Abstract
Background:
Hormonal changes in menopause might interact with the presentation of underlying autoimmune diseases, such as systemic sclerosis (SSc).
Objectives:
Our study aimed to evaluate the association of (1) current menopausal status, (2) early menopause, and (3) disease onset during fertile or post-menopausal age on SSc clinical phenotype in a large SSc cohort from the Italian Systemic sclerosis Progression INvestiGation (SPRING-SIR) registry.
Design:
Female SSc patients from the SPRING-SIR registry, fulfilling the American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (EULAR) 2013 classification criteria, with data on SSc disease onset, menopausal status, and menopausal age, were eligible. SSc onset was categorized as pre-menopausal if SSc onset happened >1 year before menopause or as post-menopausal onset if it occurred >1 year after menopause. An early menopause was defined by a menopausal age <45 years.
Methods:
Descriptive statistics and regression models were built to test the association between current menopausal status, pre-menopausal disease onset, and early menopause with SSc-related features.
Results:
At baseline, 1157/1538 (75%) patients were in menopause, 632 (50.4%) had a pre-menopausal SSc onset, and 130 (14.4%) reported an early menopause. Post-menopausal patients had more frequent limited cutaneous SSc, anti-centromere antibody positivity, interstitial lung disease, and gastrointestinal manifestations. Pre-menopausal onset cases showed more frequent diffuse cutaneous involvement and peripheral vasculopathy. Patients with early menopause had more frequent peripheral vasculopathy and interstitial lung disease, being early menopause an independent risk factor for digital ulcers and lower diffusing capacity of the lung for carbon monoxide.
Conclusion:
Current post-menopausal status and early menopause may impact SSc presentation, being associated with vascular and gastrointestinal manifestations. Menopausal status and age should therefore be thoroughly addressed, aiming at better disease management.
Introduction
Biological sex has been variably correlated to the incidence, severity, and progression of autoimmune diseases, such as systemic sclerosis (SSc), along with lifestyle, genetic, hormonal, and environmental factors.1,2 Sexual hormones exert complex effects on the immune system, although their relationship is not completely understood. 3 Menopause represents a time of meaningful hormonal change, which might interact with an underlying autoimmune disease. 4 Despite the high prevalence of autoimmune disorders among female individuals,5,6 relevant data on the impact of menopause on the course of certain autoimmune rheumatic diseases are scarce. It has been suggested that menopause may affect both the onset and the activity of these conditions, acting both directly on the disease itself and indirectly on the comorbidity burden, likely through alterations in gonadal hormone levels or androgen/estrogen ratios. 4 In SSc, previous data reported the disease to be more common in women, although men present more severe internal organ complications and higher mortality.7–12 Other reports concern the investigation of menopause as a determinant of osteopenia,13–15 vasculopathy, 16 and specifically pulmonary arterial hypertension (PAH).17,18 Despite this preliminary evidence, it is not clear whether menopausal age could be linked to disease severity. Recent data have shown early menopause to be common in patients with SSc and related it to the presence of digital ulcers (DU). 19 Moreover, hormonal therapy in transgender individuals might increase the risk of developing SSc. 20
The study aimed to evaluate (1) the role of current menopausal status on SSc clinical features, (2) the impact of early menopause on the disease clinical presentation, and (3) the effect of disease onset during fertile or post-menopausal age on the SSc clinical phenotype, investigating the large SSc cohort included in the Systemic Sclerosis PRogression INvestiGation registry of the Italian Society of Rheumatology (SPRING-SIR).
Patients and methods
Study population and data collection
The study was performed on the multicentric, national SPRING-SIR registry cohort. Each of the 37 participating centers received approval from the local Ethical Committee (approval number OSS 15.010, Comitato Etico Area Vasta Centro—Toscana) and all participants provided written informed consent.
Eligible patients from the SPRING-SIR registry were female patients, aged >18 years, classified according to the 2013 American College of Rheumatology (ACR)/European Alliance of Associations for Rheumatology (ACR/EULAR) classification criteria for SSc, 21 and with available information on the parameter “current postmenopausal status.” In addition, available information on “age at menopause” and “year of SSc onset” was required as inclusion criteria for the subsequent analysis (Figure 1).

Flow chart of patient selection process.
Baseline information collected in the SPRING-SIR database included demographic data, clinical signs and symptoms of disease, organ involvement, previous and current therapies, as previously described. 22 According to the classification of cutaneous involvement proposed by Leroy et al., 23 patients were classified as limited cutaneous (lcSSc) or diffuse cutaneous (dcSSc) subsets. SSc onset was defined as the appearance of the first non-Raynaud’s phenomenon sign or symptom; consequently, disease duration covered the time from SSc onset to the inclusion in the registry. In addition, data regarding comorbidities and the overall comorbidity burden were also evaluated, the latter using the Charlson comorbidity index (CCI).24,25 To reduce the age bias between pre- and post-menopausal patients, we used a revised version of the CCI, which did not include age. The revised EUSTAR activity index (revEUSTAR-AI) was calculated to quantify SSc-related disease activity. 26
Study design
SSc onset was categorized as pre-menopausal if the disease first manifested in the fertile age, at least 1 year before menopause onset, otherwise as post-menopausal if SSc manifested at least 1 year after menopause, otherwise as peri-menopausal onset. Early menopause was defined as menopausal age <45 years, as previously proposed. 27
In line with the different aims of the study, we considered three different exposures in our analyses: (1) pre- (vs post) menopause status at baseline; (2) early (vs regular) menopause; and (3) SSc onset before menopause (vs after). Detailed patient selection is reported in Figure 1.
All comparisons between exposed and non-exposed groups were performed, including baseline data, at the time of enrollment in the SPRING-SIR registry. For the first analysis, pre- and post-menopausal status were compared in terms of prevalence and severity of organ involvement, as well as disease activity and comorbidity burden. For the second analysis, the sole post-menopause patients were considered, and a comparison between regular versus early menopause was performed, focusing on the same abovementioned outcomes. Finally, in the third part of the analysis, the menopausal status at disease onset was considered a risk factor. With this additional categorization, we compared pre-menopausal onset and post-menopausal onset groups.
Statistical analysis
Statistical analysis was performed using SPSS version 26.0 (IBM, Chicago, IL, USA) software. Given the observational nature of the registry, without pre-defined group comparisons and effect size, a traditional a priori sample size calculation was not performed. All eligible patients were included in the analysis to ensure representativeness. In addition, all models tested were based on highly prevalent dependent variables, and an appropriate number of predictors (on a 1:10 ratio with the outcomes). Demographic and disease-related features were analyzed through descriptive statistics. Results were presented as numbers and percentages (of available data) for categorical variables, otherwise as mean (standard deviation) or median (interquartile range) according to normal or non-normal distribution. The distribution of categorical and continuous variables across groups (pre- vs post-menopausal status, pre- vs post-menopausal SSc onset, and regular vs early menopausal age) was compared using the Chi-squared test (or Fisher’s test, when required) and Student’s t-test. Linear or logistic regression models were applied to test the association between current post-menopausal status, pre-menopausal disease onset, or early menopause with SSc-related characteristics, corrected for covariates selected according to literature evidence and opinion of the investigators. These included demographic data and clinical features, that is, age, disease duration, antibody status, cutaneous subset, nailfold capillaroscopy pattern, inflammatory biomarkers, smoking exposure, interstitial lung disease (ILD), previous pregnancy, and systolic pulmonary artery pressure on echocardiography (listed in detail in the Supplemental Tables 2–15, 17–22, 24–29). Collinearity between predictors was tested through Pearson correlation, with a cutoff for collinearity >|0.7|. Statistical significance level was set at a p value <0.05. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental File). 28
Results
Descriptive analysis
Based on the abovementioned inclusion criteria, 1538/2178 SSc patients were eligible for the study. In line with previous reports, 2/3 patients were classified as limited cutaneous SSc (n = 1015, 66%), with a mean age of 59.3 ± 13.7 years and a mean disease duration of 9.1 ± 7.6 years. Anti-topoisomerase I antibodies (ATA) were positive in 500 patients (33.2%), while anti-centromere antibodies (ACA) in 495 patients (35.1%). ILD was detected on high-resolution computed tomography in 38.2% of cases, PAH on right heart catheterization in 27 patients (1.8%), while scleroderma renal crisis was reported in 16 (1.1%) cases. At baseline, 61.2% of the study population received at least one vasodilator/vasoactive treatment, and 56.6% were on immunosuppressive medications. The mean revEUSTAR-AI at baseline was 1.8 ± 1.7. Regarding the menopausal status, 1157/1538 (75.2%) patients reported to be in menopause at the time of enrollment in the registry, with a mean menopausal age of 46.8 years. A thorough description of the study population is reported in Table 1.
Characteristics of the study population and stratification of SSc disease features according to menopausal status, at the time of inclusion in the SPRING-SIR registry.

ACA, anti-centromere antibodies; ANA, antinuclear antibodies; ARA, anti RNA polymerase III antibodies; ATA, anti-Scl-70 antibodies; BMI, body mass index; CRP, C-reactive protein; cs DMARDS, conventional synthetic disease modifying antirheumatic drugs; dcSSc, diffuse skin involvement; DLco, diffusing capacity of lung for carbon monoxide; DLco/VA, diffusing capacity of lung for carbon monoxide divided by alveolar volume; DPS, digital pitting scar; DU, digital ulcers; ERA, endothelin receptor antagonists; ESR, erythrocyte sedimentation rate; FEV-1, forced expiratory volume in 1 second; FVC, forced vital capacity; ILD, interstitial lung disease; lcSSc, limited skin involvement; LVEF, left ventricular ejection fraction; mRSS, modified Rodnan Skin Score; NVC, nailfold videocapillaroscopy; NYHA, New York Heart Association; PAH, pulmonary arterial hypertension; PDE5i, phosphodiesterase 5 inhibitors; revEUSTAR-AI, revised EUSTAR activity index; RP, Raynaud’s phenomenon; sPAP, systolic pulmonary artery pressure; SRC, scleroderma renal crisis; SSc, systemic sclerosis; SSc sine scleroderma, no skin involvement; TFR, tendon friction rubs.
Bold indicate statistical significant result, with p<0.05.
Comparison between current pre-menopausal versus post-menopausal status
Post-menopausal patients were more frequently ACA positive (35.1% vs 26.1%; p < 0.001), with a lcSSc subset (70.5% vs 52.2%; p < 0.001) and had a longer disease duration (9.7 ± 8.0 vs 7.2 ± 5.8; p < 0.023) than pre-menopausal cases. In addition, post-menopausal patients had a lower prevalence of DU (18.0% vs 23.5%; p = 0.020) and digital pitting scar (DPS, 43.9% vs 50.9%; p = 0.010). Conversely, post-menopausal patients reported more frequent gastrointestinal symptoms. Both ILD and PAH were more prevalent in the post-menopausal than in the pre-menopausal group (40.4% vs 31.5%; p = 0.002 and 5.2% vs 0.8%; p = 0.027, respectively). Overall, vasodilating or vasoactive treatment was more frequent in the pre-menopausal group (67.2% vs 59.3% in the post-menopausal group; p = 0.006), as well as both biological and conventional DMARDs (8.1% vs 3.6%; and 38.1% vs 29.0%, respectively). Despite these differences in clinical presentation, the revEUSTAR-AI showed comparable values between the two groups. Even after removing the score component of age, post-menopausal patients presented a higher CCI score. Additional, detailed data are present in Table 1 and Supplemental Table 1.
When testing the current menopausal status as a risk factor of disease-associated manifestations using multivariable regression models, we could not adjust for age, given the collinearity between this parameter and menopausal status (r = 0.736). Still, after adjustment for the remaining covariates, the post-menopausal status resulted as a statistically significant, independent predictor of the presence of gastric symptoms (OR 1.447, 95% CI 1.045 to 2.002, p = 0.026) and ILD (OR 1.775, 95% CI 1.335 to 2.360, p < 0.001), while it showed a negative association with DU (OR 0.637, 95% CI 0.454 to 0.892, p = 0.009), DPS (OR 0.659, 9%% CI 0.499 to 0.870, p = 0.003), and the diffuse cutaneous subset (dcSSc, OR 0.578, 95% CI 0.424 to 0.786, p < 0.001). In addition, post-menopausal status was associated with lower values of diffusion capacity of the lung for carbon oxide (DLco) (β = −2.791, 95% CI −5.910 to −0.327, p = 0.039) and left ventricle ejection fraction (LVEF) (β = −0.965, 95% CI from −1.852 to −0.078, p = 0.033), but a higher estimated systolic pulmonary arterial pressure (sPAP) on echocardiography (β = 5.339, 95% CI 3.049 to 7.629, p < 0.001) (Figure 2(a)–(e)). Conversely, no independent association was detected with other organ involvements (additional data are presented in Supplemental Tables 2–15).
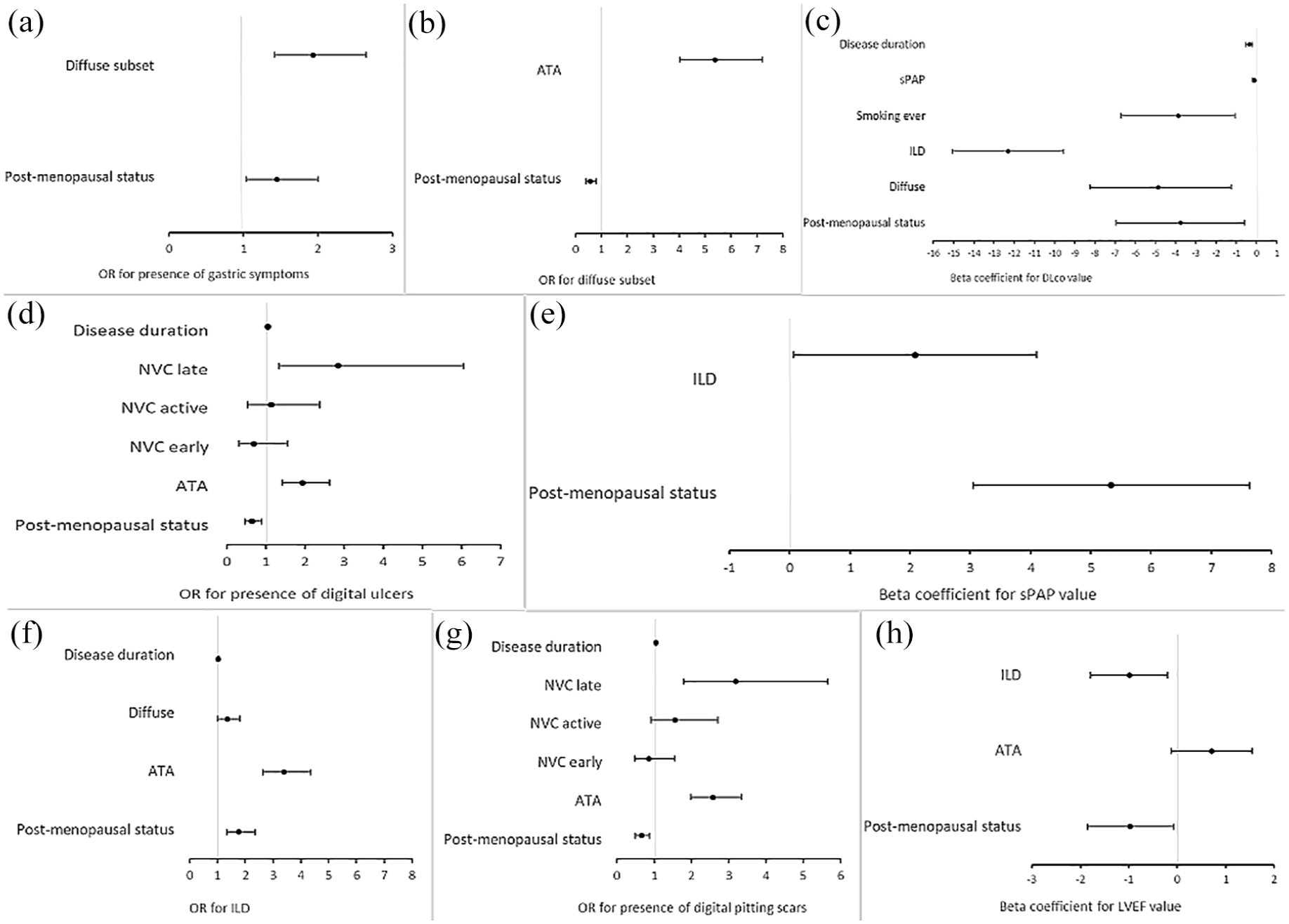
Multivariable regression models testing post-menopausal status as a predictor for SSc-related clinical features: (a) presence of gastric symptoms; (b) presence of diffuse cutaneous subset; (c) value of diffusion capacity of the lung for carbon monoxide (DLco); (d) presence of digital ulcers; (e) value of sPAP; (f) presence of ILD; (g) presence of digital pitting scars; and (h) value of LVEF.
Comparison between regular and early menopausal age
Focusing on the 902 post-menopausal cases with available data on menopausal age, an early menopause onset was reported by 130 patients (14.4%). When early and regular menopausal age groups were compared, patients with early menopausal age presented more frequently with DU (26.0% vs 16.0%, p = 0.008), history of scleroderma renal crisis (3.2% vs 0.5%, p = 0.017), and presence of ILD (47.7% vs 37.3%, p = 0.026), in line with significantly lower FVC% (97.4% vs 104.0%, p = 0.005) and DLco% values (63.9% vs 68.7%, p = 0.030), as well as more frequently complaining of dyspnea (54.4% vs 41.5%, p = 0.008). In line with the higher prevalence of lung involvement, we observed that early menopausal patients were more frequently exposed to therapy with cyclophosphamide, which is a well-known risk factor for ovarian insufficiency and early menopause. However, the exposure to this medication was not statistically different among the groups. In addition, the menopausal age of patients exposed to cyclophosphamide while fertile was lower than the patients who never received it, but again, not statistically different (47.7 ± 6.6 years vs 48.9 ± 5.09 years, p = 0.363). Detailed data are presented in Table 2, with additional information shown in Supplemental Table 16.
Characteristics of the study population and stratification of SSc disease features according to regular or early menopause, at the time of inclusion in the SPRING-SIR registry.

ACA, anti-centromere antibodies; ANA, antinuclear antibodies; ARA, anti RNA polymerase III antibodies; ATA, anti-Scl-70 antibodies; BMI, body mass index; CCI, Charlson comorbidity index; CYC, cyclophosphamide; dcSSc, diffuse skin involvement; DLco, diffusing capacity of lung for carbon monoxide; DU, digital ulcers; FVC, forced vital capacity; ILD, interstitial lung disease; lcSSc, limited skin involvement; LVEF, left ventricular ejection fraction; mRSS, modified Rodnan Skin Score; NVC, nailfold videocapillaroscopy; NYHA, New York Heart Association; PAH, pulmonary arterial hypertension; revEUSTAR-AI, revised EUSTAR activity index; RP, Raynaud’s phenomenon; sPAP, systolic pulmonary artery pressure; SRC, scleroderma renal crisis; SSc, systemic sclerosis; SSc, sine scleroderma: no skin involvement; TFR, tendon friction rubs.
Bold indicate statistical significant result, with p<0.05.
Early menopausal age was tested as a predictor of the abovementioned selected SSc features and adjusted for selected confounders. After the backward selection, early menopausal age was a statistically significant risk factor for the presence of DU (OR 1.788, 95% CI 1.006 to 3.178, p = 0.048) and lower DLco% values (β = −6.835. 95% CI −11.602 to −2.067, p = 0.005). In addition, early menopausal age showed a trend toward statistical significance as a risk factor for presence of ILD (OR 1.473, 95% CI 0.964 to 2.253, p = 0.074) and lower FVC% values (β = −4.592, 95% CI −9.403 to −0.219, p = 0.061) (Figure 3(a)–(d)). The remaining models showed a non-statistically significant association of early menopause with other clinical features (Supplemental Tables 17–22).

Multivariable regression models testing early menopause as a predictor for SSc-related clinical features: (a) presence of digital ulcers; (b) presence of interstitial lung disease; (c) value of FVC; and (d) value of DLco.
Comparison between pre- versus post-menopausal SSc onset
Among the 1254/1538 patients with available data on menopausal status, menopause onset, and SSc onset dates, 632 (50.6%) reported the onset of SSc in the pre-menopausal age, 568 (45.3%) a post-menopausal SSc disease onset, leaving a peri-menopausal onset in the remaining 54 (4.1%) cases. Given the paucity of the peri-menopausal group, this was excluded from the following analysis. In comparison to post-menopausal onset patients, pre-menopausal onset SSc cases were more frequently ATA positive (39.1% vs 26.2%; p < 0.001) and presented the dcSSc subset (22.9% vs 13.0%; p = 0.001). In addition, both disease duration and mRSS were higher in the pre-menopausal onset group (10.8 years vs 6.8 years, respectively, p < 0.001, and 7.0 vs 5.2, respectively, p < 0.001). Pre-menopausal SSc onset cases presented, on average, with a higher baseline revEUSTAR-AI (2.0 vs 1.6, p < 0.001), more frequent DPS (55.9% vs 38.6%, p < 0.001), DU (22.8% vs 14.6%, p < 0.001), more neoangiogenesis phenomena detected on nailfold videcapillaroscopy (20.3% vs 14.1%; p = 0.006), and a trend for higher prevalence of a late scleroderma pattern (27.4% vs 22.6%; p = 0.161), compared to the post-menopausal onset cases. In line with this clinical picture, the percentage of patients under vasodilator or vasoactive treatment was significantly higher (70.6% vs 54.6%, p < 0.001) in the pre-menopausal group, mainly endothelin-1 receptor antagonists (27.5% vs 15.7%; p < 0.001).
Differently, the mean values of resting estimated sPAP, as well as the PAH prevalence, were higher in the post-menopausal onset group (24.4 mmHg vs 19.5 mmHg, p < 0.001 and 6.6% vs 2.0%, p < 0.020). No statistically significant differences were found regarding the presence of ILD, although a significantly lower FVC% was detected in the pre-menopausal onset group (99.8% vs 104.4%; p = 0.001), both within the range of normality. Regarding the use of immunosuppressive treatments, pre-menopausal onset patients received more biologic DMARDS therapy (6.8% vs 2.8%; p < 0.002), in line with the higher prevalence of the dcSSc subset. The CCI score (without age) was higher in the post-menopausal onset group (0.7 vs 0.3; p < 0.001). All detailed results are reported in Table 3 and Supplemental Table 23.
Characteristics of the study population and stratification of SSc disease features according to pre- or post-menopausal SSc onset, at the time of inclusion in the SPRING-SIR registry.

ACA, anti-centromere antibodies; ANA, antinuclear antibodies; ARA, anti RNA polymerase III antibodies; ATA, anti-Scl-70 antibodies; BMI, body mass index; CCI, Charlson comorbidity index; dcSSc, diffuse skin involvement; DLco, diffusing capacity of lung for carbon monoxide; DPS, digital pitting scar; DU, digital ulcers; FVC, forced vital capacity; ILD, interstitial lung disease; lcSSc, limited skin involvement; LVEF, left ventricular ejection fraction; mRSS, modified Rodnan Skin Score; NVC, nailfold videocapillaroscopy; NYHA, New York Heart Association; PAH, pulmonary arterial hypertension; revEUSTAR-AI, revised EUSTAR activity index; RP, Raynaud’s phenomenon; sPAP, systolic pulmonary artery pressure; SRC, scleroderma renal crisis; SSc, systemic sclerosis; SSc, sine scleroderma, no skin involvement; TFR, tendon friction rubs.
Bold indicate statistical significant result, with p<0.05.
When testing pre-menopausal SSc onset as a risk factor of cutaneous and pulmonary fibrosis features, as well as pulmonary and peripheral vascular manifestations using multivariable regression models with backward selection, pre-menopausal SSc onset was neither statistically significant nor an independent predictor for any of the abovementioned parameters (Supplemental Tables 24–29).
Discussion
The analysis carried out on Italian SSc patients from the SPRING-SIR registry shows that both menopausal status and early menopausal age may have an impact on the SSc clinical features and should therefore be taken into account for both risk stratification and patient management.
Previous studies have hypothesized an increased risk of early menopause in SSc females, compared to the general population.6,13,19,29 Although in our cohort only 14.4% of post-menopausal SSc women reported an early menopause, we observed that the average menopausal age in our cohort was around 47 years, which is lower than the values reported for the general population, usually ranging between 50 and 52 years. 30 When analyzing only postmenopausal patients, our study showed that the early menopausal age was associated with a higher prevalence of DU and lower DLco% values, data that are in agreement with the suggested link between the menopause-related hormonal changes, in particular estrogens, and the control of the vascular tone.
Moreover, in our cohort, SSc patients with a pre-menopausal disease onset were more frequently ATA positive, with more prevalent fibrotic and vasculopathic involvement, although this was not detected as a significant, independent risk factor.
In line with the recent data from Liyi et al. 19 reporting that age at disease onset positively correlated with DU and menopausal age, also after adjustment for confounders, we can also observe that pre-menopausal onset patients presented with higher prevalence of DU, which goes in line with higher frequency of treatment with vasoactive drugs (70.6% vs 54.9%; p ⩽ 0.001), but a lower prevalence of PAH (2.0% vs 6.6%; p = 0.020). These data point toward the potential protective effect of vasoactive therapies that we have recently published in another analysis of the SPRING-SIR registry, 31 in which the use of bosentan for DU prevention associates with reduced risk of PAH risk. Moreover, the EUSTAR group reported that patients with late-onset SSc (after the age of 65) present more frequently with PAH, 32 further supporting the face validity of our results. As previous in vitro data have shown that estrogens can modulate the arterial tone through endothelial-dependent mechanisms, 33 the menopause-related loss of estrogen has been suggested to trigger the pathogenic mechanisms that lead to vascular damage and, consequently, to disease-related vascular manifestations, including the increased risk of developing new-onset PAH in SSc patients. 17 As a consequence, hormonal replacement therapy was hypothesized to protect against the development of isolated PAH, 34 although this was not further investigated to the best of our knowledge. Although the higher prevalence of PAH in our post-menopausal patients may be in line with this hypothesis, unfortunately, the small numbers of PAH cases in our cohort limit further analyses in this direction. However, we could still observe that the post-menopausal status is significantly associated with higher sPAP and lower DLco and LVEF% values, in line with both the higher prevalence of cardiopulmonary complications in this population and the biological association between post-menopausal status and advanced age. Conversely, we found a negative association of post-menopausal status and peripheral vasculopathy, which could be interpreted as a protective effect. This is in contrast with previous reports showing the improvement of abnormal endothelial function in postmenopausal women with Raynaud’s phenomenon secondary to SSc, with both short- and long-term therapy with conjugated estrogens having been previously reported.35,36 This result is similar to what has been described in healthy postmenopausal women after estrogen therapy. 37 Indeed, the use of conjugated estrogens in our post-menopausal population could not be excluded, given the lack of information about hormonal therapy in our dataset.
As expected, SSc patients in the post-menopausal status at baseline were older, more frequently had lcSSc, were ACA positive, and with more gastrointestinal manifestations, ILD, and PAH. These results are, in fact, in line with previous data that hypothesize an association between sexual hormone levels and clinical SSc phenotypes, mostly linked with the reported profibrotic effect of estrogens in vitro.12,17 It is well known that estrogens may increase the production of extracellular matrix protein in skin fibroblast culture of SSc patients and stimulate healthy skin fibroblasts to produce TGF-β1. 33 Our results are in agreement with previous data showing menopause influence on skin fibrosis, in particular in dcSSc. In the post-menopausal status, our SSc patients presented less frequently with the dcSSc subset and, accordingly, with a lower mean mRSS, although only the former was independently predicted after adjustment in multivariable analysis. 12
The present study has limitations. The first concerns the post hoc nature of the analysis, which included data coming from an already existing registry and not created ad hoc for our specific research question. In fact, data about hormonal levels, previous/ongoing hormonal therapy, as well as the presence of peri-menopausal symptoms, were not included in the dataset. In addition, both the menopausal status and age at menopause were self-reported by the patients, leading to a possible recall bias when trying to provide this information, and it was not confirmed with specific hormonal tests. Additional concerns might arise considering that the analyzed sample from a national registry may not be representative of the global SSc population, in particular given the vast majority of Caucasian patients and European ancestors in our cohort. However, the numerosity of the sample and the disease characteristics, in line with other international cohorts, support the representativeness of our population and the generalizability of our results. Similarly, the availability of information and the exclusion of certain cases might reflect a selection bias. However, the characteristics of the patients excluded from the analysis were in line with those included (data not shown). Another limitation is the availability of baseline data at the time of enrollment in the dataset, as well as the cross-sectional nature of the analysis. Indeed, it would have been optimal to analyze the effect of pre- or post-menopausal onset, as well as that of early menopausal age, over time during the disease course. Similarly, we could not analyze the effect of the same factor on disease presentation when menopause was happening during the disease course. In line with the second limitation, the short-term duration of the registry and the low number of new menopause events and deaths recorded, together with the loss to follow-ups, did not allow for performing in-depth longitudinal or prognostic analysis regarding both the impact on disease manifestation and mortality.
Still, our study presents strengths. We have investigated an important and unexplored area of female SSc, providing additional information about the role of menopause in SSc. Based on our data and the previous literature, we might hypothesize that menopause affects not only the disease expression but also the disease activity and associated comorbidities. In fact, although there is no independent link between pre- or post-menopausal onset of SSc with specific disease manifestations, we still observe a strong association of specific disease phenotypes with the post-menopausal status, which are additionally marked in patients with early menopause. Prospective, multicenter studies are warranted to help clarify the role of menopausal age in the development, expression, and outcome of the disease and to evaluate the role of hormones and hormonal therapies as well.
Conclusion
Our data suggest that both menopausal status and age at menopause may impact SSc clinical features, being associated with disease characteristics such as vascular, pulmonary, and gastrointestinal manifestations. The present findings underline the need for a global approach to female SSc patients, including current post-menopausal status and menopausal age in the routine clinical assessment.
A careful evaluation of sex-related issues, encompassing not only pregnancy outcomes, but also menstrual disorders, hormonal conditions, and menopausal status, is of paramount importance to tailor a personalized management of an SSc patient.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251354898 – Supplemental material for Menopause in systemic sclerosis: the impact on clinical presentation in a multicenter cross-sectional analysis from the National Registry of the Italian Society for Rheumatology (SPRING-SIR)
Supplemental material, sj-docx-1-tab-10.1177_1759720X251354898 for Menopause in systemic sclerosis: the impact on clinical presentation in a multicenter cross-sectional analysis from the National Registry of the Italian Society for Rheumatology (SPRING-SIR) by Martina Orlandi, Dilia Giuggioli, Clodoveo Ferri, Rossella De Angelis, Valeria Riccieri, Fabio Cacciapaglia, Silvia Laura Bosello, Veronica Codullo, Gianluigi Bajocchi, Lorenzo Dagna, Corrado Campochiaro, Giacomo De Luca, Giovanni Zanframundo, Rosario Foti, Giovanna Cuomo, Alarico Ariani, Edoardo Rosato, Francesco Girelli, Elisabetta Zanatta, Ilaria Cavazzana, Francesca Ingegnoli, Maria De Santis, Giuseppe Murdaca, Giuseppina Abignano, Giorgio Petitti, Alessandra Della Rossa, Maurizio Caminiti, Anna Maria Iuliano, Giovanni Ciano, Lorenzo Beretta, Gianluca Bagnato, Ennio Lubrano, Ilenia De Andres, Alessandro Giollo, Marta Saracco, Cecilia Agnes, Edoardo Cipolletta, Federica Lumetti, Amelia Spinella, Luca Magnani, Elisa Visalli, Carlo Iandoli, Antonietta Gigante, Greta Pellegrino, Erika Pigatto, Maria Grazia Lazzaroni, Franco Franceschini, Elena Generali, Gianna Mennillo, Simone Barsotti, Giuseppa Pagano Mariano, Federica Furini, Licia Vultaggio, Simone Parisi, Clara Lisa Peroni, Gerolamo Bianchi, Enrico Fusaro, Gian Domenico Sebastiani, Marcello Govoni, Salvatore D’Angelo, Franco Cozzi, Andrea Doria, Carlo Salvarani, Florenzo Iannone, Serena Guiducci, Silvia Bellando-Randone, Marco Matucci-Cerinic and Cosimo Bruni in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251354898 – Supplemental material for Menopause in systemic sclerosis: the impact on clinical presentation in a multicenter cross-sectional analysis from the National Registry of the Italian Society for Rheumatology (SPRING-SIR)
Supplemental material, sj-docx-2-tab-10.1177_1759720X251354898 for Menopause in systemic sclerosis: the impact on clinical presentation in a multicenter cross-sectional analysis from the National Registry of the Italian Society for Rheumatology (SPRING-SIR) by Martina Orlandi, Dilia Giuggioli, Clodoveo Ferri, Rossella De Angelis, Valeria Riccieri, Fabio Cacciapaglia, Silvia Laura Bosello, Veronica Codullo, Gianluigi Bajocchi, Lorenzo Dagna, Corrado Campochiaro, Giacomo De Luca, Giovanni Zanframundo, Rosario Foti, Giovanna Cuomo, Alarico Ariani, Edoardo Rosato, Francesco Girelli, Elisabetta Zanatta, Ilaria Cavazzana, Francesca Ingegnoli, Maria De Santis, Giuseppe Murdaca, Giuseppina Abignano, Giorgio Petitti, Alessandra Della Rossa, Maurizio Caminiti, Anna Maria Iuliano, Giovanni Ciano, Lorenzo Beretta, Gianluca Bagnato, Ennio Lubrano, Ilenia De Andres, Alessandro Giollo, Marta Saracco, Cecilia Agnes, Edoardo Cipolletta, Federica Lumetti, Amelia Spinella, Luca Magnani, Elisa Visalli, Carlo Iandoli, Antonietta Gigante, Greta Pellegrino, Erika Pigatto, Maria Grazia Lazzaroni, Franco Franceschini, Elena Generali, Gianna Mennillo, Simone Barsotti, Giuseppa Pagano Mariano, Federica Furini, Licia Vultaggio, Simone Parisi, Clara Lisa Peroni, Gerolamo Bianchi, Enrico Fusaro, Gian Domenico Sebastiani, Marcello Govoni, Salvatore D’Angelo, Franco Cozzi, Andrea Doria, Carlo Salvarani, Florenzo Iannone, Serena Guiducci, Silvia Bellando-Randone, Marco Matucci-Cerinic and Cosimo Bruni in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The SPRING-SIR registry was supported by the Italian Society of Rheumatology (SIR). We acknowledge the contribution of: Amato Giorgio (AOU Policlinico – Vittorio Emanuele, Catania); Benenati Alessia (AOU Policlinico – Vittorio Emanuele, Catania); Calabrese Francesca (SSD Reumatologia, Reggio Calabria); Carignola Renato (AOU San Luigi Gonzaga, Orbassano (TO)); Dall’Ara Francesca (UO Medicina Interna-Ambulatorio Reumatologia, Ospedale di Lodi); De Cata Angelo (Ospedale Casa Sollievo della Sofferenza, San Giovanni Rotondo (FG)); Doveri Marica (ASL3Genova); Girelli Francesco (Ospedale GB Morgagni, Forlì); Nicoletta Romeo (Rheumatology Unit ASO SCrocea e Carle Hospital Cuneo); Sambataro Gianluca (Azienda Ospedaliera Cannizzaro, Catania); Talotta Rossella (L.Sacco Hospital, Milan); Study Center of the Italian Society of Rheumatology (SIR): Carlo Alberto Scirè (School of Medicine and Surgery, University of Milano-Bicocca, Milan; and Epidemiology Unit, Italian Society for Rheumatology, Milan); Gianpiero Landolfi (Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy); Davide Rozza (Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy); Greta Carrara (Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy), Anna Zanetti (Epidemiology Unit, Italian Society for Rheumatology, Milan, Italy).
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
Patient and public involvement
Patients were not directly involved in setting the research question, the outcome measures, or the design of this study. However, unmet clinical needs that emerged during routine clinical practice were considered. Patients were informed of the results of this study upon request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
