communicable Diseases Network Australia (CDNA) Zika Virus working group identified “critical gaps in data at the national level,” including a lack of national reporting on microcephaly since 2003 (CDNA Zika Virus working group meeting papers to Hui, February 2016). Furthermore, while SARS-CoV-2 was not found to be teratogenic, widespread vaccine hesitancy among pregnant women led to poorer perinatal outcomes (Hui et al., 2023). If we had a timely, responsive CAR system, it could have played an important role in monitoring drug and vaccine safety during the pandemic (Hui et al., 2023). With new infectious diseases such as Mpox emerging (Nachega et al., 2024) and the likelihood of further vector-borne infections due to climate change (Logiudice et al., 2025), establishing effective early warning systems is more critical than ever. Community expectations for proactive surveillance that identifies hazards and protects reproductive health further cement the argument for making CARs a high national priority. The states/territories invest significant resources in collecting data; if we fail to ensure that its quality meets rigorous research or policy standards, we squander valuable resources and miss critical opportunities for effective intervention.
In Australia, the establishment and maintenance of CARs is the responsibility of each state/territory. Therefore, CARs have evolved (or devolved) in an ad hoc way over time, with variable standardisation. Each state/territory, historically and currently, is required to provide CA data to a national register. National reporting of CA data has been spasmodic since its inception in the early 1980s, with a couple of attempts since then to deliver consistent and high-quality CA data (Birch et al., 2004). The first national CA publication,
Standardisation to promote “quality, efficiency and cost effectiveness” (Australian Government Department of Health, Disabililty and Ageing, 2020, p. 3) of data collection is a well-recognised requirement of national minimum datasets (Australian Government AIHW, 2025), clinical quality registries (Australian Government Department of Health Disability and Ageing, 2020) and other systems for the systematic reporting of health data (Ruseckaite et al., 2023). The lack of standardisation of CARs among the Australian jurisdictions is a major challenge to timely and accurate national reporting. Addressing the heterogeneity of data sources for the NCADC is the first hurdle for improving the quality of national reporting of CAs.
Given the historical context of CARs in Australia, to facilitate the monitoring and surveillance of potential CA clusters, it is timely to consider whether the CARs are capable of addressing such issues. It is also important to understand whether they are meeting their role of contributing to new evidence on population health through use in research and policy. Therefore, this paper aimed to (i) compare and contrast the main characteristics (scope, time-frame, data sources, data quality, nature of reporting, etc.) of CARs around Australia by state/territory; and (ii) identify peer-reviewed publications which have utilised CA data by state/territory over the last 40+ years (1980–2024) to determine their utilisation and value to the research and health policy sectors.
Method
Study design
A documentary analysis (Bowen, 2009; Dalglish et al., 2020) was undertaken on website information publicly available on each state/territory-based and national CAR in Australia. A modified scoping literature review of peer-reviewed research studies that had used CAR data between 1980 and 2024 was also conducted. It incorporated modified guidelines provided by the Joanna Briggs Institute for evidence synthesis (Peters et al., 2020) and the Preferred Reporting Items in Systematic Reviews and Meta-Analysis Extension for Scoping Reviews checklist (Tricco et al., 2019).
Data collection for documentary analysis
An extraction spreadsheet was designed in Excel to capture information for the documentary analysis on the general characteristics of the CARs. The fields included were based upon (i) items that were collected for reporting in the latest Australian Institute of Health and Welfare (AIHW)
Definitions
For this study, CARs were classified as either “established” or “non-established.” An “established CAR” referred to a dataset that was specifically created and defined with the primary purpose of reporting on CA incidence and prevalence, regardless of the data source. By contrast, reporting of CAs that were derived from datasets created for other purposes, such as administrative datasets used for health service activity monitoring, was not considered an established CAR.
Search strategy
To ascertain the peer-reviewed research studies which had used either state/territory or national CAR data over the last 40+ years (1980–2024), a scoping literature review was undertaken on 28 November 2024, using four health databases: Medline/OVID, Scopus, Embase and Cumulative Index to Nursing and Allied Health Literature (CINAHL). The first 100 records in Google Scholar, arising for each of the search terms, were also reviewed. The current name and former name(s) of each state/territory CAR were used as the search terms, including expansions and abbreviations of state names (e.g. New South Wales (NSW)) (see Appendix 1). For inclusion, studies needed to incorporate the search terms in the keywords, title or abstract. Due to the potential variability in the usage of CAR titles/names in studies over the extensive time period (1980–2024), the references of within-scope articles were also manually reviewed. Due to time and resource constraints, manual reference review was undertaken by one researcher for two-thirds of the within-scope articles.
Inclusion and exclusion criteria
The 40+ year inclusion period (1980–2024) spanned the time from the commencement of the earliest Australian CAR to the present. To be included, studies needed to be peer-reviewed, published in English and have full-text available. Commentaries/notes, letters to the editor, conference presentations, lectures, non-peer-reviewed articles, grey literature and unlocatable full-text articles were excluded. Reports produced, or contributed to, by the CARs themselves were not included, as the focus of the scoping review was upon research undertaken beyond “business-as-usual” CAR reporting. Any study which used data from the Western Australian Register of Developmental Anomalies (WARDA) related to developmental anomalies, rather than CAs, was also excluded.
Eligibility screening and review
Studies from the database searches were imported into Covidence. One researcher undertook title and abstract screening (MR), followed by full-text review of eligible studies. From the articles selected from the full-text review, a manual reference search was undertaken, and the URL of any reference incorporating in its title the words “Australia” or any of the Australian states/territories, along with a specific congenital anomaly or associated general terms for CAs, was transcribed into an Excel spreadsheet. The abstracts for these articles were retrieved. Using the established inclusion and exclusion criteria, eligible articles were manually added to the within-scope studies.
Data extraction for modified scoping review
Fields included in the data extraction template were based upon data collected by Riley et al. (2023b), in their documentary analysis of government population health websites, and by Ruseckaite et al. (2023) in their scoping literature review on rare disease registries/databases. Additional fields collected were based upon the expertise of the author, who worked with clinical registries for over 20 years. The data extraction template was trialled by an independent researcher to determine ease of use and understanding of data items. Fields included the following: article title, lead author’s name, lead author organisation, year first published, journal name, the primary CAR contact involved in study, CAR involved, aim, setting, whether data linkage was involved, participants, medical condition of focus, time frame,
Analysis
Descriptive analysis using Microsoft Excel (Version 365) was undertaken to summarise the results.
Ethics
Formal approval from a Human Research Ethics Committee was not sought as all of the information used was available in the public domain.
Results
Comparison of CARs from documentary analysis
Table 1 outlines general characteristics of the state/territory-based CARs. Five Australian states (i.e. NSW, Queensland, South Australia (SA), Victoria, Western Australia (WA)) and one national body (AIHW) had
General description and characteristics of Australian Congenital Anomaly Registers. a .
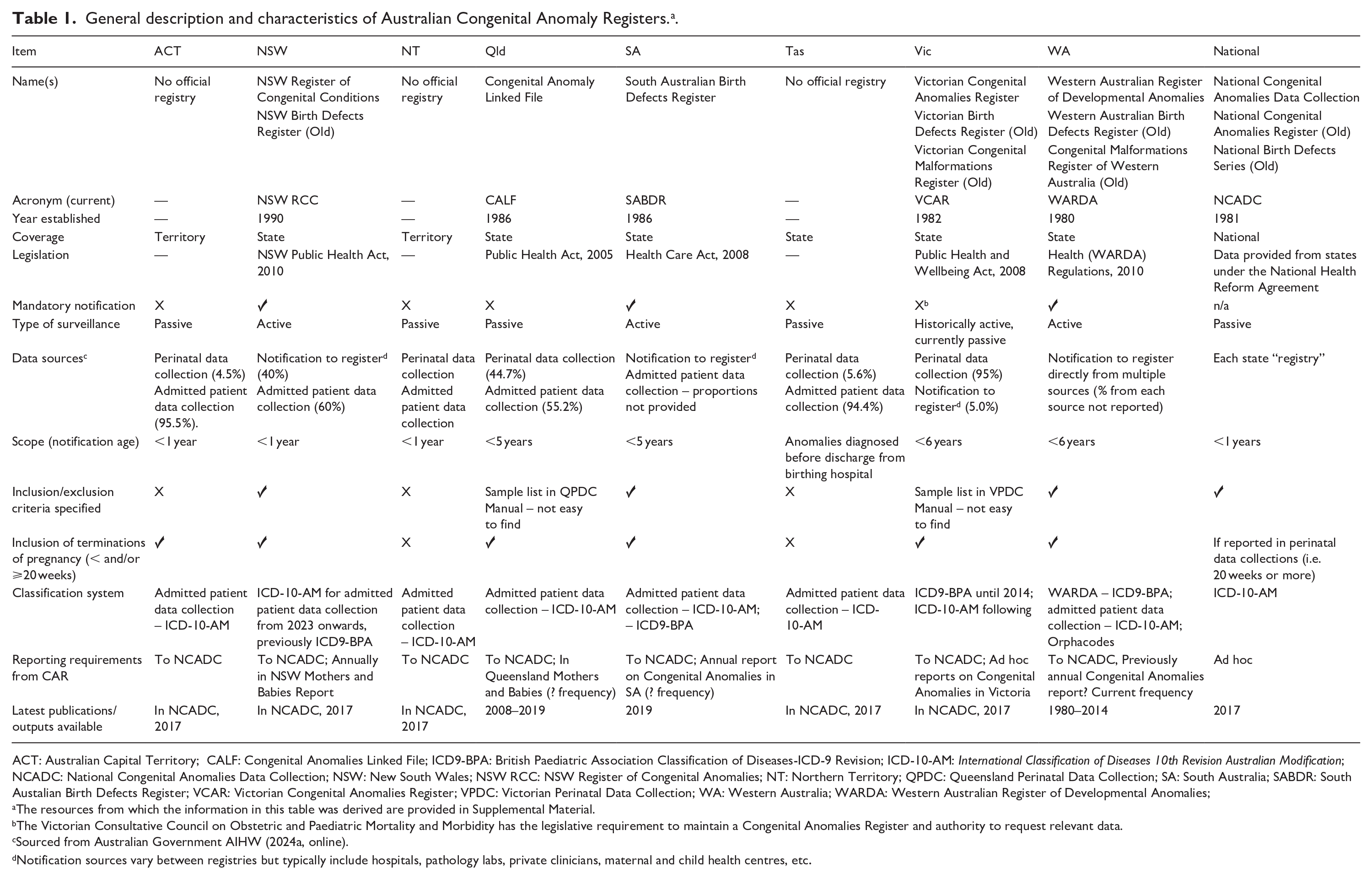
ACT: Australian Capital Territory; CALF: Congenital Anomalies Linked File; ICD9-BPA: British Paediatric Association Classification of Diseases-ICD-9 Revision; ICD-10-AM:
The resources from which the information in this table was derived are provided in Supplemental Material.
The Victorian Consultative Council on Obstetric and Paediatric Mortality and Morbidity has the legislative requirement to maintain a Congenital Anomalies Register and authority to request relevant data.
Sourced from Australian Government AIHW (2024a, online).
Notification sources vary between registries but typically include hospitals, pathology labs, private clinicians, maternal and child health centres, etc.
Most of the existing CARs were established in the 1980s, and three states (NSW, SA and WA) had legislation mandating the reporting of CAs at the time of conducting this study. The maximum
Criteria on the CA
The three most readily available state/territory-based CA inclusion/exclusion lists demonstrated inconsistencies between conditions to be reported. NSWs list of “scheduled congenital conditions” included structural malformations, chromosomal abnormalities and four medical conditions: cystic fibrosis, phenylketonuria, congenital hypothyroidism and thalassaemia major (New South Wales Health, 2018). The South Australian CA inclusions/exclusions list included, as an example only, the most common CAs grouped by body system, chromosomal anomalies, metabolic conditions or teratogenic conditions (Gibson et al., 2024). The WA CA list of inclusions/exclusions also grouped the most common anomalies by body system, but also included cerebral palsy, congenital infections, metabolic disorders and other (e.g. foetal alcohol syndrome) (Government of Western Australia, 2024). While demonstrating considerable overlap in groupings of conditions, no CARs specified the inclusion of single-gene conditions (other than the few individually named above, or those that were classified under metabolic conditions).
The three states/territories without established CARs
All Australian CARs used a version of the ICD to code CAs. Due to the increased specificity of the British Paediatric Association Classification of Diseases–ICD-9 Revision (ICD9-BPA) in
The
Published peer-reviewed studies using CA data
The scoping literature review identified 207 articles for importation into Covidence. The title, abstract and full-text review resulted in 63 studies for inclusion. Two potentially relevant articles were not included due to the inability to locate their full text. From the manual reference search of two-thirds of within-scope articles (
Almost 46% (
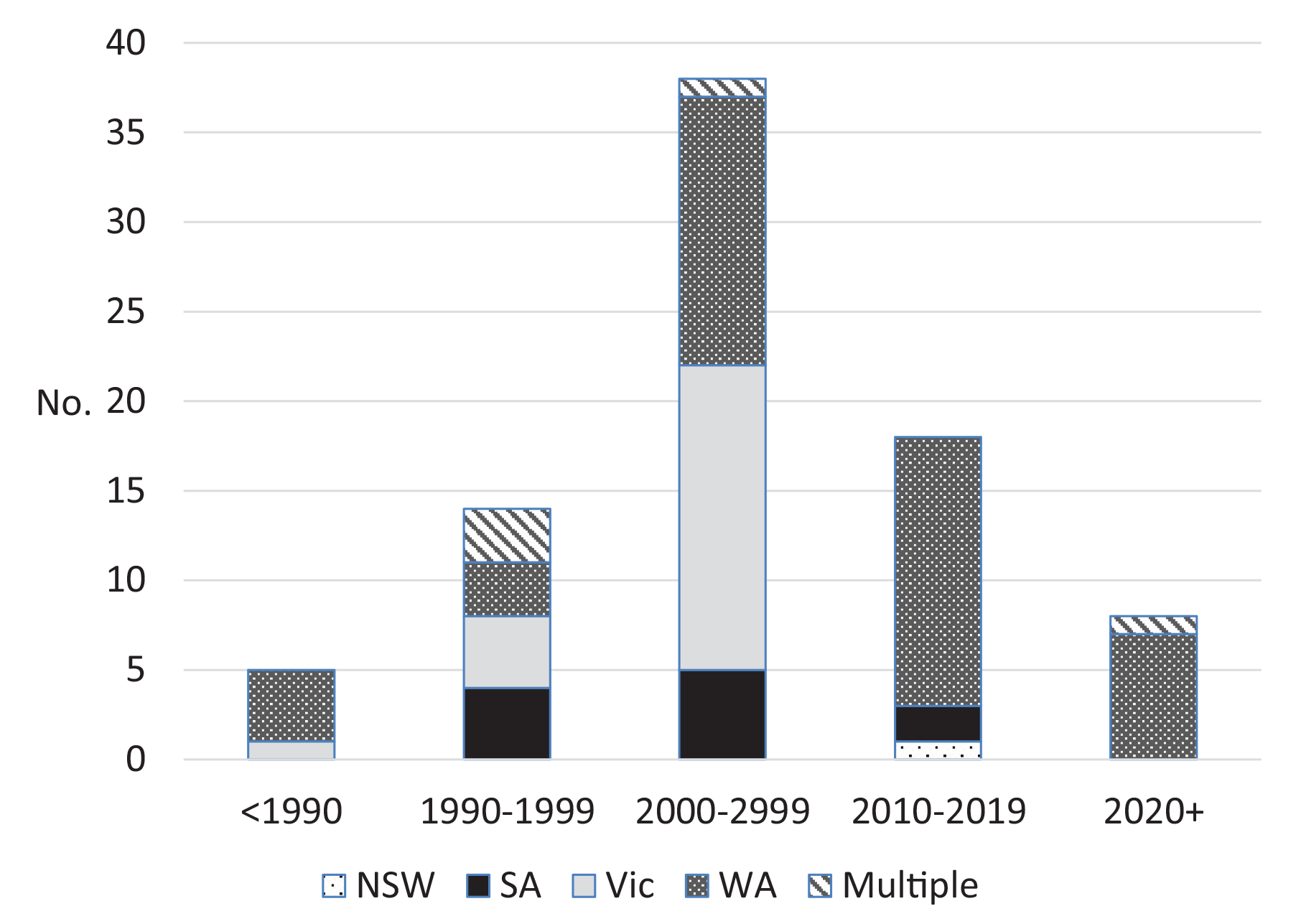
Number of studies published by established state/territory-based CARs by time period.
A common feature of the studies published by WA, Victoria and SA was the presence of a key CAR “staff member” as part of the authorship. For WA, a key member was involved in 39/44 (88.6%) of the studies, for Victoria, a key member was involved in 21/22 (95.4%) studies and in SA, a key member was involved in 7/11 (63.6%) studies.
The 83 peer-reviewed studies were published in 32 different national and international journals (see Appendix Table 1). The journals publishing the most CAR-related articles included the following:
Most of the published studies using CA data incorporated a descriptive research design (
Study designs of peer-reviewed publications of State-based BDR/CAR combined, 1990–2024.
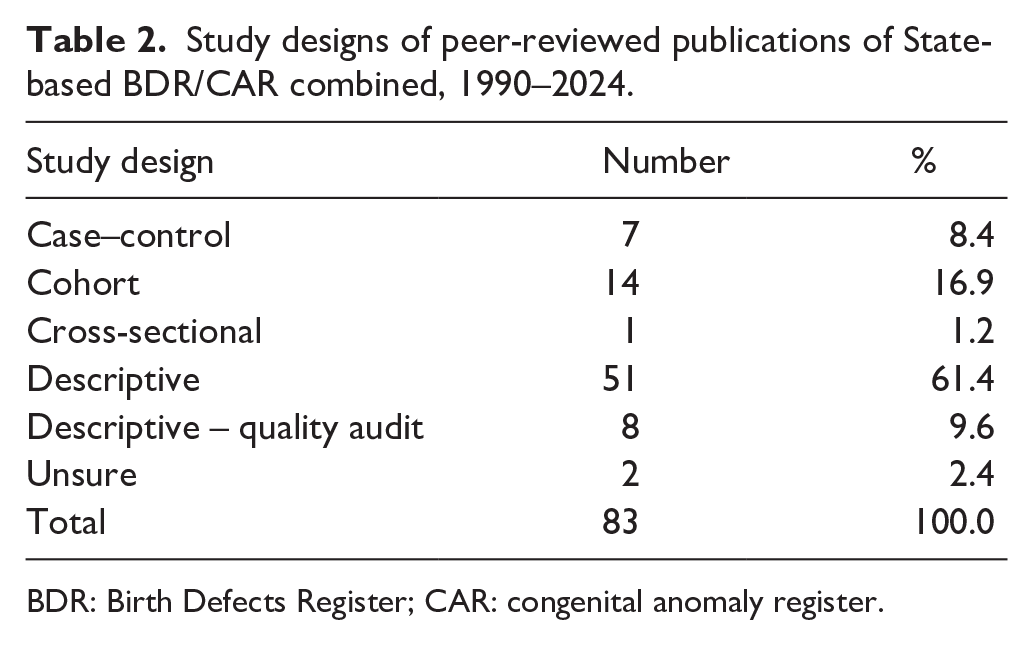
BDR: Birth Defects Register; CAR: congenital anomaly register.
The foci of the peer-reviewed studies covered a broad range of medical conditions (see Appendix Table 2). The three most common topics for CA research using state/territory-based CA data during 1980–2024 were as follows: all CAs combined (
Purpose-category for which the CA research was undertaken, 1980–2024.

CA: congenital anomalies.
Discussion
Active versus passive surveillance
The first aim of this study was to compare and contrast the general characteristics of state/territory-based CARs in Australia. Despite the expectation from the AIHW for national reporting of CA data (Australian Government AIHW, 2024a), not every state/territory maintains a specialised CAR. Two different approaches to public health surveillance were represented by the state/territory-based CARs. Passive surveillance (Nsubuga et al., 2006) that relies on existing administrative data (patient admitted data collection) or population-health datasets (e.g. perinatal data collection) was utilised by Tasmania, QLD, the ACT and the NT. By contrast, WA, SA and NSW actively sought notification from additional sources, such as pathology laboratories, hospitals, outpatient clinics, etc. This reflects active surveillance, which is known to be more accurate in case ascertainment than passive surveillance but is also more resource-intensive and expensive (Mai et al., 2019; Nsubuga et al., 2006). The Victorian CAR reflected a model that historically practiced active surveillance, but more currently reflected passive surveillance due to administrative and resourcing issues. Studies have demonstrated that under-reporting of conditions occurs when incidence/prevalence is based upon administrative data (Canaway et al., 2024; Lujic et al., 2014; Ryan et al., 2021). To foster greater consistency between CARs and to improve ascertainment of CA data, the inclusion of sources of notification beyond administrative data should be encouraged.
Legislative requirements and mandatory reporting
Three states in Australia have mandated, via legislation, the collection of CA data (NSW, WA and SA). These states currently undertake active surveillance. In Victoria, the CAR custodian has the legislative requirement to maintain a registry and also has the authority to request CA data from relevant bodies. The debate between voluntary versus mandatory notification to governments is long-standing (Mathews, 2015). Most middle- and high-income countries, including Australia, mandate the reporting of communicable diseases (National Notifiable Disease Surveillance System) and suspected child abuse or domestic violence. Given that all current active surveillance CARs in Australia have mandated reporting of CAs and that all of the passive CARs provide administrative government data on CAs that have been collected without specific patient consent, it would seem appropriate that the collection of CAs should be mandated in all states/territories. This would improve the overall ascertainment of CAs.
Scope of CAR data collection
Age at diagnosis
An inconsistency between state/territory-based CARs was the age of children to be included. To facilitate CA data consistency, the NCADA restricted national reporting to include CAs diagnosed in children up to 1 year of age (Australian Government AIHW, 2023a). This is a valid approach to harmonise the data, but there are immediate issues as the Tasmanian CA data only included conditions diagnosed within the birth episode, that is, within the first day or so of life. Based upon WA and SA analyses (Bower et al., 2010a; Gibson et al., 2016), restricting inclusion of CAs diagnosed up to 1 year of age would under-report the prevalence of CAs between 12% and 18%. Depending upon the type of CA, under-reporting could be significantly higher. For example, Bower et al. (2010a) reported that two-thirds of all cases of foetal alcohol syndrome were diagnosed after the child reached 1 year of age. They also ascertained that the resources required to collect the additional 5 years worth of diagnosis data (i.e. inclusion of CAs in children up to 6 years of age) equated to only 1.76 weeks of additional work. This was a small amount of extra work to increase CA ascertainment by 12–18% and supports the expansion of the scope of children to be captured within the CARs to 6 years of age.
Inclusion/exclusion criteria specified
To facilitate clarity on both the CA conditions and the cases (e.g. TOPs before 20 weeks) to be reported to a CAR, and to encourage notification, it is important that CARs provide readily accessible information on the inclusion/exclusion criteria within the registries. Several studies have identified that the provision of easily accessible and clear explanatory documentation surrounding government datasets is more likely to promote trust in, and use by, researchers than for datasets where this information is unavailable (Adjekum et al, 2018; Imker et al., 2021; Riley et al., 2024). Clear explanatory documentation on the CAs to be included/excluded within the CARs should be made readily accessible across all state/territory-based and national settings.
Of equal importance is the need to capture CA data, which most significantly impacts both reproductive health and children’s lives. Gjorgioski et al. (2020) demonstrated, from 2 weeks of admissions at a large paediatric hospital in Victoria, Australia, that genetic conditions contributed to over half of the inpatient stays. Currently, there is a gap in CA reporting in Australia. There is a lack of genomic diagnostic results, with most CARs reporting on trisomies but not on single-gene conditions. The data are usually unavailable from administrative/population health datasets, such as perinatal data, because the results are usually returned after the baby is discharged from the hospital. Given the high prevalence of genetic conditions in paediatric inpatient admissions, and the growing field of prenatal genomic diagnosis (Mellis et al., 2022), active surveillance mechanisms to increase the capture of genomic diagnostic test results (especially those which are unavailable during the birth episode) should be implemented, or enhanced, for all CARs.
Coding systems
As part of their approach to consistency in reporting the national CA data, the NCDAC maps all of the state/territory-based data to ICD-10-AM codes (Australian Government AIHW, 2024a). Most of the states/territories that utilise administrative data for their reporting already classify the conditions according to ICD-10-AM. The advantage for the states/territories which utilise the ICD9-BPA is the greater specificity it provides for the classification of CA. In their study into the complexity of international classification guidelines and rules, Atolagbe et al. (2021) identified extensive variation within and between countries, making the comparison of the coded data impractical for monitoring and surveillance purposes. In Australia, there are variations in state/territory-based coding guidelines that may impact the coding of CAs. Therefore, an analysis should be undertaken to identify how state-based coding guidelines impact the ICD-10-AM classification of CAs and hence their interpretation at the national level.
Only one of the state/territory-based CARs (WA) included Orphanet codes. The inclusion of this “rare diseases specific codification system” (RD-Code, 2025) provides greater specificity in the identification and classification of rare conditions. Rather than just using the ICD organ-based classification, which is not fit-for-purpose for classifying genetic conditions in light of widespread genomic testing, additional classification mechanisms, such as Orphanet codes, need to be adopted. This would increase specificity in describing genetic and other rare conditions and align with international foci and potential global collaboration (Kinsner-Ovaskainen et al., 2018).
Frequency of reporting
There is an inconsistency in the reporting of information contained within the state/territory-based CARs, and the national CAR in terms of both frequency and timeliness of reporting. Only the SABDR consistently produced dedicated CA reports, with the latest report on the 1986–2019 birth cohort published in 2024. Until recently, no national CA data had been reported since 2008. In 2019, a National Congenital Anomaly Advisory Group was formed to re-establish “a national congenital anomalies data collection” (Australian Government AIHW, 2024a, paragraph 1). Since then, national collection of CA data has occurred, resulting in the publication of CA data from the 2017 birth cohort from seven states/territories, except WA (Australian Government AIHW, 2024a). This still demonstrates a considerable time lag between data capture and reporting. Mechanisms to improve the timeliness of reporting of national CA data need to be investigated by the leadership groups and authorities responsible for collecting, reporting and managing CA data.
Peer-reviewed research using state/territory-based CARs
Over the 44 years, since the first CAR was established, 83 peer-reviewed publications incorporating CA data were identified via the scoping literature review. This equates to approximately two peer-reviewed studies per year across all of the state/territory-based CARs. Only three of the CARs (WA, Victoria and SA) demonstrated active participation in research studies, and not all of these CARs were active over the whole time period. Only WA demonstrated a consistent and continuing involvement in the use of CA data for research, particularly from 2000 onwards. From the scoping review, 44 studies involving WARDA data were identified. According to Nembhard and Bower (2016), the WARDA data had been involved in more than 235 peer-reviewed publications by 2016. This significant gap between those we identified, and the number cited in Nembhard and Bower (2016) can be partly explained by (i) the use of a scoping review method rather than a systematic review and (ii) partly by the scope of WARDA incorporating developmental anomalies (such as cerebral palsy) from 2010 onwards rather than just CA. A systematic review may be required to capture all of the peer-reviewed publications incorporating Australian CAR data.
An analysis of the authors associated with the peer-reviewed papers demonstrated the presence of leading individuals affiliated with the CARs who were involved in many of the studies during peak research time periods. Those states/territories that have not demonstrated any use of their CA data in peer-reviewed publications do not appear to have employed dedicated staff who actively used and promoted their data for research. The need for “appropriate funding and a sustainability model” (Bloom et al., 2017, p. 122) is essential for enhanced utilisation of CAR data.
Studies utilising the CA data were published in both national and international journals, demonstrating the breadth of their potential influence. Cross-country collaboration does not seem to have been the focus of more recent peer-reviewed research using CA data. To facilitate the capacity of either state/territory-based CARs or for national contributions to international CA collaborations, more resources and staffing are required at all levels.
Most of the peer-reviewed studies identified in this review employed a descriptive research design – including quality audits. The most common reasons for undertaking the research were monitoring and surveillance or undertaking observational-based research. These two purposes align with the main reasons for the establishment of CARs. Maintaining a high-quality disease register is resource-intensive (Stubbs et al, 2024), facilitating widespread use of its data would provide greater justification for the resources utilised in its support and maintenance. State/territory-based CARs should promote and develop more opportunities for increased use of their data.
Only one-third of the CA studies employed data linkage. This is reflective of the wide timeframe over which the studies were published (1980–2024). Data linkage was not common in the early years of the study period. It is expected that significantly more studies involving CA data and data linkage could be facilitated with current technologies and cross-jurisdictional networks (Smith and Flack, 2021) if the data were of sufficient quality and appropriate explanatory documentation was available. Promoting the sharing and reuse of good-quality CA data, appropriately anonymised for data linkage, should be promoted.
Research to measure the impact of public health measures to reduce CA
Two of the main foci of state/territory-based CA research over the past 40 years were NTDs and Trisomy 21. The many studies written regarding NTDs reflected the purposes for which the CARs had been established and demonstrated the importance of registers in action. Three of the state/territory-based CARs were able to actively provide monitoring and surveillance data for a serious CA, explore potential aetiological factors, provide evidence-based research for implementation of preventive measures (mandatory folate fortification) and then evaluate the success of the measures. Each of these phases is clearly evident in the peer-reviewed articles which were published in the late 1980s and early 1990s. To facilitate the primary purpose(s) for which the CARs were established (e.g. monitoring and surveillance), the state/territory governments need to re-prioritise CAR and re-affirm their commitment to the long-term viability of these collections (Box 1).
Recommendations to enhance the viability, consistency and useability of CARs in Australia.

CA: congenital anomalies; CAR: congenital anomaly register; ICD-10-AM:
Limitations
This study involved the use of two separate methods – a documentary analysis of websites and a modified scoping literature review. Both activities were undertaken by one researcher due to resource limitations. To assist with the extraction of information for the documentary analysis, most data items were based on items already defined and recorded in the technical notes of published CA reports. This minimised subjective interpretation of the data item and minimised potential researcher bias. Strict inclusion and exclusion criteria were identified prior to the commencement of the scoping literature review to help minimise selection bias. However, the manual review of two-thirds, rather than all, of the references from within-scope papers may have introduced selection bias and impacted the number of studies identified for inclusion. To promote transparency, details of all studies identified in the scoping literature review have been provided as a Supplemental Table S1.
Conclusion
This study has demonstrated some major challenges for standardisation with the state/territory-based and national reporting of CA data in Australia. Lack of consistency in key processes, lack of resourcing and, in some states/territories, lack of key leadership highlight risk to the sustainability of CA data collection in Australia. However, it also demonstrates that effective models of CA surveillance among some Australian states/territories do exist and that key research can be effectively promoted and undertaken with appropriate resourcing and leadership. This study provides several practical recommendations that could be implemented by governments to embed a “gold standard” CA monitoring and surveillance system within states/territories, and nationally, within Australia.
Supplemental Material
sj-xlsx-1-him-10.1177_18333583251343623 – Supplemental material for Congenital anomaly registers in Australia: A national challenge
Supplemental material, sj-xlsx-1-him-10.1177_18333583251343623 for Congenital anomaly registers in Australia: A national challenge by Merilyn Riley and Lisa Hui in Health Information Management Journal
Footnotes
Appendix 1: Search string for each state-based congenital anomaly register by congenital anomaly register title(s)
Accepted for publication May 5, 2025.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: LH received a clinical investigator grant (#1196010) from the MRFF Genomics Health Future Fund, under which MR was also contracted for this work.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
