Abstract
Background:
Chronic non-bacterial osteomyelitis (CNO) is a rare autoinflammatory disease characterized by bone pain and sterile bone inflammation.
Objectives:
This study aimed at describing clinical presentation, laboratory and imaging features, densitometric assessment, and therapeutic approaches in a monocentric CNO cohort.
Design:
Monocentric retrospective cohort study.
Methods:
Patients were included in the Chronic non-bActerial osteoMyELitis: A mOnocentric regisTry (CAMELOT) in case of (i) clinical diagnosis of CNO, (ii) the fulfilment of diagnostic/classification criteria, (iii) at least one medical visit at our institution between June 2004 and June 2024, and (iv) a follow-up of at least 3 months.
Results:
Fifty-one patients were included in the CAMELOT (mean age at onset 11.14 years, 68.6% females). A total of 39.2% of patients presented an autoimmune comorbidity. Bone pain was invariably present; joint involvement was demonstrated in 9.6% of patients. Most subjects (86.3%) had multifocal disease: the median number of bony lesions per patient was 4. Lumbar spine dual-energy X-ray absorptiometry (DXA) was performed in 14 patients (27.4%), showing a reduced bone mineral density (BMD) (−1.14 ± 1.21 Z-score). A total of 18 patients (35.3%) experienced a fracture on a bony lesional site; 14 patients (27.4%) presented vertebral fractures (VFs) at onset, with multiple VFs in 5 cases (35.7%). BMD Z-scores (−1.7 vs −0.5) were lower in patients with VFs, with a higher rate of “low BMD for age” (37.5% vs 16.7%) compared to patients without VFs.
Conclusion:
The high prevalence of VF observed in the CAMELOT cohort highlights the importance of enlisting CNO in the diagnostic approach to pediatric patients presenting with VF.
Plain language summary
In our cohort of 51 patients with chronic non-bacterial osteomyelitis, vertebral fractures occurred rather often (27.4% of cases). Patients with vertebral fractures have a low bone density compared to those without.
Introduction
The earliest description of the clinical picture of chronic non-bacterial osteomyelitis (CNO) dates to 1972, when Giedion observed four patients with symmetrical osteolytic lesions of the growth plate of long bones with a subacute recurrent course. 1 Six years later, a Swedish group depicted nine additional cases with a similar disease, although six subjects also presented palmo-plantar pustulosis and clavicle inflammation. 2 The histological picture consisted of sterile osteomyelitis, with predominant neutrophil infiltration. 2
The landmark clinical feature of CNO, including the severe form chronic recurrent multifocal osteomyelitis (CRMO) characterized by multiple recurrent lesions, is bone pain. Bone lesions can result in gait disturbance and limited range of motion; constitutional symptoms, such as low-grade fever and malaise, are common findings. 3 Laboratory tests are usually within the normal range with a mere slight increase in erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP). 3 Standard radiology can demonstrate osteolysis, osteosclerosis, and hyperostosis; in later stages, bone deformities can supervene due to disease damage. Given the potential polyostotic presentation, total body imaging is required: whole-body magnetic resonance imaging (WB-MRI) is now the technique of choice in defining the extent of bone involvement.4 –6
More than 50 years after its description as a distinct entity, CNO is now regarded as an autoinflammatory bone disorder.3,7 –10 Driven by innate immunity cells, the unbalanced production of cytokines, such as IL-1β and IL-10, culminates in the uncontrolled activation of osteoclasts. Consistent with the autoinflammatory pathogenic paradigm, CNO often coexists with extra-osseous manifestations such as peripheral and axial arthritis, psoriasis, palmo-plantar pustulosis, and gut inflammation. 11 Many unmet needs still exist in the CNO scenario, such as the poorly elucidated pathogenesis, the scarcely standardized therapeutic approach, and the unclear long-term course and outcome of the disease.
We hereby describe a cohort of 51 patients with CNO enrolled over 20 years at a single referral center, also providing data on bone health by means of lumbar DXA BMD Z-score and first-level bone metabolism markers. Considering the rarity of CNO,10,12 we believe our clinical experience could provide novel, valuable insights into this still enigmatic condition.
Methods
Patients were retrospectively recruited in the monocentric Chronic non-bActerial osteoMyELitis: A mOnocentric regisTry (CAMELOT) in case of fulfillment of the following inclusion criteria: (i) clinical diagnosis of CNO between June 2004 and June 2024, (ii) the fulfillment of diagnostic/classification criteria,13,14 and (iii) a follow-up of at least 3 months since diagnosis. Patients were excluded from the registry in case of (i) another diagnosis formulated after that of CNO that could better explain the whole clinical picture and (ii) a follow-up shorter than 3 months.
Demographic, clinical, laboratory, and radiological data were anonymously extracted from clinical records. Patients were sub-classified into two groups upon the decade of diagnosis (2004–2014 and 2015–2024).
Age-related reference values of weight, height, and body mass index (BMI, kg/m2) were obtained from a wide Italian pediatric population. 15 Auxological data were normalized for chronological age by conversion to standard deviation scores (SDSs).
The following clinical data were collected: bone pain, local tenderness, local swelling, constitutional symptoms, and extra-skeletal manifestations (acne, psoriasis, palmo-plantar pustulosis, inflammatory bowel disease (IBD), coeliac disease, peripheral or axial arthritis, enthesitis or sacroiliitis). Sacroiliitis and axial arthritis were diagnosed upon MRI findings. 16
Lytic, sclerotic, or hyperostotic lesions and periosteal reaction were assessed by plain radiographs. The number and sites of active lesions were investigated by WB-MRI or bone scan. MRI also allowed for the detection of soft-tissue edema surrounding bony lesions. A bone biopsy was performed upon the clinician’s judgment.
Laboratory parameters were collected. ESR and CRP were regarded as increased when above the upper cutoff of 20 mm/h and 0.5 mg/dl, respectively. Neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) were calculated, and reference ranges adjusted for age were adopted. 17 Second-to-fourth lumbar spine BMD (g/cm2) was assessed by DXA (Hologic Delphi Dexa scanner), and results were automatically converted to sex- and age-matched Z-score. 18 According to the International Society for Clinical Densitometry (ISCD), a BMD Z-score lower than −2.0 SD was classified as “low BMD for age.” 19 Vertebral fracture (VF) was defined as the reduction ⩾20% of vertebral height according to Genant’s score; VFs were further classified into mild, moderate, and severe in case of a reduction of height of 20%–25%, 25%–40%, and >40%, respectively. 20
Data regarding the following treatments were recorded: antibiotic therapy for suspected infective osteomyelitis, non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids (GCs), conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs, as methotrexate (MTX) and sulphasalazine (SSZ)), biological DMARDs (bDMARDs, tumor necrosis factor-α inhibitors (TNFi) and interleukin (IL)-6 or IL-1 inhibitors), and intravenous amino-bisphosphonates (IV-BPs). Treatment response was clinically and radiologically assessed semi-quantitatively (complete, partial, and no response). Partial radiological response was defined as the visual improvement or decrease of the number of bony lesions, while no response was defined as the persistence, the occurrence, or new ones; complete response was defined in case of pain disappearance, negative inflammatory markers, and MRI or bone scan normalization.
Statistical analysis
Demographic, clinical, laboratory, and radiological values were described as frequencies (%) if discrete and mean ± standard deviations or median (interquartile range (IQR)) if continuous with normal or without normal distribution, respectively. The normality of data distribution was evaluated by the Shapiro–Wilk test. Comparisons between the groups were investigated by T or Mann–Whitney tests for continuous data and χ2 or exact Fisher’s test for categorical ones. Correlations between variables were assessed by Pearson’s and Spearman’s tests as appropriate. Potential patterns in multiple bone localizations were assessed through the Cohen κ test. Statistical analyses were performed with SPSS-IBM (IBM SPSS Statistics 28.0.1.1, Armonk, NY, USA).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 21
Results
Of the 60 patients initially identified, nine patients were excluded (Supplemental Material), leading to a final sample of 51 patients (Table 1). In the 36 subjects (70.4%) undergoing bone biopsy, histological analysis invariably evinced sterile inflammation, osteolytic lesions, bone marrow fibrosis, and hyperostosis.
Baseline demographic details, clinical features, radiologic findings, laboratory parameters, and first-line treatment of the 51 patients included in the CAMELOT cohort.
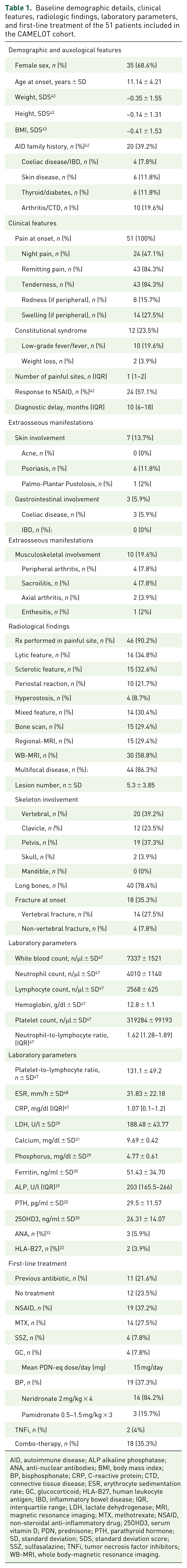
AID, autoimmune disease; ALP alkaline phosphatase; ANA, anti-nuclear antibodies; BMI, body mass index; BP, bisphosphonate; CRP, C-reactive protein; CTD, connective tissue disease; ESR, erythrocyte sedimentation rate; GC, glucocorticoid; HLA-B27, human leukocyte antigen; IBD, inflammatory bowel disease; IQR, interquartile range; LDH, lactate dehydrogenase; MRI, magnetic resonance imaging; MTX, methotrexate; NSAID, non-steroidal anti-inflammatory drug; 25OHD3, serum vitamin D; PDN, prednisone; PTH, parathyroid hormone; SD, standard deviation; SDS, standard deviation score; SSZ, sulfasalazine; TNFi, tumor necrosis factor inhibitors; WB-MRI, whole body-magnetic resonance imaging.
The average age at disease onset was 11.14 years (±4.21) with female predominance (female:male ratio of 2:1). Two patients only experienced symptom onset in adulthood (a 31-year-old female and a 26-year-old male).
Patients were subgrouped according to the quartiles of the distribution of the variable age at onset as follows: ⩽9 years (n = 18); 9–10 years (n = 9); 10–13 years (n = 13); and >13 years (n = 11).
Twenty patients (39.2%) had a family history positive for autoimmune diseases (Supplemental Table 1); the same rate of subjects (39.2%) presented concurrent autoimmune comorbidities.
Clinical presentation and imaging features
Bone pain emerged as the most common presenting symptom, being reported by all patients, especially at night (24 subjects, 47.1%), mostly with one or two painful sites; a single patient complained about diffuse aches without any specific localization. A remitting course of bone pain was described by 43 patients (84.3%). On clinical examination, tenderness at the pain site was present in 43 patients (84.3%), while local redness and swelling were detected in 8 (15.7%) and 14 (27.5%), respectively. Long bones were the most common lesion site (78.4%, Table 1), followed by vertebral and pelvis involvement (39.2% and 37.3%, respectively). The median number of bony lesions per patient was 4 (IQR 2–8; range 1–19). Five patients (9.8%) performed regional MRI alone (in toto spine (2 patients), both knees (1 patient), both ankles/foot (1 patient), and coxofemoral joint and femur (1 patient)). No specific pattern in the main skeletal localizations (long bones, pelvis, and vertebral involvement) emerged (κ for long bones–pelvis involvement: 0.075; vertebral–pelvis involvement: 0.129; vertebral–long bones involvement: −0.188). Pelvis involvement was associated with a higher number of skeletal lesions (median number of lesions: 5 (4–8) vs 3.5 (2–7), p = 0.033). Seven patients (13.7%) showed monostotic disease: two patients (3.9%) presented clavicle involvement, while four subjects (9.8%) exhibited distal femoral metaphysis, proximal tibial metaphysis, sternal body, and D5 vertebral body localization, respectively. Figures 1 and 2 visually detail non-vertebral and vertebral sites of bone pain and bone involvement.
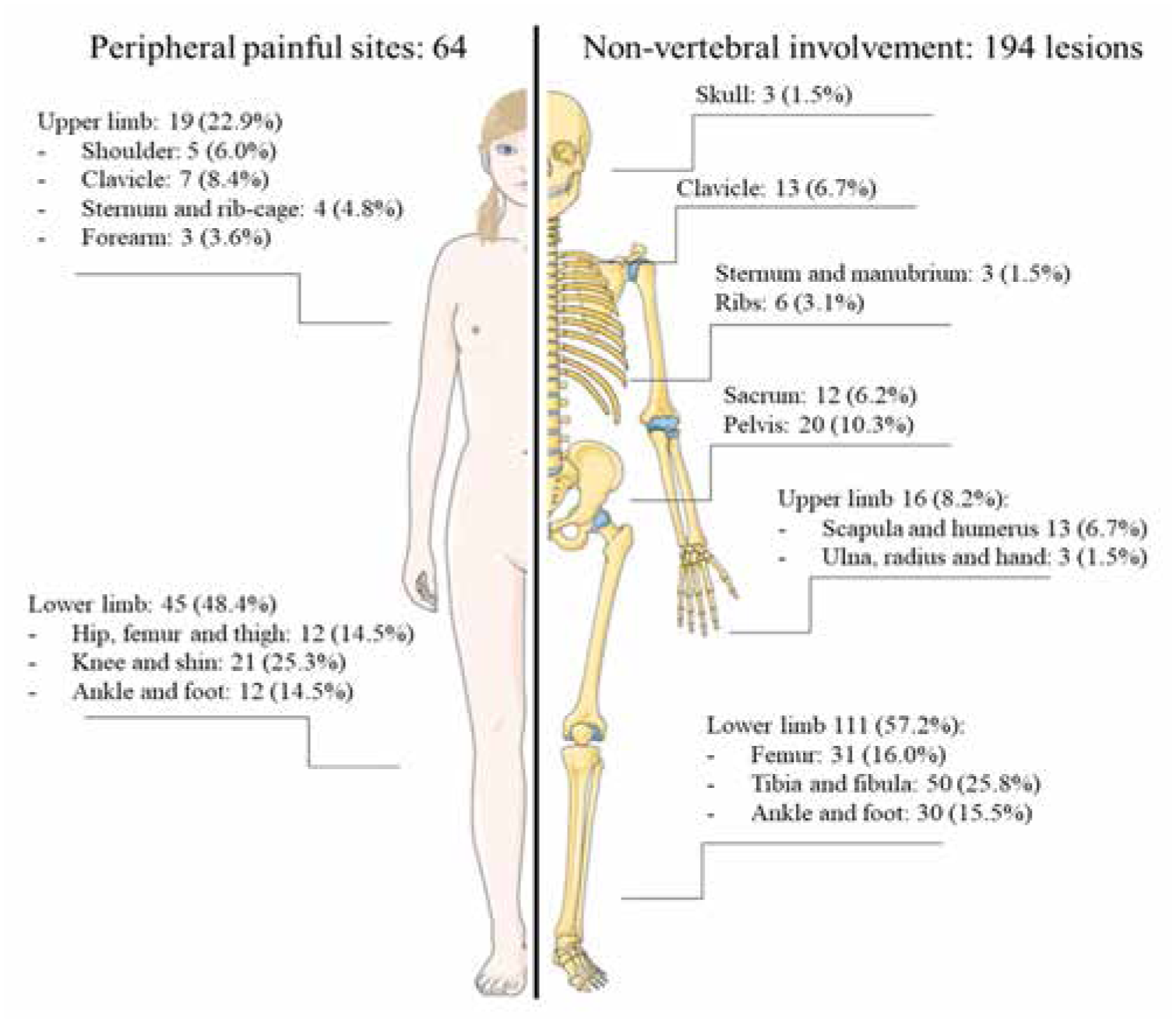
Visual representation of non-vertebral painful skeletal sites (left) and bony lesions at imaging (right).
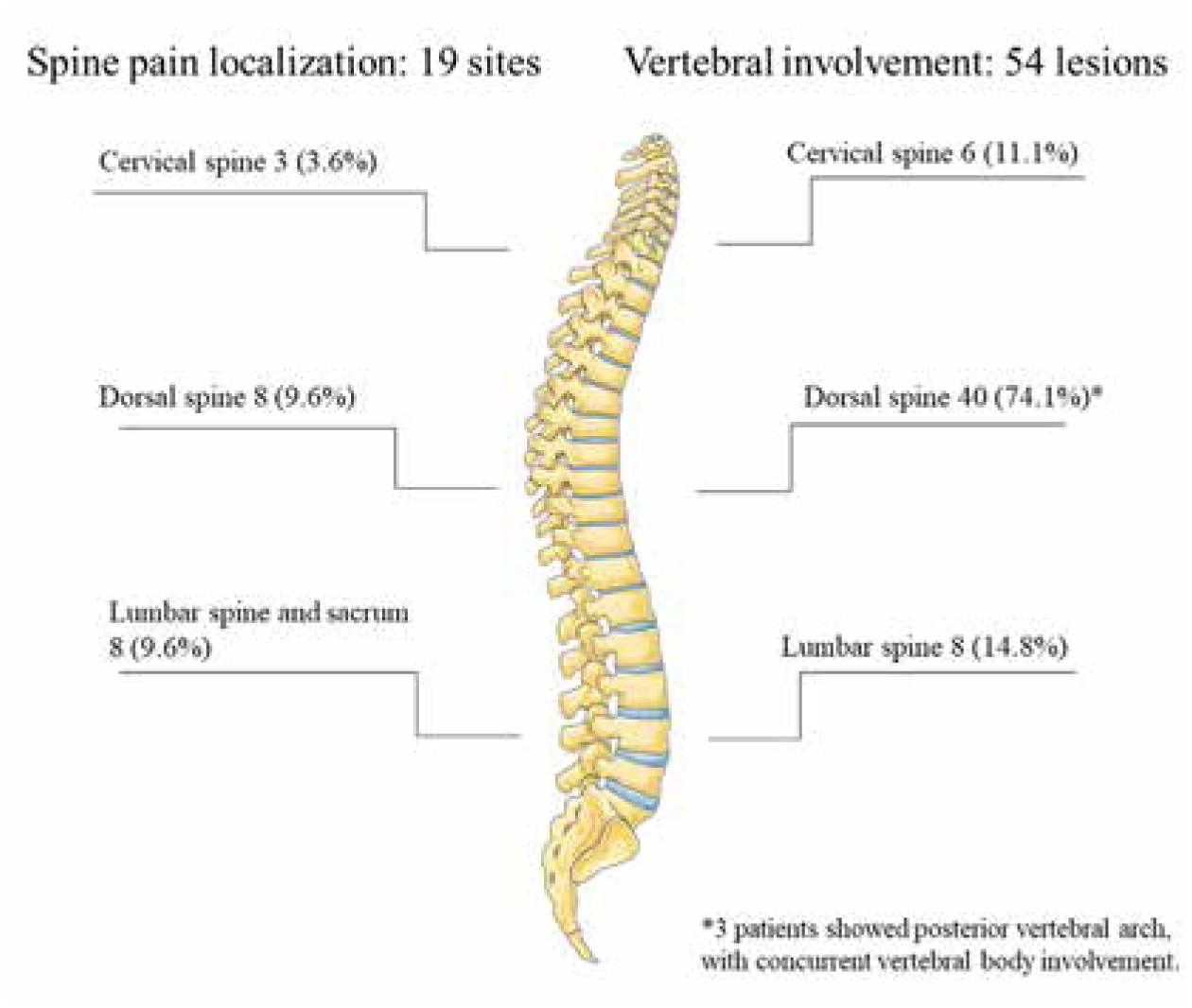
Visual representation of vertebral painful skeletal sites (left) and bony lesions at imaging (right).
Twelve patients (23.5%) complained of systemic symptoms: low-grade fever and weight loss were reported by 10 (19.6%) and 2 (3.9%) patients, respectively.
Joint involvement was the most common extra-skeletal manifestation (five patients, 9.6%). Three patients (3.8%) had peripheral arthritis (two ankle and one knee). In 7.8% of cases, peripheral arthritis and sacroiliitis coexisted, while chondro-sternal or costo-vertebral joint arthritis was associated with enthesitis in 3.9% and 2% of patients, respectively. Axial arthritis tended to be more common in patients aged 9–10 years at onset (p = 0.023) and in those with clavicle involvement (p = 0.061); psoriasis was more frequent in cases of later onset (10–13, p = 0.031).
First-line therapeutic options are detailed in Table 1 and Supplemental Table 2. Twelve patients received no treatment: in 4 patients (33.3%), pain spontaneously remitted; radiologically, one patient experienced a progressive reduction of bony lesions, whereas in the three other cases, disease did not progress. Due to radiographic progression, MTX was later introduced in three patients (25%), one with a monofocal disease and one with a multifocal disease. Two patients (16.7%) with polyostotic disease were prescribed NSAIDs as pain supervened at 3 months from diagnosis. Three patients (25%) received IV-BPs due to subsequent spine involvement.
Combo therapy (combination of more than one cs/bDMARDs) was the most common regimen adopted in patients with night pain (p = 0.020) or cutaneous erythema over bony lesions (p = 0.010). Therapeutic choices were not affected by any radiological or laboratory variable, including the number of lesions. Vertebral involvement was associated with a lower response to previous NSAIDs (28.6% vs 71.4%, p = 0.008), and higher use of IV-BP as treatment options (73.7% vs 26.3%, p < 0.001). Long bones involvement was associated with higher use of MTX (100% vs 0%, p = 0.019). Differences between patients presenting in 2004 and 2014 as compared to those diagnosed in the subsequent decade related to the therapeutic approach, as well as to clinical presentation and radiological features (Table 2). Antibiotic treatment and SSZ were more commonly prescribed in pre-2014 patients (18.2% vs 3.4%, p = 0.019 and 18.2% vs 3.4%, p = 0.019). GCs were only used in pre-2014 patients; conversely, MTX and biologics were prescribed only after 2014 (48.3% vs 0%, p < 0.001). Combination treatment was chosen for nine patients in 2014–2024, but for any in the previous decade (p = 0.003).
Clinical features of CNO in patients diagnosed before and after 2014.

AID, autoimmune disease; BMI, body mass index; BP, bisphosphonate; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; GC, glucocorticoid; GI, gastrointestinal; IQR, interquartile range; MTX, methotrexate; NLR, neutrophil-to-lymphocyte ratio; NSAID, non-steroidal anti-inflammatory drug; PLR, platelet-to-lymphocyte ratio; PPP, palmo-plantar pustolosis; SD, standard deviation; SDS, standard deviation score; SSZ, sulfasalazine; TNFi, tumor necrosis factor inhibitors; WB-MRI, whole body-magnetic resonance imaging.
Bold identifies statistical significance.
At 3–6 months after diagnosis, complete pain relief was described by 28 patients (54.9%) while radiological remission was achieved only by five subjects (9.8%). Complete response was attained in four of the 39 patients (10.3%) with available radiological follow-up (Figure 3).

Rates of overall response (a), pain response (b), and radiological response (c) to first-line treatment in the CAMELOT cohort.
Laboratory findings
ESR and CRP above the reference range were detected in 27.4% and 11.7% of patients, respectively. HLA-B27 was tested in 22 patients and found only in 2 patients (3.9%): both had a high number of active lesions at WB-MRI (eight and four, respectively). One subject showed bilateral sacroiliitis together with involvement of parietal bones and peripheral metaphysis; the other patient exhibited bilateral proximal femoral metaepiphysis and D6/D7 involvement. No abnormalities in serum calcium and phosphorus emerged in the whole population. Alkaline phosphatase (ALP) values were found to be higher than references in three patients and lower in one. Most subjects (58.6%) presented a low-normal ALP. 22 NLR and PLR were above the age reference upper limit in 2 (4.4%) and 4 (8.8%) patients, respectively. 17
Imaging investigations
Plain radiograph on the most painful site, available for 47 patients (90.4%), did not show any pathological change in 38.3% of cases (18 subjects). In the remaining cases, lytic lesions were the most common radiological abnormality, followed by sclerotic lesions and hyperostosis (Table 1). Clavicle involvement was associated with periosteal reaction (45.4% vs 14.3%, p = 0.029) and hyperostosis (27.3% vs 2.9%, p = 0.037), with a higher number of concomitant radiological abnormalities (p = 0.061). Total-body imaging was available for the whole cohort, but six patients: 15 (29.4%) performed Tc99m-Bone Scan and 30 (58.8%) WB-MRI. Total body imaging identified disease multifocality in 86.3% of cases; only seven patients presented a single lesion (13.7%). No correlation existed between the number of painful sites and the number of radiological lesions. Patients with periosteal reaction displayed higher ESR values (43.87 ± 15.99 vs 26.8 ± 20.52 p = 0.034), while a trend toward statistical significance emerged for bone sclerosis and raised inflammatory markers (ESR 39.23 ± 18.88 vs 26.97 ± 20.45 mm/h, p = 0.053 and CRP 0.85 (0.11–1.87) vs 0.2 (0.1–0.54) mg/dl, p = 0.052, respectively). Soft-tissue edema identified by regional or WB-MRI was associated with higher CRP (0.7 (0.32–1.75) vs 0.1 (0.07–0.27), p < 0.001) and ESR values, although not significantly (36.16 ± 23.26 vs 25.33 ± 19.47, p = 0.093).
Bone fractures
Bone fractures in lesional sites were a prevalent complication at presentation, being detected in 32.7% of the whole cohort (18 patients), for a total of 28 fracture events. Vertebral fractures (VFs) were the most common, with 22 VFs in 14 patients (27.4%) at onset (Table 3). Multiple VFs were detected in five patients (35.7%): three with two VFs and two with three VFs. Most commonly, VFs were localized in the dorsal spine (86.4%), while the other two fractures were localized on C7 and C6. Eight VFs were classified as mild, while two as moderate and four as severe (all involving D7 but a single one C7); only two severe fractures involved the posterior wall of the vertebral body (“vertebra plana”). All patients complained of spine pain. No patients showed neurological complications secondary to VFs; five patients were diagnosed with concurrent scoliosis, while secondary kyphosis was present in seven patients. During follow-up, two further VFs occurred after 15 months and 10 years, for a total of 24 VFs. One of these two patients showed a unifocal disorder in the distal metaphysis of the left tibia at disease onset; he then developed vertebral involvement with a first D1 VF while receiving MTX. After a switch to etanercept, the patient experienced C7 VF. The other one had multiple vertebral lesions at disease onset with a subsequent C6 VF while on MTX, neridronate, and NSAID. Six patients were prescribed with orthopedic C35 brace, one with a Milwaukee brace. Patient with C7 VF was prescribed with orthopedic rigid collar. No associations were found between the presence of VF at onset and any clinical, laboratory, or radiological baseline finding. The remaining four non-VFs (7.8%) involved the ischiopubic branch, second metatarsal bone, distal femoral metaphysis, and calcaneus.
Demographic and clinical features of patients subgrouped upon the occurrence of vertebral fractures.

AID, autoimmune disease; BP, bisphosphonate; GC, glucocorticoid; MTX, methotrexate; NSAID, non-steroidal anti-inflammatory drug; SSZ, sulfasalazine; TNFi, tumor necrosis factor inhibitors; VF, vertebral fracture; WB-MRI, whole body-magnetic resonance imaging.
Bold identifies statistical significance.
Bone health assessment
Lumbar spine DXA was available for 14 patients (Table 4). All but one patient—a 31-year-old female—received the diagnosis at a pediatric age. DXA was performed in most patients at CNO diagnosis (median months from diagnosis to DXA 0 (0–0.5)); in two cases, DXA was performed more than 6 months after diagnosis: one patient without previous fracture underwent DXA 30 months after the diagnosis was formulated, and another one with three previous VF performed DXA 52 months after the diagnosis.
Bone health parameters of 14 patients included in the CAMELOT cohort.

ALP, alkaline phosphatase; BMD, bone mineral density; BMI, body mass index; CRP, C-reactive protein; DXA, dual-energy X-ray absorptiometry; ESR, erythrocyte sedimentation rate; IQR, interquartile range; IV-BP, intravenous bisphosphonate; PTH, parathyroid hormone; SD, standard deviation; SDS, standard deviation score; VF, vertebral fracture.
Despite a high number of lesions per patient (median 4.5, IQR 2.75–8; range 1–12), none of the subjects with available DXA had involvement of lumbar vertebrae at WB-MRI. Four patients (28.6%) were classified as “low BMD for age,” presenting a BMD Z-score <−2.0.
No clear abnormality was identified in markers of bone metabolism. ALP values standardized for age were at the low end of the reference range in all but one patient (a 9-year-old female with 12 bony lesions and a VF with ALP of 125 U/l). 25-OH vitamin D3 levels were only slightly lower than optimal 23 ; serum calcium and phosphorus were both at the higher end of the reference range.
Two patients (14.3%) had received IV-BPs before DXA: a 6-year-old male was treated with IV pamidronate 0.5 mg/kg/d for three non-consecutive days 4 months before DXA, which showed a BMD Z-score of −1.4 SD. A 12-year-old male was treated with IV pamidronate 1 mg/kg/d for three non-consecutive days with a maintenance of 1 mg/kg/d monthly for five times and later quarterly for 1 year before DXA, which reported a BMD Z-score of 0.8 SD. Neither patient experienced any further fracture.
Importantly, the number of lesions was negatively correlated to BMD Z-score (ρ = −0.627, p = 0.016).
Out of the 14 subjects, 8 patients (57.1%) experienced at least one VF with a total of 15 VFs. BMD Z-score was lower—even though not significantly—in the VF group (−1.7 (−2.3 to −0.95) vs −0.5 (−2.05 to 0.52)). BMD Z-score was not associated with the presence of VF (p = 0.228) nor with the number of VF (ρ = −0.402, p = 0.155). Inflammation parameters, ESR and CRP, did not correlate with either BMD Z-score (p = 0.554 and p = 0.436, respectively) or with the number of VF (p = 0.820 and p = 0.646, respectively).
In the comparison of patients with low BMD for age to those without, interesting findings emerged in reference to the inflammatory burden: patients with low BMD for age displayed higher ESR and CRP values (61 (37.75–88) vs 21 (11.75–41), p = 0.036 and 1.75 (0.82–4.02) vs 0.5 (0.15–0.8), p = 0.034 respectively) and a higher, although not significantly, number of lesions (8 (5.75–11) vs 4 (1.75–6.5), p = 0.054); no differences among auxological parameters and disease duration at the time of DXA performance could be found.
Discussion
The monocentric CAMELOT cohort provides clinically relevant insights into a still elusive condition as CNO. First, we hereby report a high rate of VFs at disease onset, 27.4%. Such a figure appears to be the highest to date registered in literature, where prevalence ranges between 4.7% and 13.5%.8,9,24,25 In our cohort, VFs were mostly located in the thoracic spine; two VFs involved the cervical spine: a single cohort had previously described cervical spine VFs in two pediatric patients 24 while another cohort described one in a 62-year-old patient. 26 Interestingly, none of our patients developed lumbar VF. In the CAMELOT cohort, 20% of the 20 VFs could be defined as severe. Previous data can be extrapolated only from the work by Rogers, who has identified severe height loss in 6 out of 36 vertebral bodies (15%). In all our cases, VFs were symptomatic, while in the US cohort, one-fourth of the patients denied any pain. 24 Noteworthy, VFs occurred early in the disease course, warranting intensive treatment to control pain and prevent deformities due to the altered distribution of spinal loads in a growing skeleton. 27 Only two patients developed two VFs during follow-up. Vertebral involvement was registered in 39.2% of included patients, being the second most prevalent involvement site after long bones. Other cohorts observed much lower rates of vertebral involvement, ranging between 16 and 23%,3,9 peaking at 28.2% in the large and recent US study on 208 patients. 24
In our cohort, peripheral bone lesion leads to an insufficiency fracture in 7.8% of cases, but only in 2.1% of weight-bearing bones. In other studies, pathologic fractures occurred much less commonly, being registered in less than 1% of patients. 9
Bone health assessment revealed a low BMD for age in approximately one-third of the CAMELOT cohort, a finding that might be ascribed to several contributing mechanisms. The inverse correlation between BMD and the number of lesions, as well as the higher inflammatory markers and number of lesions registered in patients with low BMD for age, unveils a detrimental role of systemic inflammation underpinning disease activity; additional potential contributors enlist bone detrimental therapies and pain-induced restriction of physical activity. It should also be considered that, in children with CNO, a correct interpretation of BMD by DXA might be impinged by disease-specific bone pathological changes as hyperostosis and bone sclerosis, besides the well-known limitations affecting DXA in the general pediatric population. 28 To note, in patients with SAPHO, the adult-onset counterpart of CNO, trabecular bone score (TBS) has been demonstrated not only to be unaffected by bone artifacts but also to more reliably identify VF compared to DXA BMD. 29 Rather unexpectedly, patients with VF presented BMD Z-scores not significantly different from those not experiencing VF; a potential explanation might relate to the fact that VFs are mainly pathological fractures due to bone erosion and/or osteitis. In two patients who had previously received IV-BPs, BMD resulted within the normal range.
Literature about bone health assessment in CNO is very scarce, being limited to a single Japanese study that observed lower BMD Z-score values in almost all CNO patients compared to a sex- and age-matched population with low-for-age BMD Z-score values. In the Japanese cohort, the rate of spine involvement was much lower than in the CAMELOT (21.4%); thus, such findings might be possibly explained by the overall higher disease burden registered in this Asian population. 30
We could not replicate the Japanese observation on increased serum band 5 Tartrate-Resistant Acid Phosphatase (TRAP5b) and bone-specific ALP. 30 In our cohort, ALP, available for 10 patients only, resulted in low-normal range for age 22 in all but one patient, with a trend toward lower serum ALP and phosphorus levels for those with fractures. Together with low-for-age BMD levels, these findings might be underpinned by the activation of osteoclasts occurring in early CNO. 2
In the CAMELOT, NLR and PLR were out of range for pediatric population references in inflammatory conditions in 10% of cases. 17 These two indices have been proven useful in detecting low-grade inflammation in chronic diseases, including rheumatoid arthritis.31,32 A recent study assessed inflammatory parameters and systemic inflammation-based scores in adult CNO patients, 33 reporting similar NLR and PLR to healthy controls.
Another interesting finding that emerged in the CAMELOT registry relates to the elevated rate of polyostotic disease, 86.3%, for those with an available WB-MRI or Bone scan. This figure is higher than previous reports, where monostotic involvement was reported in 23%–48.2%,3,9 highlighting the pivotal importance of total-body techniques to precisely characterize bone involvement. WB-MRI is not only the technique of choice to assess eventual polyostotic localizations, but also allows for identification of soft-tissue inflammation surrounding osteomyelitis. 3 In our cohort, soft tissue edema was detected in more than half of the population, being associated with higher CRP. Even though bone and its periosteal shield could be considered as not metabolically connected with overlying muscles and fasciae, recent insights demonstrated that transcortical microvessels could link the intraosseous with the soft-tissue microenvironment. 34 Growth plates—the preferentially involved site in CNO 25 —are the most vascularized portion of long bones, especially in a growing skeleton, potentially accounting for the relationship with raised systemic inflammatory markers.
Differences about therapeutic approach emerged when patients were subgrouped according to the decade of diagnosis, and such changes in CNO management reflect the treatment guidelines issued in 2018. According to this consensus, patients refractory to a first-line 4-week course with NSAIDs should be candidates for csDMARDs or TNFi (with or without MTX) or IV-BPs, eventually associated with NSAIDs or GCs. 35 The same approach is adopted for subjects with active vertebral involvement, given that spinal lesions with demonstrated bone marrow edema on MRI convey a high risk of VF. In agreement with these guidelines, in the CAMELOT cohort, NSAIDs were usually administered to subjects without VF, whereas BPs were clearly the preferred option in patients with VF. This therapeutic choice translates to CNO the anti-fracturative effects that BPs have been shown to exert in multiple clinical settings, although the reduction of VF risk has yet to be demonstrated in CNO. In agreement with most recent literature, 36 BPs emerged as a valid pharmacological tool besides the anti-fracturative potential in the CAMELOT cohort: they allowed a 60% remission of pain and a 10% rate of radiological as well as overall remission, the highest we registered (Figure 3).
Limitations of the study include the limited size of the CAMELOT cohort and its retrospective design. The high prevalence of VFs in our cohort might unveil a selection bias, given that our institution is a referral center for pediatric orthopedics. Unfortunately, TBS software was not yet implemented on DXA machinery at our institution at the time of this study, thus preventing us from collecting data about TBS in CNO. We could not apply standardized clinimetric indices to evaluate disease activity. The lack of data about menarche or pubertal spurt limits the interpretation of data about growth kinetics. Preliminary data about NLR and PLR need to be confirmed in larger longitudinal studies.
Conclusion
The high prevalence of VF observed in the CAMELOT cohort highlights the importance—not only for rheumatologists—of enlisting CNO in the diagnostic approach to pediatric patients presenting with VFs. DXA might be a useful tool in CRMO, allowing the monitoring of the achievement of peak BMD to identify patients who could benefit from BP treatment.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251375183 – Supplemental material for Vertebral fractures and bone health in patients with chronic non-bacterial osteomyelitis at disease onset: insights from the monocentric CAMELOT cohort
Supplemental material, sj-docx-1-tab-10.1177_1759720X251375183 for Vertebral fractures and bone health in patients with chronic non-bacterial osteomyelitis at disease onset: insights from the monocentric CAMELOT cohort by Cecilia Beatrice Chighizola, Raffaele Di Taranto, Andrea Amati, Debora Pireddu, Stefania Costi, Francesco Baldo, Chiara Crotti, Paolo Trezza, Elisabetta Armiraglio, Maria Serena Bartoli, Massimo Varenna, Antonio Memeo, Stefano Bastoni, Achille Marino, Antonina Parafioriti and Roberto Caporali in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
