Abstract
Background:
Janus kinase inhibitors (JAKi) are effective treatments for chronic inflammatory arthritis (CIA). Tofacitinib and baricitinib are pan-JAK inhibitors, while upadacitinib and filgotinib are JAK1-selective inhibitors.
Objective:
This study aimed to compare retention rates between JAKi classes and identify predictors of discontinuation.
Design:
Retrospective observational study.
Methods:
We retrospectively evaluated patients with CIA (Rheumatoid Arthritis (RA), Psoriatic Arthritis (PsA), and Spondyloarthritis (SpA)) treated with JAKis from 2017 to 2024. The independent variable was JAKi class (pan-JAK inhibitors/JAK1-selective inhibitors). Retention was assessed using Kaplan–Meier estimates, and predictors of discontinuation were evaluated through Cox multivariable regression.
Results:
The study included 181 patients (83% women; median age 56). Diagnoses were RA (84%), PsA (9%), and SpA (7%). A total of 227 JAKi treatment courses were analyzed: 96 JAK1-selective inhibitors and 131 pan-JAK inhibitors. Overall, 118 courses (52%) were discontinued (55% inefficacy; 42% adverse events). The discontinuation incidence rate (IR) was 35.5 per 100 patient-years, with 50% stopping treatment within 1.65 years. Discontinuation was more frequent in women, patients with cardiovascular (CV) disease or risk factors, and in PsA compared to RA. Discontinuation rates were lower for JAK1-selective inhibitors (IR: 28.6; 95% confidence interval (CI): 20.5–39.8) versus pan-JAK inhibitors (IR: 39.6; 95% CI: 31.9–49.1). Multivariate analysis confirmed that pan-JAK inhibitors, female sex, PsA, CV disease, prior targeted biological and synthetic disease-modifying drugs exposure, and leflunomide co-treatment increased the risk of discontinuation.
Conclusion:
Pan-JAK inhibitors may have higher discontinuation rates than JAK1-selective inhibitors in real-world CIA patients. Clinical characteristics and comorbidities should guide JAKi selection to optimize long-term treatment retention.
Plain language summary
This study looked at how long patients with chronic inflammatory arthritis (CIA) continue to take a type of drug called a Janus kinase inhibitor (JAK inhibitor or JAKis). These drugs help reduce inflammation in diseases such as rheumatoid arthritis (RA), psoriatic arthritis (PsA), and spondyloarthritis (SpA).
There are two types of JAKis:
a) Pan-JAK inhibitors, like tofacitinib and baricitinib
b) JAK1-selective inhibitors, like upadacitinib and filgotinib
The researchers reviewed the medical records of 181 people treated with JAKis between 2017 and 2024. Most of the patients were women, with a mean age of 56 years, and most had RA. The researchers studied how often patients stopped taking these medications, and the reasons for stopping. They found that about half of the treatments were stopped, mostly due to lack of effectiveness (the drug did not work well enough) or side effects. People were more likely to stop treatment if they had other health problems, like heart disease, or if they had PsA instead of RA. Notably, patients remained on treatment longer with selective JAK1 inhibitors than with pan-JAK inhibitors. Those who were female, had prior treatment or were also taking a drug called leflunomide were more likely to stop treatment early. In summary, this study demonstrates that not all JAKis work the same in all people. Rheumatologists must consider the health status of each patient when choosing the most appropriate treatment.
Introduction
Rheumatoid arthritis (RA) and spondyloarthritis (SpA) are two of the most prevalent immune-mediated inflammatory diseases. They are chronic inflammatory joint diseases that can lead to loss of function and permanent disability. The main goal of treatment is to achieve remission or at least minimal disease activity, preserve joint function, and improve quality of life.1–3
Their prognosis has improved in recent decades and may reflect earlier diagnosis and more accurate treatment, close monitoring, the treat-to-target strategy, and the development and implementation of various targeted biological and synthetic disease-modifying drugs (b/tsDMARDs), rather than a change in the characteristics of the process itself.4–6
There are currently several bDMARDs and tsDMARDs available for the treatment of RA, SpA, including psoriatic arthritis (PsA), including the following therapeutic classes: Tumor necrosis factor-alpha inhibitors (TNFi); interleukin-6 (IL-6) inhibitors; CD20-targeted agents; IL-17 and IL-12/23 inhibitors; PD4 inhibitors (apremilast); CTLA4-Ig and Janus Kinase inhibitors (JAKi). JAKi have created a new paradigm for the treatment of inflammatory diseases, improving clinical outcomes. Several JAKi are available in Europe for the treatment of RA and SPA: baricitinib, filgotinib, tofacitinib, and upadacitinib. Due to their central role in multiple cytokine receptor signaling, such as IL-6, IL-2, IL-12/23, and interferons, JAKi act at multiple immunological levels. Attending their selectivity, tofacitinib and baricitinib are defined as pan-JAK inhibitors, while upadacitinib and filgotinib as JAK1-selective inhibitors. 7
Clinical trial evidence supports an acceptable benefit-risk profile of JAKi, 8 with efficacy comparable to other bDMARDs.9–11 Although the side effects of JAKi are diverse and somewhat known, the selectivity of JAKi may mean that the side effects are not the same for each of them. 8 After the Oral Surveillance study, based on safety signals, namely malignancy and cardiovascular (CV) disease, the European Medicines Agency recommends caution when prescribing JAKi to patients aged over 65, those with high CV risk factors, smokers, cancer risk, or thrombosis risk, and advises their use only when no suitable alternatives exist. 12 These regulatory warnings highlight the need to select well the patients to whom JAKi is prescribed; however, it raises the question of whether this also applies to other, more selective JAKi.
In real-life conditions, JAKi tend to be introduced in patients with RA who inadequately respond to bDMARDs or methotrexate (MTX) in real-world settings, often considered “difficult-to-treat” RA.13,14 RA retention rates for JAKi in real-life settings at 12 months range from 51% to 68%.15,16
Real-world studies about the retention rate of JAKi on SpA and PsA remain limited, with data showing a 1-year retention rate of 52.7% 17 until 77% at 6 months. 18 In another study including a comparison between RA and PsA, significant differences in JAKi retention were not found. 19
This study aimed to assess the retention rate and to identify factors associated with JAKi discontinuation in chronic inflammatory arthritis (CIA) patients, considering JAKi selectivity.
Methods
Study design and patient population
The study was conducted at a public reference tertiary hospital, HCSC, in Madrid, Spain. It was an observational, retrospective longitudinal study involving patients with CIA who attended the HCSC rheumatology outpatient clinic. The study was conducted on patients with CIA (RA, PsA, and SpA). All patients met the classification criteria of the European Alliance of Associations for Rheumatology (EULAR), 20 the Assessment of Spondyloarthritis International Society (ASAS), 21 or the Classification Criteria for Psoriatic Arthritis (CASPAR). 22
Only patients treated with JAKi between October 2017 and March 2024 and with complete data on initiation and discontinuation dates, as well as the reasons for discontinuation, were included in this study.
Variables and data sources
Data were retrospectively collected from patients’ records until October 2024. Information was obtained from our electronic health record (EHR), which is used routinely in all outpatient visits.
The retention of treatment was defined as the time interval between treatment initiation and definitive treatment discontinuation. The reasons for discontinuation were classified into three primary categories, as outlined below: (1) lack of effectiveness (comprising both primary and secondary failure). Lack of effectiveness was determined according to the treating rheumatologist’s clinical judgment as recorded in the EHR, without systematic use of standardized disease activity indices; (2) adverse events (AEs; infection, cutaneous or systemic reactions, and other complications or malignancies); (3) other reasons (medical decision, patient preference). Drug retention was retrospectively assessed based on the time until definitive treatment cessation.
The independent variable was JAKi classification categorized as pan-JAK inhibitors (tofacitinib and baricitinib) versus JAK1-selective inhibitors (filgotinib and upadacitinib). Baricitinib was generally prescribed at 4 mg/day, with dose reduction to 2 mg/day in selected patients, mainly due to age or comorbidities. Filgotinib was prescribed at 200 mg/day in all cases. The others were prescribed at conventional dosages.
Other covariables were: (1) Demographic (age and sex at birth); (2) Baseline disease-related (date of symptoms, laboratory parameters (anti-cyclic citrullinated peptide antibodies, antinuclear antibody, rheumatoid factor, and HLA B27)); (3) Patient disease activity at JAKi starting time, measured with erythrocyte sedimentation rate (ESR); (4) Comorbidities (hypertension, dyslipidemia, diabetes mellitus, obesity, ischemic CV disease (recorded as a composite variable, without systematic subclassification into coronary artery disease, peripheral artery disease, or cerebrovascular disease), heart failure, chronic obstructive pulmonary disease, renal disease, thyroid disease, cancer history, and depression (all collected from the comorbidity section of the EHR)); (5) Exposure to other b/tsDMARDs during follow-up, encompassing the following groups: (a) TNFi, (b) anti-IL agents (IL-6 inhibitors, IL-17 and IL-12/23 inhibitors); (c, d) CD20-targeted agents (rituximab); CTLA4–Ig (abatacept); or PD4 inhibitors (apremilast). (6) Concomitant therapy: glucocorticoid use, and conventional synthetic DMARDs (csDMARDs); and (7) Calendar time: dividing the starting time of JAKis in year intervals based on treatment strategies and the commercialization of JAKi ((a) October 2017–December 10, 2020; (b) December 11, 2020–January 10, 2022; (c) ⩾January 11, 2022). 23
Patients fulfilling the inclusion criteria were identified through the pharmacy service using a computerized system. Four physicians independently reviewed medical records within the predefined study period, resolving discrepancies by consensus. Variables of interest, including the primary outcome, potential modifiers, and confounders, were prespecified in the protocol. Sociodemographic data, diagnoses, lab tests, treatments, and antibodies were extracted directly from clinical records, while discontinuation causes and comorbidities were classified according to treating rheumatologists’ documentation.
Statistical analysis
Results are reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) recommendations. 24 A descriptive analysis was conducted. Frequency distribution was used for qualitative variables, and mean and standard deviation or median and percentiles for quantitative variables. Bivariate associations were studied using the Student’s t-test for normally distributed continuous variables and the Mann–Whitney or Kruskal–Wallis tests for non-normally distributed continuous variables. Categorical variables were analyzed with the Chi-square or Fisher Test.
The study exposure period was defined as the time from the baseline visit (the start date of the first JAKi prescription) until the occurrence of any of the following events: loss to follow-up, JAKi discontinuation, loss of follow-up, or the end of the study (October 2024). Drug persistence was calculated as the number of patients remaining on treatment divided by the number of patients at risk of discontinuation using Kaplan–Meier survival curves (excluding censored patients) and considering any of the reasons for discontinuation (e.g., loss of efficacy and AEs) as a completion event. To estimate JAKi retention rates, survival techniques were used (allowing for multiple failures per patient), expressing the incidence rate of discontinuation (IR) per 100 patients*year with their 95% confidence interval (CI). Time of observation comprised the period from the JAK1 starting until loss of follow-up, main outcome, or end of study.
Time-to-event analyses were conducted using Cox proportional hazards regression, and results were reported as hazard ratios (HRs) with 95% CI. Bivariate Cox regression analyses were performed to evaluate associations between JAKi discontinuation and individual covariates. A multivariate Cox regression model was then constructed to assess the influence of JAKi selectivity and other clinically relevant variables on discontinuation, adjusting for age, sex, calendar time, concomitant therapy, baseline disease activity, type of diagnosis, as well as all covariates with p < 0.20 in the bivariate analyses. The final multivariate model was conducted using a backward stepwise regression approach. The way in which JAKi prescription was done in real-life conditions shaped the analysis. Patients were divided into periods according to the retention rate of each JAKi, which determined the presence or not of an event in that time frame. The analysis was run considering a patient-level clustering approach. Variables such as csDMARDs, NSAIDs, glucocorticoids, and calendar time were analyzed in a time-dependent manner. All these variables were divided into periods according to the retention rate of each JAKi, which determined the presence or absence of an event in that time frame. In every period, if a patient was taking any csDMARD for more than 6 months was considered exposed. Glucocorticoids and NSAIDs were considered exposed if the patient was on this medication for at least 3 months during the period analyzed.
We considered the influence of covariates if the prevalence was >10%, and if the frequency was lower (in comorbidities or several treatments), categories were grouped based on affinity. 25 The number of variables in the multivariate model followed the rule of Freeman. 26 Missing values of more than 10% were not used in the multivariate analysis. The proportional hazard assumption was tested using Schoenfeld residuals and the scaled Schoenfeld residuals. To evaluate the overall goodness-of-fit of the Cox regression model, we examined Cox–Snell residuals. The Nelson–Aalen cumulative hazard function of the residuals was then estimated and plotted against the reference line y = x. All statistical analyses were performed using STATA software. A two-tailed p-value under 0.05 was considered to indicate statistical significance.
Results
Baseline characteristics
The study population was selected among patients with CIA in our rheumatology outpatient clinic, treated with JAKi, and finally comprised 181 patients, 83.3% of whom were females. The median age at baseline was 56 (interquartile range (IQR) 49–63) years, and had a median disease duration of 9.7 (IQR 4–15) years. Of these, 84% had RA, 9% PsA, and 7% SpA, with a total follow-up of 332.32 patient-years, and a maximum follow-up of 8.9 years. Table 1 includes a detailed description of the baseline characteristics of the patients.
Description of baseline characteristics of patients.

CV risk factors*: include Dyslipidemia, Arterial hypertension, Diabetes mellitus and Obesity.
ACPA, anti-citrullinated peptide antibodies; CI, confidence interval; CIA, chronic inflammatory arthritis; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; CV, cardiovascular; ESR, erythrocyte sedimentation rate; JAKi, Janus kinase inhibitors; NSAIDs, nonsteroidal anti-inflammatory drugs; PsA, psoriatic arthritis; RA, rheumatoid arthritis; RF, rheumatoid factor; SD, standard deviation; SpA, spondyloarthritis; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
Regarding comorbidities, 15.5% of the sample were active smokers. 8.8% of the sample had a previous diagnosis of ischemic CV disease, and 54% had some CV risk factor (dyslipidemia, hypertension, obesity, diabetes mellitus). 15.5% of the sample were active smokers. The mean lag time from diagnosis to JAKi prescription was 11 years, and as shown in Table 1 and Figure 1, most of the patients used previous b/tsDMARDs. 79.7% of patients had received at least one previous b/tsDMARD (mainly TNFi), and 46.7% had failed at least two b/tsDMARDs. Regarding concomitant therapy, 42% of patients were receiving a combination treatment with a csDMARD, with 28% using two or more csDMARDs—MTX being the most common. Additionally, glucocorticoids were co-prescribed in 55.8% of the cases.

JAKi treatment courses flowchart.
During the follow-up, patients received 227 JAKi treatment courses, 96 with JAK1-selective inhibitors, and 131 with pan-JAK inhibitors. Baricitinib was prescribed in 95 (41.8%) patients, tofacitinib in 36 (15.8%), upadacitinib in 85 (37.5%), and filgotinib in 11 (4.8%) cases. Among patients treated with baricitinib, 88% received 4 mg/day and 12% received 2 mg/day. All patients treated with filgotinib received 200 mg/day. A single JAKi course was prescribed in 137 patients, while 54 and 13 patients received two and three consecutive b/tsDMARDs, respectively. Thus, nine patients had more than three courses of b/tsDMARDs (Figure 1).
Discontinuation and retention rates for JAKi
As we see in Figure 2, overall, 118 treatment courses (52%) were discontinued. Among the reasons for treatment discontinuation, 65 treatment courses (55.08%) were due to inefficacy, 3 treatment courses were due to medical decisions (2.54%), and 1 course by patient decision (0.85%). The other 49 treatment course discontinuations (41.59%) were due to AEs, of which 25.2% were related to pan-JAK inhibitors and 16.7% to JAK1-selective inhibitors (p = 0.143). Almost 80% of the AEs were non-serious (n = 38), with infections being the most frequent (n = 14). These included five opportunistic infections (three episodes of herpes zoster and two cytomegalovirus infections), seven upper respiratory tract infections, and two additional infections. Other non-serious AEs comprised 12 cases of malaise/intolerance, 6 laboratory abnormalities, 3 superficial phlebitis, 2 cases of newly diagnosed arterial hypertension, and 1 mild renal failure. No statistically significant differences in AE severity were observed between JAKi subtypes (pan-JAK inhibitors: non-serious 94.7%, serious 5.3%; JAK1-selective inhibitors: non-serious 95.8%, serious 4.2%; p = 0.7). Serious AEs included all discontinuations associated with hospitalization or death (n = 11; 4 in group treated with JAK1-selective inhibitors class and 7 in treated with pan-JAK inhibitors). Three deaths were reported: two from SARS-CoV-2 pneumonia (one in each treatment group) and one from gastric cancer (pan-JAK inhibitors). Other serious AEs requiring hospitalization were as follows: three respiratory infections unrelated to SARS-CoV-2 (one for JAK1-selective inhibitors and two for pan-JAK inhibitors), three episodes of diverticulitis (one for JAK1-selective inhibitors and two for pan-JAK inhibitors), one bilateral pulmonary embolism (pan-JAK inhibitors), and one case of non-melanoma skin cancer (pan-JAK inhibitors).
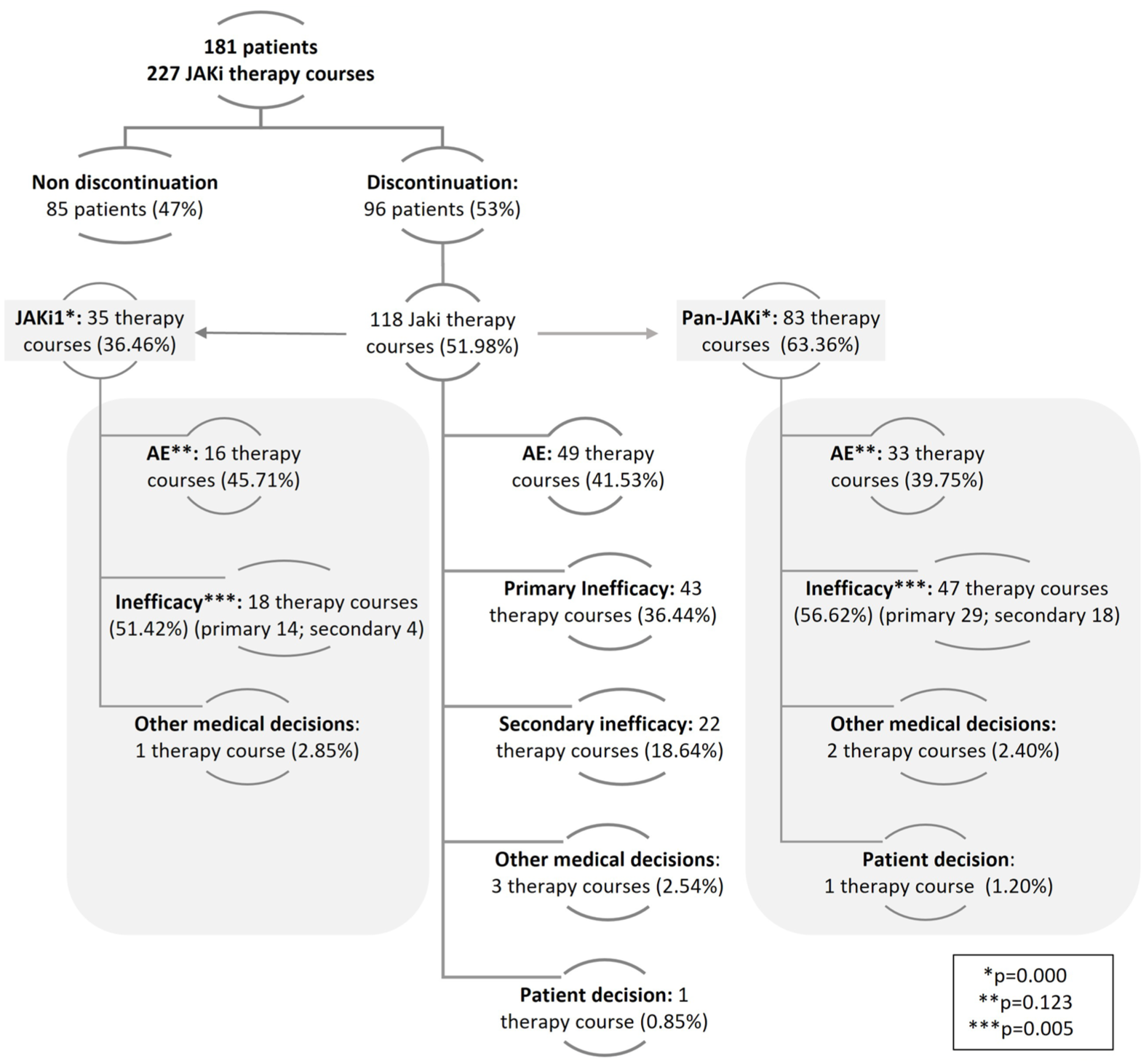
Causes of JAKi discontinuation: global and stratified.
Interestingly, 63% of patients treated with pan-JAK inhibitors discontinued compared to 36% of those receiving JAK1-selective inhibitors, and mainly due to greater inefficacy (p < 0.005; Figure 2).
The estimated IR of discontinuation was 35.5 (29.6–42.5) per 100 patients-year, with 23%, 39%, and 54% of failures at 6 months, at the first year and in the second year, respectively, and 50% of them discontinued within 1.65 years (1.2–2.2) from JAKi treatment initiation.
The IR of JAKi discontinuation was higher in women, with 39 (32–47) compared to men (IR: 18 (9.6–33.4)). Patients under 60 years of age at JAKi starting had an IR of 40.8 (32.8–50.8), those who started JAKi with an age between 60 and 70 years had an IR of 20.2 (12.4–3), and finally, patients with ages under 70 years had an IR of 39.1 (26.2–56.6). Regarding the underlying CIA diagnosis, IR of discontinuation was lower for RA (33.2 (27.3–40.4)) compared to other CIAs (57.3 (36.1–91.03)).
We found a higher IR in those patients with established CV disease (IR with: 55.2 (33.3–91.6) vs IR without: 29.7 (23.9–37)) and inpatients with CV risk factors (IR with: 34.4 (26.9–44.1) vs IR without: 28.1 (19.9–39.8)).
When stratified by JAKi classification, 35 treatment discontinuations occurred among patients receiving JAK1-selective inhibitors with an IR of 28.56 (20.50–39.78), while in pan-JAK inhibitors, there were 83 treatment discontinuations with an IR of 39.56 (31.90–49.05; Table 2).
Incidence rate of JAKi discontinuation.

CI, confidence interval; IR, incidence rate; JAKi, Janus kinase inhibitors.
Factors associated with JAKi discontinuation during follow-up
Bivariate analyses for discontinuation found that female sex ((HR 2.22 (p = 0.01)), previous courses of ts/bDMARD ((HR 1.37 (p = 0.01)), CV disease ((HR 1.94 (p = 0.009)), or dyslipidemia ((HR 1.50 (p = 0.03)), were statistically associated with JAKi discontinuation. Other baseline, comorbidity, age, or concomitant csDMARDs did not (Supplemental Table 1). JAK1-selective inhibitor showed a trend for better retention rate compared to pan-JAK inhibitors (p = 0.12; Figure 3).

Kaplan–Meier survival estimate curve.
Multivariate regression model, adjusted by calendar time, gender, age at diagnosis, CIA diagnosis, and CV risk factors, showed a higher probability of discontinuation for the pan-JAK inhibitors compared to the JAK1-selective inhibitor (HR: 1.99 (1.16–2.82); p = 0.01; Table 3). Of interest, JAKi were withdrawn more frequently in patients with higher number of previous b/tsDMARDs courses (HR: 1.25 (1.06–1.49); p = 0.01). Other factors that we found increase the risk for discontinuation were female sex (HR: 1.96 (1.01–3.81); p = 0.04), diagnosis of PsA versus RA (HR: 1.96 (1.15–3.32); p = 0.01), presence of CV disease (HR: 1.91 (1.27–2.86); p = 0.01), and combination therapy with leflunomide (HR: 1.78 (1.07–2.93); p = 0.02). Other variables did not influence and dropped off the final model. The proportionality of this regression model was tested with a p-value = 0.6, suggesting that this Cox proportional hazards model was appropriate for the data. The Cox–Snell residual plot indicates an acceptable overall fit of the Cox model. Minor deviations at higher residual values suggest a slight underestimation of risk in subjects with the highest predicted hazards.
Factors associated with JAKi discontinuation: multivariate analysis.

Adjusted by calendar time and cardiovascular risk factors.
CI, confidence interval; CIA, chronic inflammatory arthritis; HR, hazard ratio; JAKi, Janus kinase inhibitors; PsA, psoriatic arthritis; RA, rheumatoid arthritis; SpA, spondyloarthritis; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
Discussion
This study was conducted from a long-term retrospective group of patients with CIAs treated with all available JAKi at a single center in Madrid between 2017 and 2024. Our results showed that pan-JAK inhibitors (baricitinib and tofacitinib) exhibited higher discontinuation rates compared to JAK1-selective inhibitors (upadacitinib and filgotinib), regardless of other factors.
These data are in line with a recent comparative study, carried out in RA, where the overall drug retention rates were significantly lower in patients treated with pan-JAK inhibitors than in those treated with JAK1-selective inhibitors. 27 It seems that this improved retention for JAK-1 inhibitors may reflect differences in molecular selectivity and downstream cytokine signaling, which could influence both efficacy and tolerability. A study of JAK selectivity in RA supports that selective JAK1 inhibition is sufficient to drive therapeutic efficacy in RA. 28 Our observed discontinuation rate (42%) falls within the lower range of previously reported real-world data on JAKi persistence.15,16 This likely reflects the profile of our cohort, with long disease duration, prior b/tsDMARD exposure, and notable comorbidity burden, as well as changes in prescribing strategies over the long inclusion period.
Regarding age, the higher JAKi retention observed in the intermediate age group (60–70 years) is challenging to compare with other studies. This finding may reflect differences in patient characteristics, since age distribution varies by CIA diagnosis, patients have received fewer b/tsDMARD treatment courses, or treatment strategies may be less aggressive than in younger populations. Regulatory warnings regarding JAKi use in patients over 65 years may have influenced treatment discontinuation decisions, although real-world studies do not seem to reflect this impact. While JAKi safety could be affected by age-related factors—raising concerns when treating elderly patients due to comorbidities and potential changes in pharmacodynamics—JAKi retention rates appear to be comparable across age groups in clinical practice.29,30
EMA safety warnings after ORAL Surveillance likely encouraged more cautious JAKi use, especially in lower-risk patients. Recent registry data confirmed this shift and reported no excess major adverse cardiovascular event risk with JAKi versus TNFi (adjusted IR 0.89, 95% CI 0.63–1.25). 31 Such regulatory changes may have influenced retention patterns. Regarding gender as a factor influencing the IR of JAKi discontinuation, previous studies reported that female patients with CIA showed significantly worse disease activity and lower response to treatment.32,33 A higher rate of JAKi discontinuation for female patients with RA was already described in the ANSWER cohort. 34 These differences may be explained not only by physiological factors, but also by disparities in the perception and reporting of symptoms, as women tend to report worse outcomes in patient-reported outcome measures, which may influence clinical decision-making and perceived treatment effectiveness. 35
Interestingly, our findings suggested that patients with PsA had a higher risk of JAKi discontinuation compared to those with RA. Possible reasons could be that the number of approved JAKi for PsA is more limited and that PsA is frequently associated with a higher burden of comorbidities such as hypertension, diabetes, dyslipidemia, obesity, and other CV manifestations, which may impact both tolerability and clinical decision-making. 36 Consistent with our findings, a previous study reported lower JAKi survival rates in PsA compared to all other available b/tsDMARDs. 37 The varying responses to JAKi in RA, SpA, and PsA highlight that, despite shared pathogenic mechanisms, treatment should be tailored to each patient, considering the specific disease, patient profile, and treatment history.
CV disease has emerged as a concerning factor associated with JAKi discontinuation, and CV status should therefore be carefully assessed prior to initiating JAKi therapy. However, it must be kept in mind that real-world patients often present with a higher burden of comorbidities compared to those enrolled in randomized controlled trials.
Regarding our findings on JAKi discontinuation, we observed that withdrawal was more frequent among patients with a higher number of prior b/tsDMARD courses. These results are in line with a previous study reporting that prior failure of bDMARDs was a determinant of poor response to JAKi in RA. 38 These underscore the potential impact of treatment history on both the effectiveness and persistence of JAKi.
According to EULAR recommendations, MTX (or another csDMARD) should ideally be continued when starting treatment with JAKi, following a careful assessment of associated risks. 39 In our study, although not statistically significant, JAKi treatment persistence tended to be higher when combined with MTX. Conversely, the combination therapy with leflunomide was associated with a higher rate of discontinuation. While previous case series in RA have reported favorable outcomes regarding the efficacy and safety of the leflunomide–JAKi combination. 40 It is important to consider that in our study, the number of patients receiving leflunomide was quite small. Moreover, these patients had a longer disease duration, suggesting a more treatment-refractory profile, which might have influenced our results. Our findings also have health policy implications, as real-world retention data can inform regulatory decisions, reimbursement, and guideline development, especially regarding patient selection and safety considerations.
The main limitations of this study are those inherent to any retrospective observational study. We did not stratify our analysis by dose strength; although a small proportion of patients received baricitinib 2 mg/day, all filgotinib cases were prescribed at 200 mg/day. Differences in dosing may affect efficacy and safety and should be taken into account when interpreting our real-world findings. Prescription bias cannot be completely ruled out. In addition, given the extended follow-up, clinical practice has changed over time as different JAKi therapeutic options have become available, depending on the periods of commercialization. To mitigate this imbalance, we included the calendar time variable in the analysis. Another limitation is that this was a single-center study, with a considerable size difference among the JAKi treatment groups, reflecting the smaller sample for more recently approved drugs; therefore, grouping into broader categories was required. Finally, we did not report direct parameters of disease activity and instead used ESR as a surrogate marker. In addition, discontinuations due to lack of effectiveness were based on physician judgment rather than standardized disease activity indices, reflecting real-world decision-making. The main strength of this study lies in its longitudinal design with an inclusion period of 7 years, including the first, the second, and all subsequent JAKi courses until the end of the study, allowing a whole perspective of the disease course. As this is an observational study, we have included a broad non-selected real-world CIA patient’s spectrum with a variety of clinical and therapeutic options included in the multivariate analyses, allowing the adjustment for confounders to minimize potential bias. With all this in mind, we consider that these results add to the current knowledge on the real-world management of patients with CIA.
To date, there are very few studies that have compared the retention rates of JAKi in patients with CIA under real-world conditions (Supplemental Table 2). Therefore, findings provide novel insights into the differential persistence of JAKi classes, offering clinically relevant information that may support therapeutic decision-making in clinical practice. These results highlight the importance of considering both patient characteristics and prior treatment history when selecting a JAKi. Further studies are warranted to assess which JAKi type is most suitable for specific patient subgroups.
Conclusion
In summary, this real-world study makes a meaningful contribution by providing evidence on the incidence of JAK inhibitor discontinuation, which is in line with and expanding upon previous research. Our findings underscore that pan-JAK inhibitors (baricitinib and tofacitinib) may be associated with higher discontinuation rates compared with JAK1-selective inhibitors (upadacitinib and filgotinib) among patients with CIA. Importantly, we also observed that patients with PsA appear to experience higher discontinuation rates than those with RA, highlighting clinically relevant differences across disease subtypes. Taken together, these results not only reinforce the value of real-world data but also advance our understanding of JAKi treatment patterns, emphasizing the need for individualized therapeutic strategies that integrate diagnosis, comorbidities, and prior treatment history.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251393130 – Supplemental material for JAK1-selective inhibitors versus pan-JAK inhibitors: comparative real-world study of drug retention in chronic inflammatory arthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X251393130 for JAK1-selective inhibitors versus pan-JAK inhibitors: comparative real-world study of drug retention in chronic inflammatory arthritis by Leticia Leon, Pedro Pablo Bermejo, Leonor Laredo, Jose Alberto Peña Pedrosa, Maria Teresa Benítez Gimenez, Dalifer Freites, Clara de Miguel and Lydia Abasolo in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251393130 – Supplemental material for JAK1-selective inhibitors versus pan-JAK inhibitors: comparative real-world study of drug retention in chronic inflammatory arthritis
Supplemental material, sj-docx-2-tab-10.1177_1759720X251393130 for JAK1-selective inhibitors versus pan-JAK inhibitors: comparative real-world study of drug retention in chronic inflammatory arthritis by Leticia Leon, Pedro Pablo Bermejo, Leonor Laredo, Jose Alberto Peña Pedrosa, Maria Teresa Benítez Gimenez, Dalifer Freites, Clara de Miguel and Lydia Abasolo in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
