Abstract
Background:
Severe forms of systemic sclerosis (SSc) can be treated with autologous hematopoietic stem cell transplantation (aHSCT), but transplant-related mortality (TRM) can be considerable.
Objectives:
To describe the long-term outcomes after aHSCT with a focus on clinical and immunological parameters.
Design:
Retrospective single-center cohort study.
Methods:
The overall survival, course of skin fibrosis and lung function, need for immunosuppressive treatments, and immunophenotyping with flow cytometry of peripheral blood of SSc patients after aHSCT were analyzed.
Results:
Thirty-three patients were included with a mean follow-up time of 7.0 years (maximum 15.7 years). TRM was 6.1%, and seven deaths occurred during follow-up. Skin fibrosis improved, and lung function (forced vital capacity and diffusion capacity of carbon monoxide) stabilized in the long term. 34.5% never needed immunosuppressive therapy after aHSCT. Of patients who needed immunosuppression, 80% were treatment-free at the time of 15 years after aHSCT. The immunological reset after aHSCT persisted in the long term: Memory B cells declined for 2 years, and T helper cells were reduced for 5 years.
Conclusion:
aHSCT appears to promote positive long-lasting effects for SSc patients. Long-term data on safety are reassuring, considering that only the most severely affected and refractory SSc patients undergo aHSCT.
Keywords
Background
Systemic sclerosis (SSc) is the inflammatory rheumatic disease with the highest case mortality. 1 Immunosuppressive therapy, antifibrotics, and vasodilative agents are the mainstay of treatment. But the therapeutic armamentarium or treatment algorithms are limited by the heterogeneous nature of the disease, by the plethora of manifestations with differing treatment responses, and the complex pathogenesis comprising inflammation, fibrosis, and endothelial damage. Although ongoing studies explore new and ambitious treatment modalities for SSc (including B cell inhibition, bispecific antibodies, and chimeric antigen receptor (CAR)-T-cell therapies), 2 autologous hematopoietic stem cell transplantation (aHSCT) is one of the therapies for severe SSc forms with the best evidence and recommended in national and international guidelines. 3 Three randomized controlled clinical trials showed the superiority of aHSCT versus cyclophosphamide,4–6 but also showed a high transplant-related mortality (TRM) ranging from 6% to 10.1% in the two larger multicenter trials; in the single-center trial, no TRM was reported. To date, there is little data about the long-term outcomes and the immune reconstitution of SSc patients after aHSCT.
The present study aimed to describe the long-term outcomes of SSc patients after aHSCT from our center with a focus on clinical and immunological parameters.
Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 7
Patients and transplantation procedure
Thirty-three patients were recruited for this retrospective cohort study. These were all patients who were followed up at the University Hospital of Würzburg. Most underwent aHSCT in Würzburg, some at the University Hospital of Tübingen. All fulfilled the American College of Rheumatology/European Alliance of Associations for Rheumatology classification criteria for SSc. 8 All patients had progressive disease despite receiving conventional immunosuppressive therapy. The immunoablative aHSCT procedure was previously described 9 and was performed according to the autologous stem cell transplantation international scleroderma (ASTIS) trial. 5 Briefly, hematopoietic stem cells were mobilized after cyclophosphamide with granulocyte-colony stimulating factor (G-CSF) and collected via leukapheresis. A cluster of differentiation 34 (CD34+) selection of the graft was performed in all patients (apart from 1 due to low cell numbers). The conditioning regimen included cyclophosphamide (apart from 1, who received melphalan due to impaired renal function) and rabbit anti-thymocyte globulin (ATG; cumulative dose: 30 mg/kg body weight), followed by re-infusion of the autologous hematopoietic stem cells.
Before undergoing aHSCT, all of our patients were carefully evaluated to exclude patients with highly impaired organ functions, resulting in increased treatment-related toxicity/mortality. The evaluation recommendations we followed were first published in 2017 by the European Society for Blood and Marrow Transplantation (EBMT). 10 Only patients refractory to at least one (in most cases multiple) immunosuppressive treatment regimens were selected for aHSCT, including but not restricted to mycophenolate, cyclophosphamide, and rituximab. Disease progression after aHSCT was defined as restart of immunosuppression due to the deterioration of SSc manifestations that already existed before aHSCT.
Flow cytometry
The staining and measurement of peripheral lymphocytes was previously described. 11 The lymphocyte subsets were defined for the B-cell subdifferentiation as follows: B cells (CD19+), transitional B cells (CD19+/CD10+/CD38+), post-switch memory B cells (CD19+/CD27+/immunoglobulin D (IgD−)), pre-switch memory B cells (CD19+/CD27+/IgD+), naïve B cells (CD19+/CD27−/IgD+), and circulating plasmablasts (CD19+/CD38++/CD27++/IgD−). For the T-cell subdifferentiation: T cells (CD3+), T helper cells (CD3+/CD4+), naïve T helper cells (CD3+/CD4+/CCR7+/CD45RA+), cytotoxic T cells (CD3+/CD8+), natural killer cells (CD3−/CD56+), and γδT cells (CD3+/TCRγδ+). The used antibodies with isotypes and conjugated dyes are shown in Table S1.
Statistical analysis
Calculations were done with Prism (V10; GraphPad Software, Boston, MA, USA). For continuous variables, differences between-paired groups were examined with Wilcoxon signed-rank tests and differences between unpaired groups with Mann–Whitney U tests. For metrical variables differences, between-unpaired groups were calculated with Fisher’s exact tests. Kaplan–Meier curves were used for survival analysis, and log-rank tests (Mantel–Cox) to assess whether or not factors with known risk for worse outcomes might have influenced overall survival. Differences were considered significant when two-tailed p-values were less than 0.05. Excel (Microsoft, Redmond, WA, USA) was used to collect the data. Figures were grouped with paint.net (V5; dotPDN LLC, Kirkland, WA, USA).
Results
Patient characteristics
Thirty-three patients underwent aHSCT from December 2008 to May 2024. The median age at the time of aHSCT was 47.0 years (range 23–64), and 17/33 (51.5%) were female. Concerning disease extent, 29/33 (87.9%) had a diffuse cutaneous form with a mean modified Rodnan skin score (mRSS) of 23.1 points (range 2–44), 24/33 (72.7%) were Scl70-antibody positive, 26/33 (78.8%) had SSc-associated interstitial lung disease (ILD), 14/33 (42.4%) had cardiac involvement (defined as elevated troponin plus either positive cardiac MRI or positive myocardial biopsy), and 7/33 (21.2%) had pulmonary arterial hypertension (PAH), which was well controlled at the start of aHSCT. The main indication for aHSCT was progressive ILD (in 69.7%) despite previous immunosuppressive therapy. The previous immunosuppressive treatment was not part of this study, as these data for most of our patients were already published.12,13 Most of the patients (96.7%) received cyclophosphamide for the conditioning regimen (two patients who underwent aHSCT before 2011 received an older transplant protocol including thiotepa 5 mg/kg body weight on 2 days, in addition to cyclophosphamide), only one patient received melphalan (50 mg/m2 on 2 days) due to impaired renal function. All patients also received anti-thymocyte rabbit ATG (Table 1).
Characteristics of the study population.
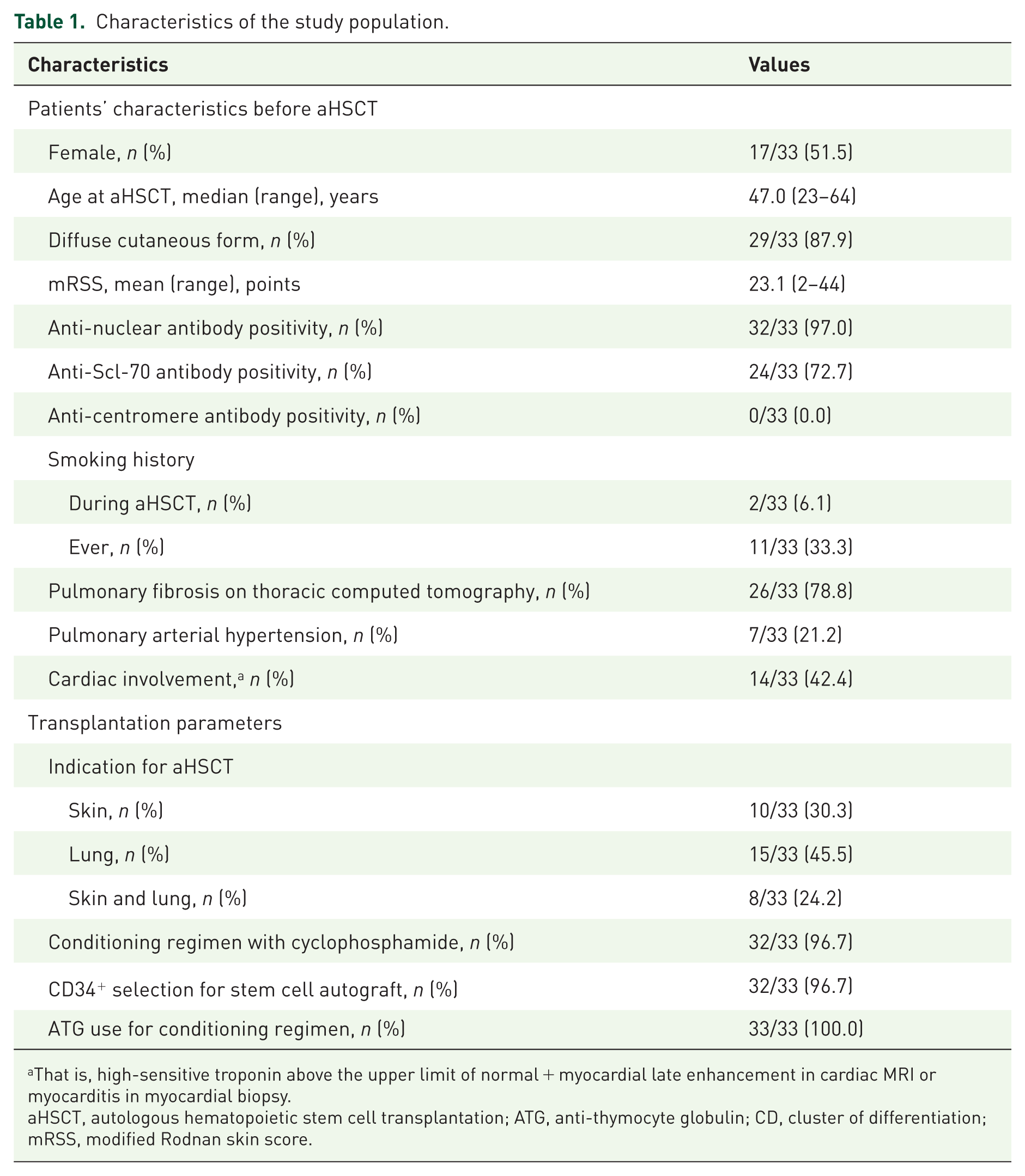
That is, high-sensitive troponin above the upper limit of normal + myocardial late enhancement in cardiac MRI or myocarditis in myocardial biopsy.
aHSCT, autologous hematopoietic stem cell transplantation; ATG, anti-thymocyte globulin; CD, cluster of differentiation; mRSS, modified Rodnan skin score.
Long-term survival and treatment-related mortality
The median follow-up time of the whole population was 83.0 months (range 1 month–15.7 years). One patient was lost to follow-up after 3 years. Twenty-three patients (69.7%) are alive by June 2025; of these, 17 patients were followed for over 5 years, and 6 patients had a follow-up shorter than 5 years (Figure 1).

Follow-up times of the population, including deaths and periods, when immunosuppressive treatment was taken (blue).
The treatment-associated mortality, which was defined as death within 100 days after aHSCT, was 2/33 (6.1%). One patient died due to acute heart failure (supposedly caused by cyclophosphamide and ATG, without preexisting cardiac involvement of SSc) in combination with a gram-negative septic shock. The second patient suffered a stroke due to middle cerebral artery occlusion + respiratory insufficiency (supposedly caused by progressive preexisting SSc-ILD). Seven deaths occurred during the follow-up after a median of 29.0 months after aHSCT and were due to a cardiogenic shock (at month 4; cardiac SSc-involvement was present before aHSCT), a septic shock (at month 10), progress of PAH (at month 28; PAH was present before aHSCT), occurrence of progressive multifocal leukoencephalopathy (at month 29), respiratory insufficiency (at month 31; PAH was present before aHSCT), congestive heart failure (at month 119; cardiac-SSc involvement was present before aHSCT), and bronchial carcinoma (at month 151; Table 2).
Long-term outcomes after aHSCT.

Deaths due to septic shock, progressive multifocal leukoencephalopathy, respiratory insufficiency, congestive heart failure, bronchial carcinoma, cardiogenic shock, progression of pulmonary arterial hypertension; treatment-related mortality not included.
aHSCT, autologous hematopoietic stem cell transplantation.
The overall survival is shown in Figure 2(a). No significant differences between subpopulations with known risk factors for worse outcomes could be detected with log-rank (Mantel-Cox) tests for Scl70 antibody-positivity (p = 0.274; Figure 2(b)), presence of ILD (p = 0.999; Figure 2(c)), heart involvement (p = 0.512; Figure 2(d)), or presence of PAH (p = 0.0805; Figure 2(e)).

Long-term survival after aHSCT. (a) Kaplan–Meier graph of the overall survival of the whole cohort. No significant differences in the survival after aHSCT could be detected in the subpopulations with Scl70-antibody positivity (b, purple), with ILD (c, blue), heart involvement (d, red), or PAH (e, green).
Need for immunosuppressive therapy after aHSCT
Twenty-nine patients could be followed for at least 1 year to describe the need for a restart of immunosuppressive therapy. 10/29 (34.5%) never received immunosuppressive therapies after aHSCT, 10/29 (34.5%) needed immunosuppressive therapies only temporarily and were able to discontinue them again, and 9/29 (31.0%) took immunosuppression at the end of the follow-up. Restart of immunosuppressive therapies occurred at a mean time of 1.3 years. The highest immunosuppression intake was observed in the second year (15/28 (53.6%)) and was lowest up to 15 years after aHSCT (2/10 (20.0%) needed immunosuppression; Figure 3). Reasons for a restart of immunosuppression after aHSCT were progressive disease (progressive skin fibrosis 9/19 (47.4%), progressive lung involvement 4/19 (21.1%), arthritis 1/19 (5.3%), and myositis 1/19 (5.3%)) or appearance of immunological adverse events (4/19 (21.1%), comprising secondary microscopic polyangiitis, immune thrombocytopenia, secondary Sjögren’s disease, and eosinophilic dermatitis).

Need for immunosuppressive therapy after aHSCT. Shown are percentages with 95% confidence intervals.
Course of lung and skin parameters in the long term after aHSCT
Twenty-six patients could be followed for the course of their skin and lung involvement for at least 1 year after aHSCT. Skin fibrosis improved in the long term, as the mRSS showed a sustained decrease after aHSCT (p = 0.003 in a Kruskal–Wallis test; Figure 4(a)). The pulmonary function stabilized, as forced vital capacity (FVC) exhibited stable values (p = 0.363; Figure 4(b)), as well as diffusion capacity for carbon monoxide (DLCO) did (p = 0.266; Figure 4(c)).
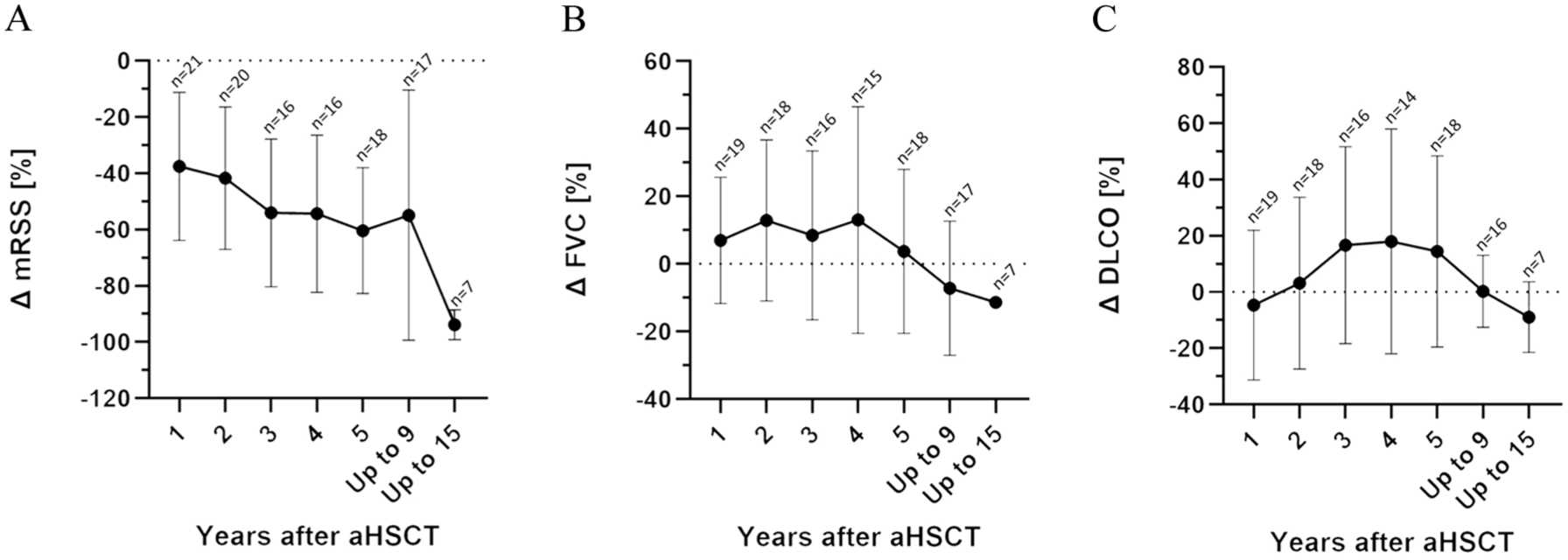
Course of skin and lung parameters after aHSCT. (a) Improvement of the mRSS. (b) Changes in FVC and (c) of DLCO. Shown are means ± standard deviation, each in comparison to the values before aHSCT.
Immunological reset after aHSCT
To characterize the long-term immunological reset of the aHSCT procedure, 26 patients who received at least 2 flow cytometric analyses were analyzed. The previously described reduction of memory B cells 9 was present until 2 years after aHSCT: Mean post-switch memory B cells were 8.3% (range 0.7–23.7) before aHSCT versus 1.9% (range 0.0–3.7) 2 years after aHSCT (p = 0.002). An increase in naïve B cells was present until 1 year after aHSCT: Mean naïve B cells were 79.8% (range 55.0–96.8) before aHSCT versus 84.5% (range 0–96.5) 1 year after aHSCT (p = 0.036). Within the T-cell compartment, a long-lasting reduction in T helper cells after aHSCT could be detected: Mean T helper cells were 46.4% (range 24.1–58.4) before aHSCT versus 31.9% (range 22.2–40.7) 5 years after aHSCT (p = 0.009). A long-lasting increase in cytotoxic T cells could be detected: Mean cytotoxic T cells were 18.6% (range 6.0–41.0) before aHSCT versus 27.1% (range 20.0–41.1) up to 15 years after aHSCT (p = 0.040; Figure 5).

Flow cytometric long-term characterization of lymphocytes after aHSCT. (a) B cells, (b) pre-switch memory B cells, (c) post-switch memory B cells, (d) naive B cells, (e) transitional B cells, (f) circulating plasmablasts, (g) T cells, (h) T helper cells, (i) naive T helper cells, (j) cytotoxic T cells, (k) NK cells, and (l) γδT cells. Numbers of patients: 1 year after aHSCT = 19, 2 years = 19, 3 years = 15, 4 years = 15, 5 years = 14, up to 9 years = 15, up to 15 years = 7. Shown are means ± standard deviation.
Discussion
In this study, we describe the long-term outcomes of 33 patients with refractory SSc who underwent aHSCT. Early TRM amounted to 6.1%, which has to be taken into account during patient selection. The extent of skin fibrosis improved after aHSCT, and the lung parameters stabilized in the long term. About one-third of the patients remained off immunosuppressive therapy after aHSCT. Long-lasting changes in the lymphocyte composition indicate a reset of the immune system achieved by aHSCT.
TRM in the larger multicenter trials ASTIS and scleroderma: cyclophosphamide or transplantation trial (SCOT) were stated at 10.1% and 6%, respectively. In our cohort, we found a TRM of 6.1%, which is also in line with a large non-interventional prospective multicenter trial from the EBMT, which found a TRM of 6.25%, defined as 100 days non-relapse mortality. 14 The fairly high TRM in the ASTIS trial is thought to be caused by the reduced possibilities in supportive therapy. That means, at the start of the long recruitment period of ASTIS (from 2001 to 2009), there was limited knowledge on SSc-specific complications after aHSCT, like renal crises, G-CSF-induced alveolitis, cardiac complications, or severe opportunistic infections. This knowledge could be applied in our cohort, which may have positively influenced TRM. On the other hand, patients who would have been excluded from the ASTIS or SCOT trial were also transplanted with reduced intensity after thorough cardiopulmonary assessment according to EBMT guidelines.
Some patients after aHSCT need a restart of immunosuppressive therapy, 12 partly due to progressive SSc disease, or due to engraftment syndromes and new onset of secondary autoimmune disorders, which represent specific treatment-related immunological adverse events. 13 In our cohort, 34.5% never received immunosuppressive therapy after aHSCT.
Due to its treatment toxicity, alternate treatment approaches should be carefully considered before performing an aHSCT, keeping in mind that delaying aHSCT can lead to organ deterioration, making an aHSCT no longer feasible. To date, only a few trials and no randomized controlled trial comparing aHSCT to other treatment regimens other than cyclophosphamide are available. A retrospective case–control study showed superiority of aHSCT versus rituximab treatment and conventional immunosuppression over 60 months. 15 In a retrospective case–control study, the comparison of aHSCT against the emerging combination therapy of RTX plus MMF in severe SSc cases showed a non-inferiority in mRSS and FVC improvements after 24 months. 16 Interpreting these results, it has to be taken into account that the number of patients was low, baseline characteristics were not matched for most parameters, and the extent of SSc was rather more severe in the aHSCT group. Nevertheless, this combination therapy exhibits fewer treatment-associated adverse events than aHSCT and should be tested in future trials. Also emerging cellular therapies, like CAR-T-cell therapies or bispecific antibodies, should be evaluated against aHSCT, as to date, cyclophosphamide is the only comparator in randomized controlled trials.
Data on long-term outcomes are scarce; two studies describe the outcomes for a median follow-up of 7.5 years each.17,18 Overall survival after 5 years was about 85% in both studies, and thus similar to the finding in our study. In these studies, Georges et al. described 22 aHSCT patients, and Maltez et al. presented a matched analysis of 41 aHSCT patients versus 85 patients with non-transplant treatment regimens. There was a trend toward superior overall survival in the aHSCT group without reaching statistical significance. Response according to mRSS and FVC after 2 years was superior in the aHSCT group.
aHSCT aims to achieve a reset of the immune system, which was described for the first year after aHSCT 9 but can also be detected in the long term. The reduction of memory B cells was present until the second year after aHSCT. Within the T-cell compartment, there were long-lasting reduction of T helper cells long-lasting increase in cytotoxic T cells.
Another monocentric study found a significantly lower number of T helper cells in SSc patients responding to aHSCT. 19 On the other hand, the reduction of T helper cells is a well-known risk factor for certain opportunistic infections. Hence, the long-lasting reduction of T helper cells shows the importance of regular follow-up visits in the long term.
In our study, memory B cells were reduced until 2 years after aHSCT, which was already described for the first year after aHSCT by our group 9 and in a monocentric study in Brazil. 20 In the latter, B-cell reconstitution of 22 SSc patients was assessed up to 360 days after aHSCT. Naïve B-cell frequencies increased from 60 to 360 days post-aHSCT compared with pre-transplantation. Conversely, memory B-cell frequencies decreased during the same period. Both groups found an increase in interleukin-10 producing regulatory B cells, which illustrates the reset of the immune system after aHSCT.
Limitations
Limitations of our study are the single-center and retrospective design. Diverse immunosuppressive therapies after aHSCT, including rituximab, were used, which influences and complicates the interpretation of the lymphocyte regeneration.
Conclusion
This is one of the largest studies on long-term outcomes after aHSCT in SSc. aHSCT appears to promote positive long-lasting effects for SSc patients with reassuring safety data, considering that only the most severely affected and refractory SSc patients undergo aHSCT.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251397179 – Supplemental material for Clinical and immunological long-term outcomes after autologous hematopoietic stem cell transplantation in systemic sclerosis: a retrospective single-center experience
Supplemental material, sj-docx-1-tab-10.1177_1759720X251397179 for Clinical and immunological long-term outcomes after autologous hematopoietic stem cell transplantation in systemic sclerosis: a retrospective single-center experience by Michael Gernert, Jörg Henes, Patrick-Pascal Strunz, Hannah Labinsky and Marc Schmalzing in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Appendix
Acknowledgements
We thank Katharina Eckert, Vanessa Drescher, Silke Killinger, Anette Koss-Kinzinger, Isabelle Kuntzsch, Heide Pototzky, and Melissa Väth for expert technical assistance and advice.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
